Abstract
Six ovine fetal brains were harvested 33 to 35 days postchallenge from 5 ewes, each of which was given 3000 Toxoplasma gondii oocysts on day 90 of pregnancy. Histopathologic examination of transverse sections taken at 13 levels in the fetal brains revealed the presence of toxoplasmosis-related lesions in all 6 brains. However, lesions were not randomly distributed (P = .007); they were most numerous at the level of the optic tract, the rostral margin of the pons, and 4 mm caudal to the ansate sulcus and were absent in all sections at the level of the caudal cerebellum. Lesion distribution may be due to hemodynamic factors, differences in the expression of endothelial surface receptor molecules at the level of the blood-brain barrier, or the presence of localized permissive/inhibitory factors within the brain. The results have implications for the selection of areas of brain from aborted ovine fetuses to be examined histopathologically for laboratory diagnosis.
Toxoplasma gondii is a globally ubiquitous protozoan parasite that infects many warm-blooded species. 10,17 Intrauterine infections of the placenta and fetus occur in humans and sheep where the consequences include placentitis, fetal death and resorption, abortion, and stillbirth. 6 In Ireland, T. gondii is the most commonly diagnosed cause of abortion in sheep, 1 and toxoplasmosis is estimated to cost the UK sheep industry £11 million annually. 4
In experimental and field cases of ovine T. gondii infection, characteristic histopathologic lesions are found in the placenta and fetal brain and less frequently in the fetal heart, lung, and liver. 6,17 The diagnosis of field cases is usually made by the identification of the typical histopathologic lesions in fetal brain and/or placental tissue; as placental tissue is commonly omitted from laboratory submissions, diagnosis is frequently based on the presence of the characteristic central nervous system (CNS) lesions alone. 25 Typical CNS lesions in ovine fetal brain consist of gliotic foci, which may occur in either gray or white matter, and some of these surround central areas of necrosis that are occasionally mineralized. 17 Intralesional T. gondii organisms may or may not be visible in sections stained with hematoxylin and eosin (HE). Nonsuppurative leptomeningitis, sometimes involving the submeningeal brain parenchyma, is described in some cases. 17
Although the characteristic appearance of the T. gondii–induced lesions has been described, the distribution of these lesions within the ovine fetal brain has not been documented. Such knowledge may not only contribute to our understanding of the pathogenesis of ovine fetal toxoplasmosis but also prove useful in the identification of sites at which lesions occur with higher frequency and increase the diagnostic sensitivity of histopathologic examination of fetal brain. The present study was designed to determine whether lesion density varies significantly among different areas of the fetal brain in experimental congenital T. gondii infection of sheep.
Materials and Methods
This study was conducted using animal tissue samples generated as part of a larger, multidisciplinary investigation of experimental toxoplasmosis in pregnant sheep. 16,23,33
Animals
Nine adult female sheep (Cheviot and Suffolk cross) seronegative for T. gondii antibodies were included in the study. Synchronized breeding was conducted as previously described. 16 Pregnancy was confirmed in all 9 ewes by transabdominal ultrasound scanning on day 70 of gestation. The ewes were housed and penned individually from day 83 of gestation. Ewes were randomly assigned to control (n = 4) and challenge groups (n = 5). The experimental use of animals was licensed by the appropriate Irish authorities and conducted in compliance with the policies operated at the Central Veterinary Research Laboratory of the Department of Agriculture, Fisheries and Food.
Experimental Infection
T. gondii oocysts were harvested from the feces of cats that had been fed infected mouse brain tissue (Moredun 4 strain). The Moredun 4 strain was originally isolated in mice inoculated with fetal brain material from a field case of ovine abortion. Oocysts were allowed to sporulate suspended in 2% sulfuric acid at 22°C for 7 days and were stored in the same solution at 4°C for 3 months, until required. To prepare the inocula, the stock oocyst suspension was agitated to disperse the oocysts. Sporulated oocysts were then identified and enumerated in the suspension using a Neubauer hemocytometer. Individual challenge doses of 3000 sporulated oocysts were prepared and made up to 5 ml with distilled water. The 3000-oocyst dose was selected on the basis of the results of a previous temperature response study in T. gondii–seronegative male sheep challenged with oocysts from the same stock suspension (data not shown).
Ewes in the challenge group were orally inoculated with 3000 sporulated oocysts of the Moredun 4 T. gondii strain at day 90 of pregnancy. The control ewes were administered a placebo (5 ml of distilled water) by mouth.
Clinical assessment of ewes was carried out daily, and maternal venous blood samples were collected from the ewes on the day of challenge (day 0) and at 14 and 21 days postchallenge (dpc) as well as on the day of euthanasia (33 or 35 dpc). Sera were separated on the day of collection and stored frozen at –80°C. A commercial T. gondii antibody ELISA test (IDScreen Toxoplasmosis Indirect, ID-Vet, Montpellier, France) was used to screen maternal sera.
Ewes were euthanized at 33 (n = 5) or 35 dpc (n = 4). Four fetuses from challenged ewes and 2 fetuses from control ewes were obtained on 33 dpc, while 2 fetuses from challenged ewes and 2 fetuses from control ewes were obtained on 35 dpc. Upon opening the calvarium, the meninges were dissected using a new disposable scalpel and forceps for each fetus. A sample of brain tissue approximately 1 cm3 was excised from the rostral aspect of the right cerebral hemisphere and frozen at –80°C for subsequent testing by polymerase chain reaction (PCR). The remainder of the fetal brain was fixed in 10% neutral buffered formalin solution.
Neurohistopathology
To facilitate serial sectioning using a macrotome, each formalin-fixed brain was immersed in molten (at 45 deg C) proprietary bacteriology agar (Bacto Brucella Agar, Difco Laboratories, Detroit, Michigan), prepared according to the manufacturer’s instructions (equivalent to 1.5% agarose), and then allowed to solidify. Four-millimeter-thick transverse sections of agar-encased whole brain were prepared for each of 13 sample sites defined by anatomical features on the brain surface (Fig. 1). These transverse sections were divided between 2 or 3 histology cassettes, if necessary, so that all of each transverse brain section was processed routinely to paraffin-embedded tissue blocks. Tissue orientation was maintained during processing to ensure that the face of each tissue block was the caudal surface of a transverse section. Five-micrometer-thick tissue sections were cut from each block, mounted on glass slides, and stained routinely with HE. The sections were examined by one pathologist who was blinded to the infection status of the fetuses. The number of discrete lesions attributed to T. gondii infection per tissue section was recorded, and the total lesion score for each brain level per fetus was calculated.
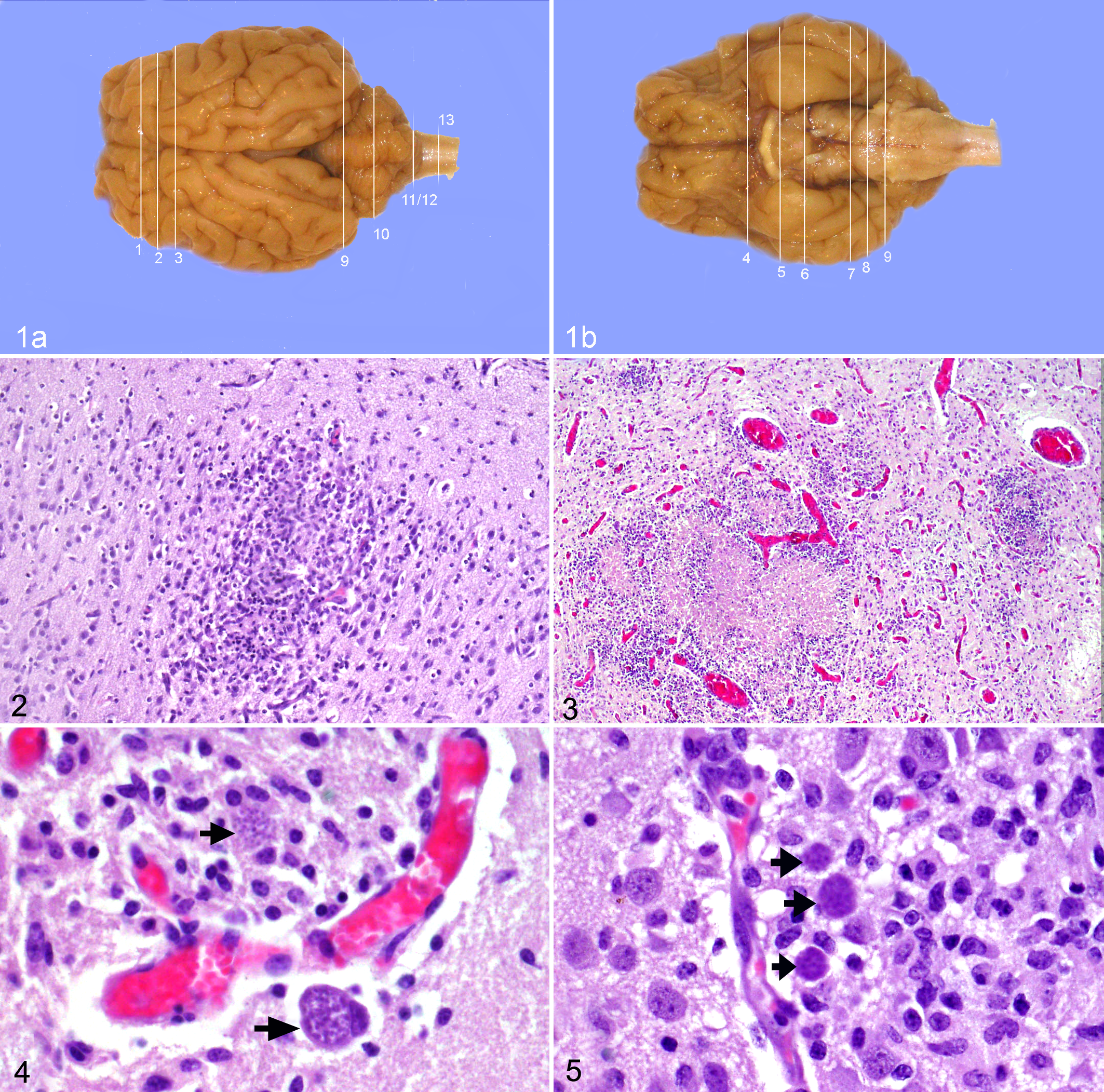
(a) Brain; ovine fetus; dorsal surface, gross. (b) Brain; ovine fetus; ventral surface, gross. The 13 sampling sites were defined by anatomical structures on the dorsal and ventral surfaces of the ovine fetal brain: (1) cruciate sulcus, (2) ansate sulcus, (3) 4 mm caudal to ansate sulcus, (4) level of the optic chiasm (the optic chiasm is absent from this specimen), (5) optic tract, (6) mammillary body, (7) rostral margin of pons, (8) midpons, (9) rostral cerebellum (junction of the pons with the medulla, just rostral to the root of the abducens nerve), (10) midcerebellum, (11) caudal cerebellum, (12) obex, (13) cranial spinal cord.
The area of each HE-stained tissue section was measured using Image J software (imagej.nih.gov/ij). Briefly, the HE-stained tissue sections were photographed on a white background using a tripod-mounted digital camera. With Image J, each image was first converted to 8-bit black-and-white; the scale of the image was then set with reference to a photographic scale within each image. (Details of the procedure used to measure the area of tissue sections are available as a supplementary file to the online version of this article, available at http://vet.sagepub.com/supplemental.) For each brain, the total area for each transverse brain section was determined by summing the areas measured for the individual tissue sections from the same section level. The lesion density for each whole transverse brain section was calculated by dividing the total lesion count for the section by its area.
Real-time PCR
DNA was extracted from 20 mg of brain tissue using the High Pure PCR Template Preparation Kit (Roche). The T. gondii real-time PCR technique has been described. 16 The target sequence was the 529–base pair repeat element of T. gondii. 13 Each sample was tested in triplicate. Every test run included positive and negative control samples in addition to DNA extracts from fetal brain tissue samples. Results were expressed as copy numbers per milligram of sample.
Statistics
To test the hypothesis that T. gondii lesion density does not vary significantly within the fetal brain, the lesion density (number of lesions per cm2) of the 13 sampling levels were compared using the Friedman repeated measures test within Minitab 16.0 statistical software (Minitab.com). Post hoc testing was conducted where significant differences were detected in the data to identify the transverse sections with significantly different mean ranks, as previously described. 9
Results
Serology
All 9 ewes were seronegative for antibodies to T. gondii at the initial prebreeding time point and on the day of experimental challenge (0 dpc). Four of the 5 challenged sheep seroconverted by 14 dpc, and all 5 seroconverted by 21 dpc (data not shown). All the control ewes remained seronegative throughout the study.
Neurohistopathology
Encephalitis was present in all 6 fetuses collected from the experimentally infected ewes; lesions of encephalitis were not detected in any of the unchallenged control fetuses. The lesions attributable to T. gondii included foci of gliosis without a central area of necrosis (Fig. 2), foci of necrosis surrounded by a margin of reactive glial cells (Fig. 3), and focally extensive areas of nonsuppurative encephalitis. Each discrete focus was counted as a single lesion whether intralesional protozoa were apparent or not. However, protozoan forms frequently seen in association with these lesions included tachyzoites, tachyzoite pseudocysts, and bradyzoites (Figs. 4, 5). The lesion density results are listed in Table 1.
Lesion Density by Transverse Section Level, Total Lesion Number, and Real-time Polymerase Chain Reaction Results for Toxoplasma gondii–Infected Fetal Brains a
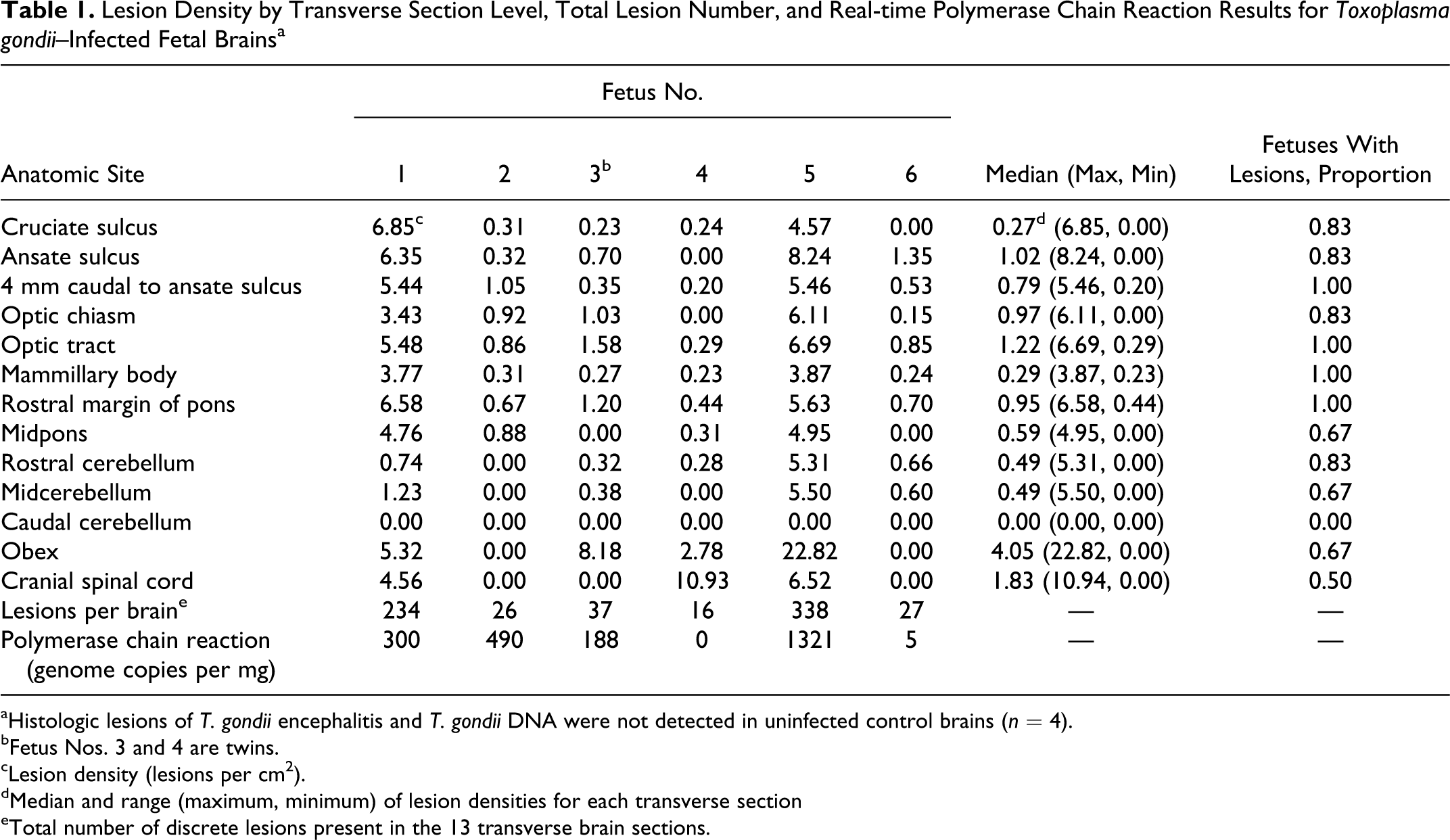
aHistologic lesions of T. gondii encephalitis and T. gondii DNA were not detected in uninfected control brains (n = 4).
bFetus Nos. 3 and 4 are twins.
cLesion density (lesions per cm2).
dMedian and range (maximum, minimum) of lesion densities for each transverse section
eTotal number of discrete lesions present in the 13 transverse brain sections.
Other lesions observed in infected fetal brains but not included in the lesion count were focal aggregates of mononuclear cells in the leptomeningeal space, usually overlying foci of nonsuppurative encephalitis or gliosis (Fig. 6). Similar perivascular infiltrates were present around some blood vessels adjacent to parenchymal lesions. The mononuclear infiltrate was composed of lymphocytes and macrophages, although eosinophils were also present in some of the meningeal and perivascular infiltrates. Leukoencephalomalacic foci of hypereosinophilic necrotic white matter were present in low numbers in the periventricular cerebral white matter of 2 of the infected lambs.
Cerebrum; ovine, fetus No. 2. Transverse section, 4 mm caudal to ansate sulcus. A focus of coagulative necrosis surrounded by a narrow margin of gliosis in the cerebrum with adjacent nonsuppurative meningitis; Toxoplasma gondii encephalitis. HE.
Lesion Distribution
Lesions were detected in 12 of the sampling levels in at least 3 of the 6 infected fetuses. A significant difference in lesion density among section levels was identified (Friedman test, P = .007, n = 6, df = 12). Post hoc testing calculated that the difference in mean ranks was significant when ≥ 3.8 (post hoc experimentwise α = 0.05). Two section levels (the optic tract and rostral pons) had significantly greater mean ranks than 7 other section levels (Fig. 7). Sections from 4 mm caudal to the ansate sulcus, the optic tract, and the rostral margin of the pons had the highest lesion densities, and every infected brain had lesions at these 3 sampling sites. Although lesions were present in all infected fetal brains at the level of the mammillary body, lesion density was significantly lower at this site than in the optic tract and rostral margin of the pons sections. Lesion density was generally high at the level of the ansate sulcus, but not all infected fetal brains had lesions at this site. Lesions were not detected in sections of caudal cerebellum in any of the infected fetuses.
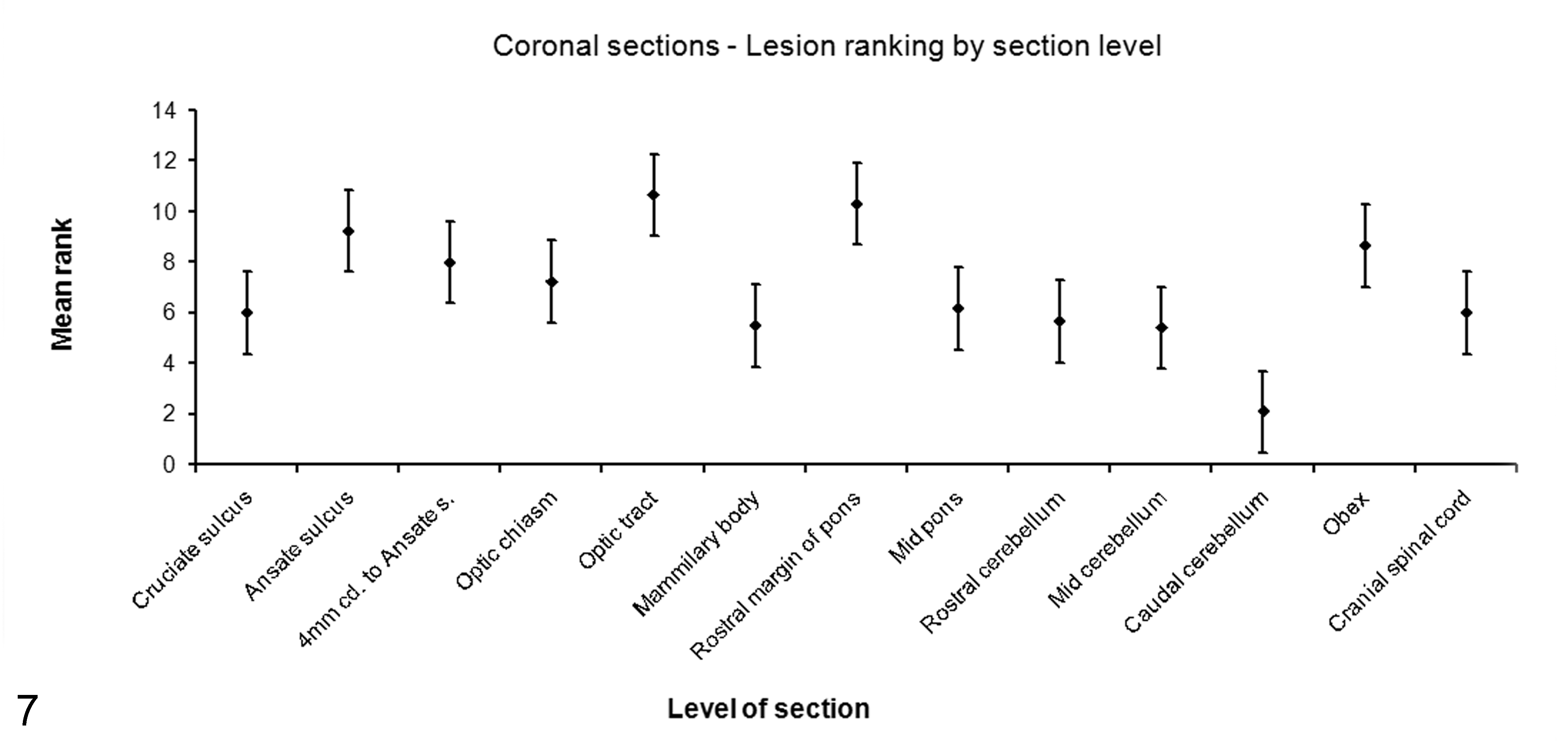
The mean ranks of Toxoplasma gondii lesion density in 13 transverse brain sections from infected fetuses. Error bars are ± half the significant difference in mean rank. Transverse sections with nonoverlapping error bars are significantly different. Friedman test, P = .007, post hoc significant difference in mean ranks ≥ 3.8, α = 0.05.
Real-time T. gondii PCR
Five of the 6 challenged fetal brains tested positive for T. gondii–specific DNA. All 4 brain tissue samples from control fetuses tested negative. The median number of repeat-element targets per milligram was 244 copies (range, 0–1321; Table 1). The results of the T. gondii real-time PCR were reported as part of a larger study. 16
Discussion
The findings of this study indicate that the lesions associated with T. gondii encephalitis occur in greatest number at the level of the optic tract, rostral margin of the pons, or 4 mm caudal to the ansate sulcus and that they are absent from the caudal cerebellum. Routine histopathologic examination of the brain in veterinary diagnostic laboratories typically involves examination of sections of cerebrum, midbrain, medulla, obex, cerebellum, and cranial spinal cord. The results of the present study indicate that the precise location that the cerebral section is taken from may have a determining influence on whether a diagnosis of T. gondii encephalitis can be made. In 3 of the 6 infected fetuses, lesions were apparent in only one-third of those transverse sections located caudal to the rostral margin of the pons. Therefore, the results support the recommendation that sections taken at the level of the optic tract, the rostral margin of the pons, or 4 mm caudal to the ansate sulcus be used for screening ovine fetuses for toxoplasmosis.
The histopathologic lesions identified in the brains from the offspring of the experimentally challenged ewes were undoubtedly due to toxoplasmosis. The lesions, which were absent from the control lambs, were typical of those described in the naturally occurring 17 and experimental 6 disease. Organisms whose morphology was typical of T. gondii tachyzoites and bradyzoites were visible within and near some lesions in infected fetal brains. T. gondii DNA was detected in fetal brains from challenged ewes only, not in any of the control fetuses. Finally, the timing of the maternal serologic response to challenge in this controlled study was similar to that described in sheep in previous T. gondii challenge studies. 22,24,25
This study involved counting discrete lesions typical of T. gondii encephalitis per unit area within a 2-dimensional plane. The categorization of the 3 “types” of lesion enumerated in this study is somewhat arbitrary because the lesions observed in 2 dimensions were components of larger 3-dimensional phenomena; therefore, “type” of lesion observed depends on the plane of section. Although the approach adopted in the present study ignored the 3-dimensional nature of lesions and the variation in individual lesion size and although it did not attempt to distinguish between single and coalescing lesions, it proved adequate for addressing the broad question of lesion distribution being investigated here. More rigorous stereologic methodology was not warranted in this study.
Other reports of ovine congenital toxoplasmosis have described leukoencephalomalacic foci in the cerebral cortices of lambs, suggesting that these lesions could be the result of placental insufficiency and fetal hypoxia. 6,17 Similar lesions are described in lambs of ewes with placentitis due to Chlamydophila abortus. 5,27 However, the pathogenesis of intrauterine white matter injury in sheep is not yet fully understood. Fetal hypoxia due to placental insufficiency does not fully explain the pathogenesis of white matter injury in the ovine fetus, as subcortical white matter injury can result from chronic exposure to endotoxin in the absence of hypoxia. 12
Different strains of T. gondii are known to vary in their virulence and pathogenicity, a phenomenon observed in mice and related to T. gondii genetic type. 18,19 Studies of T. gondii isolates of ovine origin indicate that type II strains are the most common11,20,26 and that some differ in virulence when inoculated into mice. 20 Whether variations in virulence in mice correspond to differences in pathogenicity or to parasite dissemination in sheep infected with these strains is currently not known but cannot be ruled out. The potential for differences in virulence among strains of T. gondii must therefore inform any extrapolation of the results of this study.
T. gondii is disseminated within lymph and/or hematogenously.8,15,30 Therefore, significant differences in lesion density between levels of the neuraxis may be due to factors that influence the probability of the parasite crossing the blood-brain and/or blood–cerebrospinal fluid (BCSF) barrier or to differences in permissive or inhibitory factors within the areas of the brain. Important determinants may include local variations in hemodynamic factors, including vascular density and/or tissue-specific endothelial cell surface molecule expression. Capillary density varies among regions in rat brain and is correlated with glucose metabolism in brain tissue. 2 Similar differences are likely to be present in other species. The relative contributions of non-cell-associated tachyzoites versus tachyzoite-infected leukocytes to T. gondii penetration of the blood-brain barrier has not yet been elucidated, 21 but such information could significantly increase our understanding of the pathogenetic mechanisms involved in establishing CNS infection by T. gondii.
Factors affecting the infection of endothelial cells or blood-brain barrier by T. gondii have been studied using free tachyzoites and in vitro methods. T. gondii tachyzoites have been shown to have differing tropisms for endothelial cells, depending on tissue type. T. gondii tachyzoites can bind to retinal endothelium, 7 and tachyzoites multiply significantly faster in human retinal endothelial cells than in aortic, umbilical vein, or dermal microvasculature endothelium. 29 Whether tachyzoites have a similar tropism for blood-brain barrier or BCSF barrier endothelium is currently unknown. The intercellular adhesion molecule 1 expressed by endothelial and other cells is involved in the penetration of biological barriers by T. gondii tachyzoites in vitro. 3 Expression of intercellular adhesion molecule 1 by cultured human brain endothelial cell is increased by inflammatory cytokines, including interferon-γ and TNF-α. 32 Although both cytokines are important in the host’s immune response in preventing parasite replication, they may act to facilitate T. gondii crossing the blood-brain barrier. 21
Intraleukocytic T. gondii tachyzoites now appear to have a significant role in parasite dissemination in vivo, and T. gondii can infect and exploit monocytes to reach the perivascular space in the brains of mice. 8 Furthermore, in mice injected intravenously with both intraleukocytic and non-cell-associated tachyzoites, the intraleukocytic tachyzoites disseminated more efficiently to the brain. 31 Therefore, the interaction of infected leukocytes and endothelium may be a more significant determinant of T. gondii’s apparent tissue tropisms and ability to traverse the blood-brain barrier in vivo than the interaction of circulating extracellular tachyzoites with endothelial cells. In an in vitro murine blood-brain barrier model, the majority of T. gondii–infected cells crossing the blood-brain barrier were CD11b+/CD11c+/- antigen–presenting cells, and almost no extracellular tachyzoites crossed the blood-brain barrier in this model. 21 Tachyzoites using leukocytes as “Trojan horses” would be dependent on the interaction of leukocytes and endothelium to traverse the blood-brain barrier. Increased expression of intercellular adhesion molecules by leukocytes and endothelium facilitates leukocytes crossing the blood-brain barrier. 14 Whether the level of constitutive expression of any cell adhesion molecule by blood-brain or BCSF barrier endothelium varies in different areas of the noninfected brain is not known. Higher levels of vascular cell adhesion molecule 1 and activated leukocyte cell adhesion molecule expression were associated with more severe CNS lesions and greater parasite burden in a T. gondii–susceptible mouse strain when compared to a resistant one, 28 findings that highlight the role of genetics in determining susceptibility to lesion development in the T. gondii–infected host.
Despite the high sensitivity of the PCR test, 1 brain (fetus No. 4) with a total of 16 histologic lesions had a negative PCR result. This negative result was most likely due to the absence of T. gondii DNA in brain tissue at the site sampled, as only single or no microscopic lesions were detected in sections of rostral cerebrum from the majority (66%, n = 6) of infected brains (data not shown).
This article describes, for the first time, significant variations in T. gondii lesion density among regions of the ovine fetal brain. In this study, the animals were euthanized by 125 days of gestation; therefore, the ultimate clinical outcomes and neuropathology of the infected fetuses may be surmised on the basis of these results but cannot be known with certainty. Caution must be exercised when attempting to extrapolate experimental results to the investigation of field cases of ovine abortion. However, this experiment was designed to mimic naturally acquired intrauterine infection with T. gondii as closely as possible by the choice of the route and timing of challenge of the pregnant ewe, the form of the parasite (oocysts) in the challenge dose, and the use of the Moredun 4 strain, which was originally isolated from fetal brain tissue from a field outbreak of toxoplasmosis in a sheep flock. Though differences due to host or parasite genetics have not been studied here and cannot be ruled out, we suggest that it is reasonable to infer that the results of this study are likely to reflect the distribution of encephalitis lesions in cases of naturally occurring congenital toxoplasmosis in aborted ovine fetuses. Furthermore, the results could be used to inform a sampling plan for brain tissue from the ovine aborted fetus in the diagnostic laboratory. Sampling from the sites with significantly higher lesion density could be expected to improve the diagnostic sensitivity of neurohistopathology and/or PCR for the diagnosis of toxoplasmosis in aborted ovine fetuses.
Footnotes
Acknowledgements
We thank Brian Cloak and the staff at the Regional Veterinary Laboratory, Athlone, and the Central Veterinary Research Laboratory Farm for their assistance. This project was funded by a grant from the Research Stimulus Fund of the Irish Department of Agriculture, Fisheries, and Food.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support in the form of a research grant from the Research Stimulus Fund of the Department of Agriculture, Fisheries, and Food (Government of Ireland) was received to fund a research project on selected causes of infectious ovine abortion. The grant provider has no contractual right to influence and has not in any way attempted to influence this contribution. P. Marques, J. O’Donovan, and A. Proctor received stipendary income as postgraduate students from the grant. J. Gutierrez received salary as a postdoctoral researcher from the grant. J. Nally, B. Markey, H. Bassett, D. Sammin, D. Buxton, S. Maley, S. Worrell, M. McElroy, and C. Brady did not receive any remuneration from the grant for this project.
