Abstract
Embryonic stem cells have had a significant impact on understanding gene function and gene interactions through the use of genetically engineered mice. However, the genetic context (ie, mouse strain) in which these modifications in alleles are made may have a considerable effect on the phenotypic changes identified in these mice. In addition, tissue- and time-specific gene expression systems may generate unanticipated outcomes. This article discusses the history of embryonic stem cells, reviews how mouse strain can affect phenotype (using specific examples), and examines some of the caveats of conditional gene expression systems.
Keywords
Our present use and knowledge of embryonic stem (ES) cells derived from earlier work on cancer cells. Initial understanding and proof of pluripotent/multipotent cells derived from studies using the undifferentiated cells within mouse teratomas (tumors containing endodermal, mesodermal, and ectodermal structures; see Fig. 1
), or embryonic carcinoma (EC) cells.
111
In the late 1950s, Leroy Stevens at the Jackson Labs began to notice that 129 mice had a proclivity for developing teratomas.
110,112
Stevens bred these 129 mice to select for this characteristic so that he could better study and understand this unusual tumor. He found that injection of the undifferentiated cells from the tumors into mice would result in teratomas that contained undifferentiated cells, which could again be used to passage teratomas into other mice.
110,112
By injecting the tumor cells into the peritoneum, he discovered what he termed embryoid bodies: aggregates of cells that resembled embryos.
110,111
Stevens put forth the idea that within the tumors were cells that had pluripotential capacity—EC cells.
83,110
EC cells are aneuploid and have regenerative capacity, generating not only malignant EC cells but also differentiated (nonmalignant) somatic and extraembryonic cells.
3
Advancing the findings by Stevens, Kleinsmith and Pierce used the undifferentiated cells from Stevens’s embryoid bodies to demonstrate that EC cells could be grown in culture and that a colony derived from a single EC cell could cause a teratocarcinoma when injected into a mouse.
59
Their work proved that a single cell can proliferate and develop into all 3 germ cell layers—a stem cell.
59,82
Mouse, teratoma: photomicrographs of the 3 germ cell layers contained with a teratoma (a) endoderm, (b) ectoderm, and (c) mesoderm.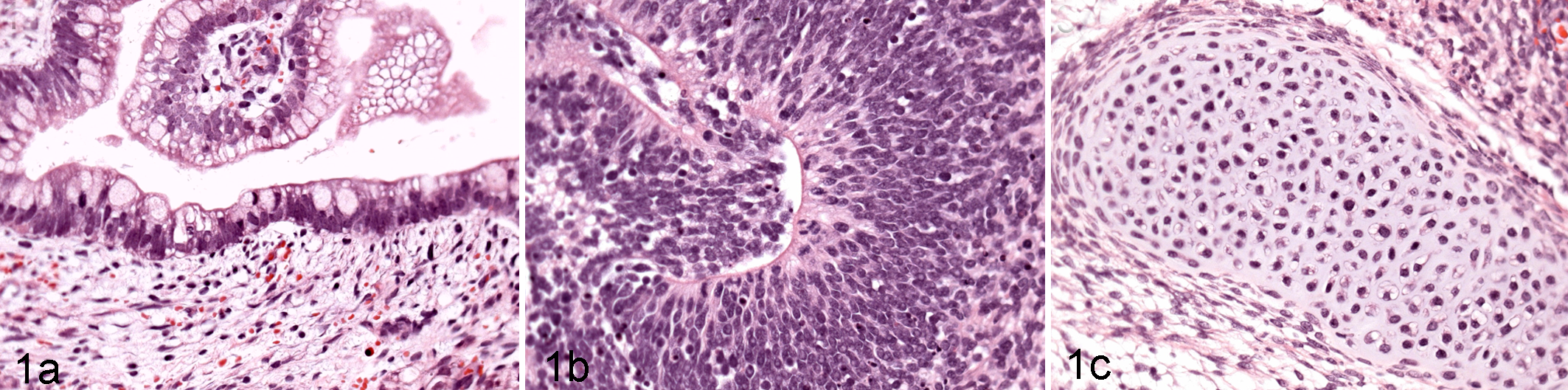
The concept of the stem cell had a long-standing history in the scientific community. Concurrent with the studies of EC cells, efforts were also focused on identifying stem cells in normal adult animals. The concept of adult stem cells was initially given in vivo validity by Till and McCulloch in 1961. 9,123 Their studies demonstrated that mice injected with as few as 10 000 bone marrow cells 1 day after supralethal whole-body irradiation developed regenerative hematopoietic nodules containing both myeloid and erythroid cell lineages in the spleen. 9,123 Their data, along with later studies, 9 demonstrated that cells exist within adult bone marrow that are capable of regenerating the myeloid and erythroid cell lineages.
Stevens’s focus on the similarities between the embryoid bodies derived from EC cells and the histologic appearance of early embryos led him to investigate the pluripotential nature of embryonic cells. Stevens began grafting 1-, 3-, 6-, and 9-day old embryos into the testes of mice to see if the cells would grow and differentiate and if undifferentiated cells could then be harvested from these grafts. 109 The results of grafting 1-day-old embryos were limited, with rare cell growth. In those grafts that did grow, the masses generally comprised undifferentiated clusters of cells. In contrast, 3-, 6-, and 9-day embryo grafts developed into teratomas. Interestingly, though, day 3 tumors had more undifferentiated cells (which could develop into teratomas when grafted into other mice) than day 6 embryos. 109 By day 9, the embryo grafts contained no undifferentiated cells, and Stevens concluded that by day 9, all the cells of the mouse embryo were committed to a single lineage. 109 Using the undifferentiated cells from the embryonic grafts, Stevens created transplantable teratoma cell lines. This work clearly revealed that embryos contain pluripotent stem cells and that the earlier the embryo, the more pluripotent stem cells present.
The ongoing association between the embryo and stem cells increased the attention on the inner cell mass of the mouse blastocyst as a source of pluripotent stem cells. However, it was not until 1981 that this goal was achieved. Evans and Kaufman at the University of Cambridge grew, for the first time in culture, cells derived from 129/SvE strain mouse blastocysts. 37 While others had attempted, without success, to grow cells from the inner cell mass in vitro, Evans and Kaufman succeeded by developing tissue culture conditions that promoted proliferation rather than differentiation. Unlike EC cells, these cells had a normal karyotype and, most important, could develop into teratomas in vitro and after inoculation in vivo. These studies conclusively demonstrated the pluripotent nature of a single cell derived from a blastocyst: an ES cell.
Consequently, ES cells from different species were derived. In 1995, the first article on nonhuman primate–derived ES cells was published, 119 followed by publications on human-derived ES cells. 98,118 Further advances in ES cell culture conditions have significantly increased the strain and species for which ES cells are available. Many strains of mouse and several strains of rat ES cells have been maintained in serum-free conditions containing leukemia inhibitory factor and bone morphogenetic protein 4 in combination with inhibitors of glycogen synthase kinase 3 and FGF-MAPK (termed the 2-inhibitor culture). Inhibition of FGF-MAPK signaling notably reduces differentiation of ES cells, and inhibition of glycogen synthase kinase 3 helps maintain the pluripotent state (reviewed in Blair et al 11 ). Since C57BL/6 is currently the most widely used mouse strain in biological research, the use of C57BL/6 ES cells have many advantages, particularly in that their use precludes the need to backcross into the C57BL/6 background to create strain congeneity. At the present time, even with rapid congenic techniques, the time to create a congenic strain is generally 1 to 2 years. 129
Introduction of Foreign Genes Into ES Cells
Transgenic technology began in the early 1970s with injection of SV40 DNA into blastocysts 52 and matured with a pivotal study by Costantini and Lacy in which a cloned rabbit β-globin gene was introduced into mouse germ cells. 27 However, the use of homologous recombination to generate gene targeted mutant mice was developed much later. It was not until the mid-1980s that methods were developed to optimize the targeting of cloned DNA into specific regions of the genome through homologous recombination. 38,39,106,107,116 In 1984, a breakthrough publication changed the entire field of ES cell technology: Embryo-derived pluripotent cells were injected into mouse blastocysts to produce germ cell chimeras. 13 Together, these works created the foundation for the development of the “knockout” mouse. In 1989, several authors reported germline transmission of mice containing genes modified in ES cells using homologous recombination, 60,117,133 thus beginning a new era in genetic research through gene targeting.
The Importance of Understanding ES Cells and Mouse Strains
With the development of transgenic and knockout mice, the importance of understanding the normal phenotypic variation of the strains utilized in these studies has become imperative for the appropriate interpretation of gene effects. Many knockout studies utilize ES cells derived from 129 mouse strains. These became a favorite for gene targeting for several reasons: First, 129 ES cells were the first to be described and used for genetic manipulation of the mouse genome; second, they are easily derived and maintained; and, third, they have a higher frequency of creating germline-competent cells. 104 Having an understanding of the 129 strains is essential for understanding failures in recombination events (ie, using a DNA library from one 129 strain in ES cells from another 129 strain) and variations in phenotypic outcomes in genetically engineered mice (GEM).
It’s complicated
Although the 129 strains are all grouped under the same umbrella, there is a remarkably high degree of genetic variability within this line. 104,121 To this point, the Jackson Laboratory performed an extensive historical and molecular evaluation of 129 strains and ES cells. Their recommendations were (1) to backcross to the same 129 substrain from which the ES cells originated, (2) to use a genomic library derived from the same strain as the 129 ES cell line, and (3) to understand and take into consideration the genetic traits of the 129 substrain prior to initiating studies (for an up-to-date listing of the current classifications of 129 strains, see http://www.informatics.jax.org/mgihome/nomen/strain_129.shtml). 104
The 129 pedigree is divided into 4 groups: parental origin, steel origin, teratoma susceptible, and X (a genetically contaminated line with only limited similarity to the other 129 substrains). The parental strain is the strain from which the other strains were derived. However, except for strains derived from 129/Re (now 129P1/ReJ; eg, 129/Ola substrains), all other 129 substrains derive from the 129 colony in Stevens’s lab (129/SvJ, now 129X1/SvJ). The sublines that Stevens generated to study teratomas developed into the steel and teratoma-susceptible groups. The steel substrains were originally outcrosses of 129 to the C3H/Hu line, which had the steel-J allele. These were backcrossed to be congenic with 129X1/SvJ mice. These lines are now 129S2/SvPas (breeding colony from Jean-Louis Guenet) and 129S9/SvEvH (breeding colony of Martin Evans). 129S6/SvEvTac is the Taconic outcross of 129X1/SvJ to 129S5/SvEvBrd (129/SvEv-Gpi1c , a marker of chimerism introduced by Evans). Ter substrains were derived from an outcross of 129X1/SvJ to hybrid (WC × C57BL/6) to give a dominant spotting mutation (KitW , formerly W). 129X1/SvJ and 129S4/SvJae are very different (Mouse Genome Informatics). In addition, there is evidence of contamination of 129X1/SvJ and 129P2/OlaHsd with non-129 strains. 104
The 129 substrains are so different from one another that skin grafting from 129P3/J, 129X1/SvJ, and 129S9/SvEvH between themselves or other 129 substrains results in graft rejection. 104 There are numerous other examples of differences between the 129 substrains 16,21,23,78,90 ; however, the essential point of these details is to understand that the 129 substrains are genetically dissimilar from one another.
The effects of flanking DNA and modifier genes
Individual alleles generally do not work alone to create a phenotype but are modified by other genes that are different between mouse strains. 29 Hence, the phenotypic outcome of gene modification in the genetically engineered animal is profoundly affected by the background genotype of the strains involved as well as the environment in which the animal is housed. Of particular importance is the DNA flanking the allele of interest, which cosegregates with the modified allele; thus, the flanking DNA and the allele are linked. The effects of these linked genes may lead to false conclusions on the effect of a given gene on a phenotype, even in congenic strains (backcrossed into a single strain for 10 generations; by theoretical calculations, the gene content is 99.8% backcross strain). Flanking DNA has the potential to modify the phenotype if modifier genes cosegregate with the allele of interest. 96 In addition, small segments of the donor strain will be incorporated randomly into the genome. 103
The importance of flanking DNA in phenotype was noted in a study comparing mice with Rab3a mutations. One mouse (C57LB/6J) was identified in an ENU screen to have a point mutation in the Rab3a gene (Rab3a Ebd), while the other was a knockout mouse derived from 129/Sv ES cells. Both types of mice were backcrossed into C57BL/6J for 3 generations. Interestingly, the phenotype in the ENU-derived mice was more severe than the GEMs with a mutation in the same gene. The difference was attributed to the presence of residual 129/Sv DNA in the GEM. 131 Flanking DNA from the original ES cell strain, estimated to be approximately 50 cM containing 178 genes, likely had a significant effect on the phenotype. 131
While congenic strains theoretically may have 99.8% of their DNA derived from the backcrossing strain (this will be somewhat variable), these mice may still manifest the effects of the flanking DNA. 35 Schalkwyk and colleagues 96 termed this flanking DNA the congenic footprint and estimated that the chance of having > 3.3 cM on each side of the target allele was 95% in congenic strains. 96 This means that the flanking DNA may include 110 genes or more (in their study, the region included 573 genes). While there are strategies to expunge this flanking DNA, most GEM contain ES cell–derived flanking DNA. Additionally, the finding that fragments of donor DNA are present throughout the “congenic” genome indicates that completely eliminating the ES cell DNA from a strain may be impossible. 35
Strain Differences and Phenotype
Much has been reported on the routine background histopathology findings in various strains of mice. 8,15,48 It is important to understand how these background lesions could affect an anticipated phenotype. 32,36,63,77,94 By the 1990s, it was becoming clear that GEMs developed by different groups at different facilities often had notably dissimilar phenotypes. Groups began looking into the source of these differences, and it turned out that phenotypes vary by ES cell strain, backcrossing strain (modifier genes), degree of backcrossing (eg, evaluation of hybrids), region deleted, and factors that have epigenetic effects (eg, maternal behavior, uterine environment, hormones). While myriad examples exist of how background strain genetics can influence the mouse phenotype after selected gene modification, this review simply demonstrates a few select examples.
Corpus Callosum Conundrum
Several strains of mice have been found to have disruptions in, or agenesis of, the corpus callosum (CC), which is a band of axons that connect the left and right hemispheres, allowing cross talk between the 2 sides of the brain. 67–69,125 The appropriate targeting of these fibers requires normal development of the midline structures, normal development of the neurons in layers 2/3 and 5 of the cerebral cortex, cingulated pioneering axons, hippocampal structures, and glia, which secrete guidance factors. 22,87,89 Thus, abnormalities in the development of the CC are an effect of defects anywhere along the pathway.
In the early 1980s, Wahlsten 125 published an article comparing the differences between commonly used mouse strains in the degree of presence or absence of the CC. Strains that had no notable abnormalities in the CC were the A/J, C57BL/6J, and DBA/2J strains. However, he found that the 129/J strain had a 71% frequency of CC defects and that over 30% of BALB/c mice from 2 vendors had CC defects, ranging from agenesis to a slight reduction in CC fibers. Subsequent studies have confirmed these findings 69 and demonstrated that at least 2 different loci were associated with CC abnormalities in 129 and BALB/c strains. In many cases, the CC abnormalities were associated with the malformation in the anterior and ventral hippocampal commissures. 67,71,126
Interestingly, all 129 substrains have a deletion polymorphism of Disc1, which is the disrupted in schizophrenia 1 gene and implicated as a gene important in schizophrenia. 74,92 DISC1 is thought to interact with multiple proteins in the brain that are important in neurite extension, signal transduction, the cytoskeleton, and neurotransmission. 20,84 In mice not prone to CC defects (CBA/CaCrl and C57BL/6JCrl), expression of a mutated form of Disc1 resulted in abnormalities of the CC development, 99 suggesting that aberrations of this gene may be important to the underlying CC defects in 129 mice.
The 129 mice also have deficits in behavioral phenotyping studies; whether this is related to the CC is uncertain. Mice of the 129 strains are generally more anxious and have poorer spatial learning, balance, and coordination when compared to strains with normal development of the CC. 4,20,23,124 In fact, there are also notable differences in behavioral profiles between the highly divergent 129 substrains (for a detailed review, please see Cook et al 23 ). Given the plethora of well-reported defects in the central nervous system of 129 mice, they should not be considered an appropriate choice for developing GEMs anticipated to have neurologic and/or behavioral deficits.
Regardless, incomplete development or agenesis of the CC, often with the identification of hippocampal abnormalities and behavioral defects, are commonly reported phenotypes in GEMs developed on a 129 background. Unfortunately, many of these studies have been performed in mice that were incompletely backcrossed into C57BL/6 (or other strain). Because the CC abnormalities often occur at a higher incidence in homozygous mutant mice than heterozygous and wild-type control littermates, the finding is frequently reported as a phenotype. 17,34,56,108,131 However, there is evidence that when the mice are completely backcrossed into C57BL/6 or other strain without genetic CC abnormalities, the CC phenotype decreases or disappears. While the mutations may in fact compound the underlying defect, detailed studies to explain this observation have not been performed, and the lesion is still attributable to the 129 background strain.
GEMs and CC Defects Initially Attributed to Gene Mutations but Actually Artifacts of the 129 Strain
Emx1 mutant mice
Emx1, the mouse homologue of the Drosophila empty spiracles gene, is thought to play an important role in the patterning of the central and peripheral nervous systems. 47,97 Emx1 mutant mice generated by targeted disruption in JM-1 ES cells (129/SvJ) were initially reported to have defects in the development of the CC, which was consistent with its role in neuronal patterning in Drosophila. 85 However, when Emx1 mutant mice were backcrossed into a C57BL/6 background, they had essentially normal CCs (as well as normal indusium griseum and taenia tecta), with only a mild reduction (8%) in the CC cross-sectional area as compared to the wild-type littermates. 47 The researchers concluded that inactivation of the Emx1 gene was not responsible for the brain phenotype originally reported.
β-Amyloid precursor protein mutant mice
Alzheimer disease is a neurodegenerative disease characterized by cognitive defects and by the histologic presence of neurofibrillary tangles in the cytoplasm of neurons and central nervous system plaques composed largely of β amyloid. 45 Abnormalities in the expression and processing of β-amyloid precursor protein, with deposition of β amyloid into central nervous system plaques, have been suggested to play an important role in the cognitive defects associated with Alzheimer disease. 43–45,55 Initially, disruptions in the App (β-amyloid precursor protein) gene were reported to have reductions or agenesis of the CC in homozygous mutant mice (as well as behavioral deficits), which were rarely present in wild-type or heterozygous mice. 79 Using 129S9/SvEvH ES cells, the gene was disrupted through the insertion of a neomycin cassette with a transcription termination sequence into exon 2. The initial phenotyping was carried out on second-generation matings (ie, 129S9/SvEvH;C57BL/6 hybrids). However, when the mice were backcrossed to a C57BL/6 strain to congeneity, the hippocampal commissure and CC defects disappeared. In contrast, the frequency and severity of these brain abnormalities increased notably when the App mutants were on the 129/SvEv or 129/Ola background. 71 The negative data were consistent with the findings in App deletion mutants from other groups. 66,71,132
Because more researchers using GEM are aware of the 129 CC and hippocampal defects, many have begun to carefully backcross mice derived from 129 ES cells for > 10 generations to C57BL/6 (eg, Satoh et al 95 and Shu et al 100 ). However, recent reports on Il18r1 and Apbb1 (formerly FE65) mutant mice suggest that backcrossing alone may not be enough to guarantee that flanking 129 DNA will not affect the phenotype. 24,35 However, despite the established knowledge of the central nervous system and behavioral defects in 129 mice, publications are common of abnormalities in the CC and hippocampus in 129-derived mice incompletely backcrossed into unaffected strains. 56,108,131 The general reason is that, as previously mentioned, homozygous mutant mice on the 129 background often have (for unknown reasons) more CC and hippocampal defects than their wild-type and heterozygous littermates.
Other Background Strains Altering GEM Phenotypes
Col4a3: Mouse model of Alport glomerulonephritis
Alport syndrome is the result of mutations in the genes encoding collagen IV chain α3, α4, or α5, resulting in basement membrane defects. 62,76 While the disease is associated with deafness and anterior lenticonus, the most devastating lesions result from a progressive glomerulopathy, which leads to end-stage renal disease. 7,76 The glomerular lesions are characterized by splitting of the glomerular basement membrane, with effacement of the podocyte foot processes. 25,26 All 3 chains must be present and normal to form the appropriate α3α4α5(IV) collagen triple helix to lay down as part of the adult glomerular basement membrane. 55 The most common form of Alport syndrome is due to an X-linked mutation in the COL4A5 gene; 7,53 however, autosomal recessive mutations in COL4A3 and COL4A4 also result in this syndrome. 76
In 1996, two groups published findings in GEMs that had Col4a3 mutations. The mice from both groups were on a mixed 129X1/SvJ background. These mice developed end-stage renal disease around 10 to 14 weeks of age 26,75 and were generally deceased by 16 weeks of age. However, if mice with Col4a3 defects were backcrossed into the C57BL/6 background, histologic lesions and end-stage renal disease developed much later than when the mutation was on a 129X1/SvJ background (end-stage renal disease at 194 vs 66 days, respectively). 2,55 Further studies also revealed that in C57BL/6 mice, COL4A5 and COL4A6 deposition into the basement membranes was increased, but mice on the 129 background strain showed very little glomerular basement membrane deposition of COL4A5 and COL4A6. 55 Findings suggested that COL4A5 and COL4A6 deposition in the glomerular basement membrane might help delay the onset of the glomerulopathy and that this ability to switch to the α5α6(IV) isoform might be an important component to the strain differences in these mice. 55,120 The modifier genes for these differences were mapped to chromosomes 9 and 16. 2
Egfr1
The effect of genetic background strain on the phenotypes of epidermal growth factor 1 receptor (Egf1r) gene mutations in mice has been well described. In 1995, Threadgill et al published an article that demonstrated what many had noticed—namely, that the phenotype of a targeted mutation was not identical between groups focusing on the same gene target. 120 These studies focused on the targeted disruption of the mouse epidermal growth factor receptor. The original germline chimeras were developed by inserting a neo cassette into exon 2, which resulted in 2 splice variants—1 that formed a nonfunctional protein and 1 that had an in-frame mutation to remove domain 1 of the extracellular region. These coisogenic mice were then bred into random-bred CF-1 and CD-1 lines from a closed colony. The 3 different backgrounds with the same mutation had notable differences in their phenotype. Embryos derived from the CF-1 background died around implantation, with defects in the inner cell masses and no clear endoderm. The coisogenic 129/Sv embryos died in midgestation due to placental abnormalities characterized by a marked reduction in the spongiotrophoblast layer. Mice on the CD-1 background mutant mice survived as long as postnatal day 18, dying with abnormalities in the skin, kidney, brain, liver, and gastrointestinal tract.
Further evaluations of the Egfr mutations in mice were published by Sibilia and Wagner, who modified a different region of the Egfr gene. 102 As with Threadgill et al, the results in a 129 background suggested placental insufficiency, but most organs were histologically normal at embryonic day 13.5 (although reduced in size). When these mice were bred into a C57Bl/6 background, a small number of mutant fetuses developed to term. However, in a 129 (unknown classification) × C57Bl/6 × MF1 cross, some mice, while growth retarded, survived until postnatal day 20 but lacked whiskers and eyelids and had thin, fibrotic corneas. In addition, the epidermis was thin with poorly defined layers and scant keratinization. Hair follicles were also abnormal, and the hair coat did not develop normally. The researchers did not identify the kidney, brain, liver, and intestinal abnormalities described by Threadgill et al. Clearly in this example, the genetic background and the location of the mutation in the gene may have an important impact on the phenotype and, subsequently, the understanding of the gene function during development.
TGFβ1
Transforming growth factor β1 (Tgfb1) mutant mice represent another well-described example of the effect of strain differences on study outcome (for an excellent review, see Doetschman 32 ). Tgfb1 mutant mice were originally generated on a 129X1/SvJ background. 61 Approximately half the homozygous mutants survived to birth and died within a few weeks of autoimmune disease. 61,101 The cause of embryonic lethality in those mice that did not survive to parturition was studied, and it was found that there were 2 points at which TGFβ1 played an important role: first before implantation and then during yolk sac vasculogenesis and/or hematopoiesis. 54 Full penetrance of embryonic lethality was significantly affected by the background strain utilized; mutant mice bred into the C57BL/6 background all died preimplantation. 54 When the mice were bred to a mixed 129 × C57BL/6J/Ola × NIH/Ola background, embryonic lethality was not completely penetrant, 80,81,113 and the embryos died as a result of yolk sac vasculogenesis and/or hematopoietic abnormalities. 31 In 1997, Bonyadi et al 12 identified an important modifier gene on mouse chromosome 5; subsequently, a modifier on chromosome 1114 has been identified. While many believe that using C57BL/6 cells will be the answer to many background strain problems, it is clear that if Tgfb1 mutations had originally been generated in C57BL/6 ES cells, the autoimmune phenotype may not have been identified before the generation of conditional gene knockout systems was developed.
There are numerous other examples of genetic background affecting phenotype: Sod1, 49 Krt 8, 5,6 and Cftr, 57,72,91 to name only a few. All these differences are attributable to variations in modifier genes.
Zinc Finger Nuclease Technology
ES cell technology may in part be replaced by zinc finger nuclease technology, in which ES cells are bypassed and targeted modifications occur more rapidly. The zinc finger nuclease technology utilizes specific DNA-binding zinc finger proteins fused with a nuclease domain. Thus, the pair of zinc finger proteins targets a specific location on the DNA, and the nuclease domains come together to create a double-stranded break. Repair may be either through high-fidelity homologous recombination utilizing the inserted modified DNA or by nonhomologous end joining, which is a highly error-prone mechanism of double-strand break repair in DNA. Nonhomologous end-joining repair of the double-stranded DNA break often results in gene disruption through deletions or insertions. 18,64 This method allows the use of 1-cell fertilized eggs rather than ES cells. Zinc finger nuclease mRNA is injected into eggs, which are then transferred to pseudopregnant females. Thus, all embryos developing from those eggs will have germline transmission. However, off-target nuclease activity may cause unanticipated findings. This technology opens the field of genetically engineered animal models to almost any species or mouse strain without the use of ES cells. Because the mutation is made in the specific species or strain of choice, there is no need for backcrossing to create a strain congenic for the mutation. While the Zinc finger nuclease technology may reduce many of the issues associated with the use of ES cells, ES cell–derived mice will continue to be heavily utilized, and the issues of mouse strain background will persist regardless of the technology utilized.
Tissue-Specific and Temporal Gene Regulation
Manipulation of genes in a temporal and tissue-specific manner has enhanced the understanding of gene function and allowed for the disruption of genes that would otherwise have caused embryonic lethality. 14 Through the use of various expression systems, genes may be selectively expressed or deleted from particular tissues and/or at precise times and can be turned on and off in a tissue-specific or global fashion. These systems are not perfect, and it is essential to understand the potential for unanticipated effects, including the generation of hypomorphic alleles (alleles with reduced levels of gene activity) or neomorphic alleles (gain in function dissimilar to their normal function). Examples of other potential unanticipated effects of gene modification are discussed below, but for a more complete discussion, see Matthaei 73 or Adams and van der Weyden. 1
One of the most common methods for tissue/time-specific gene deletion is through the use site-specific recombinase (SSR) technology. 14 Transcriptional control using SSRs in a tissue-specific manner has improved the understanding of localized gene function. However, “tissue-specific” promoters may not be entirely tissue specific or may be “leaky,” resulting in unanticipated phenotypes. 30,40
The most frequently utilized SSRs are the Cre-LoxP and Flp systems. SSR technology requires the use of 2 genetically modified mice, which are subsequently bred together; 1 mouse is generated using homologous recombination (inserted DNA has bacteriophage LoxP or FRT sites flanking the desired region of gene disruption), and 1 is generated using DNA insertion by random integration (Cre or Flp recombinase, whose product excises DNA between LoxP or FRT sites, respectively). Other systems, such as the phiC31-attP-AttB SSR 10,41,128 and specific Cre and Flp recombinase variants, are not addressed.
Cre recombinase also has issues as a result of cryptic lox sites in the mouse genome and has been associated with chromosomal aberrations and sister chromatid exchanges. 50,51,70,122 Interestingly, Cre recombinase or Cre recombinase RNA expression in the cytoplasm of oocytes may resulting in excision at the LoxP sites very early in development in all offspring, including those that were genetically Cre recombinase negative (epigenetic effect). 73,86 Cre expression in the oocyte may also result in global deletion of the floxed gene rather than deletion in a tissue-specific manner. It is therefore recommended that Cre males only be used during crossbreeding with LoxP mice. However, oocyte-specific expression of Cre is being used to ubiquitously delete a gene region. One such example is the ZP3-cre mouse line, in which Cre is expressed using the zona pellucida 3 gene promoter. 65 As an aside, epigenetic effects are not limited to the oocyte: Rassoulzadegan et al reported that RNA can be transmitted via sperm into nontransgenic offspring, resulting in the expression of the transgenic phenotype. 88
Temporal control of SSR or gene expression may be achieved through the use of promoters that are activated by specific compounds or as the result of endogenous cell signaling pathways. These compounds include tetracycline (doxycycline), IPTG, tamoxifen, and RU486, as well as endogenous mediators such as cytokines. 14,73 One of the most commonly used systems is the tetracycline/doxycycline response system, 28,42,58 in which the gene (SSR or other gene of interest) is induced by administration of tetracycline/doxycycline (tet-on) or blocked by administration of tetracycline/doxycycline (tet-off). Present systems for tet-on and tet-off are improved over those of the past but may still have issues of prolonged activation of the promoter after (1) tetracycline removal in tet-on systems and (2) aberrant expression of the gene while on tetracycline in the tet-off system. 19,58 Additionally, tetracycline is an antibiotic that may alter the intestinal flora, potentially altering the phenotypic outcome, particularly as bacterial flora have recently been demonstrated to have a role in phenotype. 33,93,127,130 Tetracycline also incorporates into bone, and its effect on the bone has not been extensively evaluated, although it has not been thought to have untoward effects. 46 A second system commonly used is that of the LacO/LacIR system, which is based on the Escherichia coli lac operon. 28 By adding IPTG (a lactose analogue), the promoter is derepressed and the gene of interest transcribed. In this system, the gene expression is inhibited to less than 10% of normal, which increases to 80% normal expression 48 hours after the addition of IPTG. This system has been suggested to be a better method of controlling gene expression or deletion than the tetracycline-based systems. 73
While not the focus of this article, it is important to understand that aberrant phenotypes may also develop in transgenic mice (eg, mice expressing Cre or Flp recombinase) as a result of not only mouse genetic background but also the random integration of the transgene. The most common effects are due to (1) disruption of unrelated genes due to arbitrary insertion, (2) copy number or “gene dosage” effects, (3) unanticipated expression due to adjacent promoter activity at site of insertion, and (4) downstream transcriptional events as a result of strong promoters. 73
The Future of the Mutant Mouse
Generating and acquiring mutant mice is an expensive and time-consuming scientific endeavor that may not result in a phenotype. As a result, many researchers may be discouraged from pursuing forward-genetics studies. Because of this, large-scale gene trap and gene-targeting programs have been developed to generate mutant mice with a maximum in gene coverage and a minimum in effort overlap using a single strain of mouse (C57BL/6). The International Gene Trap Consortium (http://www.genetrap.org/) and the International Knockout Mouse Consortium (http://www.knockoutmouse.org/) are committed to developing C57BL/6 mouse ES cell lines with every possible expressed gene knocked out and to making the database and cells available to researchers for a nominal fee. International Gene Trap Consortium member institutions utilize high-throughput approaches to establish insertional mutations throughout the genome in mouse ES cells to generate standard loss-of-function alleles. To date, over 450 000 ES cell lines have been generated; the database may be reviewed and cells ordered for a basic handling fee. The International Knockout Mouse Consortium includes the Knockout Mouse Project, an initiative supported by the National Institutes of Health, the European Conditional Mouse Mutagenesis Program, the North American Conditional Mouse Mutagenesis Project, and the Sanger Institute’s Mouse Genetics Program, as well as others, to minimize redundancy in efforts while maximizing the amount of the genome modified. To date, the International Knockout Mouse Consortium project has knocked out 17 000 genes in ES cells. 105,115
Conclusions
Stem cell and DNA manipulation technologies have significantly affected the scientific understanding of gene effects and gene interactions. Mouse models of disease and gene function will continue be an essential component of biological research. Thus, it is essential for the researcher and pathologist alike to understand mouse genetics and the caveats of the tools utilized to alter gene expression for accurately and appropriately phenotyping GEM.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
