Abstract
Cytotoxic lesions of the corpus callosum are lesions secondary to different medical conditions. Radiologically, lesions are identified on magnetic resonance imaging as a hyperintense signal on diffusion-weighted imaging and decreased apparent diffusion coefficient values of the splenium of corpus callosum. Signal changes are reversible in almost totality of the cases. Previous cases of cytotoxic lesions of the corpus callosums have been associated with several metabolic disturbances, but ketotic hyperglycemia has never been reported. We here discussed the case of 28-year-old patient with complex visual hallucinations presenting with cytotoxic lesions of the corpus callosums and type I diabetes. Treatment of hyperglycemia was followed by full clinical recovery and complete regression of the radiological abnormalities at 3-month follow-up. Elevated levels of circulating pro-inflammatory mediators associated with ketotic hyperglycemia in type I diabetes support an implication of cytokines in the pathophysiology of the cytotoxic lesions of the corpus callosums.
Keywords
Introduction
Cytotoxic lesions of the corpus callosum (CLOCCs) are a clinical and radiological entity characterized by reversible lesions of the splenium of corpus callosum (CC) presenting as hyperintense signal on diffusion-weighted imaging (DWI) sequences of magnetic resonance imaging (MRI) due to cytotoxic edema. 1 These lesions have been previously called “MERS,” “reversible splenial lesion syndrome,” “reversible splenial lesion,” and “transient splenial lesions.” 1 According to the type of anatomical involvement of CC, three patterns of CLOCCs have been described. 1 Radiological findings generally disappear within few weeks, without sequelae or atrophy.
The pathophysiology is still debated. Cytotoxic edema is considered to be the result of an activation of a cytokines cascade, blood–brain barrier leakage, and massive release of glutamate. This neurotransmitter binds N-methyl-
CLOCCs have been reported as secondary lesions in several clinical conditions ranging from metabolic disturbances, infections, drug therapy, and malignancy to trauma. 1 Patients of all ages can be affected, and pediatric cases 2 have been reported as well.
A broad spectrum of symptoms has been associated with CLOCCs 3 wandering from headache, blurred vision, and vertigo to more severe conditions such as seizures and altered mental status. We reported here a case of CLOCCs in a female patient presenting with complex visual hallucinations (CVHs) associated with hyperglycemia and ketoacidosis revealing type I diabetes.
Case report
A 28-year-old woman was admitted to neurology ward because of faintness and episodes of CVH. She presented several short-lasting (20–30 s) lifelike episodes from her personal past life with no emotional component. They were binocular and persisting after eye closing. She referred no episodes of speech disturbances or loss of consciousness. Outside of CVH, the patient had no visual impairment. She did not complain of headache. Personal past medical history was notable for thyroiditis but she had no treatment at home. She drank 5–10 alcohol units a week, and urine drug screen was negative at admission. General clinical examination was unremarkable. Blood tests at the admission revealed normal blood count, slight inflammatory syndrome (c-reactive protein (CRP) = 6.5 mg/L, normal values = (n.v.) < 5 mg/L) and electrolyte abnormalities (sodium = 133 mmol/L (n.v. = 136–145 mmol/L); chloride = 95 mmol/L (n.v. = 98–107); bicarbonate = 18 mmol/L (n.v. = 23–29 mmol/L)). Hepatic and renal functions were normal. Serum protein immunoelectrophoresis was normal. Antinuclear antibody (ANA) and antineutrophilic cytoplasmic antibody (ANCA) antibodies were negative. Serology for HIV, syphilis, Lyme’s disease and hepatitis B and C was negative. Immunoglobulin G (IgG) for herpes virus-1 and varicella zoster was positive without IgM. Electroencephalogram (EEG) performed twice, one of which during several CVH events, was normal without background activity abnormalities nor paroxysmal discharges.
Considering sex, age, and previous history of thyroiditis, an autoimmune encephalitis was first suspected, and a lumbar puncture was performed, revealing in cerebrospinal fluid (CSF) analysis 9 lymphocytes, with normal level of proteins, lactic acid, and glucose. IgG index was normal. Oligoclonal bands were absent in CSF. Serum and CSF panel for antibody-associated encephalitis was negative (anti-NMDAr, anti-AMPAr, anti-GABAr, anti-DPPX, anti-CASPR2, and anti-LGI, EUROIMMUN AG, Germany). Cytokines levels were not available in serum and CSF analysis. Microbiological direct exam and viral/bacterial multiplex polymerase chain reaction (PCR) were also negative. In contrast, fasting blood glucose was 352 mg/dL (adjusted natremia according to Katz’s formula was 137 mmol/L, within the normal range), glycate hemoglobin was 10.9%, ketones were present in urines, anti-GAD antibodies were absent, but C-peptide was reduced (0.194 nmol/L (n.v. = 0.370–1.470 nmol/L)), allowing a diagnosis of de novo type I diabetes. Diabetic ketoacidosis needed a treatment of intravenous (IV) insulin and intravenous fluids, and the patient was admitted to intensive care unit. Brain MRI showed one rounded posterior splenial lesion with restricted diffusion on DWI imaging, low apparent diffusion coefficient (ADC), and hyperintense signal on fluid-attenuated inversion recovery (FLAIR) (Figure 1(a), (b), (c)), highly suggestive of CLOCCs. Two days after hyperglycemia correction and insulin continuous therapy, the patient reported resolution of CVH. Immunotherapy was not retained due to favorable clinical outcome. She was dismissed with subcutaneous insulin therapy and
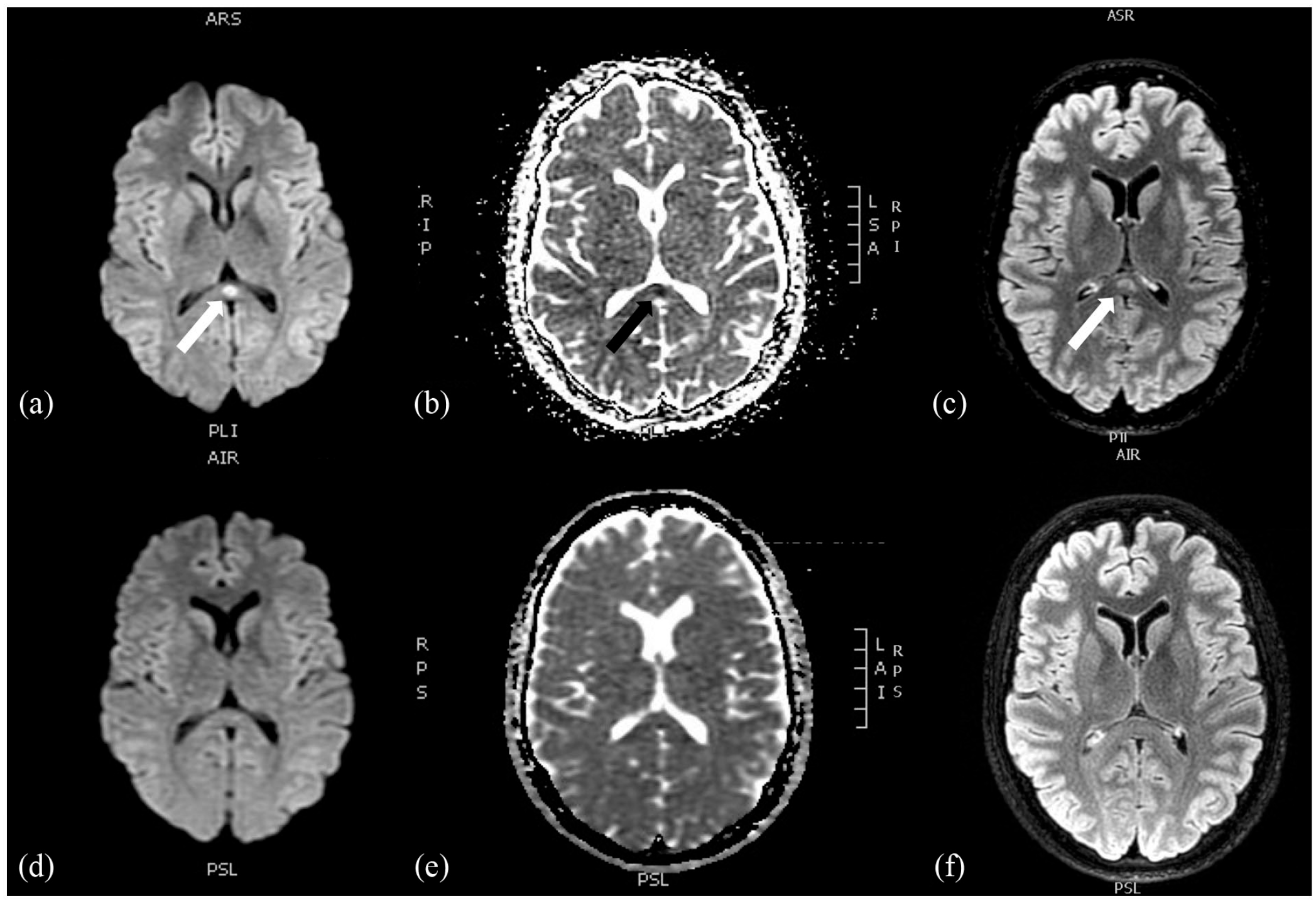
Brain MRI revealing the presence of an oval lesion within the splenium of the corpus callosum. (a) Restricted diffusion displayed by an area of high signal intensity on diffusion-weighted imaging (DWI) and FLAIR (c) and low signal intensity on apparent diffusion coefficient (ADC) map (b). There is no hemorrhage on gradient echo T2-weighted imaging and no gadolinium enhancement (not showed). Three-month control MRI showed complete resolution of the lesion on DWI/ADC (d and e) and FLAIR (f) sequences.
Discussion
This case report adds type I diabetes with ketoacidosis to the long but not yet exhaustive list of CLOCCs etiologies.
Pathophysiology of these lesions is still unknown, but more recent studies suggested a crucial role of cytokines. 2 In type I diabetes, especially during hyperglycemic state, 4 high level of circulating pro-inflammatory cytokines and dysregulation of pro- and anti-inflammatory cytokine balance 5 are prominent features, and their major role can be suspected in pathophysiology of CC lesions found in our patient. Even if cytokines levels are not routinely tested in our institution for diabetic patients, elevated levels of pro-inflammatory cytokines (such as tumor necrosis factor α (TNFα), interleukin (IL)-6, IL-1β), as demonstrated in the clinical context of ketotic hyperglycemia according to the previous literature,4,5 can be reasonably assumed as the most probable link between the two conditions. Conversely, in hyperosmolar hyperglycemia state associated with type II diabetes, the hyperosmolar state itself, alone or synergistically with hypernatremia, can explain per se cytotoxic edema generation. 6
In our patient, considering the female sex and the young age, with a history of autoimmune disorders (thyroiditis), we first suspected an autoimmune encephalitis. CLOCCs have been interestingly associated also with voltage-gated potassium channel (VGKC) and glutamate-receptor antibody encephalitis, 1 but antineuronal antibody screening was negative and the patient did not fulfill the diagnostic criteria for antibody-negative encephalitis. 7
CVH reported by the patient may result from a direct lesional effect or a secondary epileptic origin. Stroke involving CC can present with transient formed visual hallucinations, 8 but isolated CC lesions are extremely rare. In a large series of CC strokes, 9 “minimal” or “discrete” lesions of CC were always multifocal or associated with posterior cerebral artery (PCA) infarcts. CC is also less vulnerable to lacune and small vessels ischemic disease due to histological differences between CC and centrum semiovale arteries. 9
Hallucinations are frequently described in CLOCCs 10 and may be due to CC direct involvement. However, they are not specific as they have been reported in callosal lesions of other etiologies.8,11 This observation has been supported by radiological studies on schizophrenia, where neuropsychiatric symptoms have been associated with splenium of CC structural abnormalities (reduced size or lower diffusion tensor imaging anisotropy) 12 or agenesis. 13 This highlights the important role of CC in regulation of cognitive, affective, and self-regulative behaviors and can explain the neuropsychiatric manifestations in patients with CLOCCs.
However, an epileptic etiology of hallucinations cannot be ruled out in our patient. Even if epileptic activities, due to occipital or temporal lobe seizures, were not detected by EEG of the scalp, normal findings are reported in a low proportion of epileptic patients due to deep or mesial temporal seizure localization. 14
Concerning therapeutical approach to CLOCCs, no evidence-based recommendations have been established. Wide heterogeneity of etiologies associated with CLOCCs explained the absence of guidelines in the management of this affection. Antibacterial/antiviral therapy, correction of electrolyte, or metabolic disturbances are reported in CLOCCs associated with infectious or systemic disease, respectively. Corticotherapy or IV immunoglobulins have been sometimes administrated in meningoencephalitis cases. In the cases of undefined underlying systemic disorder, no purposeful treatment was given, 15 and still good outcome was observed. Given these observations and potential side effects, immunotherapy should be accurately discussed case by case.
Conclusion
To the best of our knowledge, we reported the first case of CLOCCs associated with type I diabetes ketotic hyperglycemia. As reported for previous conditions associated with CLOCCs, elevated levels of circulating pro-inflammatory mediators, highly present in hyperglycemic state, suggest a role of cytokines in the pathophysiology of the CLOCCs. Further investigations, such as cytokine panel analysis in blood and CSF, may clarify the pathologic mechanism leading to CLOCCs.
Footnotes
Author contributions
Pasquale Scoppettuolo contributed to investigation, data curation, validation, writing—original draft/review and editing. Laura Sinkunaite contributed to validation, investigation, and writing—review and editing. Michaela-Felicia Topciu contributed to investigation, validation, and writing—review and editing. Joachim Schulz contributed to supervision, validation, and writing—review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
