Abstract
Primary sclerosing cholangitis is a chronic and progressive cholestatic liver disease that has been extensively documented in the human literature. Although it shares many features in common with chronic lymphocytic cholangitis in cats, primary sclerosing cholangitis has never been reported in a nonhuman primate. Primary sclerosing cholangitis is characterized by the presence of intrahepatic and/or extrahepatic inflammation and concentric fibrosis of bile ducts, eventually leading to cirrhosis and hepatic failure. The pathogenesis and cause remain unknown, but the disease likely involves a multifactorial mechanism with genetic- and immune-mediated components. The authors report 2 cases that histologically resemble the condition in humans; they consist of 2 adult male baboons with a clinical history of chronic elevated liver enzymes. In both cases, the liver was histologically characterized by thick bands of fibrosis and mild lymphoplasmacytic periportal cholangiohepatitis with concentric periductal fibrosis, resulting in atrophy and loss of bile ducts. Immunohistochemical analysis revealed positivity of hepatocytes to cytokeratin 7. Masson stain demonstrated marked biliary fibrosis. This is the first report that resembles sclerosing cholangitis in a nonhuman primate, and it suggests that the baboon may provide a useful animal model for this condition in humans.
Two intact male Baboons in adequate body condition were part of the nonhuman primate colony at the Southwest National Primate Research Center at the Texas Biomedical Research Institute. Both animals were captive bred. Case 1 was an 11-year-old that had been part of a study group related to diet and imaging analysis. The baboon had a 2-year history of elevated liver enzymes (alanine aminotransferase, alkaline phosphatase; Table 1) and a 1-month history of severe hepatomegaly and splenomegaly. At the time of necropsy, liver enzymes were markedly elevated. The baboon was also hypercholesterolemic and had a prolonged PPT time. Case 2 was a 6-year-old with a 3-year history of colitis and elevated liver enzymes (alanine aminotransferase, alkaline phosphatase, γ-glutamyltransferase); a moderate (conjugated) hyperbilirubinemia was present at the time of euthanasia. The baboon had no history of any experimental procedures performed. At necropsy, both livers were firm and contained multifocal nodular areas (Fig. 1). On cut section, prominent bile ducts surrounded by abundant fibrous connective tissue were evident (Fig. 2). In case 1, the distal half of the gallbladder and all associated draining ducts with extension into the liver were markedly thickened and partially obstructed. In case 2, the caudate and quadrate lobes and the gallbladder contained thickened fibrotic biliary tracts.
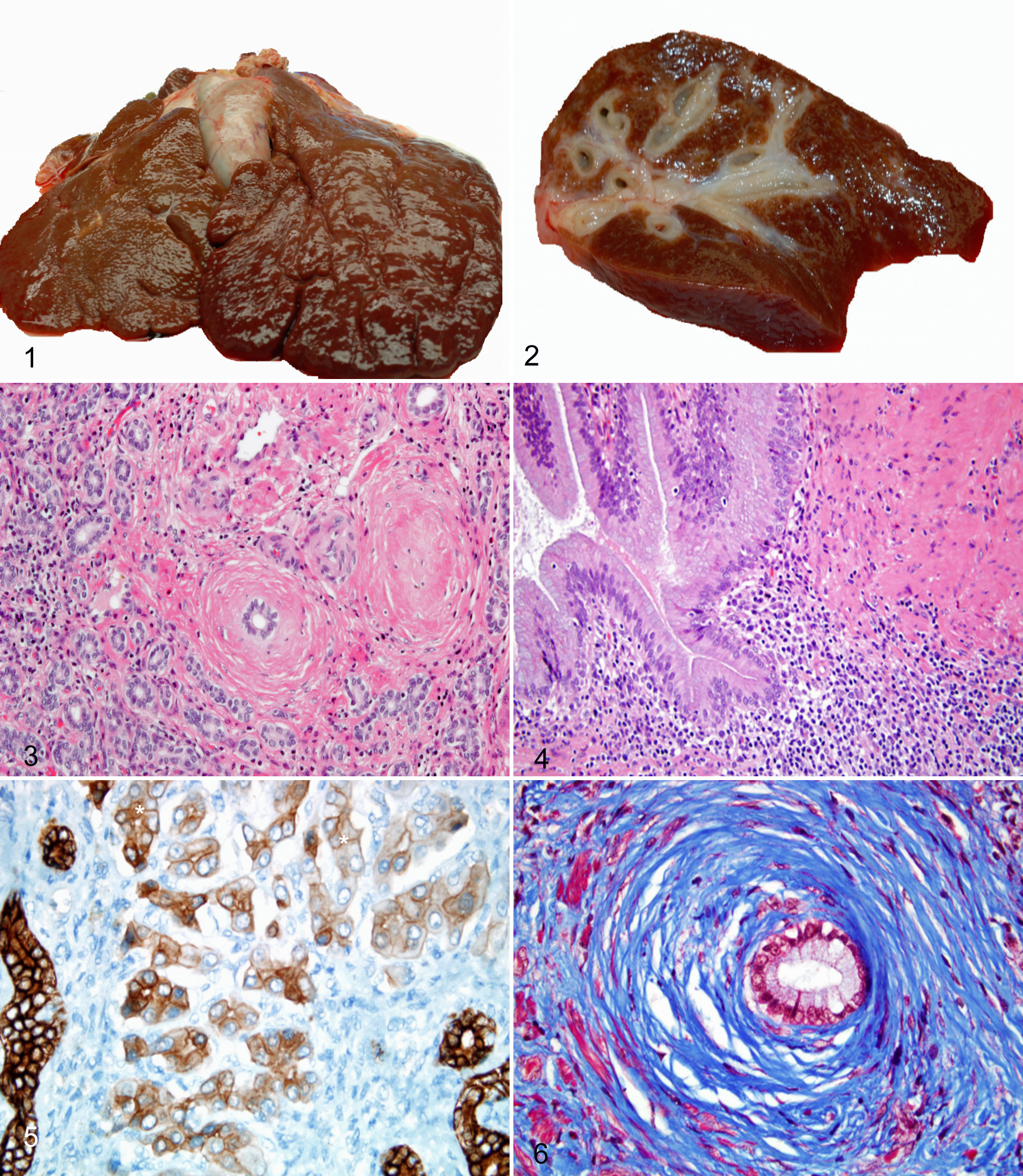
Liver; case 2. Grossly, the liver is firm with multifocal nodular areas.
Clinical Biochemistry in 2 Baboons Resembling Primary Sclerosing Cholangitis a

aALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; GGT, γ-glutamyltransferase.
Samples of the livers were fixed in 10% neutral buffered formalin, processed conventionally, embedded in paraffin, cut at 5 μm, stained with hematoxylin and eosin, and reviewed by light microscopy. Unstained sections from both cases were cut at 5 μm, deparaffinized, immunostained for cytokeratin 7 antigen (Dako, Denmark), and stained with Masson trichrome, silver stain, Rhodanine, Victoria blue, and Hall method for bilirubin.
Histologic examination of the liver in both cases revealed expansion of the portal tracts by fibrosis, a moderate increase in numbers of bile ducts, and an inflammatory infiltrate composed primarily of lymphocytes. Concentric bands of fibrosis surrounding bile ducts were observed (Fig. 3). Larger bile ducts contained lymphoplasmacytic infiltrates in the lamina propria and a small number of intraepithelial lymphocytes along with marked biliary hyperplasia (Fig. 4). Hepatocytes stained positively with cytokeratin 7, consistent with chronic cholestasis (Fig. 5). 15 Masson trichrome staining confirmed the marked biliary fibrosis (Fig. 6). Rhodanine and Victoria blue stains did not reveal copper, and the Hall method did not reveal bilirubin, suggesting that copper storage and bile stasis were not responsible for the lesions observed. Based on the gross and histologic appearance, special stain results, immunohistochemical analysis, and lack of an identified cause, the lesions resemble the condition in humans referred to as primary sclerosing cholangitis (PSC).
Discussion
PSC is a well-recognized disease in humans, histologically characterized by the presence of intrahepatic and/or extrahepatic biliary duct obliterative fibrosis that eventually progresses to cirrhosis and liver failure. 9,10 The diagnosis is made by the exclusion of secondary causes, as the disease has no specific clinical, biochemical, or histologic findings. The term secondary sclerosing cholangitis is used for a condition with similar clinical features to PSC but in which a direct cause for the process is known. 8 PSC is believed to have a multifactorial cause. 4,11 The disease appears to involve genetic factors, inadequate activation of the immune system, and a deleterious effect of bile acids on hepatocytes that leads to extensive fibrosis. 4,7,16
The existence of a predisposition of PSC among relatives reflects the hereditary component of the condition. Genetic factors clearly play an important role in the disease. Siblings of PSC patients are more likely than the overall population to develop the disease. 2 According to breeding colony records, the baboons in these cases were not related. Additionally, PSC manifests more commonly in males than females, with up to a 50% increased incidence. 1 Interestingly, the 2 cases reported here were both males, suggesting a similar predisposition, although an increased number of cases are warranted to corroborate this finding. Immunogenetic studies have demonstrated an association between the human leukocyte antigen complex and PSC. 6,17 Studies regarding this area remain to be elucidated in animals.
PSC is strongly associated with inflammatory bowel disease. Seventy-five to ninety percent of patients with PSC have a history of coexistent ulcerative colitis or Crohn disease. 1 It has been postulated that inflammatory cells are activated in the bowel of inflammatory bowel disease patients and then recruited to extraintestinal sites due to aberrant expression of the adhesion molecules MAdCAM 1 and CCL25 by the liver. 8,11 Bacteria that enter the portal circulation through an inflamed and highly permeable intestine could activate the innate immune system and serve as an inciting event in PSC. Activation of innate immune responses could lead to bile duct loss, progressive cholestasis, and fibrosis. Interestingly, 1 baboon had evidence of chronic colitis, and perhaps this inflammation played a role in the development of PSC.
Recent studies have showed an increased prevalence of Helicobacter pylori and other nongastric Helicobacter spp. in the livers of patients with PSC; however, the significance of this is still uncertain. 3,13 Lymphocytic cholangitis in cats is a condition that has striking similarities to PSC. 14 Interestingly, Helicobacter spp. has been recently implicated in feline lymphocytic cholangitis. 3,5 Further studies regarding the association of Helicobacter spp. and PSC are needed. Silver stains were performed and were negative in both cases described here (data not shown).
Multiple studies have elaborated on the hypothesis of “bile toxicity” in PSC.4 It has been proposed that bile acids that accumulate in different areas of the liver in PSC have a deleterious effect on hepatocytes and further contribute to the development of cirrhosis. In humans, one of the major pathways involved in the detoxification of bile is by hydroxylation and conjugation, leading to renal elimination. One of the major determinants in this pathway is the expression of the SXR gene.16 Patients with particular genetic variants in the SXR gene are prone to more severe disease, illustrating the effect of cholestasis on disease progression. In the cases reported here, both baboons had a 2-year history of elevated levels of alkaline phosphatase, an enzyme specific for cholestasis (3019 U/L and 1033 U/L; reference value, 0–555 U/L). Whether cholestasis is a cause or a consequence of PSC in these animals remains unknown.
PSC should be considered in the differential diagnosis for baboons with a chronic history of elevated liver enzymes. In humans, the diagnosis is made by the exclusion of secondary causes because there are no specific clinical, biochemical, or histologic findings. It is important to mention that in baboons, causes of secondary sclerosing cholangitis must be evaluated before a diagnosis of PSC is suspected. The most commonly described causes include parasitism, cholelithiasis, pancreatitis, and trauma. 12 To our knowledge, this is the first report of a condition that shares histopathologic features of PSC of humans. Further investigations to demonstrate if the syndrome in baboons is the same as in humans are still needed.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
