Abstract
Erysipelothrix rhusiopathiae septicemia, associated with an increased mortality of captive psittacines in a mixed-species aviary, was diagnosed by histopathology, Gram staining, bacterial culture and sequencing, immunohistochemistry, and real-time polymerase chain reaction (PCR). Over a period of 23 days with no premonitory signs, 2 rainbow lorikeets and an eclectus parrot died. Of these birds, one lorikeet and the eclectus were submitted for necropsy. The main pathologic findings were thrombosis (2/2), bacterial embolism/thromboembolism (2/2), necrotizing hepatitis (2/2), necrohemorrhagic myocarditis (1/2), fibrinohemorrhagic and heterophilic visceral coelomitis (1/2), submandibular necrosuppurative dermatitis with necrotizing vasculitis and bacterial and fungal thromboembolism (1/2), and locally extensive rhabdomyonecrosis with bacterial embolism (1/2). Intralesional bacteria were positive by Gram staining and immunohistochemistry in both cases. E. rhusiopathiae was isolated by routine bacterial culture from the liver of the lorikeet, which was also positive by real-time PCR. This report is one of the rare descriptions of erysipelas in psittacines, and to the authors’ knowledge, it appears to be the first in the described species using immunohistochemistry and real-time PCR on avian paraffin-embedded tissues for the diagnosis.
Erysipelothrix rhusiopathiae is a small, nonsporulating, Gram-positive rod found worldwide and may resist up to 5 years in the external environment (soil, surface waters). 4 It can infect a number of domestic and wild animal species including mammals, birds, amphibians, reptiles, fish, and even invertebrates such as ectoparasites of warm-blooded animals. The disease caused by E. rhusiopathiae in animals is called erysipelas, whereas in humans it is referred to as erysipeloid. 20 Human infection is commonly related to occupational exposure to sick or infected animals (anthropozoonosis) and frequently presents as an acute cellulitis at the portal of entry and uncommonly as a systemic infection typically involving the endocardium (subacute endocarditis) or joints. 7,20
In birds, E. rhusiopathiae infection has been described in poultry, turkeys, ducklings, and a variety of captive and free-ranging avian species including a laughing kookaburra (Dacelo novaeguineae), ring-necked pheasants (Phasianus colchicus), quails (Coturnix spp. and C. coturnix japonica), malleefowl (Leipoa ocellata), emus (Dromaius novohollandiae), a little blue penguin (Eudyptula minor), little swifts (Apus affinis), chukars (Alectoris chukar), white Pekin ducks (Anas platyrhynchos domestica), guinea fowl (Numida meleagris), and a Hawaiian crow (Corvus hawaiiensis). 1 –3,5,8,11,12,14,16 –19,22,24 –26,29 –31 Reports of E. rhusiopathiae infection in psittacine birds appear to be rare and include a description in translocated, endangered kakapos (Strigops habroptilus) and captive, recently imported lovebirds (Agapornis personata). 10,15 In birds, infection is most important in turkeys, which seem to be especially susceptible to septicemia. 9 Turkeys typically show signs of systemic infection characterized by thromboembolism and bacterial endocarditis. 25 This report describes the pathological, histochemical, bacterial culture, immunohistochemical, and polymerase chain reaction (PCR) findings of an outbreak of fatal peracute mortality associated with E. rhusiopathiae infection in a mixed-species aviary.
Over a period of 23 days, 2 rainbow lorikeets and an eclectus parrot peracutely died with no premonitory signs. Of these birds, one lorikeet and the eclectus were submitted for necropsy. Sections of all organs were immersed in 10% neutral-buffered formalin, routinely processed for histopathology, stained with hematoxylin and eosin (HE), and evaluated microscopically. Gram staining technique was performed in all tissues from both parrots. In addition, routine bacterial culture was performed on frozen sections of liver from both animals. A real-time PCR and an immunohistochemistry (IHC) assay with a polyclonal antiserum for E. rhusiopathiae serotypes 1a, 1b, and 2 were also performed on paraffin-embedded tissues in both cases, as previously described. 21,23
The lorikeet was found on the floor with ataxia and unable to fly, and it died shortly thereafter. At necropsy, the adipose tissue was moderately atrophied. The main gross findings were moderate to severe dehydration, severe hepatomegaly (Fig. 1), a large area of dark red discoloration in the left ventricle (Fig. 1), mild accumulations of yellowish transparent fluid in the coelomic and pericardial cavities, mild splenomegaly, and mild bilateral thyroid enlargement. Microscopically, the myocardial interstitium and epicardium were multifocally suffused with abundant erythrocytes (Fig. 2). Multifocal acute necrosis of cardiomyocytes and numerous intravascular fibrinocellular thrombi and colonies of rod-shaped bacteria in affected areas were noted (Figs. 2 and 3). The endothelial cells lining some affected vessels contained bacterial organisms within their cytoplasm (Fig. 3). The liver had rare, randomly distributed foci of coagulative and lytic hepatocellular necrosis infiltrated with heterophils; rare intralesional bacilli were observed. Many sinusoids were distended by abundant hypereosinophilic plasma, fibrin thrombi, leucocytes, and erythrocytes, and portal areas were mildly infiltrated by lymphocytes. Mild to moderate bacterial embolism was noted in the kidneys, gizzard, and proventriculus. Rare, small foci of necrosis and heterophilic inflammation with fibrin thrombi were present in the visceral adipose tissue. Mild thrombosis of pulmonary capillaries was also noted. The bone marrow had myeloid hyperplasia. Other findings included mild to moderate acute pulmonary hemorrhage and edema, mild to moderate diffuse hyperplastic goiter, and acute congestion of the liver, kidneys, and parathyroid glands. The spleen, visceral nervous ganglia, small intestine, cloaca, skin, skeletal muscle, brain, testes, adrenals, and pancreas were histologically within normal limits.
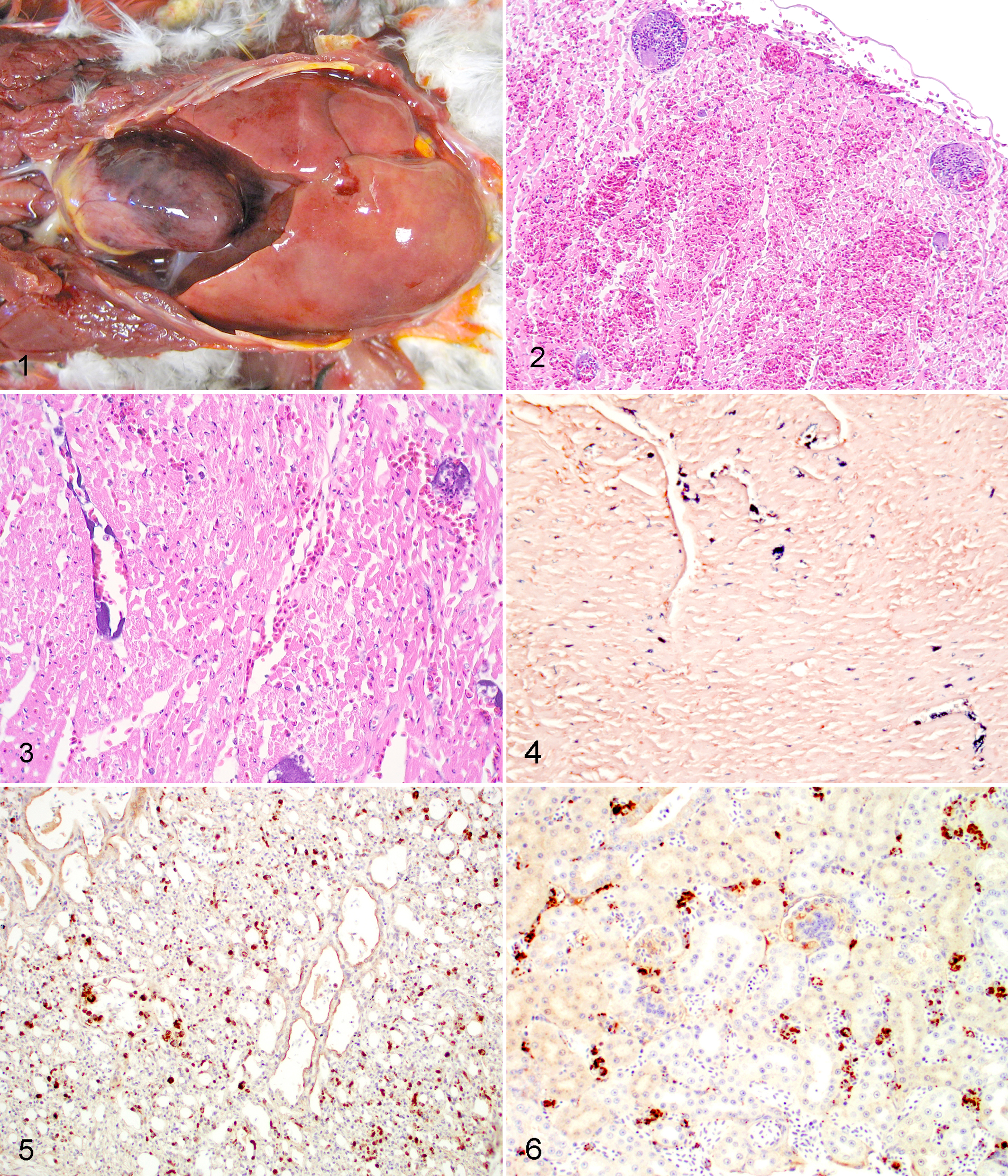
Rainbow lorikeet, coelomic cavity. Severe hepatomegaly and a large area of dark red discoloration in the left ventricle are noted.
The eclectus parrot was isolated from the aviary because of cagemate aggression, and it was treated with dexamethasone, vitamins, and meloxicam. It died 3 days later with no premonitory signs. Grossly, this parrot had an appropriate body condition. Major lesions were severe dehydration, the presence of whitish plaques adhered to the submandibular skin, moderate multifocal hemorrhage of the serosal surface of the esophagus and trachea, whitish foci in the liver, whitish discoloration of deep pectoral musculature, and mild proventricular dilation. Microscopically, the liver had randomly distributed foci of coagulative and lytic hepatocellular necrosis with dilatation of sinusoids by hypereosinophilic plasma and fibrin thrombi that occasionally contained emboli of bacilli. Erythrophagocytosis with hemosiderosis and phagocytosis of bacteria was observed in Kupffer cells. Bacterial thromboembolism or embolism was noted in the lung, heart, kidney, skin, skeletal muscle, and spleen. The submandibular cutaneous lesions consisted of areas of epidermal and dermal necrosis with heterophilic inflammation, thrombosis of blood vessels in the dermis and panniculus, and intralesional bacterial and fungal embolism with necrotic vasculitis and invasion of surrounding tissues by fungal hyphae. The crop, trachea, and visceral adipose tissue had multifocal to coalescing fibrin deposits with extravasated erythrocytes and heterophils and intralesional bacteria (bacilli) on the serosal surfaces. Pectoral muscle lesions consisted of acute rhabdomyonecrosis with dystrophic mineralization of fibers and intralesional bacterial embolism. Additional microscopic findings included mild to moderate acute necrosis of cardiomyocytes and mild renal tubular mineralization. The pancreas, small intestine, proventriculus, brain, gizzard, bone marrow, visceral and sympathetic periadrenal nervous ganglia, testes, adrenal, and thyroid were histologically within normal limits.
Intralesional bacteria stained positive with Gram in areas of thromboembolism and necrotizing and inflammatory lesions in both birds (Fig. 4). In the lorikeet, a pure growth of E. rhusiopathiae was obtained from the liver by bacterial culture, whereas Escherichia coli was isolated from the liver of the eclectus parrot. The isolate was referred to the Spanish National Centre of Microbiology and Taxonomy Reference Laboratory. The identification was confirmed using a BIOLOG GP2 panel with 95 carbon sources (BIOLOG, Hayward, CA, USA) with a similarity of 98% (T: 0,712) and by 16sRNA sequencing of 1386 bp fragment. The fragment showed a homology of 99.4% with E. rhusiophatiae sequence of Gene Bank accession no. AB055908 (strain ATCC 19414). Immunohistochemistry for E. rhusiopathiae antigen revealed mild (eclectus parrot) to marked (lorikeet) dark brown (positive) staining of bacteria-like organisms in both psittacines (Figs. 5 and 6). Immunolabeling appeared to involve mostly endothelial cells and circulating leukocytes in a variety of tissues including the lung, heart, kidney, and brain. E. rhusiopathiae-specific bacterial DNA was detected by real-time PCR in paraffin-embedded tissues from the rainbow lorikeet. Tissues from the eclectus parrot were negative by real-time PCR.
This report is one of the rare descriptions of mortality associated with septicemic E. rhusiopathiae infection in parrots; the diagnosis was based on the pathologic findings, Gram stain, bacterial culture, IHC, and real-time PCR on avian paraffin-embedded tissues. Only 2 other reports of erysipelas in psittacines were found in the literature reviewed by the authors. 10,15
The use of IHC and PCR on paraffin-embedded tissues of confirmed and suspected cases of erysipelas in birds has not to been reported previously in the literature reviewed by the authors. Different protocols for PCR have been used to diagnose E. rhusiopathie infection. 12,23 In this report, correlation of real-time PCR and IHC staining with bacterial culture in the lorikeet indicated that these methods were reliable for the diagnosis of erysipelas in paraffin-embedded tissues and could be used for retrospective investigation of archived suspected cases in which frozen tissues are not available. Although erysipelas was suspected in the eclectus parrot, based on the pathologic findings and the results of Gram staining and IHC, it could not be entirely confirmed by bacterial culture and PCR, which yielded negative results for E. rhusiopathiae. The sensitivity of this PCR assay on formalin-fixed tissues is unknown, and the fixation procedure used may have interfered with the assay. 27 The specificity of the immunohistochemical test was determined to be 100% in a previous study, since there was no evidence of cross-reaction with other pathogens tested. 21 Disagreement between direct bacterial isolation and the IHC assay was found in 25 of 170 (14.7%) swine cases. 21 This result might be similar to the findings in the eclectus parrot, in which the anti-inflammatory drugs used may have interfered with bacterial infection.
Different pathological presentations of erysipelas infection have been described in birds. Thrombosis, hemorrhage, necrosis, and inflammation observed in several tissues in the lorikeet with previous reports in several bird species, although the extensive splenic and proventricular damage described in such reports was not noted in this case. 13,20,22,31 E. rhusiopathiae experimentally infected chickens showed similar pathological findings to those of the lorikeet in this study, including congestion, organomegaly (liver, spleen, and kidney), and vascular damage in several organs (intravascular fibrin thrombi), highly suggestive of disseminated intravascular coagulation and shock. 28
Routes of transmission for E. rhusiopathiae are believed to include direct horizontal transmission by asymptomatic carriers such as pigs, turkeys, or contaminated fish food, as well as indirect horizontal transmission by the means of fomites such as contaminated soil or mechanical vectors such as arthropods. 6,20 Genotyping homologous studies between strains coming from different outbreaks support the cross-infectivity between species. 22,23 The source of infection in the parrots in this report remains undetermined.
In conclusion, E. rhusiopathiae septicemia was demonstrated in an outbreak of mortality in captive psittacine birds. This report is one of the rare descriptions of erysipelas in psittacine birds and the first using PCR on paraffin-embedded tissues and IHC in birds.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
