Abstract
A retrospective review of mortality records of Key Largo woodrats (Neotoma floridana smalli) in a captive breeding program revealed chronic renal disease in 5 of 6 woodrats older than 4 years of age. Two of the 5 woodrats with chronic renal disease also had clinical evidence of diabetes mellitus. Kidneys from all 5 woodrats were examined via light microscopy, histochemical staining, immunohistochemical staining, and transmission electron microscopy. The dietary histories of the affected animals were examined as well. The most striking histopathologic abnormality in the affected kidneys was the presence of large protein casts within cortical and medullary tubules in combination with lesions of membranous glomerulopathy and glomerulosclerosis. Transmission electron microscopy revealed thickening and undulation of the tubular and glomerular mesangial basement membranes with the variable presence of electron-dense deposits within the capillary endothelial basement membrane. Patchy glomerular immunoreactivity for IgG was noted in 2 cases, but IgA and IgM immunoreactivity were not present. The pathologic changes in the kidneys of the Key Largo woodrats mirrored many of the features of chronic progressive nephropathy commonly diagnosed in laboratory rats. Woodrats in the captive population were fed an ad libitum high-protein diet similar to diets that have been shown in laboratory rats to exacerbate the development and progression of chronic progressive nephropathy. It is concluded that Key Largo woodrats develop glomerulonephropathy with features similar to chronic progressive nephropathy described in laboratory rats. Age, concomitant disease, and dietary factors may contribute to the development and severity of this potentially age-limiting disease in Key Largo woodrats.
The Key Largo woodrat (KLWR), Neotoma floridana smalli, is an endangered subspecies of the Eastern woodrat, Neotoma floridana, with a range currently limited to the northern portions of Key Largo, Florida. 21 Adult KLWRs are brown, with males being significantly larger than the females (males average 258 g in body weight and females average 210 g). 21 Fewer than 500 individuals are thought to remain in the wild, and the biggest threats to the species may include a combination of issues, including predation by free-roaming domestic cats, raccoons (Procyon lotor), black rats (Rattus rattus), fire ants, habitat loss, and disease. 12 A population analysis suggests that at the current rate of decline, the KLWR could be extinct by the year 2016. 12
A captive breeding program was started in 2002, with 6 male and 6 female KLWRs initially being housed at Lowry Park Zoo in Tampa, Florida. Breeding success was limited in that only 14 offspring were produced in the first several years of the program. 10 A second captive breeding facility was added to the program in August 2005, when 11 woodrats were brought to Disney’s Animal Kingdom in Orlando, Florida. A retrospective examination of mortality records from 13 adult KLWRs necropsied at Disney’s Animal Kingdom revealed causes of death to include complications of neoplastic disease, bacterial infections associated with trauma (eg, bite, fight, mating), and chronic renal disease. Among the older animals that had been necropsied (those older than 4 years of age, n = 6), 5 cases of chronic renal disease were identified. The high incidence of chronic renal disease led to a further examination of these cases.
Chronic progressive nephropathy (CPN) is a common age-related disease in laboratory rats, Rattus norvegicus. A variety of factors are involved in this disease, including age, sex, genetics, and diet. This report describes the pathologic features of a chronic renal disease in aged KLWRs that mirrors many of the features of CPN of laboratory rats. Factors that influence the development of glomerulonephropathy in the KLWR may include advancing age, the presence of concomitant disease, and diet.
Materials and Methods
Animals
Thirteen adult KLWRs (greater than 1 year old) were necropsied from August 2005 to April 2009. Seven animals were 4 years old or younger at the time of death and showed no evidence of renal pathology. Six animals were 4.5 years of age or older at the time of death, and 5 had evidence of severe renal pathology.
Case No. 1 was a female that was at least 5.5 years old and weighed 280 g on the date of her death. She died spontaneously. The animal was wild caught, so her exact birth date was unknown. Approximately 10 days before her death, caretakers noticed a change in the color of her urine. A sample was collected from the bottom of the cage, and a urinalysis was performed. No urine abnormalities were noted aside from fecal contamination of the sample. Blood work was not performed on this animal at any time.
Case No. 2 was a female that was 5 years old and weighed 260 g on the date of her death. She was euthanized because of a declining quality of life. She had presented approximately 9 months before her death, with a primary complaint of polyuria/polydipsia and glucosuria. She was treated for diabetes mellitus using glipizide (Glucotrol, Pfizer Inc., New York, New York; 0.5 mg/kg orally twice a day) for the 9 months before euthanasia. Urine glucose ranged from 1000 to 2000 mg/dl with occasional samples measuring 4000 mg/dl; specific gravity ranged from 1.008 to 1.032; trace ketones were occasionally noted; and proteinuria (300-2000 mg/dl) was consistently present. Blood work was not performed on this animal at any time.
Case No. 3 was a male that was 5.5 years old and weighed 411 g on the date of his death. He presented 1 year before his death with a history of polyuria/polydipsia and was treated for diabetes mellitus based on clinical evidence of glucosuria. He was treated with glipizide (Glucotrol, Pfizer Inc.; 0.5 mg/kg orally twice a day) similar to case No. 2. At initial presentation, the urine glucose measured > 2000 mg/dl; the urine protein measured 2000 mg/dl; and the specific gravity was 1.034. A urine protein:creatinine ratio was determined on the initial presentation urine sample and was 7.9 (urine protein = 254 mg/dl, urine creatinine = 32 mg/dl). Following 2 months of Glucotrol therapy, the glucosuria resolved, but the urine protein continued to range between 300 and 2000 mg/dl on serial urine sampling. The woodrat was euthanized after being discovered hunched over and nonresponsive. Blood work was not performed on this animal at any time.
Case No. 4 was a male that was 4.5 years old and weighed 280 g on the date of his death. Ten days before euthanasia, this woodrat presented with multiple subcutaneous abscesses that were likely the result of mating (biting) injuries from an exhibit mate. The abscesses were treated with surgical debridement and medical management. He was euthanized because of a lack of response to treatment and because of the progressive nature of the subcutaneous wounds. A complete blood count and chemistry panel analyzed immediately before euthanasia revealed leukocytosis (16 200 cells/μl, reference range = 6790–13 990 cells/μl) and neutrophilia (8200 cells/μl, reference range = 560–2560 cells/μl). 11 The chemistry panel was unremarkable. Urinalysis was not performed on this animal at any time.
Case No. 5 was a male that was 4.5 years old and weighed 275 g on the date of his death. One month before death, he presented for polyuria and polydipsia. Urine analyzed at that time had a specific gravity of 1.009 with no evidence of glucose or protein. The woodrat was euthanized after presenting with clinical evidence of severe dehydration, dyspnea, and weakness progressing to nonresponsiveness. A complete blood count and chemistry panel analyzed immediately before euthanasia revealed relative neutrophilia (5355 cells/μl, reference range = 560–2560 cells/μl), toxic neutrophils, hypoglycemia (< 10 mg/dl, reference range = 146–198 mg/dl), and mild hypoalbuminemia (2.4 mg/dl, reference range = 2.6–4.4 mg/dl). 11
Clinical Pathology
Urine samples submitted for analysis were collected from the bottom of the cage utilizing wax paper to allow for collection above the wire mesh. Urinalysis (measuring specific gravity, glucose, bilirubin, ketones, erythrocytes, pH, protein, urobilinogen, nitrite, and leukocytes) was performed in our on-site hospital laboratory by dipstick (Multistix 10SG, Siemens Healthcare Diagnostics, Tarrytown, New York). Complete blood counts and serum chemistry panels submitted for 2 woodrats were performed by a commercial veterinary diagnostic laboratory utilizing standard techniques.
Necropsy and Histopathology
A necropsy was performed in all cases by the authors (S.P.T. and F.C.O.) following standard protocol. A complete set of tissues was collected for each case. Tissues were fixed in 10% neutral buffered formalin and trimmed within 72 hours. Tissues were then routinely processed, trimmed at 5 μm, mounted on frosted glass microscopy slides, and stained with hematoxylin and eosin. Periodic acid–Schiff (PAS), trichrome, and Congo red stains were applied to slides containing sections of kidney from each animal. Sections of kidney were also trimmed at 3 μm and stained with PAS/hematoxylin stain.
Transmission Electronic Microscopy
Samples of formalin-fixed kidney from 2 woodrats were embedded in Poly/Bed 812-Araldite resin. Semithin sections were stained with toluidine blue for light microscopy, and ultrathin sections were stained with uranyl acetate and lead citrate and examined using a JEOL100 CXII electron microscope.
Immunohistochemistry
Sections of kidney from all 5 woodrats were submitted for immunohistochemical staining for IgA, IgM, and IgG. Specimens were processed, embedded in paraffin, and sectioned on a rotary microtome at 4 μm. Sections were placed on slides coated with 2% 3-aminopropyltriethoxysilane and dried at 56°C overnight. The slides were subsequently deparaffinized in xylene and hydrated through descending grades of ethyl alcohol to distilled water. Slides were placed in Tris Buffered Saline, pH 7.4 (Scytek Labs, Logan, Utah), for 5 minutes for pH adjustment. Following Tris Buffered Saline, slides underwent heat-induced epitope retrieval utilizing citrate buffer, pH 6.0 (Scytek), in a vegetable steamer for 30 minutes at 100°C, allowed to cool on the counter at room temperature for 10 minutes, and rinsed in several changes of distilled water. Endogenous peroxidase was blocked utilizing a 3% hydrogen peroxide–methanol bath for 30 minutes, followed by running tap and distilled water rinses. Following pretreatment, standard avidin–biotin complex staining steps were performed at room temperature on the DAKO autostainer. All staining steps are followed by 2 rinses in Tris Buffered Saline + Tween 20 (Scytek). After blocking for nonspecific protein with Normal Horse Serum (Vector Labs, Burlingame, California) for 30 minutes, sections were incubated with an avidin/biotin blocking system for 15 minutes each (Avidin D, Vector Labs; d-Biotin, Sigma, St Louis, Missouri). Primary antibody slides were incubated for 60 minutes with the polyclonal goat anti-rat IgA diluted at 1:500 or polyclonal goat anti-rat IgM diluted at 1:4000 (AbD Serotec, Raleigh, North Carolina) in Normal Antibody Diluent (Scytek). Biotinylated horse Anti-Goat IgG (H + L; Vector) prepared at 11.0 μg/ml in Normal Antibody Diluent + 2% Normal Rat Serum (Vector) was incubated for 30 minutes, followed by RTU Vectastain Elite ABC Reagent (Vector) incubation for 30 minutes. Primary antibody slides were incubated for 60 minutes with the polyclonal rabbit anti-rat IgG (Biotin Conjugated; Novus Biologicals, Littleton, Colorado) diluted at 1:100 in Normal Antibody Diluent, followed by RTU Vectastain Elite ABC Reagent incubation for 30 minutes. Reaction development utilized Vector Nova Red peroxidase chromogen incubation of 15 minutes, followed by counterstain in Gill Hematoxylin (Thermo Fisher, Kalamazoo, Michigan) for 1.5 minutes, differentiation, and dehydration, clearing and mounting with synthetic mounting media.
Results
Necropsy
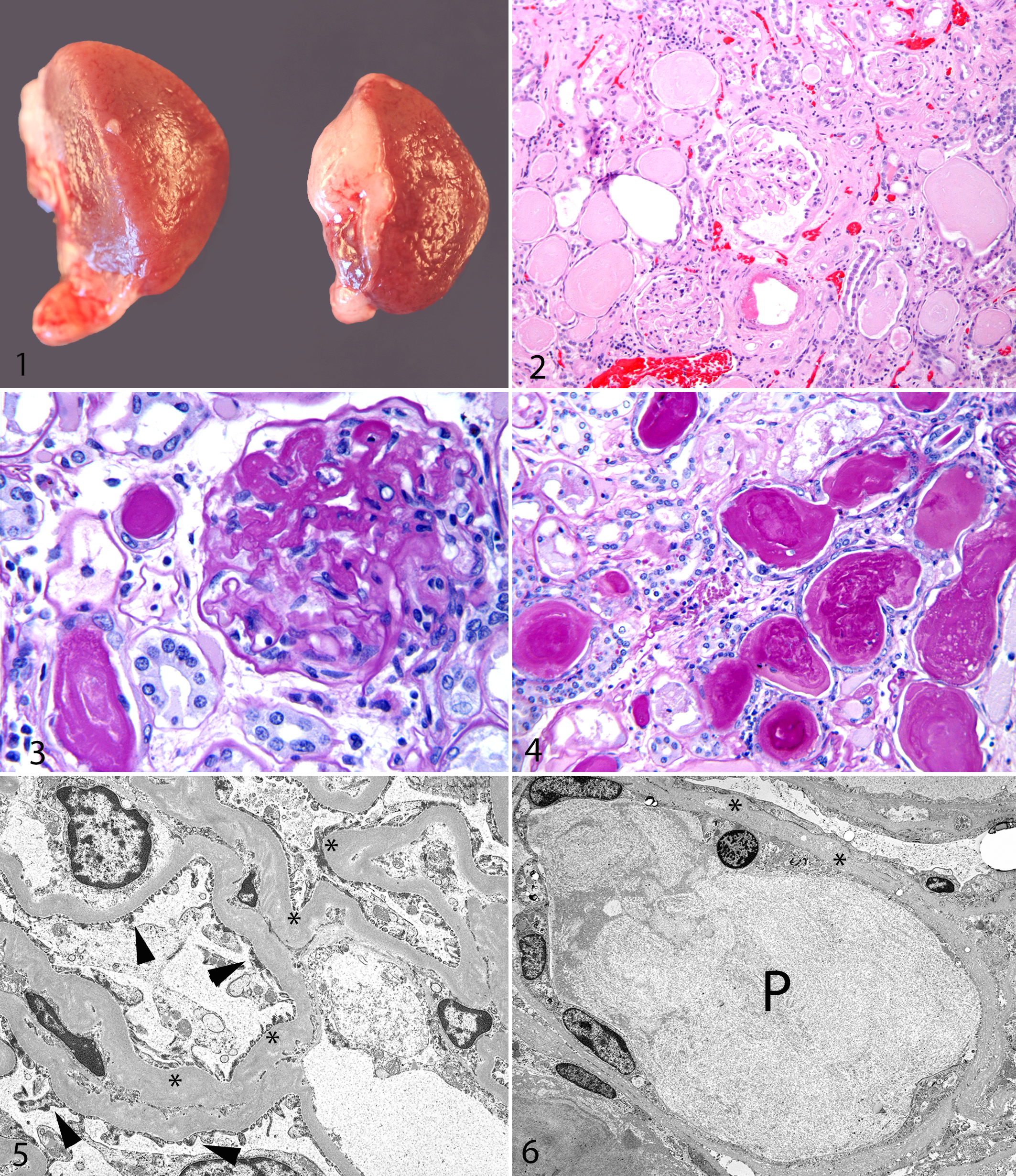
Despite their state of disease at the time of death, all 5 animals weighed more than the average weights of their wild counterparts. Adult KLWR males average 258 g in body weight and females average 210 g. 20 KLWR Nos. 1–5 weighed 280 g (female), 260 g (female), 411 g (male), 280 g (male), and 275 g (male) on the date of death. The kidneys of all 5 animals had a similar gross appearance at necropsy. The kidneys were enlarged and swollen. The capsular surface was pitted and irregular, and cortical tissue bulged from the capsule on cut section (Fig. 1 ). The cortical tissue was uniformly pale. Additional lesions noted at necropsy included the following: KLWR No. 1 had both pleural and abdominal effusion that was clear to pale yellow; KLWR No. 4 had multiple large chronic subcutaneous abscesses up to 1.5 cm in diameter filled with purulent exudate (chronic bite wounds); and KLWR No. 5 had bilateral consolidation and reddening of the cranioventral lung consistent with acute bronchopneumonia.
Histopathology
The histopathologic changes in the kidneys were similar in all 5 animals examined. The capsular surface was irregular and undulant. The cortical interstitium was distorted by proliferation of mature fibrous tissue with occasional small aggregates of lymphocytes and plasma cells (Fig. 2.). Tubular degeneration was evident, with cortical tubules being dilated, sometimes markedly so, and lined by attenuated flattened epithelial cells. Pynotic epithelial cells were also occasionally present in the degenerate tubules. Scattered tubular regeneration was identified and characterized by increased basophilia of tubular profiles with hypertrophy and hyperplasia of tubular epithelial cells. Rare mitotic activity within the hyperplastic tubular epithelial cell population was noted. There was multifocal thickening of the cortical tubular basement membrane by amorphous eosinophilic material that stained PAS positive. Numerous tubules contained hyaline or granular eosinophilic proteinaceous casts (Fig. 3). Glomeruli were often enlarged, although some sclerotic small glomeruli were usually present as well. Glomeruli exhibited segmental thickening of the basement membrane of Bowman capsule as well as the glomerular capillary loops by a homogenous amorphous eosinophilic material (also PAS positive) (Fig. 4). Portions of the kidney were severely affected (often as much as 75% of the renal parenchyma), while other areas within the same kidney showed minimal changes. Amyloid was not present in any sections examined with Congo red stains. A diagnosis of diffuse global glomerulopathy and glomerulosclerosis was made in all 5 cases examined.
Additional histopathologic lesions included the following: For KLWR No. 1, a well-demarcated nodular mass composed of well-differentiated hepatocytes was diagnosed as a focal hepatoma. Cortical lymphoid hyperplasia was evident in multiple lymph nodes and in gastrointestinal mucosal-associated lymphoid tissue. Mild to moderate focal subpleural histiocytosis was present in the lungs. For KLWR No. 2, multifocal mild chronic myocardial degeneration and fibrosis were observed, as well as diffuse acute pulmonary edema. For KLWR No. 3, lesions in organs other than the kidneys were not noted. For KLWR No. 4, there was evidence of multifocal to focally extensive areas of severe suppurative dermatitis and cellulitis with intralesional gram-positive bacteria. For KLWR No. 5, there was evidence of focally extensive severe necrosuppurative bronchopneumonia and pleuropneumonia with intralesional gram-negative bacteria. Inflammation, necrosis, and bacterial colonization also extended to the pericardium and myocardium. Random foci of acute necrosis were noted in the liver.
Transmission Electron Microscopy
The ultrastructural features of the kidney in the 2 animals examined were similar. Cortical tubules and glomeruli from both animals were examined. Glomeruli had evidence of redundant, folded, and thickened capillary basement membranes with multifocal electronic-dense deposits in the thickened membranes (Fig. 5). There was loss of pedicles and coverage of the basilar membrane by hypertrophied or irregularly shaped podocytes. The basement membrane of Bowman capsule was also thickened in some areas, with evidence of hyperplasia of epithelial cells lining Bowman space. Tubular changes included thickening and undulation of the basement membrane, attenuated tubular epithelial cells, degeneration of tubular epithelial cells with variable loss of microvilli, and tubular granular proteinaceous casts (Fig. 6).
Immunohistochemistry
Sections of kidney from all 5 woodrats examined exhibited negative immunoreactivity for IgA and IgM (data not shown). Control tissues for KLWR IgA and IgM were not available, so the validity of this staining profile could not be confirmed. The kidneys of 3 woodrats were negative for IgG, but patchy segmental staining of the glomerular capillary basement membrane was noted in 2 woodrats (Nos. 4 and 5).
Discussion
Light microscopic and ultrastructural changes in the kidneys from these 5 KLWRs were diagnosed as glomerulopathy and glomerulosclerosis. The morphologic features mirror those seen in aged laboratory rats with CPN. Two of the 5 animals had clinical evidence of diabetes mellitus, so the possibility of diabetic nephropathy (DN) playing some role in the renal pathology cannot be completely ruled out in those cases. Microscopically, CPN is characterized by thickening of the glomerular basement membranes, an increase in the number of mesangial cells, cystic dilation of Bowman capsule, atrophy of the glomerular tuft, and glomerulosclerosis.3,4,6,8,16,20 Tubules are often markedly dilated or cystic with tubular proteinaceous casts. Tubular epithelial degeneration, atrophy, and hyperplasia may be seen, and tubular basement membranes are often thickened. 6 Interstitial changes may also be evident and consist of interstitial edema or fibrosis with a mononuclear inflammatory infiltrate.6,9,13 The most striking histopathologic abnormality in the affected KLWR kidneys was the presence of large proteinaceous casts within both cortical and medullary tubules. These proteinaceous casts were seen in combination with glomerular and interstitial changes similar to those described in CPN. Likewise, the ultrastructural changes in the KLWR kidneys were identical to those seen in CPN. Transmission electronic microscopy revealed thickening and undulation of the tubular and glomerular basement membranes with the variable presence of electron-dense deposits within the capillary endothelial basement membrane.
Clinical pathology data were sparse from this population of KLWRs due to husbandry limitations on handling of this species as well as difficulty obtaining diagnostic samples. Proteinuria and hypoalbuminemia are both commonly seen in laboratory rats with CPN. 3 Urinalysis was consistently performed on only the 2 animals with diabetes mellitus. In those animals, urine protein measured up to 2000 mg/dl by the dipstick method. Single point-in-time urinalyses performed on KLWR Nos. 1 and 5 revealed no evidence of proteinuria, a surprise given the evidence of severe tubular proteinuria on histopathologic examination of the kidneys from all 5 cases. Mild hypoalbuminemia was noted in a blood sample from KLWR No. 5, while the albumin was within normal limits for the only other KLWR from which blood was analyzed, No. 4.
CPN (also known as age-related nephropathy, chronic nephritis, chronic nephrosis, spontaneous glomerular sclerosis, progressive glomerulonephrosis) is a common age-related disease in laboratory rats. The specific cause and pathogenesis of CPN is generally not known, although a variety of factors in addition to age (strain, sex, diet) are known to play a role in the disease.2–4,6,8,11,16–18 Greater than 75% of rats of susceptible strains (Sprague-Dawley and Fischer 344 strains) approaching 3 years old may show lesions of CPN. 16 The disease is more common and more severe in males.6,8,16 High-protein and high-calorie diets contribute to the severity of disease.6,8,10,16–18 Although IgM may be observed in the mesangium of affected glomeruli, immune complex deposition and autoantibody production do not appear to play a role in the glomerular pathology.5,16,19
Age-related chronic renal disease that is morphologically similar to CPN has been diagnosed in a variety of laboratory rodent species, including mice, hamsters, gerbils, and guinea pigs. 3 However, CPN is unique to rats because basement membrane thickening occurs in the absence of vascular lesions. 3 A study of renal pathology in adult wild Norway rats (Rattus norvegicus) found no evidence of CPN in 39 animals. 7 Arteriosclerosis of the afferent and efferent renal vessels was not observed in any of the KLWRs in this report.
There is an association with strains of domestic laboratory rats and CPN. CPN has been diagnosed in all rat strains used in safety evaluation studies but is most commonly diagnosed in rats of the Sprague-Dawley and Fischer 344 strains. 20 Similar to the domestic laboratory rat, the KLWR appears to be a species that is prone to the development of chronic renal disease when other contributing factors are present—namely, species, age, and diet. The small number of animals examined (3 males, 2 females) prevents us from making any conclusions about the effect of sex on this disease in KLWRs.
From a taxonomic perspective, the KLWR (Neotoma floridana smalli) is a “close cousin” of the domestic laboratory rat, Rattus norvegicus. Both are in the order Rodentia and the superfamily Muroidea. The taxonomic link stops at the family level. The laboratory rat is in the family Muridae (true mice and rats, gerbils, spiny mice, and crested rats), and the KLWR is in the family Cricetidae (New World mice and rats, voles, and hamsters).
Age is a well-documented contributing factor in laboratory rats with CPN. Initial lesions are noted as early as 2 months and progress over time. As noted above, greater than 75% of rats of susceptible strains approaching 3 years old may show lesions of CPN. 16 CPN is considered an age-limiting disease in laboratory rats. 16 The mean age of the affected animals in this small study group was 4.5 years old. Significant renal pathology was not noted in any KLWRs that died at 4 years of age or younger in this population (n = 7). The expected life span of KLWRs in the wild or in captivity is not definitively known. A retrospective review of husbandry records from the captive-breeding population indicate that 5.5 to 6 years of age may be the maximum life span for this species. It is concluded that the animals in this report were of advancing age for this species and that chronic renal disease is an age-limiting condition.
A variety of studies have demonstrated a relationship between protein and caloric intake in laboratory rats and development/severity of CPN.2,11,17,18 Rats fed high-protein diets or ad libitum diets develop CPN earlier in life and have significantly more severe renal pathology than do rats on lower protein diets or those placed on dietary caloric restriction.2,11,17,18 Decreasing the dietary protein concentration from 23% to 15% in Fischer 344 rats decreased the severity of CPN lesions. 17 Dietary caloric restriction, even in the face of high-protein concentrations (21%), may slow the development and progression of CPN. 11 Dietary caloric restriction may also have beneficial effects in rats with diabetes (Nos. 2 and 3 in this report), as caloric restriction inhibits diabetic renal injury in a type 2 DN rat model. 14 The woodrats in this report were fed an ad libitum diet that included mixed vegetables with a commercially available rodent block (Mazuri rodent pellets, Cat 5663, St Louis, Missouri). The composition of the KLWR rodent block was 26% protein, 7.7% fat, 33% starch/carbohydrate, and 10% neutral digestible fiber. Table 1 compares the KLWR rodent block with high- and low-protein diets used in published studies examining the impact of protein and caloric intake on CPN. Compared with the published “high-protein” diets fed in those studies, the KLWR pellet contained higher protein, higher fat, lower starch/carbohydrate, and higher fiber. Wild diet items of the KLWR are generally lower in protein, lower in starch, and significantly higher in neutral digestible fiber than the diet fed to the animals in this report (personal communication, Eduardo Valdes, 2010). At the time of this report, it was believed that high dietary protein increased the reproductive potential of the KLWRs in the captive breeding program. This was important because of the need to maximize reproduction of this endangered species. However, given the fact that KLWRs appear susceptible to development of CPN, the high-protein rodent block fed to the animals in this study may exacerbate the onset and severity of disease in this captive population. The animals described in this report each exceeded the average weight of their wild counterparts, some significantly so. It is likely that high-protein diets and ad libitum feeding contributed to excess weight gain, as well as the renal pathology, in this captive population.
A comparison of the diet of KLWRs with chronic renal disease with diets in published studies of the effects of high and low protein diets on CPN in laboratory rats.
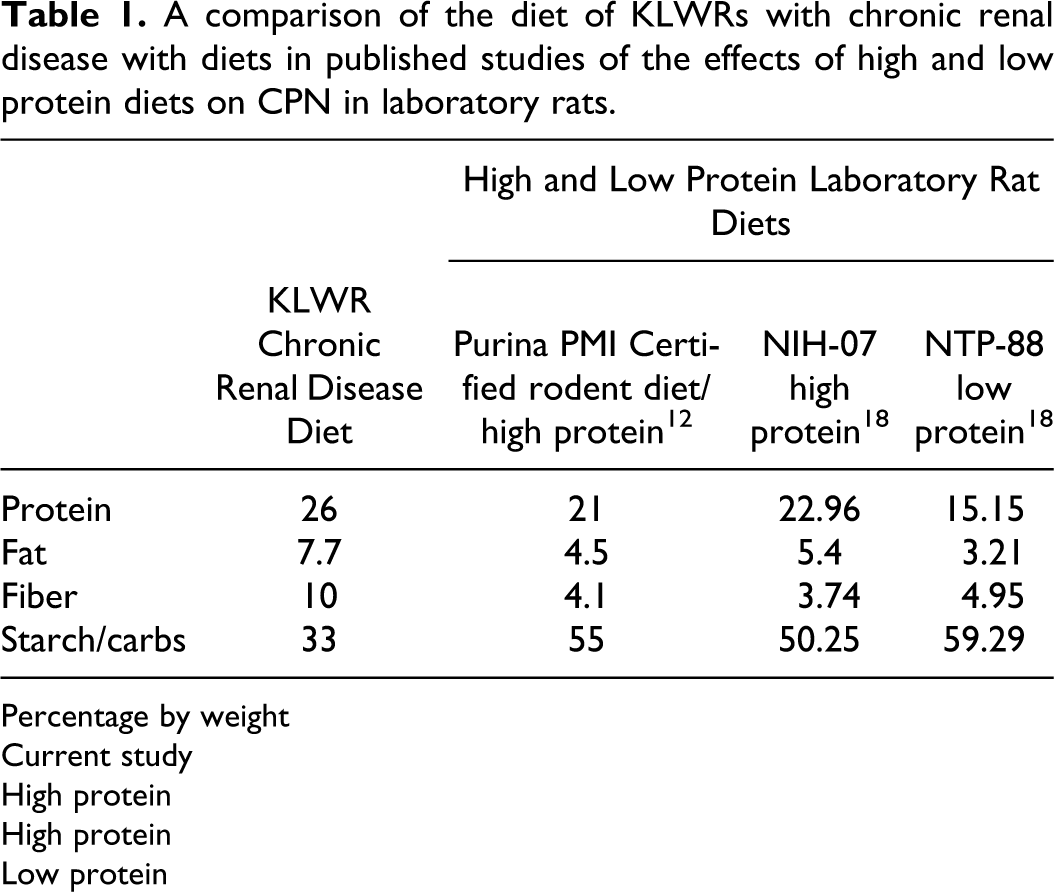
Percentage by weight
Current study
High protein
High protein
Low protein
Two KLWRs described in this report were diagnosed with diabetes mellitus 9 months and 12 months before death. Histopathologic examination of the pancreas from the 2 affected woodrats revealed adequate numbers of normal-appearing islets of Langerhans. This fact, in combination with the diet and weight history in these animals, suggests that they were suffering from a type 2 diabetes-like disease.
Rats are susceptible to renal damage associated with diabetes mellitus.14,15,22 DN is a condition associated with both type 1 and type 2 diabetes mellitus. Approximately 40% of human patients with diabetes mellitus will develop DN. 1 DN affects all compartments of the kidney, including the glomerulus, tubulointerstitium, and vasculature. 1 The characteristic features of DN are diffuse and nodular glomerulosclerosis as well as arteriosclerosis of afferent and efferent arterioles. 1 Nodular glomerulosclerosis is caused by proliferation of glomerular mesangial cells and mesangial matrix. The mesangial proliferation results in formation of nodules visible on light microscopy as sclerotic acellular accumulations of eosinophilic material (also known as Kimmelstiel-Wilson nodules). 1 Nodular glomerulosclerosis was not a feature of the renal pathology in any of the KLWRs examined. Likewise, arteriosclerosis was not identified in any of the animals. Diffuse glomerulosclerosis due to DN can be difficult to differentiate from other conditions that result in glomerulosclerosis; therefore, the possibility that DN played a role in the renal pathology in 2 KWLRs cannot be excluded.
Factors known to be associated with the development and severity of CPN in laboratory rats (age and diet) may also play a role in the development of this disease in KLWRs. The woodrats in this report were considered aged animals and were fed an ad libitum diet high in protein. Since age and inherent susceptibility of the species to this disease cannot be controlled, dietary manipulation may be an important management tool for CPN in KLWRs. Although it was believed that the high-protein diet increased the reproductive potential of the captive KLWR population, this diet may have inadvertently limited the life span of the adult animals and exacerbated the development of renal pathology. Reduction of dietary protein and/or restriction of caloric intake decreases the progression and severity of CPN in laboratory rats and should be considered for captive populations of KLWRs. For those animals in which diabetes mellitus may have played a role in renal pathology, caloric restriction may prove valuable as well by reducing the incidence of obesity and by inhibiting diabetic renal injury. Even with dietary manipulation, chronic renal disease may be a significant age-limiting condition in captive populations of this endangered species.
Footnotes
Acknowledgements
We thank the cast members/staff at Disney’s Animal Kingdom for their assistance in the day-to-day husbandry and medical care of these animals, and we would like to specifically thank the “Nights” animal care team for its dedication to this nocturnal species. The Investigative Histopathology Laboratory at Michigan State University was invaluable in optimizing immunohistochemistry protocols, and Ralph Common provided electron microscopy expertise. We also thank Dr Eduardo Valdes for assistance with nutritional information and wild diet data as well as review of the article.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
