Abstract
The authors describe genital alterations and detailed histologic findings in baboons naturally infected with Treponema pallidum. The disease causes moderate to severe genital ulcerations in a population of olive baboons (Papio hamadryas anubis) at Lake Manyara National Park in Tanzania. In a field survey in 2007, 63 individuals of all age classes, both sexes, and different grades of infection were chemically immobilized and sampled. Histology and molecular biological tests were used to detect and identify the organism responsible: a strain similar to T pallidum ssp pertenue, the cause of yaws in humans. Although treponemal infections are not a new phenomenon in nonhuman primates, the infection described here appears to be strictly associated with the anogenital region and results in tissue alterations matching those found in human syphilis infections (caused by T pallidum ssp pallidum), despite the causative pathogen’s greater genetic similarity to human yaws-causing strains.
At Lake Manyara National Park (LMNP), a disease of unknown etiology, first reported in 1994, 37 produces severe anogenital lesions in olive baboons (Papio hamadryas anubis). Field observations indicate that the disease solely affects primates of the genus Papio and that its features resemble the manifestations of human syphilis infections.
Although treponematosis in wild nonhuman primates is not a new phenomenon and was documented decades ago,17,18 there is a paucity of information on its pathology. Historically, infections in nonhuman primates have been reported only from areas of West Africa. Clinical signs, when present at all, were usually described as small ulcers on the muzzle.3,4,17,19 The disease in LMNP’s baboon population is characterized by clinical manifestations that dramatically differ from those previously described in West Africa.16 –18 The majority of information regarding enzootic infection in wild nonhuman primates is based on serologic screening methods, and only a single strain collected from a baboon in Guinea has been subjected to genetic and clinical studies in the laboratory. 11
The aim of this study is to give a detailed description of the clinical appearance and characteristic histologic skin alterations in olive baboons with genital ulcerations at LMNP. The identification and characterization of the responsible pathogen, Treponema pallidum, is described.
Materials and Methods
Field Survey and Sampling
The total population size of olive baboons at LMNP is estimated at 2,500 individuals. Only one-third of Lake Manyara’s protected area (330 km 2 ) is covered by land, crowding the existing primate population to an extremely high density. Baboons can be found in all parts of the park, but the groundwater forest and villages bordering LMNP in the north attract most of the animals. The disease is present in all parts of the national park, based on informal sightings of visibly infected animals. Baboons with genital ulcerative lesions visible to the naked eye were categorized as clinically affected, and animals without any genital lesions were classified as clinically nonaffected. Those animals with genital lesions were further divided according to initial, moderate, or severe stages, depending on the extent of macroscopic visible ulceration. In 2007, clinically affected baboons (case Nos. 21–60, 65; n = 41) and clinically nonaffected baboons (case Nos. 1–20, 61–64; n = 24) throughout LMNP (Table 1 ), of all age classes and both sexes, were temporarily immobilized for short-duration anaesthetic procedures using a mixture of ketamine and xylazine (n = 63 individuals; 2 animals were recaptured). 30
Cross-Tabulation of Tests Used and Sample Size in 63 Immobilized Olive Baboons a at Lake Manyara National Park, n
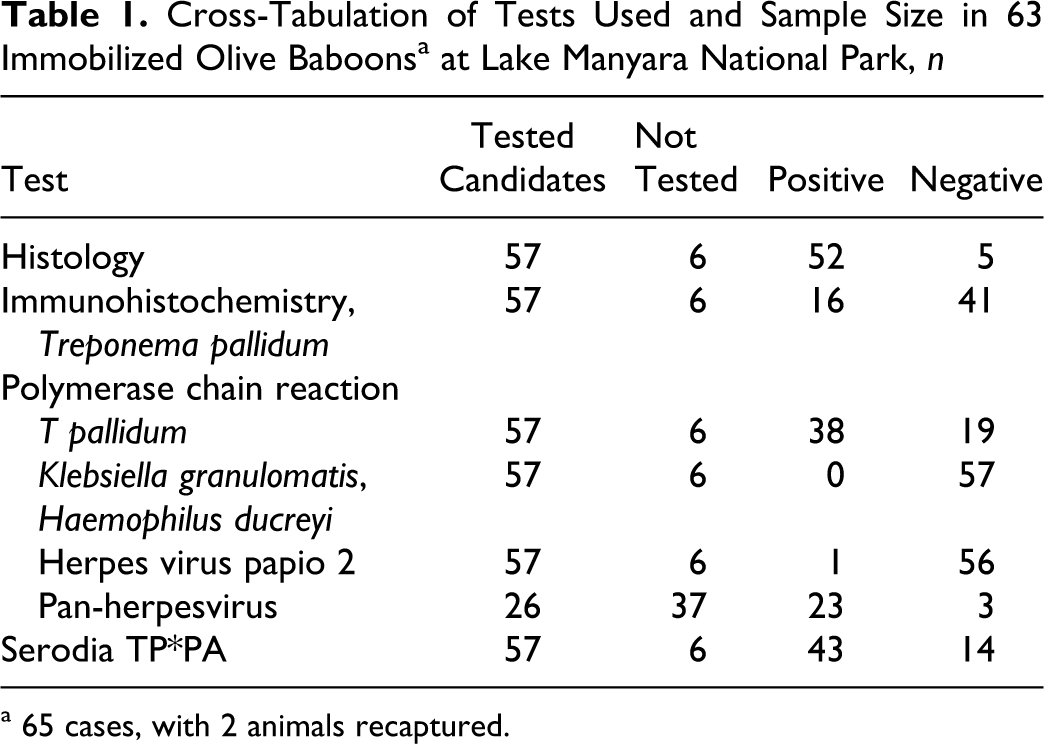
a 65 cases, with 2 animals recaptured.
Samples collected included blood, genital swabs, and skin tissue biopsies. During a 3-month time, a total of 63 olive baboons were sampled (case Nos. 56 and 60 as well as Nos. 58 and 59 are from the same individuals; ie, both animals were sampled twice). Individuals were temporarily marked by shaving parts of their tails. Skin tissue biopsies were taken with a sterile 6-mm biopsy punch (Heiland MED Vertriebsgesellschaft, Hamburg, Germany). In males with dermatitis affecting the corpus of the penis, tissue samples were carefully taken with a sterile scalpel blade to prevent damage to the corpus cavernosum penis. Under general anesthesia, clinically affected individuals were sampled for one piece of tissue from the edge of the alteration and another one from adjacent healthy-looking skin tissue. In clinically nonaffected baboons, a single skin tissue sample of the anogenital region was collected. These samples, which included all 3 layers of the skin (epidermis, dermis, and subdermis), were immediately preserved in 10% buffered neutral Lilli’s formalin. Resulting skin wounds were sutured in a simple interrupted manner with absorbable 3/0 Surgicryl PGA (SMI, Hünningen, Belgium) and covered with silver spray (Henry Schein, Melville, NY). Sampled animals received a shot of long-acting antibiotics (Duphamox LA, Fort Dodge GmbH, Würselen, Germany) to prevent secondary infections at the side of retained biopsy wounds.
Tissue Samples and Histology
Formalin-preserved tissue samples were cut into 2 vertically asymmetrical pieces and embedded into paraffin (Hypercenter XP, Thermo Shandon GmbH, Darmstadt, Germany). The paraffin blocks were subsequently cut into 4-μm-thick layers with a microtome (Mikrotom HM 400R, Microm International GmbH, Walldorf, Germany) and submerged in a 40°C bath containing distilled water. After being transferred onto a slide (Superfrost Plus, Gerhard Menzel Glasbearbeitungswerk GmbH & CoKG, Niedersachsen, Germany), specimens were allowed to dry over night at 37°C.
Hematoxylin and eosin staining (Thermo Shandon GmbH; Merck KGaA, Darmstadt, Germany) and periodic acid–Schiff staining (Merck KGaA) were performed in all specimens with a fully automated process (Varistain Gemini, Thermo Shandon GmbH). Warthin–Starry silver staining was also performed in 63 individuals (case Nos. 1–63, case Nos. 64 and 65 were not sampled for skin tissue). Giemsa staining (Carl Roth GmbH & CoKG, Karlsruhe, Germany) was applied in all cases in which bacterial structures were suspected. Staining followed the manufacturer’s protocol. Each slide was examined by 2 researchers on an independent basis in 2 separate rounds, under light microscopy using a standardized meandering course, with starting magnification of 100×, followed by increasing magnifications up to 1000×.
Immunohistochemistry
Paraffin-embedded tissue sections were heat pretreated with citrate buffer (pH 6.0) and followed by immunohistochemical analyses with a rabbit polyclonal antibody against T pallidum diluted 1:500 (No. GTX40465, Biozol Diagnostica Vertrieb, Eching, Germany). Primary mouse-anti-human B-cell CD20 antibody (M755, Dako, Hamburg, Germany) and rabbit-anti-human T-cell CD3 antibody (A452, Dako) were used in dilutions of 1:300 and 1:50, respectively, to differentiate B cells and T cells. Biotinylated secondary antibodies streptavidin and diaminobenzidin (No. 760-091, View DAB Detection Kit, Ventana, Hamburg, Germany) were applied as chromagen according to the supplier’s instructions. A negative control for the specificity of the antibody was performed by replacing the primary antibodies with sheep serum on a slide. A tissue section from a patient infected with primary-stage syphilis served as positive control. Mayer hematoxylin (Sigma-Aldrich Chemie GmbH, Munich, Germany) was used to counterstain. A semiquantitative estimation of the proportion of B cells and T cells (in percentage) was used to describe the present lymphocyte population.
Molecular Biological Tests
DNA was extracted from skin tissue biopsies conserved in RNA-Later (Applied Biosystems Inc, Foster City, CA). Table 1 and 2 give an overview of the sample size and tests used. Skin samples were cut into small pieces and ground with Precellys-Keramik beads (peQlab Biotechnologie, Erlangen, Germany), followed by incubation with proteinase K for 4 to 6 hours at 56°C. Remaining steps followed the manufacturer’s protocol (NucleoSpin Tissue Extraction Kit, Macherey-Nagel, Düren, Germany). All washing steps were carried out twice.
Clinical Manifestation, Histologic Signs of Infection, Immunohistochemistry, and Polymerase Chain Reaction Results in 57 Olive Baboons From Lake Manyara National Park
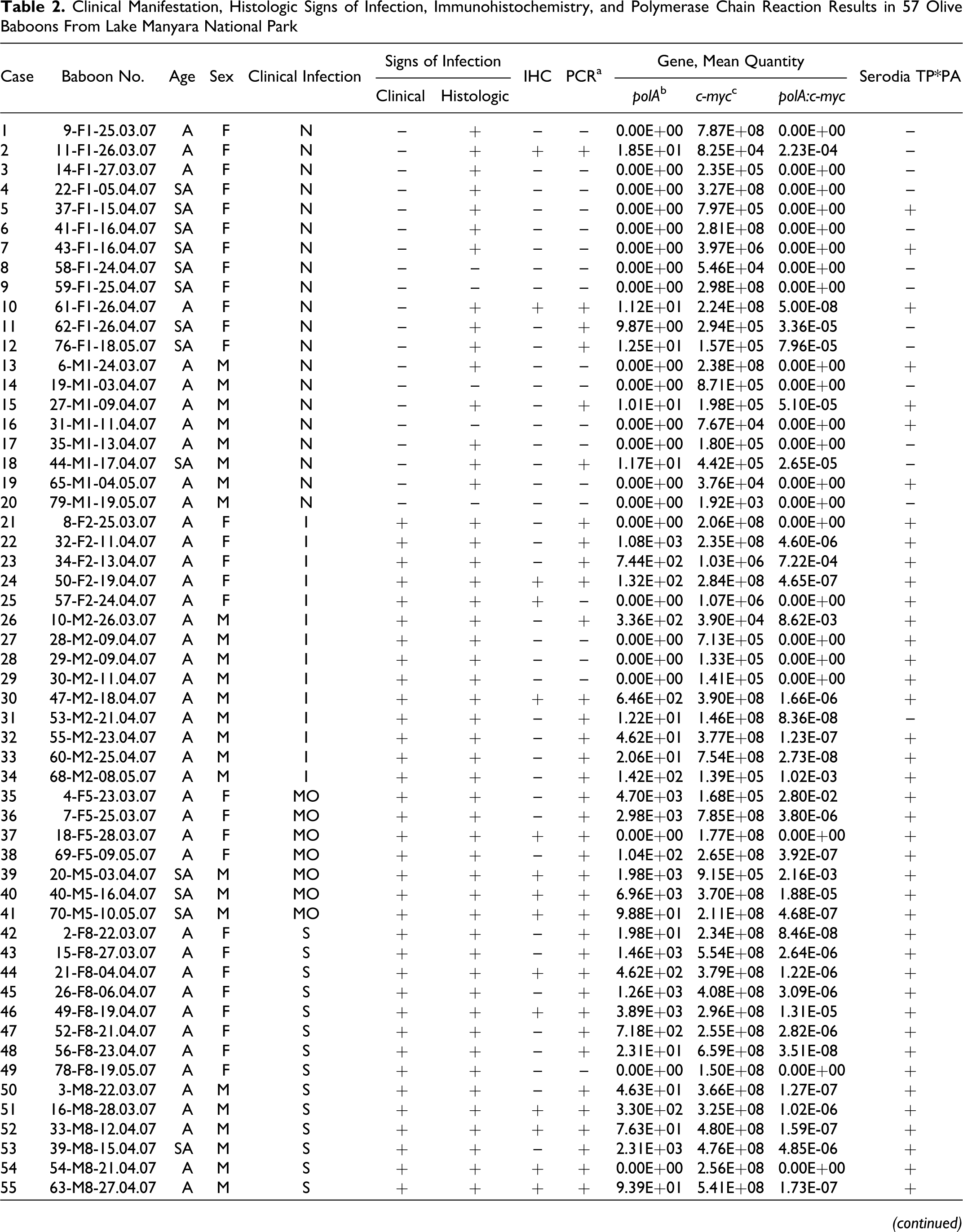
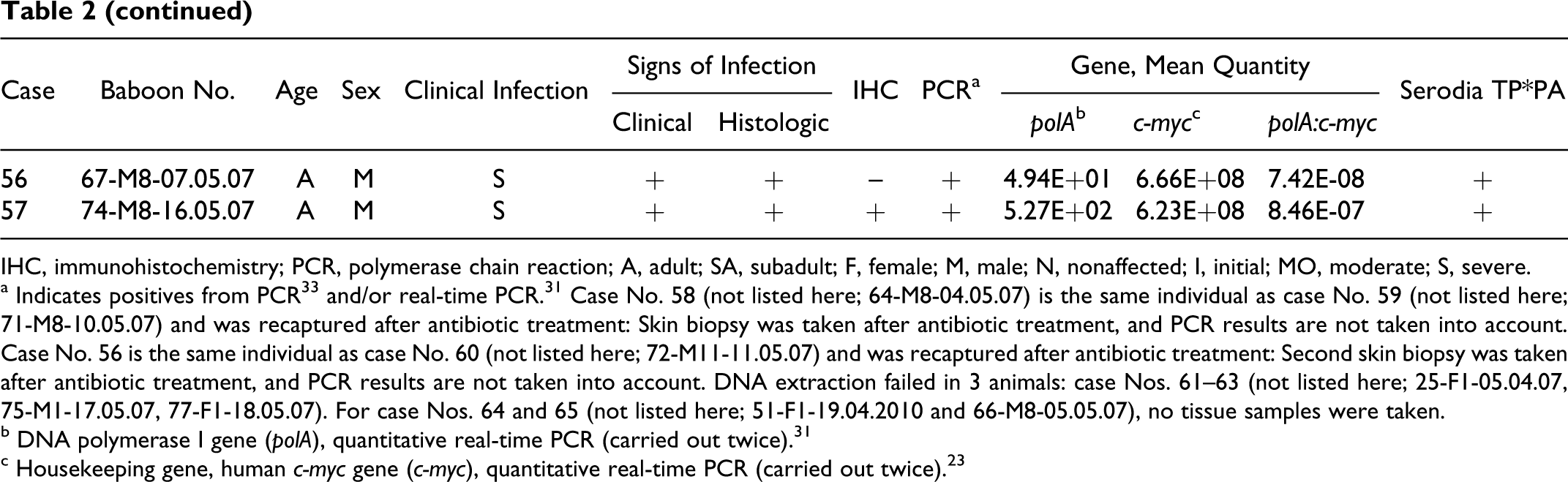
IHC, immunohistochemistry; PCR, polymerase chain reaction; A, adult; SA, subadult; F, female; M, male; N, nonaffected; I, initial; MO, moderate; S, severe.
a Indicates positives from PCR 33 and/or real-time PCR. 31 Case No. 58 (not listed here; 64-M8-04.05.07) is the same individual as case No. 59 (not listed here; 71-M8-10.05.07) and was recaptured after antibiotic treatment: Skin biopsy was taken after antibiotic treatment, and PCR results are not taken into account. Case No. 56 is the same individual as case No. 60 (not listed here; 72-M11-11.05.07) and was recaptured after antibiotic treatment: Second skin biopsy was taken after antibiotic treatment, and PCR results are not taken into account. DNA extraction failed in 3 animals: case Nos. 61–63 (not listed here; 25-F1-05.04.07, 75-M1-17.05.07, 77-F1-18.05.07). For case Nos. 64 and 65 (not listed here; 51-F1-19.04.2010 and 66-M8-05.05.07), no tissue samples were taken.
b DNA polymerase I gene (polA), quantitative real-time PCR (carried out twice). 31
c Housekeeping gene, human c-myc gene (c-myc), quantitative real-time PCR (carried out twice). 23
Polymerase chain reactions (PCRs) were run in 57 animals with successful DNA extraction (Table 2), to detect T pallidum and to rule out the presence of Klebsiella granulomatis, Haemophilus ducreyi, and herpesvirus papio 2, all known to cause compatible clinical signs. Negative extraction and PCR controls were included in each assay. A real-time PCR assay specific to T pallidum and, for control and standardization, the housekeeping c-myc gene was carried out twice for each tissue sample. In addition, 26 randomly chosen baboons (case Nos. 1, 4, 7, 9, 10, 12, 17, 21, 24, 26, 30, 31, 35, 37, 38, 40–42, 44–46, 48–49, 52, 54, 59) were tested for the presence of any other herpesviruses with pan-herpes consensus PCR.12,39 PCR conditions for herpesvirus papio 2 followed the same protocol as that used to detect the other herpesviruses. 13 A 16S rRNA gene PCR 38 was used to test for a broad range of different prokaryotes in 6 baboons (case Nos. 17, 21, 26, 37, 49, 59). All primers are summarized in Table 3 .
Primers Used in the Study
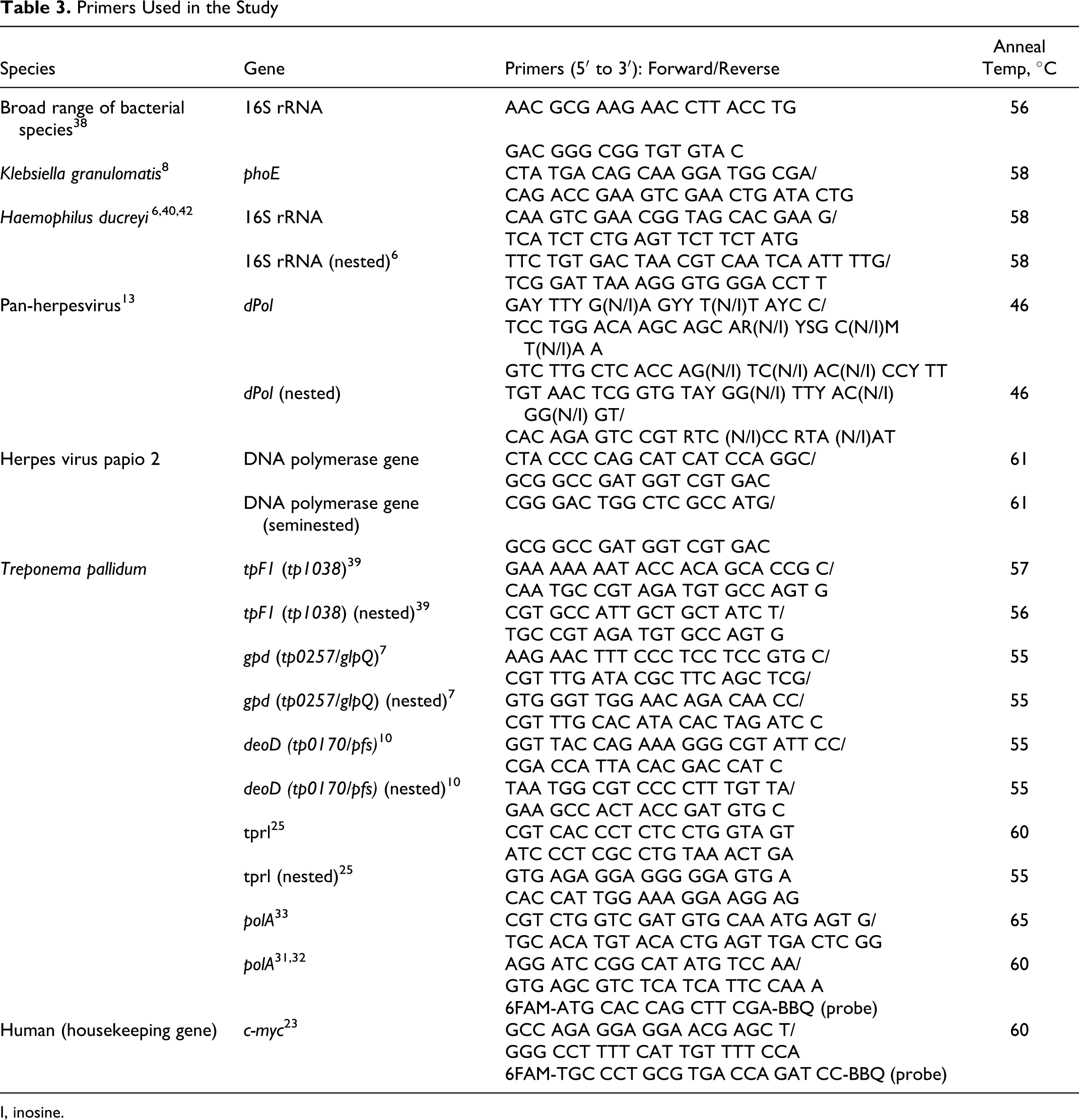
I, inosine.
Products were gel extracted and sent to the sequencing lab of the Robert Koch Institute for sequencing with the PRISM Ready Reaction DiDeoxy Termination Cycle Sequencing Kit (Applied Biosystems) according to the manufacturer’s instructions. Resulting sequences were processed using BLASTN 2.2.18+. 1
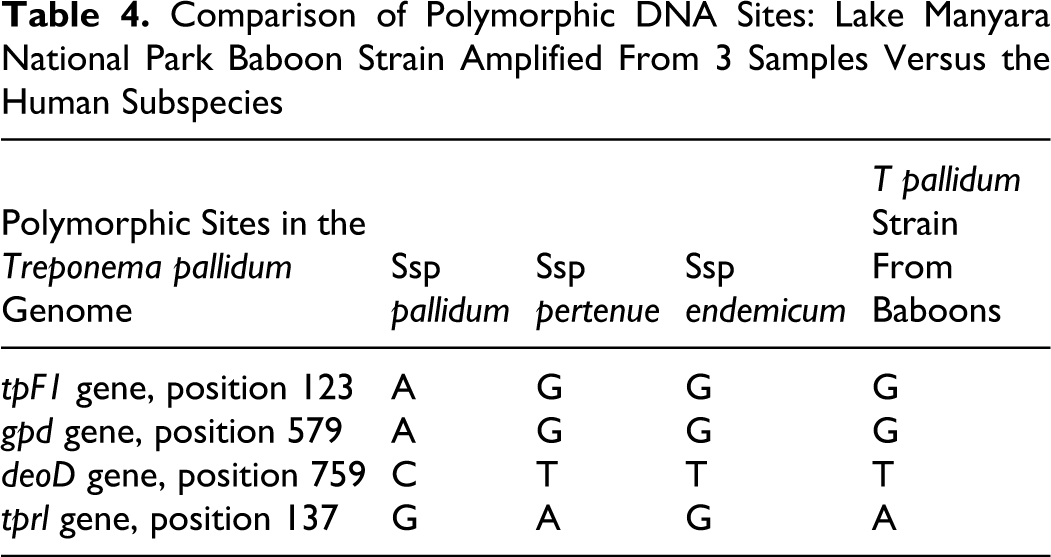
Products of PCRs were also characterized in 4 polymorphic regions (tpF1 [ tp1038], 39 gpd [ tp0257/glpQ], 7 deoD [ tp0170/pfs], 10 and tprl 25 ) previously determined to aid in differentiation between the T pallidum ssp (Table 4 ); sequences of polymorphic regions were deposited in GenBank (accession Nos. FJ896487, EU683034, FJ896486, and FJ896489, respectively).
Comparison of Polymorphic DNA Sites: Lake Manyara National Park Baboon Strain Amplified From 3 Samples Versus the Human Subspecies
Serology
Baboon sera were tested for the presence of antibodies to T pallidum. The qualitative gelatin particle agglutination assay Serodia-TP*PA (Fujirebio Diagnostics, Inc, Malvern, PA) was used in 57 sera (case Nos. 1–57) following the manufacturer’s protocol. Tests included positive and negative controls.
Statistical Analyses
A 2-way frequency table was used to describe and test for the association between variables. Analyses were performed with BMDP Statistical Software (Release 7.0, Statistical Solutions, Saugus, MA). 12 The analysis of the association between clinical infection and T pallidum PCR results made use of 2-tailed Fisher exact test, whereas the association between the clinical infection stage (nonaffected, initial, moderate, and severe) and T pallidum PCR results utilized the exact Wilcoxon–Mann–Whitney U test (Testimate 6.0.65 Software, Statistics.com, Arlington, VA). The statistical significance of the relationship between the clinical infection and histology, as well as the clinical infection and immunohistochemistry results, was assessed with the 2-tailed Fisher exact test. The association between age and clinical infection stage was tested with BMDP Statistical Software using the Spearman rank correlation coefficient. Finally, Kruskal–Wallis one-way analysis of variance and Spearman rank correlation were used to test for the association between the number of T pallidum genome copies per housekeeping c-myc gene (standardization of extraction success) obtained from real-time PCR and different genital ulceration stages. P values were considered statistically significant at .05 or less.
Results
Gross Pathology
Healthy female olive baboons show sexual swelling of the anogenital area that is correlated with hormonal changes in the course of their sexual cycles. 29 In estrus, the sexual turgescence reaches its maximum. The labia vulvae are closed, and the vulva is separated from the anus by a 1- to 2-cm long perineum. In healthy males, the penis is nonpigmented. The glans penis is well demarcated and similar in shape to that of humans. We observed that in clinically affected individuals, the skin of the anogenital region displays a moderate to severe necrotizing dermatitis with severe mutilation of the outer genital structures. In the course of infection, a cloacae may appear in severely affected females (case No. 46; Fig. 1), whereas males tend to lose the glans or even the complete corpus of the penis (case No. 52; Fig. 2). Progressive scarification of the tissue leads to a vagina and anus that are permanently ajar, while in males it promotes phimosis. The age of the baboon was not significantly correlated with the extent of genital ulceration (n = 57, Spearman r S = –.105, P = .44).
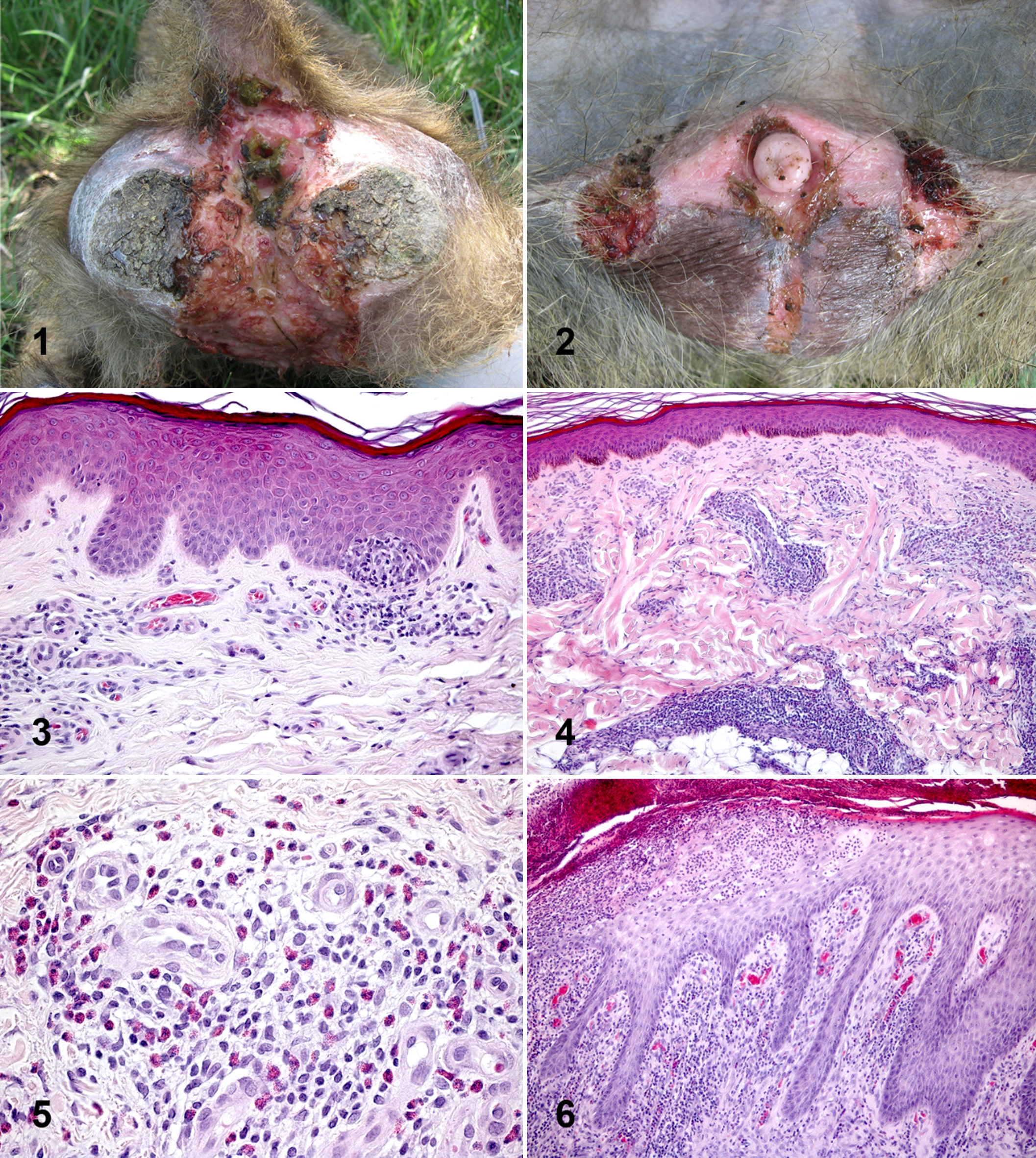
Female Olive baboon, case No. 46, anogenital region. Severely affected female with massive destruction of the outer genitals. Macroscopic view.
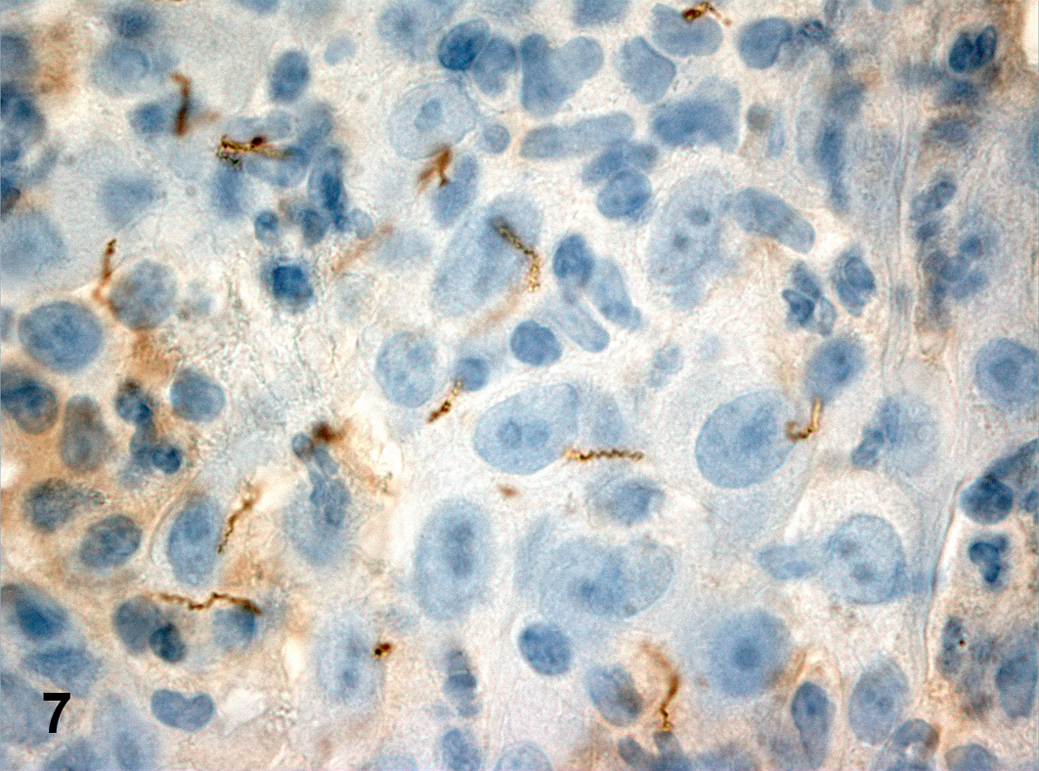
Skin tissue biopsy of the genitals, case No. 39. Immunohistochemical detection of Treponema pallidum. The characteristic spiral-like shape of the spirochete is visible. Rabbit polyclonal antibody against T pallidum, with epithelial cells slightly counterstained with Mayer hematoxylin.
Histopathology
The microscopic findings displayed a uniform manifestation (case Nos. 1–7, 10–13, 15, 17–19, 21–57). Skin lesions were characterized by irregular epidermal proliferation of different extent (Fig. 3). The epidermis developed hyperkeratosis and hypertrophy of the epidermal rete pegs, which branch and project deeply into the corium. The exaggerated elongation of rete ridges was accompanied by acanthosis, acantholysis, and exocytosis. In some cases, small intraepidermal microabscesses were found. In the underlying dermal layer, a moderate to severe mixed-cell infiltration was present (Fig. 4), composed mainly of lymphocytes, histiocytes, and single giant cells. The cellular reaction was most marked around the dermal blood vessels, resulting in superficial and deep perivascular dermatitis. In several cases, a moderate to severe tissue eosinophilia was demonstrable (case No. 50; Fig. 5). In severe cases, the overlaying epidermis was ulcerated (case No. 44; Fig. 6), and a chronic granulomatous reaction ensued, starting from the ulcer margins. The center of the lesion was filled with cell detritus, neutrophil granulocytes, and macrophages. The surface was covered with a dried serosanguineous discharge. No inclusion bodies of type Cowdry A were found. Fungal infections could be excluded by the results of periodic acid–Schiff and Warthin–Starry silver staining. The histochemical silver staining method according to Steiner failed to demonstrate intralesional bacteria. A relatively high percentage of clinically healthy animals showed clear signs of a beginning dermatitis, characterized by mild epidermal hypertrophy and elongation of the rete ridges (case Nos. 1–7, 10–13, 15, 17–19).
Immunohistochemistry
Immunohistochemistry demonstrated small numbers of spirochete-like bacteria in 16 of 63 cases (Table 2; case Nos. 58–63 were negative and are not displayed; case Nos. 64–65 had no skin tissue biopsy). Two animals (case Nos. 2 and 10) had no macroscopic genital lesions but proved to be positive for T pallidum by PCR. The remaining 14 animals (case Nos. 24–25, 30, 37, 39–41, 44, 46, 51–52, 54–55, 57) had signs of genital ulceration. Treponemes were significantly more often detected in clinically affected than in clinically nonaffected baboons (n = 57, 2-tailed Fisher exact test, P = .032). Spirochetes were mainly distributed in the center of the lesion, showing a clear tropism for the epidermis–dermis region (epitheliotropic pattern) and displaying a vasculotropic pattern. They were arranged in small clusters. A single organism was about 6.0 to 15.0 μm in length and 0.15 μm in diameter. A characteristic spiral-like shape with 8 to 15 corkscrew spirals was clearly visible (case No. 39; Fig. 7).
An immunologic differentiation between B cells and T cells was done to characterize the inflammatory cell reaction. In all cases, more T cells than B cells were present among the lymphocytic inflammatory cell population; 10 to 20% of the inflammatory cells were CD20-positive B cells and 70 to 80%, CD3-positive T cells. The T cells were diffusely distributed in the altered dermis, frequently found in close association with the epidermal–dermal junctions, or infiltrating the inflamed epidermis. B cells were found to a lesser extent. They were located in the deeper dermal parts and accumulated around blood vessels or formed follicular aggregates. Follicular aggregates with clear CD20-positive centers and CD3-positive mantel zones were frequently found in deeper dermal parts.
Molecular Biology
Thirty-eight baboons tested positive by PCR 33 and/or real-time PCR31,32 for T pallidum (Table 2). The bacterium was detected by PCR in 6 of 20 asymptomatic animals (case Nos. 2, 10–12, 15, 18), 10 of 14 animals in the initial stage of the disease (case Nos. 21–24, 26, 30–34), 7 of 7 animals with moderate disease (case Nos. 35–41), and 15 of 16 animals in the severe stage of the disease (case Nos. 42–48, 50–57). Treponemes were more likely to be detected in animals with advanced infection stages. Baboons with positive T pallidum PCR results showed clinical signs of infection significantly more often than did those that were PCR negative (n = 57, 2-tailed Fisher exact test, P < .0001). A comparison of the different clinical infection stages with T pallidum PCR results revealed significant differences between the described genital ulceration stages (n = 57, exact Wilcoxon–Mann–Whitney U test, P < .0001), with treponemes detected more often in the advanced infection stages of the disease. Olive baboons with histologic signs of an infection were PCR positive for T pallidum infection significantly more than were those without characteristic histologic alterations (n = 57, 2-tailed Fisher exact test, P = .004).
The number of T pallidum genome copies per housekeeping gene was significantly correlated with the severity of infection (n = 57, Kruskal–Wallis one-way analysis of variance, P = .043). Baboons that showed a moderate clinical infection stage (mean rank sum, 40.21) had the highest number of T pallidum gene copies, followed by severely affected animals (mean rank sum, 32.43) and animals in the clinical initial stage (mean rank sum, 29.75). Nonaffected baboons had the lowest number of T pallidum genome copies (mean rank sum, 21.80).
Particularly noteworthy is that 30% of the olive baboons without genital ulceration (ie, clinically nonaffected) were PCR positive for T pallidum (case Nos. 2, 10–12, 15, 18). Comparison to previously published information regarding polymorphic sites useful in distinguishing the subspecies indicated that all isolates of the LMNP simian strain from 3 individuals were genetically most closely related to the nonvenereal human T pallidum ssp pertenue, which causes yaws (Table 4).
In contrast to the T pallidum findings, molecular biological tests for K granulomatis and H ducreyi were negative. The PCRs targeting herpesvirus were positive in 23 of 26 tested individuals (Table 1): 19 clinically affected (17 of those PCR positive for T pallidum) and 7 clinically nonaffected (2 of those PCR positive for T pallidum). The PCRs revealed a variety of sequences, ranging from only 1 animal infected with cercopithecine herpesvirus 16 (herpesvirus papio 2, case No. 45) to 20 infected with Papio hamadryas lymphocryptovirus 1–3 (case Nos. 1, 7, 9, 10, 12, 21, 24, 26, 30, 35, 38, 40–42, 44, 46, 48, 52, 54, 59) and 2 with Baboon cytomegalovirus (case Nos. 31, 49). Only 3 animals (case Nos. 4, 17, 37) were PCR negative for herpesvirus infection (Table 1). The 16S rRNA generated a range of different bacterial sequences (including those of common skin commensals) but no cluster or specific pathogen associated with genital ulceration. Primer sequences are listed in Table 3.
Serology
Baboons with genital ulceration were positive for antibodies to T pallidum significantly more often than were those without macroscopic visible lesions (n = 57, 2-tailed Fisher exact test, P = .0018). Only 1 of 38 clinically affected animals tested negative in the qualitative gelatin particle agglutination assay (case No. 31). Furthermore, PCR-positive individuals were significantly more often positive for antibodies to T pallidum than were animals with negative PCR results and/or real-time PCR results (n = 57, 2-tailed Fisher exact test, P = .0083). Data are summarized in Table 2.
Discussion
T pallidum is an obligate pathogen that requires inoculation with only 50 organisms to initiate an infection in humans. 35 The demonstration of T pallidum in clinical specimens is a challenging task: This bacterium cannot be grown on artificial media; it cannot be maintained in tissue culture for more than a brief period; and it replicates slowly in animal models. 41 Furthermore, it is highly susceptible to drying 41 and thus requires high-quality sampling. In this article, the presence of T pallidum in the tissue of baboons affected by a genital disease was demonstrated with both immunohistochemistry and advanced molecular biological tests specific to the bacterium, and infection was confirmed using serology.
Although T pallidum has a long-term history of genital ulceration in humans,20,26 treponemal infections in wild nonhuman primates have only once been suspected to cause similar outward signs. 45 In this study, hematoxylin and eosin and Giemsa staining were used to evaluate the presence of tissue alterations typical of treponemal infection. The main characteristic findings were irregular epidermal proliferation with acanthosis and acantholysis, epidermal ulceration, and perivascular dermatitis with mixed-cell infiltration and tissue eosinophilia. All these observations can be found in humans infected with T pallidum ssp pallidum.15,36
Syphilis and yaws infection can be partitioned into 3 stages: primary, secondary, and tertiary.2,9,15,36,41,46 Currently, we are not able to categorize the different grades of infection observed in the olive baboons at LMNP into the defined stages established in humans. It would be too hypothetic to assume that within our sample size of baboons, all potential stages of the infection were represented. However, at LMNP, the clinical manifestation appears strictly limited to the anogenital region; clinically affected baboons are significantly more often positive for T pallidum; and no alterations outside this area were seen (eg, rash in different parts of the body, soft raised condyloma lata, or deformation of the bones). Maybe the simian strains described here lack the ability to disseminate or cause clinical manifestations elsewhere in the body, even though in the context of what is known from human treponemal infection, this seems unlikely; alternatively, perhaps baboons respond to T pallidum infection differently than do humans. It is also open to question whether the baboon’s life span allows the disease to progress to a level comparable to the tertiary stage in humans or African apes. 34 Future studies should include the utilization of PCR on blood samples and immunohistochemistry of lymph node tissue. Sampling procedures and protocols necessarily need to be adjusted for proper preservation of T pallidum. The results should contribute to the understanding of disease transmission and the course of the disease.
In this study, PCR proved the most sensitive means of detecting T pallidum. The most reliable and sensitive T pallidum–specific PCR assay demonstrated that 86% of clinically affected baboons (with genital lesions; 32 of 37) and 30% of clinically nonaffected baboons (without genital lesions; 6 of 20) were positive for the bacterium. Spirochetes were demonstrated immunohistochemically in 39% of PCR-positive animals (15 of 38) and 5% of PCR-negative animals (1 of 19). It is possible that the relatively low positive rate achieved using rabbit polyclonal antibodies against T pallidum could be caused by antigenic differences between the baboon strains and those used to develop commercially available tests; because simian strains have been little studied thus far, the sensitivity and specificity of the tests available have not been validated for them. However, since many of the most immunogenic antigens are invariant among the human subspecies as well as the baboon-derived Fribourg-Blanc strain, it seems highly unlikely that polyclonal anti– T pallidum antibodies would fail to react with the treponemes of the LMNP simian strains. Finally, in the past, silver staining was most commonly used for the detection of Treponema sp in histologic specimens. Since detection rates are extremely variable but in general low, ranging anywhere from 0%5,46 to 33%, 27 41%, 44 and 50%, 36 it is not surprising that in our case, Warthin–Starry silver staining failed to detect the spirochetes. Given the very low sensitivity and specificity of Warthin–Starry silver staining, some authors have made use of Dieterle staining (Sigma-Aldrich Chemie, Germany) with better results. 5 However, at least in Germany, Dieterle staining has been taken off the market and is therefore no longer available.
In both human yaws and syphilis, spirochete numbers can change significantly over the course of infection.15,36,46 This is also true of the baboons at LMNP, in which the quantity of treponemes in the skin tissue biopsy differed significantly between clinical infection stages. Fluctuation in the number of Treponema, as paired with clear macroscopic and microscopic signs of chronic infection in the affected baboons (granulomatous tissue alterations, plasma and giant cell infiltration, collagen fiber production), might explain the relatively low number of spirochetes found in some of the tissue biopsies.
RNA-Later, as a nucleic acid fixative especially designed for RNA protection, can also be used to conserve DNA samples (protocol is available from the manufacturer). Under field conditions, it is often necessary to minimize sample size while enhancing sample usability. RNA-Later allows the protection of RNA and DNA in a single sample, which was proved by adequate yields of DNA from the tissue extracts used for PCR screening.
The combination of laboratory results with field observation data allows maximal diagnostic interpretation. Animals that are without clinical and histologic signs of infection and that are negative for treponemes in immunohistochemistry, PCR and serology can be diagnosed as T pallidum noninfected (n = 4, case Nos. 8–9, 14, 16, 20). In contrast, animals with genital ulceration, histologic findings that indicate an infection, and positive immunohistochemistry, PCR, as well as serology results can be classified as T pallidum positive (n = 13, case Nos. 24, 30, 37, 39–41, 44, 46, 51–52, 54–55, 57). Baboons, which are positive according to all methods except for immunohistochemistry, can also be classified as T pallidum positive (n = 18, case Nos. 21–23, 26, 32–36, 38, 42–43, 45, 47–48, 50, 53, 56). As previously discussed, the sensitivity of immunohistochemistry seems low, and false-negative animals are likely. In the case of animals without clinical signs of infection but with characteristic histologic findings as well as a positive PCR result for T pallidum (n = 6, case Nos. 2, 10–12, 15, 18), it is plausible that the infection is in its prodromal stage or, more likely, the animals are in a latent stage of infection, based on the natural history of treponemal infections.
Olive baboons with or without clinical signs of infection but positive histology and negative PCR results and immunohistochemistry for T pallidum (n = 13, case Nos. 1, 3–7, 13, 17, 19, 27–29, 49) need to be interpreted carefully. In these cases, serology can be used to enlighten the true infection stage. The detection of antibodies specific to T pallidum indicate that an adult baboon was in contact with the pathogen and either recovered or still hosts an infection. In the latter case, it is possible that sample quality may have deteriorated during storage in the field, making it impossible to detect the spirochete. The conservation of samples in formalin is much less defective under field conditions than it is for cryoconservation, which unfortunately limits the sample usage for advanced molecular biological screening methods. Another plausible scenario that explains PCR-negative cases with active infection is that of sampling error: The biopsy taken for histology may have included clusters of treponemes in the skin, while the biopsy for PCR failed to incorporate clusters of spirochetes. We encountered a single case in which a female showed clinical and histologic signs of infection with positive serology results and treponemes visualized by immunohistochemistry (n = 1, case No. 25) but was PCR negative. For the reasons discussed above, this animal should be considered T pallidum positive.
A negative serology can help rule out the rare cases in which a local trauma with secondary infection might mimic a T pallidum–associated genital ulceration. Last, serology is an important tool with which to rule out infection in animals without clinical, histologic, or molecular evidence of T pallidum. However, it is possible that infected animals fail to seroconvert. Therefore, parallel methods of testing are of invariable value.
Three subspecies of T pallidum are recognized on the basis of clinical characteristics of the diseases they cause in humans: yaws (ssp pertenue), bejel (ssp endemicum, also known as endemic syphilis), and syphilis (ssp pallidum). T carateum causes pinta and is currently recognized as another species. Although the clinical manifestations of these diseases vary widely, the different subspecies of the spirochete T pallidum are so closely related that only in recent years has genetic differentiation between them become possible.10,11,22,24 The nonsexually transmitted subspecies (pertenue and endemicum) are recognized as diseases of early childhood,2,28 while ssp pallidum causes syphilis in humans and is the only known subspecies of the T pallidum complex adapted to a venereal transmission mode. In this context, it is interesting that the LMNP simian strains are genetically most closely related to nonvenereal human T pallidum strains (ssp pertenue), although the clinical manifestation in baboons differs from what would be expected on the basis of genetic similarity. Although Mlengeya found only sexually active baboons to be clinically affected, 37 the transmission mode is not yet clear, and the age distribution of clinically affected baboons observed in the field, compared to the different stages of genital ulceration, was not statistically significant in this study. The genetic results, but not the description of pathology, are consistent with analysis of a T pallidum strain obtained decades ago from a baboon in Guinea. 25
Our results demonstrate that herpesviruses do not play a causal role in the infection present in baboons at LMNP. Although herpesvirus papio 2 had been considered the causative agent for the genital disease in olive baboons at LMNP,21,37 in our study it was only found in one individual. Thus, there was no correlation between herpesvirus papio 2 prevalence and clinical manifestations, therefore making it unlikely that herpesviruses play a role in the pathology of this disease. The high number of baboons positive for lymphocryptovirus 1–3 is not surprising, as the prevalence of latent lymphocryptovirus infections in wild nonhuman primates is known to be high. 14 Lymphocryptoviruses in nonhuman primates cause symptoms similar to Epstein-Barr virus infections in humans and have never been documented in association with genital ulceration. Furthermore, lymphocryptoviruses were found to be equally prevalent in infected versus uninfected animals at LMNP.
In conclusion, we present a detailed description of histologic findings in wild baboons consistent with syphilis-like clinical signs, which are associated with the consistent finding of a strain of T pallidum most closely related to the human strains of T pallidum ssp pertenue. Epidemiologically, it is interesting that the prevalence of the disease (based on histologic findings) is much higher than expected from macroscopic observations in the field (the range in different baboon troops at LMNP has been estimated at 7.1 to 29.1% 37 ). The histologic findings (Langhans and giant cells, granulomatous tissue, irregular epidermal proliferation and scarification) indicate that the disease is chronic in many animals. Further studies are needed to describe the pathogenesis of this disease in baboons, from inoculation to the appearance of the first clinical signs, until recovery or death. According to what is known from human inoculation experiments with the Fribourg-Blanc simian strain, there is a potential for primate-to-human transmission, 43 underscoring the necessity for additional research to gain insight into the disease’s transmission dynamics, as well as its temporal and spatial distribution.
Footnotes
Acknowledgment
This study was carried out in accordance with the Tanzania Wildlife Research Institute’s Guidelines for Conducting Wildlife Research (2001) and with permission of Tanazania National Parks (TNP/HQ/E.20/08B). Additional research permission was granted by the Commission for Science and Technology in Tanzania (2007-56-NA-2006-176).
For support during the study, we thank the Tanzania Wildlife Research Institute, especially J. Keyyu; Tanazania National Parks; Lake Manyara National Park headquarter staff, especially Chief Park Warden M. Mtahiko; as well as the Tanzania Commission for Science and Technology. For assistance, we thank E. Kiwele, K. Mollel, D. M. Kambarage, R. A. Muhala, P. Wambura, A. Lawrenz, S. Tacke, Tierpark Berlin, G. Armelagos, and B. Steiner. S. Bruisten and A. Weber are thanked for providing positive controls and N. Köchelmann and K. Kaiser-Jarry as well as W. Henkel for technical support in the lab. S. Frischmann is thanked for his generous help and support in terms of serology.
This work was supported by the German Academic Exchange Program (D/06/43974), the Christian Vogel Fond (2006), the World Association of Zoos and Aquariums (07002), the University of Leipzig, the Justus Liebig University of Giessen, Scil animal care company, Telinject Inc, Translogistic Inc, Wuppertal Zoo, and the Robert Wood Johnson Foundation Health and Society Scholars Program. Analyses were supported by the German Primate Centre and Robert Koch Institute; Serodia TP*PA tests were sponsored by Mast Diagnostica GmbH.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
