Abstract
An 8-year-old male Nubian goat was presented with a peracute history of straining to urinate and unilateral mandibular swelling. At necropsy, the rostral half body of the left mandible was distorted by fusiform swelling that, on sagittal section, had marked medullary bone loss and replacement with a well-demarcated fibrous mass. Histologically, the mass comprised streaming spindloid cells with whorls and storiform patterns, interspersed with numerous multinucleated giant cells. Bone formation was not present in the neoplasm. The mandibular mass was diagnosed as nonossifying fibroma, a relatively common tumor in children but seldom reported in domestic animals.
Nonossifying fibroma (NOF) has been infrequently reported in animals 5 but is a common benign neoplasm in children. 16 In human patients, NOF is often diagnosed radiographically, and it is differentiated from a radiographically similar lesion—namely, fibrous cortical defect (metaphyseal fibrous defect)—by its larger size, elongated shape (parallel to the long axis of the bone), and medullary involvement. 7 Three cases of NOF have been reported in young Thoroughbred horses: in the proximal tibiae of a lame yearling filly, 5 in the first phalanx of a lame 8-month-old gelding, 2 and in the mandible of an 8-month-old-filly. 12 Cases of NOF have also been reported in the femur of a 6-year-old-cat and the rib of a sheep. 4 In domestic animals, NOF is histologically characterized by sheets, whorls, and interlacing bundles of fibroblast-like spindle cells with numerous thin-walled blood vessels and varying numbers of multinucleated giant cells.2,4,5 Osteoblastic differentiation or bone production by the spindle cells has not been observed. 5
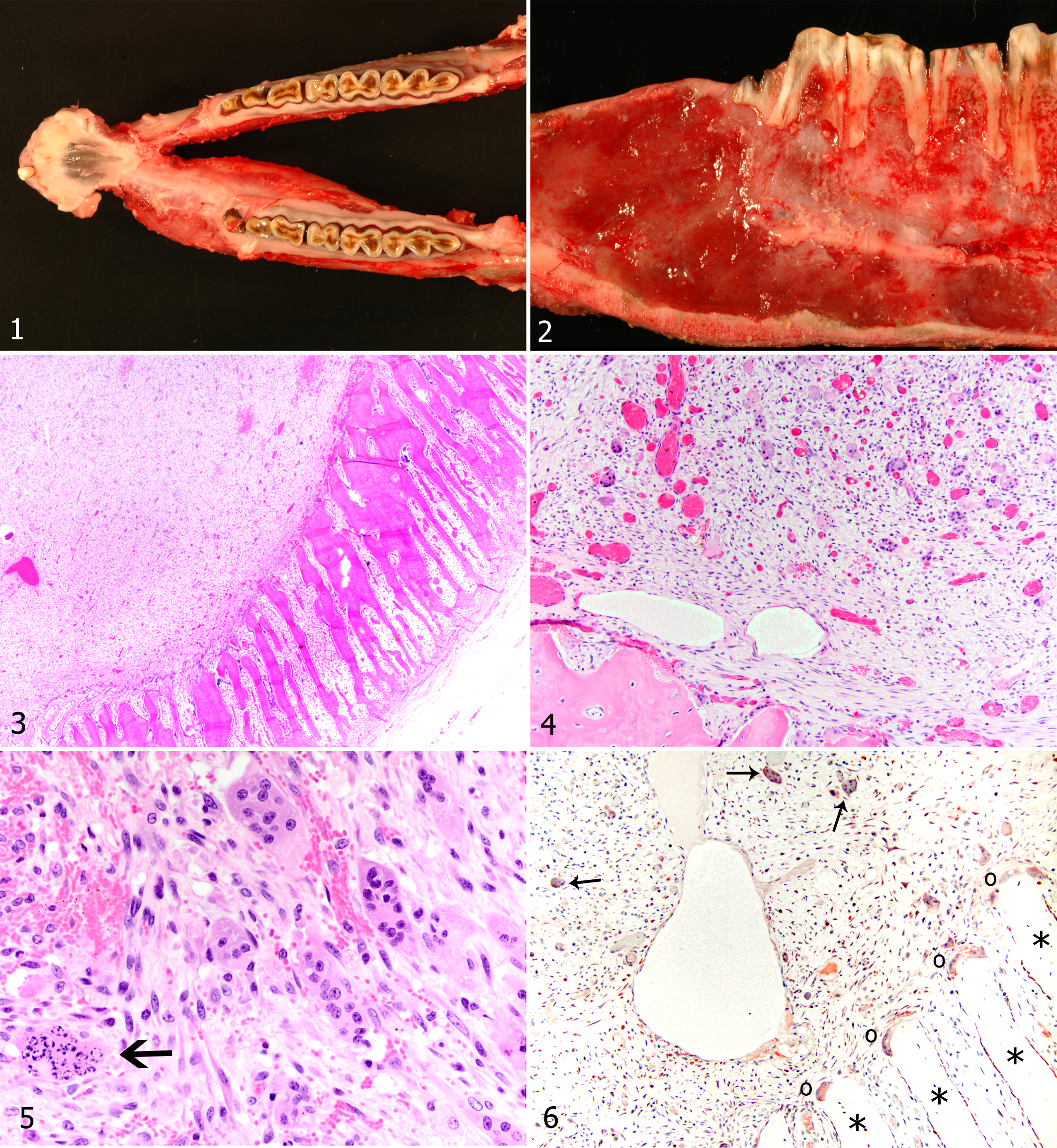
An 8-year-old male Nubian goat in good nutritional condition was presented to the University of California Veterinary Teaching Hospital with a peracute history of straining to urinate. Approximately 1 month before presentation, the owner noted firm swelling of the left mandible that had become progressively larger. There was no history of mandibular pain or dysphagia. The goat was euthanized and a complete necropsy performed. Mandibular incisor teeth were absent except for the left first incisor, which was mobile and surrounded by a sulcus 0.3 cm wide and 0.6 cm deep. The loss of incisor teeth was attributed to dental attrition due to age. The left mandible had fusiform swelling, 9 cm in length and 3 cm in diameter, that extended from just caudal to the mandibular symphysis to the molar region (Fig. 1). The mandibular cortical bone was distorted and hard. On sagittal section, a well-demarcated, soft, glistening, and pale fibrous mass expanded the mandibular medullary cavity (Fig. 2). The alveolar bone subjacent to the most rostral premolar root was largely absent, although alveolar bone sparing was evident around the caudal cheek teeth.
Sections of bone were decalcified with 15% formic acid for 5 days before histologic processing and embedding in paraffin. Immunohistochemistry was performed on 4-μm serial sections using a routine streptavidin–biotin–horseradish peroxidase detection system 10 with the following modifications. Individual slides were pretreated with Protease K (S3202, Dako Corp, Carpinteria, CA) for 6 minutes at room temperature before blocking with 10% normal horse serum. Primary antibody, rabbit polyclonal anti–human lysozyme (1:1000, A0099, Dako Corp), was applied for 60 minutes. Immunoreactivity was visualized using AEC (3-amino-9-ethylcarbazole, Invitrogen, San Francisco, CA) as the chromogen, with Mayer’s hematoxylin as counterstain (Sigma Chemical Co, St Louis, MO). Negative controls were prepared by replacing the primary antibody with pooled rabbit immunoglobulin G (Invitrogen) at the same dilution as the primary antibody.
Histologically, the mandibular cortex was replaced by parallel interconnected plates of lamellar and woven bone, oriented perpendicular to the former cortex (peripheral reactive bone formation; Fig. 3). Multinucleated osteoclasts were clustered at the interface between neoplasm and bony tissue. The mass was composed of numerous individualized multinucleated giant cells admixed with a variably dense population of mononuclear spindle cells that formed streams, whorls, and storiform patterns in a loose fibrillar fibrovascular matrix (Fig. 4). Multinucleated giant cells within the mass were more numerous than the osteoclasts clustered at the neoplasm–bone interface. The multinucleated giant cells were 15 to 50 μm in diameter with distinct cell borders, scant to abundant amphophilic cytoplasm, and 6 to 20 central oval nuclei. Mitotic figures were not observed. Many multinucleated giant cells had pyknosis, nuclear fragmentation, and increased cytoplasmic eosinophilia (Fig. 5). The multinucleated giant cells within the mass were faintly labeled by the antilysozyme antibody, as were the osteoclasts in adjacent cortical bone (Fig. 6). In a caprine splenic control tissue, macrophages had strong expression of lysozyme. The neoplastic spindle cells had scant to moderate amount of pale amphophilic cytoplasm with indistinct cell borders (Fig. 5). Mitotic figures were fewer than 1 per 10 high-power fields. Multiple small foci of hemorrhage and numerous dilated, empty, thin-walled vessels were in the tumor. Neither hemosiderin nor hematoidin was identified, suggesting that the hemorrhage was acute. Within the mass, there was diffuse loss of cortical and trabecular bone with replacement by neoplastic tissue. Neoplastic cells had eroded through the cementum and penetrated the dentin of the first premolar root.
The histologic features of NOF are whorled-to-storiform fibrous stroma, spindle-shaped fibroblasts, multinucleated giant cells, and lack of bone production. 11 Bone formation is absent in NOFs, except for peripheral reactive bone formation, 3 as seen in this case. Foam cells may be present in human NOFs. 17 We regard the absence of foam cells in this caprine NOF as a species difference because, in our experience, foam cells that are present in human tumors or other disorders are often absent in the analogous bone lesions of animals.
Several alternative diagnoses were considered in this case, including fibrosarcoma, intraosseous myofibroma of the mandible, desmoplastic fibroma of bone, fibrous dysplasia, ossifying fibroma, giant cell reparative granuloma, and giant cell tumor of bone. The neoplasm lacked the cellular density and anaplastic features of a fibrosarcoma; furthermore, the numerous multinucleated giant cells present in this neoplasm are not typical of fibrosarcoma. The neoplasm in this goat lacked the relatively low cellular density and histologic features of an intraosseous myofibroma of the mandible, a rare human spindle cell tumor composed of interlacing bundles of spindle cells that express smooth muscle markers and that are believed to originate from myofibroblasts. 1 Intraosseous myofibromas are mainly soft tissue tumors and are not associated with giant cell formation. Desmoplastic fibroma of bone would be expected to be more cellular at the periphery of the expanding lesion and to demonstrate a dense, collagenous, nearly acellular desmoplastic central region. 8 The neoplasm in this goat lacked the cellular density and bone spicule formation characteristic of fibrous dysplasia and ossifying fibroma. 15 In fibrous dysplasia, spicules of poorly formed, woven bone arise within thin, linear condensations of fibrous tissue without transition to recognizable osteoblasts. 15 In ossifying fibroma, spindle cells that resemble fibroblasts undergo transformation to osteoblasts at the margins of the developing bone spicules. 15 In addition, neither ossifying fibroma nor fibrous dysplasia would have the numbers of multinucleated giant cells present in the neoplasm of this goat. Finally, this lesion lacks the density of giant cells to be a giant cell reaction (synonym: giant cell reparative granuloma) or giant cell tumor of bone. Additionally, intralesional bone formation, which was not present in the neoplasm described here, is common in giant cell reparative granulomas.3,14
The precise histogenesis of the intralesional multinucleated giant cells was not determined. However, the tumor-associated multinucleated giant cells were weakly positive for lysozyme expression, as were adjacent bone-associated osteoclasts. The weakness of the lysozyme immunoreactivity in the NOF compared to splenic macrophages in the control tissue sections was presumed to be the result of the decalcification procedure. In 54 human lesions containing giant cells (giant cell tumor of bone, fibrous histiocytoma, NOF, and giant cell tumor of the tendon sheath), cells with immunoreactivity for lysozyme were rarely found, except in giant cell tumors of the tendon sheath. 6
Human NOFs do not generally cause pain, and many are diagnosed according to localized swelling or after pathologic fracture. 5 Some researchers suspect that benign neoplastic activity results in the transformation of fibrous cortical defect lesions to NOF. 4 NOF has a characteristic radiographic appearance of eccentric, well-defined, radiolucent, circular, or oval lesions with smooth lobulated edges and surrounding sclerotic bone. Cortical expansion and thinning may be present.3,7 NOF often appears in children at 4 to 8 years of age and is typically in the metaphyseal region of developing long bones, such as the femur.7,16 Human NOF is generally a self-limiting lesion with spontaneous regression over the course of several years.3,4,16 However, pathologic fracture is a risk once the NOF occupies more than 50% of the diameter of the bone.13,16 Spontaneous regression in domestic animals has not been reported; however, no clinical or radiographic evidence of residual neoplasia was detected in 1 horse after surgical excision of a NOF. 2 A cytogenetic abnormality (reciprocal chromosomal translocation) was associated with a NOF in the tibia of an 18-year-old skeletally mature woman. 9 Although NOF is most often identified in the long bones, the tumor has been identified in the mandible of 11 human patients 3 and 1 horse. 12 The relevance of this anatomic distribution is unknown. This caprine NOF resulted in mandibular distortion as well as resorption of cortical and trabecular bone. Interestingly, NOF appears to be a tumor of young people and horses, whereas the goat of this report was 8 years old. It is possible that this caprine neoplasm was present in the mandible for years before detection.
Footnotes
Acknowledgment
We thank Dr Kirsten Murphy for draft editing and Barry Puget, Diane Naydan, and the University of California–Davis Histopathology Laboratory for their technical expertise.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
