Abstract
Enterococcus hirae infections are reported to cause growth depression, encephalomalacia, endocarditis, and septicemia in chickens. This report describes osteomyelitis in the proximal femur of a 3-week-old broiler chicken that also suffered from valvular endocarditis and liver necrosis. Histologically, clusters of gram-positive coccoid bacteria were found in many organs, including bone lesions. In tissues from 5 of 6 examined chickens from the same flock, E hirae was isolated in large numbers. To the authors' knowledge, this is the first report of spontaneous bacterial osteomyelitis where E hirae was cultured from bone and where coccoid bacteria consistent with Enterococcus spp were simultaneously demonstrated within bone lesions.
Bacterial infections are the cause of a great number of losses in broiler chicken production. Enterococcus hirae is a bacterial species that is isolated relatively often, but its significance is less understood than the more well-known Escherichia coli and Staphylococcus aureus infections. Infections with E hirae have been a relatively common finding in Norway, since first being observed a decade ago. In 2009, there has been an increase in the number of infections caused by this bacterium.
The genus Enterococcus comprises gram-positive coccoid bacteria belonging to the family Streptococcacea. Enterococci are considered part of the normal intestinal flora and are opportunistic pathogens in birds and mammals.
E hirae was first described in 1985 based on strains originally named Enterococcus faecium, which caused growth depression in chickens.8,9 E hirae is found to be common in the intestinal flora of several domestic animals,3,4,6 although in chickens the bacteria are usually found in the small intestine only in 3- to 4-week-old birds. 5 E hirae infections are reported to cause encephalomalacia, septicemia, and endocarditis in chickens. 15
This report describes bacterial osteomyelitis in a proximal femur of a 3-week-old broiler chicken in Norway, where E hirae was cultured from bone and where gram-positive coccoid bacteria consistent with Enterococcus spp were demonstrated intralesionally.
Clinical History
Eight 3-week-old commercial broiler chickens from a flock of 9,900 animals were submitted for examination to the Section of Pathology, National Veterinary Institute, Oslo, Norway. The death rate in the flock had increased after 2 weeks of age, from an accumulated mortality of 1.75% over the first 2 weeks of age to 1.9% during the third week and 1.3% during the fourth week. The chickens were slaughtered at 30 days of age—the average slaughter age in Norway. Case Nos. 1 to 3 were found dead, whereas case Nos. 4 to 8 were euthanized by a blow to the head followed by decapitation. Case Nos. 4 to 6 were found lying on their backs, whereas case Nos. 7 and 8 exhibited signs of lameness.
Gross Pathology
At necropsy, the broilers were in good condition; however, their weights ranged from 565 g to 1085 g. The lungs were congested and edematous, and splenomegaly was present in all individuals. In 5 chickens, vegetative valvular lesions were found on the right atrioventricular valves. The lesions were yellow-white with a cauliflower appearance. Both femoral heads were separated from the shaft when the coxofemoral joints were disarticulated in No. 7, and a fracture of the right femoral neck surrounded by hemorrhage was present in No. 8. There were no further macroscopical lesions on the sagittally cut surface of any bones, and no other joint lesions were observed. Table 1 summarizes the macroscopic lesions of the 8 chickens.
Macroscopic Lesions
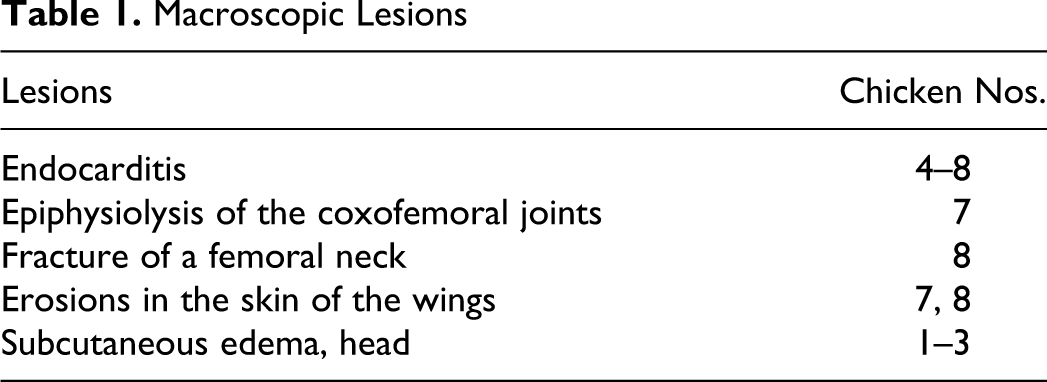
Histopathology
Tissue samples were fixed in 10% formalin for 24 hours; then, the femur was decalcified in 10% formic acid for 24 hours. The decalcified bone from case No. 7 was routinely processed, as were the heart, lung, liver, and kidney from all chickens except case Nos. 3 and 6. Tissue sections were stained with hematoxylin and eosin for histopathologic evaluation and with Gram stain to investigate bacterial presence.
Microscopically, epiphyseal clefts with remnants of blood and cellular debris were present in the proximal femur of chicken No. 7. The vessels in the growth plate were variably filled with blood and thrombi. There were no pale or eosinophilic areas around the vessels demonstrating degenerative or necrotic cells; however, the chondrocytes of the epiphyseal plate were disorganized. In the metaphysis, there were accumulations of an exudate that contained nuclear debris—possibly, degenerating heterophils (Fig. 1). Clusters of gram-positive coccoid bacteria were seen within the inflammatory exudate and within foci of necrosis in some metaphyseal trabeculae (Fig. 2). The synovium of the coxofemoral joint exhibited small villous formations with few inflammatory cells.
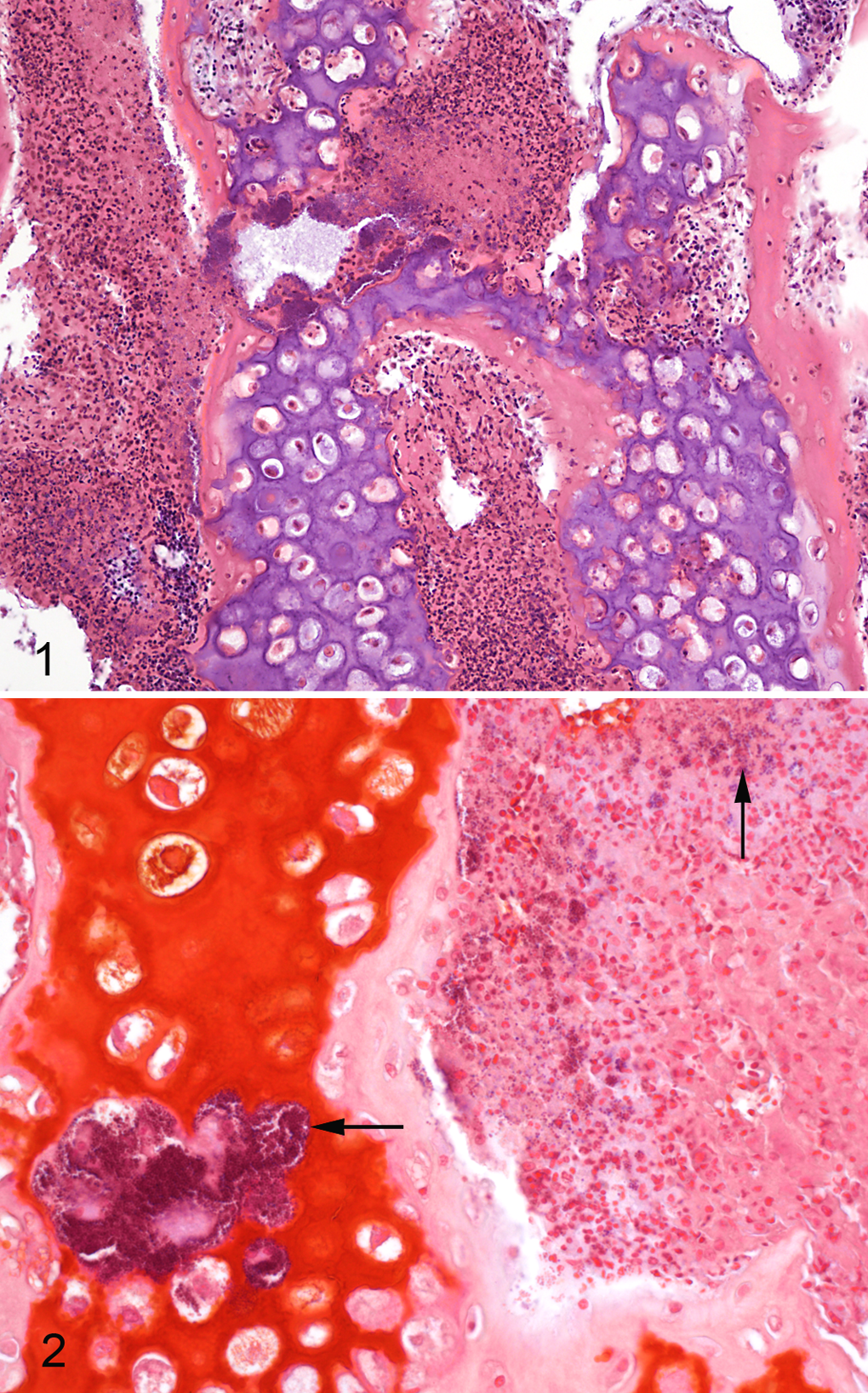
The valvular lesions (case Nos. 4–8) consisted of large numbers of bacteria, as well as a chronic-active inflammatory response extending into the myocardium, where a multifocal inflammation was noted, as dominated by mononuclear cells. Congestion and edema were present in the lungs of all cases. There was congestion in the liver tissues, and in case Nos. 1 and 7 multifocal areas of necrosis and inflammatory cells were seen. Gram-positive coccoid bacteria were present in the histologic sections of myocardium, lung, liver, and kidney in 4 of 5 examined chickens (case Nos. 1, 4, 7, 8), including bone of No. 7.
Bacteriology
Upon bacteriological examination, E hirae was isolated in large numbers from 4 of 6 livers (case Nos. 1, 6, 7, 8), 2 of 4 lungs (case Nos. 4 and 6), and 3 of 3 valvular endocardial lesions (case Nos. 4, 6, 7). Culture of bones from the 2 animals exhibiting lameness (case Nos. 7 and 8) resulted in the isolation of E hirae in large numbers.
The initial identification of E hirae was made on the basis of the following traits: The isolate produced α-hemolytic colonies on blood agar and were negative for catalase and oxydase. The bacteria were gram-positive and coccoid in shape, and they reacted with Lancefield group D antisera. Biochemical reactions showed acid production from sucrose but not from mannitol or glycerol, and the strain was arginine dehydrolase positive and hippurate negative.
For positive identification, 16S rRNA sequencing and polymerase chain reaction using E hirae–specific primers were performed. 10 The isolates were sensitive to ampicillin and tetracycline, the two antibiotics approved for use in poultry in Norway. It was concluded that E hirae was the primary cause of the septicemia, endocarditis, and the osteomyelitis.
Discussion
Bacterial osteomyelitis is a common cause of lameness in broiler chickens and was first reported by Nairn and Watson in 1972. 14 The causative organism most frequently cultured is coagulase-positive S aureus; however, E coli may also produce similar bone and joint lesions. 13 Concomitant synovitis, arthritis, and chondronecrosis are often present. When chondronecrosis is present, the condition is called bacterial chondronecrosis with osteomyelitis. 12
Osteomyelitis may be recognized on gross examination by sagittal cleavage, and the gross lesions caused by S aureus are described as focal yellow areas of caseous exudate or lytic areas, making the bone thin and fragile. No such macroscopic lesions were seen by sagittal sawing of the femoral bones. However, macroscopic lesions may be present in only 40% to 67% of cases,7,11 and many osteomyelitic lesions are detectable only by histologic examination, 15 as in this case.
The histologic findings in the proximal femur correspond to findings in staphylococcal and E coli osteomyelitis; however, chondronecrosis was not seen. The reason for this could be that (1) E hirae does not affect chondrocytes in the growth plate in the same way as S aureus or E coli, (2) the chicken was killed at a time before E hirae affected chondroid tissue, or (3) lesions present in tissue were not observed because of their placement in relation to the sections examined. Clusters of gram-positive coccoid bacteria were found localized in inflammatory exudates in the metaphysis of the proximal femur of chicken No. 7.
In many reports, bacteria are identified in histologic bone lesions in poultry. However, bacteria other than E hirae have been cultured from the confirmed histologic lesions.1,2 E hirae has been isolated from bone marrow and hock joint in natural and experimental studies, although to our knowledge, no bone lesions have been described. 16 Reasons for this may be that (1) bone sections were not studied macroscopically or microscopically or (2) histologic examination was not performed owing to a lack of macroscopic lesions and microscopic lesions were thus not observed.
Enterococcus spp were reported in a review article by McNamee 13 as being involved in “femoral head necrosis” in chickens 9 ; however, no such lesions were proven in our case. The localization of the osteomyelitis in the proximal femur was compatible with the studies of Nairn 14 and McNamee, 12 who demonstrated that the proximal end of the femur was most commonly affected in naturally occurring bacterial chondronecrosis with osteomyelitis in poultry, followed by proximal tibiotarsus. The osteomyelitis in the proximal end of the femur of No. 7 and the femur fracture in No. 8 may explain the erosions on the wings, as the chickens used the wing tips for support under locomotion. The epiphysiolysis in No. 7 may be caused by the bacterial osteomyelitis. 1
Naturally occurring endocarditis in chickens caused by E hirae has been reported, and in this context, the bacteria was isolated from different tissues, including bone marrow. However, no histologic description of bone was mentioned. This case illustrates that the absence of macroscopic bone lesions in lame chickens should encourage histopathologic and bacteriologic examination of the proximal part of both femur and tibiotarsus to confirm a diagnosis and prevent underdiagnosis, as described by Emslie et al 7 and McNamee et al. 11 Osteomyelitis was histologically identified in the proximal femur of a lame chicken, and detection of gram-positive coccoid bacteria—as consistent with Enterococcus spp within the bone lesions and the pure culture of E hirae from the bone—are reported for the first time, suggesting that E hirae was the cause of the osteomyelitic lesions.
As such, E hirae should be included in the differential diagnosis of bacterial osteomyelitis in broiler chickens.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
