Abstract
Tumor cell invasion into the surrounding nervous tissue is one of the histologic hallmarks of anaplastic meningiomas. To identify other possible markers for aggression in canine meningiomas, the relationship between histologic features and the expression of molecules involved in cell adhesion, cell proliferation, and invasion was examined. Immunohistochemistry for epithelial cadherin (E-cadherin), neural cadherin (N-cadherin), β-catenin, doublecortin (DCX), and Ki-67 was performed for 55 cases of canine meningioma. DCX was preferentially expressed in tumor cells invading the brain parenchyma (12 of 14 cases), suggesting its involvement in the invasion process. Regardless of the histologic type, E-cadherin and N-cadherin expression was observed in 31 of 55 and 44 of 55 cases, respectively. There was a significant positive correlation between DCX and N-cadherin expression and a significant negative correlation between E-cadherin and N-cadherin expression, suggesting that decreased E-cadherin and increased N-cadherin expression induce DCX expression. Typical membranous β-catenin expression was observed in 10 of 55 cases, whereas nuclear translocation was observed in 33 cases. Nuclear β-catenin expression was frequently found in anaplastic meningiomas (12 of 14 cases). The Ki-67 labeling indices were significantly higher in anaplastic meningiomas than in other types. These findings indicate that the expression of N-cadherin and DCX and the nuclear translocation of β-catenin are closely associated with the presence of invasion and anaplasia in canine meningiomas. Notably, granular cell meningiomas were negative for almost all the molecules examined, suggesting that they have a different tumor biology than other meningiomas.
Meningiomas originate from the meningothelial cells covering the central nervous system 10 and constitute about 40% of all primary intracranial tumors in dogs. 4,22 Canine meningiomas are histologically diverse and can undergo mesenchymal and epithelial differentiation, similar to their human counterparts. 17,23 The World Health Organization (WHO) classification for brain tumors of domestic animals includes 9 histologic types of canine meningiomas: meningothelial, fibrous (fibroblastic), transitional, psammomatous, angiomatous, papillary, granular cell, myxoid, and anaplastic (malignant). 10 All except the anaplastic type are slow growing and have a relatively benign progression. In domestic animals, the assessment of meningioma malignancy is still based on morphologic criteria, such as a high mitotic rate and cellularity, patternless growth, and invasion into the surrounding nervous tissue. 10,14 In human meningiomas, tumor cell invasion into the brain parenchyma is the most crucial, 18,24 although this is not a factor in the WHO criteria. 28 Few indicators for malignancy are available for canine meningiomas, 14 and further studies are necessary to identify molecules that may correlate with tumor invasion.
Doublecortin (DCX), which plays a crucial role in neuroblast migration during the development of the cerebral cortex, is highly expressed in invasive brain tumors, including malignant meningiomas in people. 3 The expression of DCX protein is stronger at the margin of a tumor lesion than at the center, 3 suggesting that DCX would be a good marker for invasion. In human meningiomas, Ki-67 is a standard marker of cell proliferation activity and is widely used to evaluate tumor grade and malignancy during pathologic diagnosis. 18 Epithelial cadherin (E-cadherin) is expressed in normal arachnoidal tissue as well as in epithelial tissue and is the main cadherin protein expressed in human meningioma cells. 1,21 The E-cadherin cell adhesion system includes β-catenin, which indirectly connects cadherin to actin filaments. 2,15,25 Altered expression of β-catenin occurs in several human tumors, including gastric, colon, hepatocellular, and esophageal tumors. 15,25 Suppression of E-cadherin expression may result in cells acquiring a mesenchymal morphology and increased motility. 26 The expression of neural cadherin (N-cadherin) is often increased in place of E-cadherin. 8,26 One of the etiologic hallmarks of human meningiomas is the loss of chromosome 22. 16,19 Additional hallmarks in human meningioma include the loss of heterozygosity of the neurofibromatosis type 2 (NF2) gene and the loss of the NF2-encoded protein merlin, which functions as a tumor suppressor factor by controlling cadherin-mediated cell–cell adhesion. 2,5,11,16,19 Although the expression of cadherin molecules are believed to play a role in the morphogenesis of human meningiomas, 2,6 there has been no such finding for canine meningiomas.
The objective of the present study is to analyze the expression of a series of cell adhesion and associated molecules in canine meningiomas and compare their expression in benign and anaplastic cases. The molecules examined were relevant to cell adhesion (E-cadherin, N-cadherin, and β-catenin), cell invasion (DCX), and cell proliferation (Ki-67). This data will provide new information regarding the histogenesis and histologic grade of canine meningiomas.
Materials and Methods
Tissue Samples and Histopathology
Fifty-five canine meningiomas were collected from 13 necropsy and 42 surgical biopsy cases during the last 5 years (2005–2009). The tumor samples included 21 meningothelial, 4 fibrous, 10 transitional, 3 psammomatous, 1 angiomatous, 2 granular cell, and 14 anaplastic meningiomas. Table 1 shows the breed, sex, age, tumor location, histologic subtype, and clinical evidence of parenchymal invasion for each case.
Canine Meningiomas Examined in the Present Study
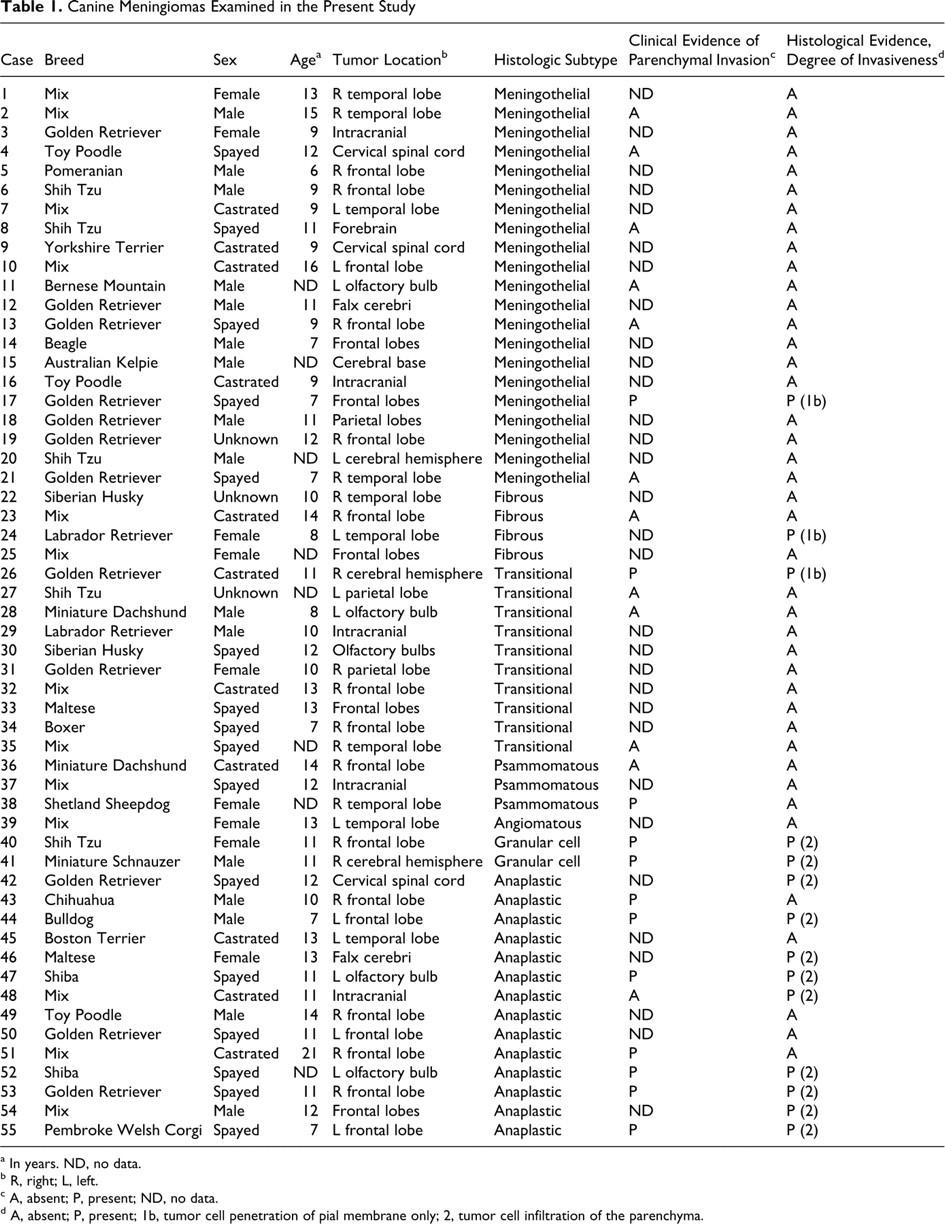
a In years. ND, no data.
b R, right; L, left.
c A, absent; P, present; ND, no data.
d A, absent; P, present; 1b, tumor cell penetration of pial membrane only; 2, tumor cell infiltration of the parenchyma.
The tissue samples were routinely fixed in 10% phosphate-buffered formalin solution. Paraffin sections (2 to 4 μm thick) were stained with hematoxylin and eosin. Histologic classification of canine meningiomas was performed according to the current WHO classification guidelines for domestic animal tumors. 10
Immunohistochemistry
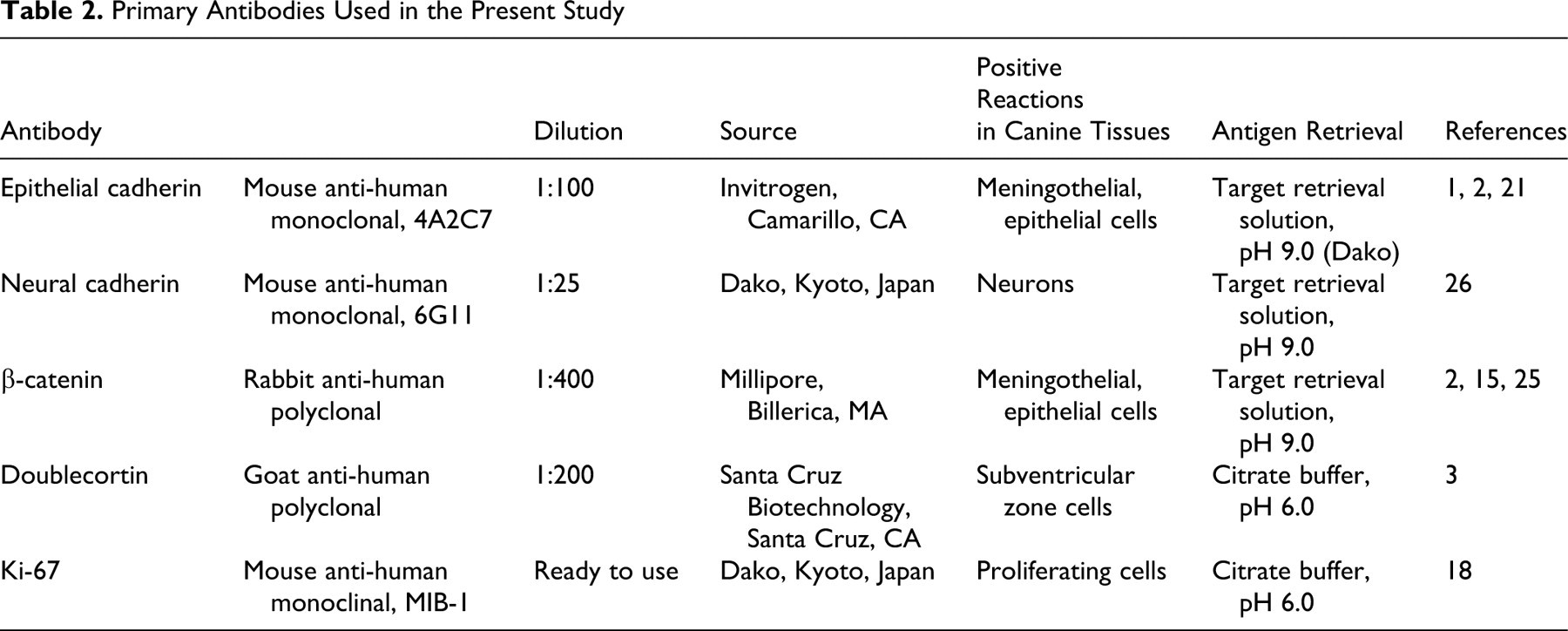
Immunohistochemistry was performed using the labeled streptavidin biotinylated antibody method. Table 2 lists the primary antibodies used and the canine tissues with positive reactions. Deparaffinized sections were first autoclaved at 120°C for 10 minutes for antigen retrieval. The tissue sections were then treated with 1% hydrogen peroxide / methanol for 30 minutes and incubated in 8% skimmed milk / Tris-buffered saline at 37°C for 40 minutes to block nonspecific reactions. Sections were then incubated at 4°C overnight with the primary antibodies. After being washed 3 times in Tris-buffered saline, the sections were incubated with biotinylated anti-mouse, anti-rabbit, or anti-goat immunoglobulin G (1:400, KPL, Guildford, UK) at 37°C for 40 minutes and then with horseradish peroxidase–conjugated streptavidin (1:300, Dako Japan, Kyoto, Japan) at room temperature for 40 minutes. Finally, the reaction products were visualized with 0.05% 3-3′-diaminobenzidine and 0.03% hydrogen peroxide in Tris/HCl buffer, before being counterstained with Mayer’s hematoxylin.
Primary Antibodies Used in the Present Study
The percentage of immunopositive cells was defined as the ratio of strongly labeled tumor cells to the total number of cells examined in 5 high-power (400×) fields. Sampled fields consisted of randomly selected nonoverlapping areas, particularly tumor borders, with highest labeling in foci of invasion into brain parenchyma. Box and whisker plot analyses were performed on the minimum number of counted cells per antigen.
Statistical Analyses
All data analyses were performed with SPSS 15.0. First, a chi-square test was performed to determine the statistical differences between each immunohistochemical stain score and histologic tumor type and between each immunohistochemical stain score and canine meningioma tumor location (frontal lobe, temporal lobe, parietal lobe, olfactory bulb, and cervical spinal cord). Based on these statistical data, comparisons between benign and anaplastic meningiomas were performed with the chi-square test. Then, linear discriminant analyses were performed for association between immunohistochemical parameters and their discriminant values in anaplastic meningiomas. Pearson correlation coefficient analysis was used to assess whether there was any correlation between the immunostaining scores of cadherin molecules and those of nuclear β-catenin, DCX, and Ki-67. A P value of .05 was considered to be significant for all analyses performed.
Results
Histopathology
Table 1 summarizes histologic findings and degree of tumor invasiveness. Histologic evidence of invasive behavior was found in 14 of 55 cases (25%). In the 14 invasive cases, tumor cell infiltration into the parenchyma was observed in 11 cases (79%), and penetration of tumor cells into the pial membrane was observed in 3 cases (21%). Tumor cell dissemination along Virchow-Robin spaces was not observed in any case.
Immunohistochemistry
Table 3 summarizes immunohistochemistry results for each marker molecule. First, the immunoreactivity for E-cadherin, N-cadherin, and β-catenin in normal arachnoidal tissue was examined. Normal arachnoidal samples were collected at necropsy from a 5-year-old male Beagle dog without neurologic disease. E-cadherin was detected along the cell membranes of normal arachnoidal cells, whereas N-cadherin was not (Fig. 1a, 1b). β-Catenin was also distributed along the cell membranes but not in cytoplasm and nuclei of intact arachnoidal cells (Fig. 1c). Arachnoidal cells were completely immunonegative for DCX and Ki-67.
Immunohistochemical Expression of Cell Adhesion and Other Related Molecules in Canine Meningiomas a
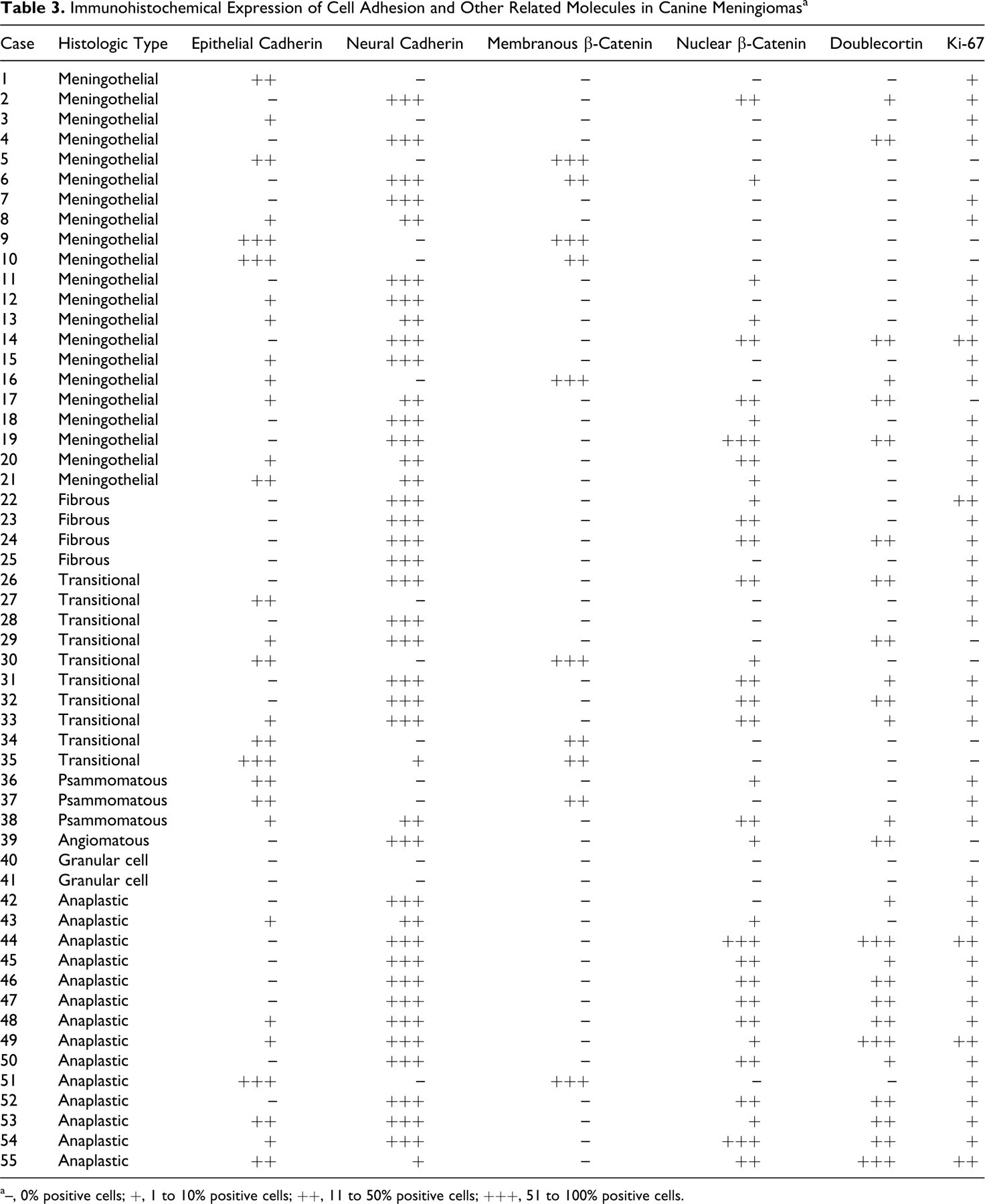
a–, 0% positive cells; +, 1 to 10% positive cells; ++, 11 to 50% positive cells; +++, 51 to 100% positive cells.
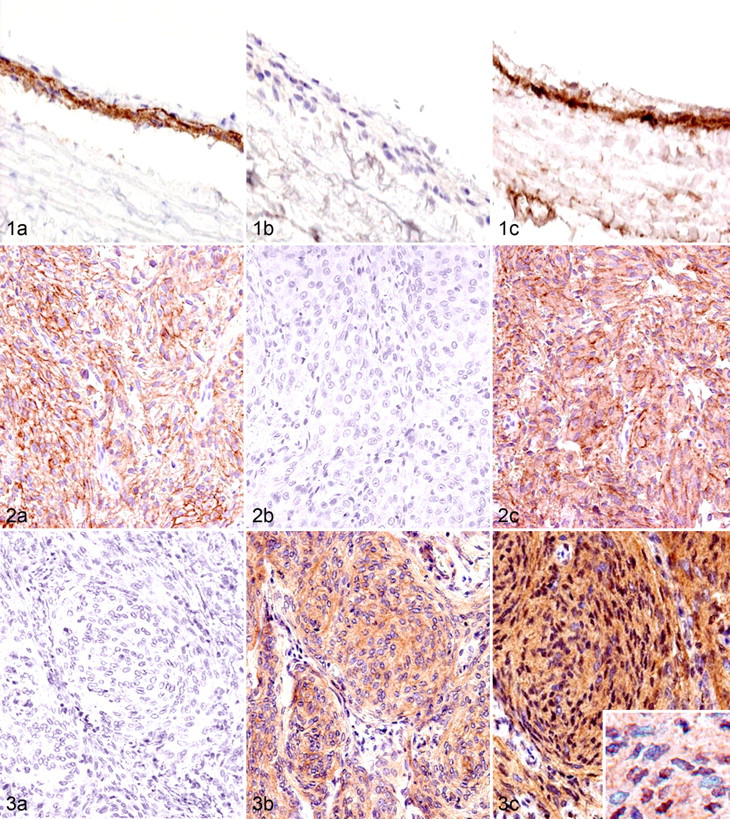
In canine meningiomas, regardless of the histologic features, the single expression of E-cadherin and N-cadherin was observed in 12 of 55 cases (22%) and 24 of 55 cases (44%), respectively (Figs. 2, 3), whereas their combined expression was observed in 17 cases (31%). The expression of β-catenin along the cell membrane was observed in only 10 cases (18%), with cytoplasmic expression in most cases. The translocation of β-catenin into the nucleus was detected in 33 cases (62%). DCX expression in tumor cells was detected in 26 cases (47%) and was more frequently observed in anaplastic meningiomas than in other types. DCX was preferentially expressed in the tumor cells at foci of invasion into the brain parenchyma (Fig. 4a), and DCX-positive cells were always detected at tumor borders when the histologic samples contained the marginal regions. Tumor cells invading the subpial nervous tissue tended to exhibit more intense immunoreactivity for DCX (Fig. 4b). The number of cells with Ki-67-positive nuclei varied from 0 to 18.1%. Figure 5 presents the results of box and whisker plot analyses for the minimum number of counted cells per antigen. Notably, granular cell meningiomas had a different immunohistochemical profile from other types of meningiomas, showing no immunoreactivity to any of the examined molecules.
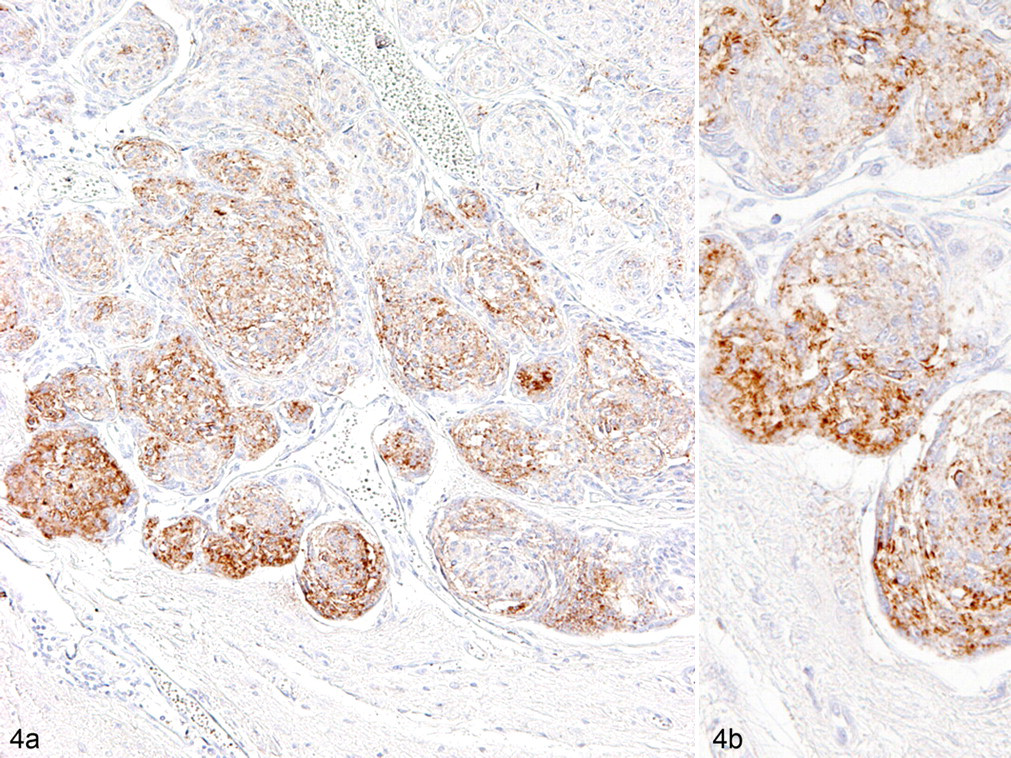
Anaplastic meningioma; dog No. 46. Doublecortin-immunopositive tumor cells are mainly observed at the tumor margin adjacent to the brain parenchyma (a). Tumor cells invading the subpial nervous tissue tend to exhibit more intense immunoreactivity for doublecortin (b). Labeled streptavidin biotin method. Counterstained with Mayer’s hematoxylin.
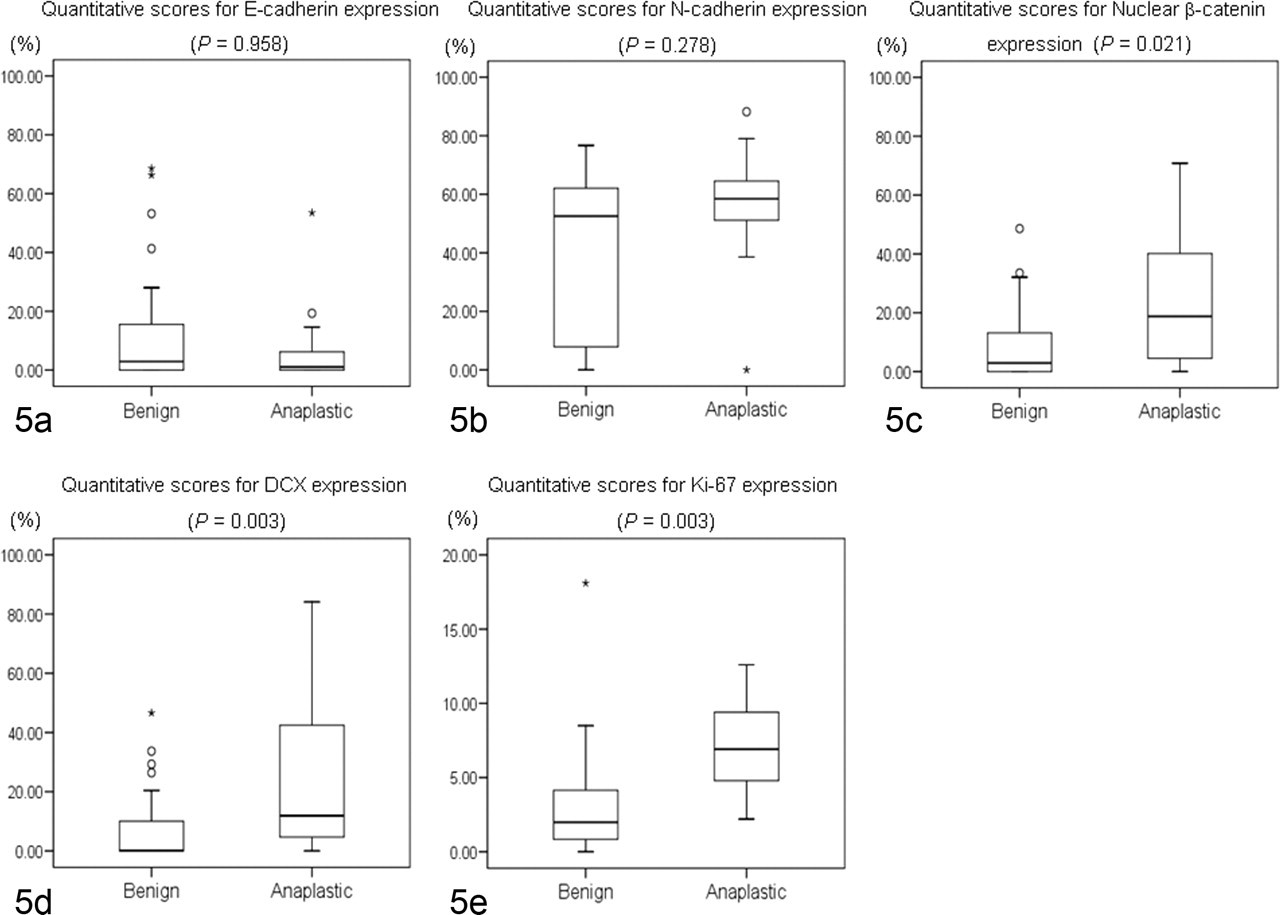
Box plots of quantitative scores for each molecule examined in canine meningiomas. No significant differences are detected in the expression of epithelial cadherin (E-cadherin) (a) and neural cadherin (N-cadherin) (b) between benign and anaplastic meningiomas, but significant differences are found for nuclear β-catenin (c), doublecortin (DCX) (d), and Ki-67 (e). Values in box plots reflect the 25th, 50th (median), and 75th percentiles. The whiskers represent approximately 90 to 95% of the values. °, moderate outliers; *, extreme outliers.
Statistical Analyses
The relationship between the histologic type of canine meningioma and the expression of each molecule was examined. Significant differences were found between fibrous and psammomatous types for both E-cadherin and N-cadherin expression (both P = .030) and between psammomatous and anaplastic types for N-cadherin expression (P = .027). Granular cell meningiomas were differentially classified in the present study because they did not express any cell adhesion molecules. No significant differences were observed on the basis of tumor location. Significant differences in the expression of DCX, nuclear β-catenin, and Ki-67 were found between benign and anaplastic meningiomas (Table 4 ). However, no significant differences were found between benign and anaplastic meningiomas for E-cadherin or N-cadherin expression.
Relationship Between Immunohistochemical Scores of Examined Molecules and Histologic Malignancy in Canine Meningiomas
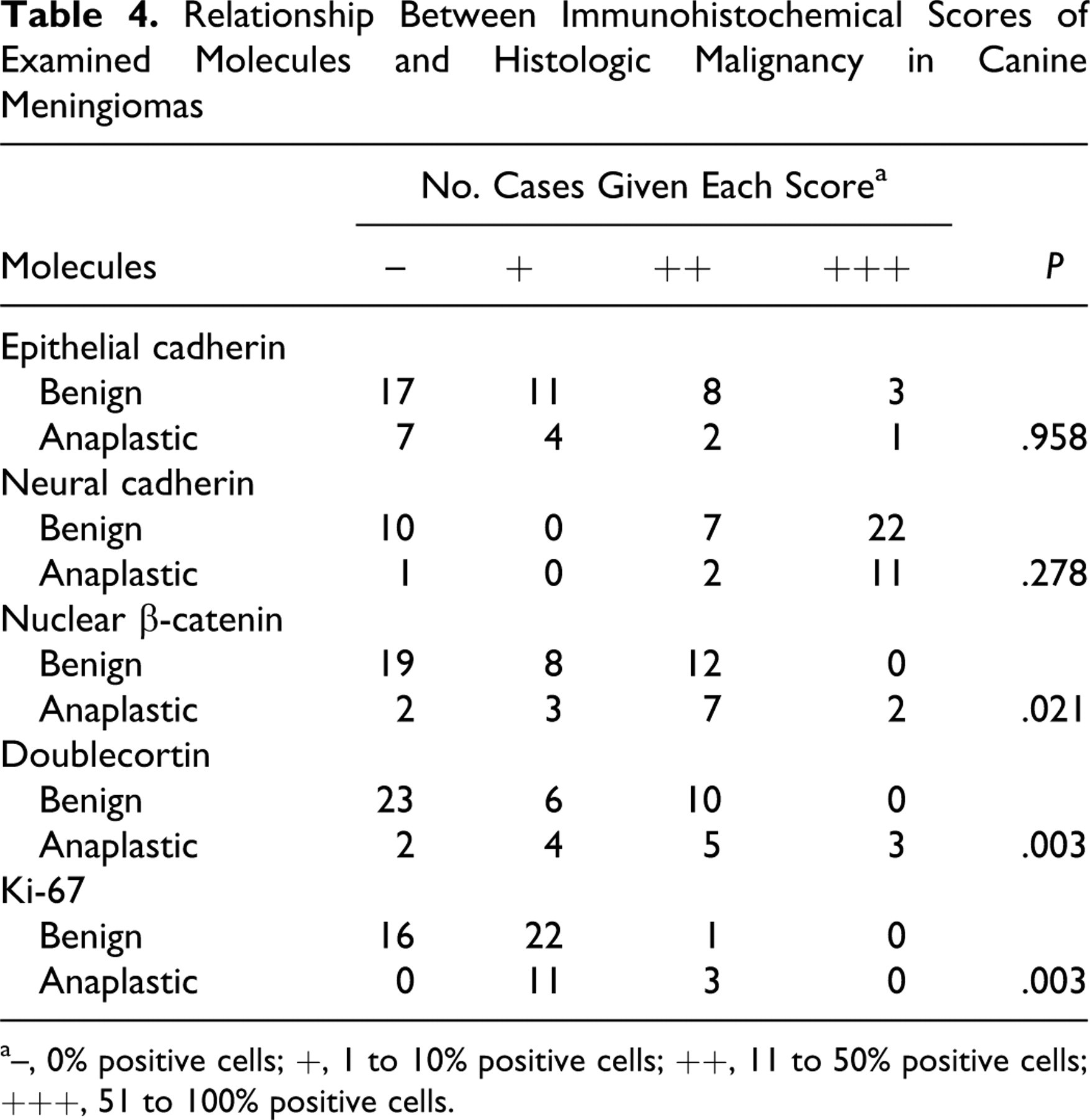
a–, 0% positive cells; +, 1 to 10% positive cells; ++, 11 to 50% positive cells; +++, 51 to 100% positive cells.
Table 5 presents results of linear discriminant analyses between immunohistochemical parameters and their discriminant values. In terms of their expression, nuclear β-catenin, DCX, and Ki-67 appeared to be reliable single predictive molecules, and accuracy scores for predicting the histologic grade were 73.6%, 71.7%, and 77.4%, respectively. Adding the results of E-cadherin and N-cadherin to that of nuclear β-catenin did not increase the predictive value of histologic grade (73.6%), whereas combining all results increased the predictive accuracy to 84.9% .
Linear Discriminant Analyses Between Immunohistochemical Parameters and Their Discriminant Values

a The number represents the diagnostic concordance rate of histologic grade.
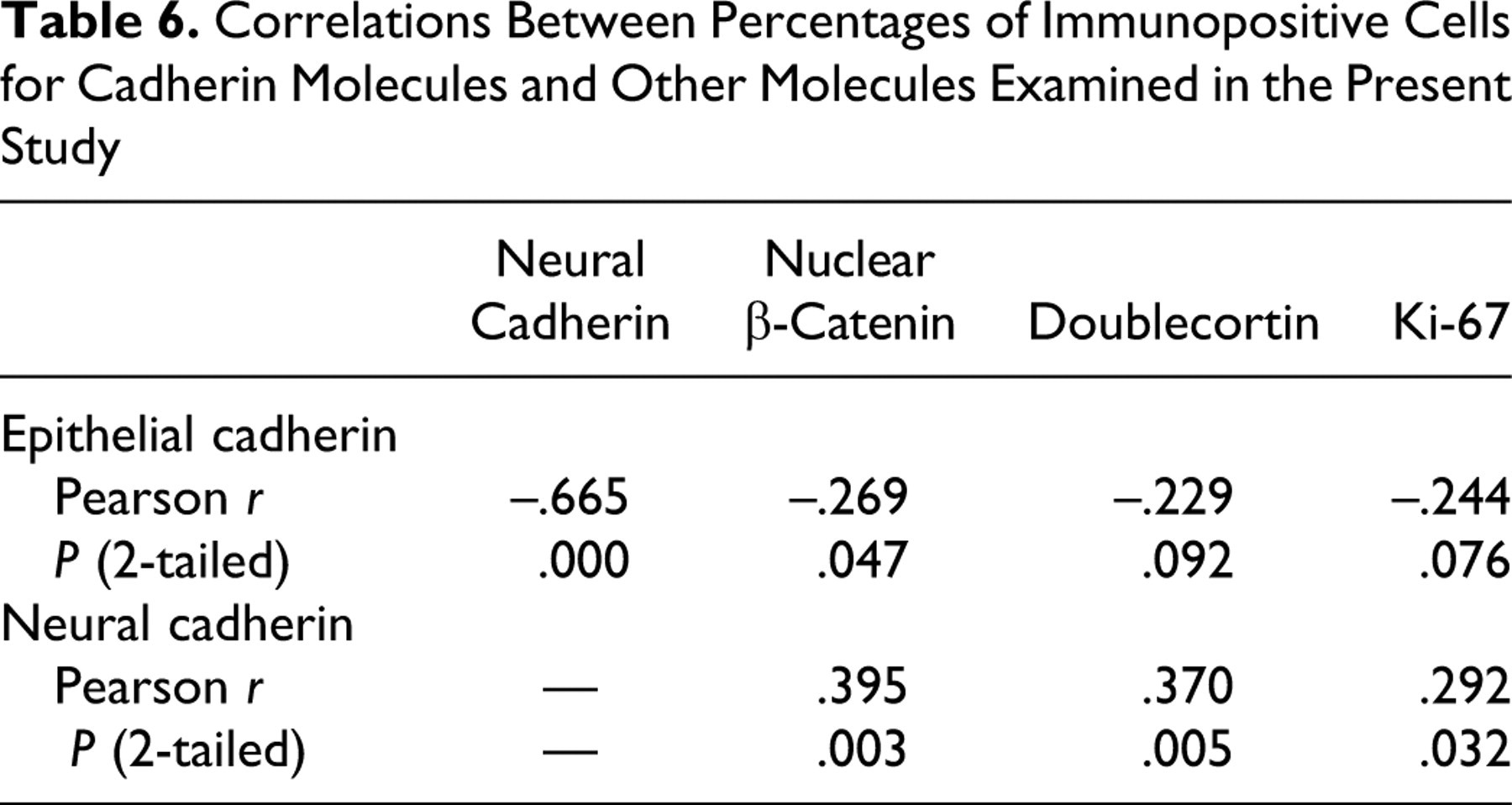
Table 6 summarizes the correlation of the percentage of positive cells for cadherin molecules in relation to all other molecules examined. A significant negative correlation was found between E-cadherin and N-cadherin and between E-cadherin and nuclear β-catenin, as shown by Pearson correlation coefficients. A significant positive correlation was found between N-cadherin and nuclear β-catenin, between N-cadherin and DCX, and between N-cadherin and Ki-67.
Correlations Between Percentages of Immunopositive Cells for Cadherin Molecules and Other Molecules Examined in the Present Study
Discussion
There are several novel findings concerning the expression of molecules involved in cell adhesion, cell invasion, and cell proliferation in these canine meningioma cases. The percentages of nuclear β-catenin-positive, DCX-positive, and Ki-67-positive cells were significantly higher in anaplastic meningiomas than in benign meningiomas. By linear discriminant analyses, these 3 molecules appeared to be of value for predicting the histologic grade of canine meningiomas in terms of single-factor analyses. Although combining the results from the cell adhesion molecules (E-cadherin, N-cadherin, and nuclear β-catenin) did not increase the predictive value of the histologic grade, the use of all 5 parameters was the most reliable predictor. These results indicate that the expression patterns of these molecules can be used for assessing the anaplastic nature of canine meningiomas. However, granular cell meningiomas showed a distinct cell adhesion molecule expression pattern, and so this tumor is considered to be very different from other types of meningioma. With respect to subtype-related differences, fibrous and psammomatous types had statistically significant differences in the expression of both cadherin molecules, whereas psammomatous and anaplastic types had statistically significant differences in N-cadherin. No significant differences were observed in respect to tumor locations. However, findings must be viewed in relation to the small sample size for each histologic subtype and in each site.
The expression levels of E-cadherin and N-cadherin in canine meningiomas were not significantly different between benign and anaplastic cases, which is similar to findings in human meningiomas. 6,7,21 However, a significant negative correlation between E-cadherin and N-cadherin expressions was noted. These results suggest a complementary expression in canine meningiomas regardless of the histologic type. N-cadherin expression in tumor cells is regarded as a sign of invasive behavior. 1,9,12,27 Like E-cadherin, the N-cadherin molecule generally interacts with other N-cadherin molecules expressed in nearby cells in a homophilic manner. 26 Consequently, N-cadherin expressed on a tumor cell may increase its affinity for stromal cells, such as fibroblasts that normally express N-cadherin. This association seems to help tumor cells invade into stroma. 26 The shift of the expression from E-cadherin to N-cadherin in canine meningiomas may facilitate invasion into the surrounding nervous tissue that normally expresses N-cadherin.
The cytoplasmic tail of the E-cadherin molecule binds to β-catenin to form the cadherin–actin filament complex. 2 Such complex formation is necessary for functional cell–cell adhesion. 2 β-Catenin also plays a decisive role in the Wnt signaling pathway, which has been implicated in the pathogenesis of various human cancers. 13 The activation of the Wnt pathway leads to the stabilization of β-catenin and its translocation into the nucleus. Such nuclear accumulation of β-catenin is thought to induce the transcription of some oncogenes through the formation of a complex containing β-catenin and a transcription factor such as Tcf/Lef1. 20 The present study showed the more frequent nuclear localization of β-catenin in anaplastic meningiomas compared to their benign counterparts, as well as a significant positive correlation between β-catenin nuclear localization and the expression of N-cadherin. Therefore, increased expression of N-cadherin and nuclear localization of β-catenin may be involved in the progression to more anaplastic canine meningiomas.
Tumor cell invasion into the surrounding nervous tissue, which is a hallmark of anaplasia in the WHO criteria, 10 was present in most canine anaplastic meningiomas examined. DCX expression was significantly increased in anaplastic meningiomas compared to benign meningiomas. DCX expression was particularly observed at the tumor margin adjacent to normal nervous tissue, and the tumor cells invading to the subpial nervous tissue tended to exhibit more intense immunoreactivity for DCX. Whereas DCX expression could be an accurate predictor of anaplastic meningiomas, its utility may be limited when the border area between the tumor and surrounding nervous tissue is not obtained surgically. In addition, DCX expression had a significant positive correlation with N-cadherin. In tumors, invasive behaviors are a sign of progression. 1,9,12,27 Therefore, the mechanism by which canine meningiomas invade into the surrounding nervous tissue might be related to the expression of DCX and N-cadherin.
The present study revealed the expression of a series of cell adhesion and related molecules in canine meningiomas by immunohistochemistry. All these findings indicate that the expression of N-cadherin and DCX and the nuclear translocation of β-catenin are closely associated with progression of canine meningiomas.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
