Abstract
To establish a simple and uniform classification of bovine peripheral nerve sheath tumors (PNSTs), 63 tumors from 44 cattle were examined histologically and immunohistochemically with antibodies against S100 protein and 2′,3′-cyclic nucleotide-3′-phosphohydrolase (CNPase). Immunohistochemically, all the tumors were positive for S100 protein, CNPase, or both. Four types of PNST were recognized: 35 schwannomas, 9 neurofibromas, 14 hybrid (neurofibroma–schwannoma) tumors, and 5 malignant PNSTs. Axons were identified by immunohistochemistry for neurofilament in a proportion of tumors of each type of PNST. In conclusion, bovine PNSTs commonly have both schwannomatous and neurofibromatous areas. Moreover, the Schwann cell markers S100 protein and CNPase, in combination with antibodies against neurofilament, are valuable diagnostic tools to classify bovine PNSTs.
Keywords
Bovine peripheral nerve sheath tumors (PNSTs) are some of the most frequently found neoplasms at meat inspection. 5,8,9,11,20,22,26 They are mainly diagnosed in cattle older than 5 years, and they seem to affect all breeds, although Herefords and Holsteins are overrepresented. 8,22,26 The classification of bovine PNSTs is based mainly on the system used to classify human PNSTs, in which PNSTs are classified as schwannomas, neurofibromas, or perineuriomas. 15 Perineuriomas have histologic and immunohistochemical characteristics that differ considerably from the 2 other PNSTs, whereas some schwannomas and neurofibromas share several histologic characteristics. Human schwannomas are composed of abnormally proliferating Schwann cells that displace adjacent nerve fibers. In contrast, neurofibromas consist of a more heterogeneous cell population, including Schwann cells, perineural cells, fibroblasts, and mast cells. 15,16 In neurofibromas, axons tend to be mixed with tumor cells. Both schwannomas and neurofibromas express S100 protein and Leu-7 immunohistochemically, but in neurofibromas, only a proportion of the neoplastic cells is labeled. 15,16 Human neurofibromas and schwannomas may arise spontaneously or as part of inherited disorders. Heritable tumors are most often seen in patients with the neurofibromatosis syndrome 1 or 2. 16
In the veterinary literature, standardization is lacking in the nomenclature and classification of PNSTs. Designations such as neurilemmoma, neurinoma, Schwann cell tumor, and schwannoma are used for nerve sheath tumors that consist mainly of Schwann cells. 3,6,7,13,21,23 Depending on the involvement of nerve fibers and other cell types, such as perineural cells and fibroblasts, the designation neurofibroma is used. 3,7,14,21,32 Finally, some authors have incorporated all benign variants in the broad category of benign PNST. 5 There is also some disagreement on the criteria to distinguish benign and malignant PNSTs. 2,4,19,23,24,36,40 The purpose of this study was to standardize the classification of bovine PNSTs by evaluating morphologic and immunohistochemical features, using 2′,3′-cyclic nucleotide-3′-phosphohydrolase (CNPase) as a specific Schwann cell marker in conjunction with S100 protein and neurofilament.
Materials and Methods
The material encompassed 63 PNSTs from 44 cattle that were submitted from abattoirs for diagnosis at the Department of Veterinary Disease Biology (University of Copenhagen, Copenhagen, Denmark) from 1998 to 2008. One or 2 tumors were selected from each animal for examination. The formalin-fixed, paraffin-embedded tissues were sectioned at 2 to 3 μm and stained with hematoxylin and eosin. Selected sections were stained with a modified Gordon Sweet technique (no gold chloride toning) to differentiate reticulin fibers from other collagen fibers. 1
For immunohistochemistry, sections from each tumor were mounted on adhesive-coated slides (Superfrost Plus, Menzel-Glaser, Braunschwaig, Germany), processed through xylene, and rehydrated in ethanol. Antigen retrieval was by boiling in a microwave oven (700 W) twice for 5 minutes in Tris–EDTA buffer—1.21 g Tris base (A 1379, Applichem, Darmstadt, Germany) and 0.372 g EDTA (8418, Merck, Darmstadt, Germany)—in 1 liter of distilled water, pH 9. Endogenous peroxidase was blocked with 0.6% (v/v) H2O2 in Tris-buffered saline (TBS; pH 7.6) for 15 minutes at 20°C before the antibodies were applied to CNPase, S100 protein, or neurofilament (Table 1). In accordance with manufacturer’s instructions (Lab Vision, Thermo Fisher Scientific, Fremont, CA), the UltraVision ONE HRP polymer detection system and the UltraVision LP HRP polymer detection system were applied (Table 1). Slides were given two 5-minute washes in TBS, pH 7.6, before addition of each new reagent. Before being mounted with glycerol–gelatin, the sections were counterstained in Mayer’s hematoxylin for 10 seconds and washed for 1 minute in tap water and 4 minutes in distilled water. For negative controls, run in parallel, the primary antibody was replaced by 5% normal swine serum or 1% bovine serum albumin in TBS and with an irrelevant polyclonal or monoclonal (matching isotype) antibody of the same immunoglobulin G concentration as the primary reagent. Normal bovine central and peripheral nervous tissues were used as positive controls.
Details of Immunohistochemical Methods
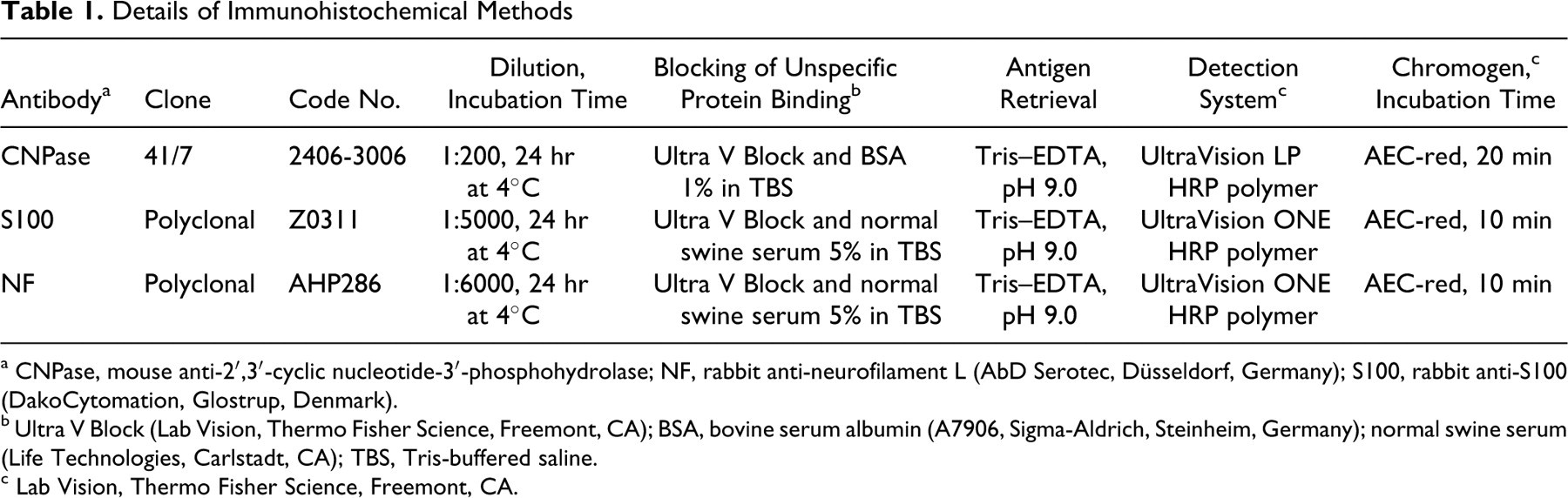
a CNPase, mouse anti-2′,3′-cyclic nucleotide-3′-phosphohydrolase; NF, rabbit anti-neurofilament L (AbD Serotec, Düsseldorf, Germany); S100, rabbit anti-S100 (DakoCytomation, Glostrup, Denmark).
b Ultra V Block (Lab Vision, Thermo Fisher Science, Freemont, CA); BSA, bovine serum albumin (A7906, Sigma-Aldrich, Steinheim, Germany); normal swine serum (Life Technologies, Carlstadt, CA); TBS, Tris-buffered saline.
c Lab Vision, Thermo Fisher Science, Freemont, CA.
Both the intensity of the immunolabeling with CNPase and S100 antibodies and the proportion of positive neoplastic cells were graded from 0 to 3. The intensity score represented the predominant immunoreactivity, in comparison to that of the positive control. The percentage of immunopositive cells, independent of intensity, was assessed as follows: 0, < 1%; +1, 2 to 9%; +2, 10 to 50%; +3, ≥ 50% of the tumor.
The tumors were classified as schwannoma, neurofibroma, hybrid neurofibroma–schwannoma, or malignant PNST (MPNST). The lack of expression of S100 protein or CNPase in > 20% of the tumor cells, plus the presence of entrapped axons, was used to distinguish neurofibromas from schwannomas. Tumors with distinct areas of schwannomatous and neurofibromatous differentiation were classified as hybrid neurofibroma–schwannoma. Finally, invasive tumors with necrosis, hemorrhage, cellular disarray, and nuclear atypia were classified as MPNSTs.
Results
The tumors were solitary or multicentric, 0.5 to 40.0 cm in diameter, gray or white-yellow, fleshy to firm, generally encapsulated or circumscribed, and often lobulated. Some tumors had focal mineralization, cysts, or hemorrhage. Of the cattle with PNSTs, 59% were of the Danish Holstein breed. The tumors mainly affected the heart (30%), mediastinum (22%), nerves near the thoracic vertebrae (11%), intercostal nerves (10%), and brachial plexus (8%). Less common locations included the tongue, skin, and lungs. Three cows had PNSTs in the vertebral canal and were reported to have had clinical signs. The age of cows with PNSTs ranged from 3 to 17 years (mean, 8.5 years); the age of cows with MPNSTs ranged from 4.0 to 4.5 years (mean, 4.0 years).
The 63 tumors were divided into 4 types by histopathologic and immunohistochemical characteristics. Schwannomas constituted 35 of the 63 tumors (56%). The schwannomas were well demarcated and often surrounded by a thin fibrous capsule, but one schwannoma infiltrated adjacent lingual musculature. Most schwannomas had areas with the characteristic Antoni A pattern, in which the neoplastic cells were closely packed and arranged in whorls, bundles, or palisades, with Verocay bodies. The cells were generally elongated with indistinct cell borders and a round or oval central nucleus with finely stippled chromatin and 1 prominent nucleolus. In intervening neoplastic tissue, areas resembling the Antoni B pattern, with rounded and loosely arranged cells, were sometimes seen. In 12 of 35 schwannomas, the cells were arranged in a more disorganized fascicular pattern, and most of these tumors completely lacked the characteristic whorls and palisades (Fig. 1). The tumors with a fascicular pattern were identified as schwannomas by their diffuse immunoreactivity for S100 protein and CNPase. In a few schwannomas, hemorrhage, mineralization, and necrosis were seen; in others, the cells were moderately pleomorphic. Thick-walled vessels were seen in some schwannomas, especially in areas with Antoni A pattern, and perivascular lymphocytes were frequently observed. In 9 of 35 schwannomas, axons entrapped in the tumor were identified by immunoreactivity for neurofilament.
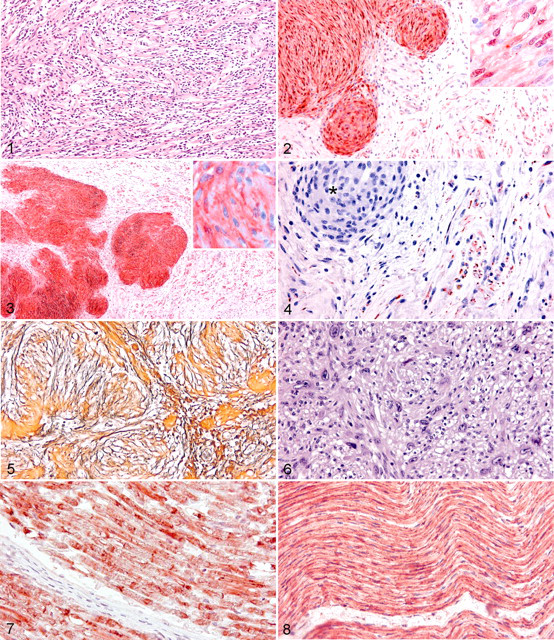
Bovine schwannoma. Fascicular pattern. The cells are arranged in fascicles without whorls or palisades. HE.
Neurofibromas made up 9 of 63 (14%) PNSTs. The neurofibromas were discrete, but 2 cardiac tumors infiltrated the myocardium. In comparison with schwannomas, neurofibromas were less cellular, and the cells were arranged more loosely into bundles or short interwoven fascicles. The cells also were more heterogeneous, and not all tumor cells were positive for S100 protein and CNPase (Figs. 2, 3). Furthermore, axons were entrapped in most tumors, as revealed by immunohistochemistry for neurofilament (Fig. 4).
Tumors composed of both neurofibroma and schwannoma were classified as hybrid neurofibroma–schwannomas; they constituted 14 of 63 (22%) tumors. The hybrid tumors were well demarcated, and in most, the schwannoma component was predominant, constituting 60 to 90% of the tumor. In the areas of schwannoma, both the Antoni A and the fascicular patterns were recognized, and the transition between these areas and those of neurofibroma was usually abrupt (Fig. 3). The areas of neurofibroma were typically dominated by a collagenous stroma, as opposed to the areas of schwannoma, which were dominated by reticulin fibers (Fig. 5).
The mitotic activity in all types of benign PNST was very low, with less than 1 mitotic figure per 10 high-power (400×) fields. More than 1 benign PNST subtype was identified in half of the 19 cows from which 2 tumors were examined. There was no apparent difference among neurofibromas, schwannomas, and hybrid neurofibroma–schwannomas in anatomic location, size, or gross characteristics.
Five MPNSTs from 4 cows were included in the study. All MPNSTs were infiltrative and densely cellular, and all had widespread areas of necrosis and hemorrhage. The disorderly neoplastic cells were pleomorphic; furthermore, anisocytosis, anisokaryosis, giant tumor cells, nuclear molding, and abnormal macronucleoli were frequently seen (Fig. 6). The mitotic activity was low, with 1 mitotic figure per 10 high-power fields. Only 1 of the MPNSTs had metastasized (to the lungs, kidneys, and adrenal glands).
Immunoreactivity for S100 protein was cytoplasmic and/or intranuclear (Fig. 2), whereas that for CNPase was confined to the cytoplasm (Fig. 3). The Schwann cells of bovine myelinated and nonmyelinated nerves (the sciatic and vertebral nerve, respectively) had intense immunoreactivity for both S100 protein and CNPase (Figs. 7, 8). Immunoreactivity for neurofilament was intracytoplasmic in neuronal cell bodies and in axons adjacent to or entrapped in the tumor.
Immunohistochemically, all the tumors were positive for S100 protein, CNPase, or both. Immunohistochemistry for S100 protein was positive in 61 of 63 (97%) tumors; in 51 of 63 (81%), intensity was +2 or +3 and widely distributed. The 2 tumors that did not express S100 protein were both MPNSTs. Immunohistochemistry for CNPase was also positive in 61 of 63 (97%) tumors, with 2+ or 3+ intensity and wide distribution. The CNPase negative tumors were a schwannoma and a MPNST.
Immunoreactivity for S100 protein and CNPase was observed in the same cells in all 4 types of tumors, although the intensity was not always equally strong with both antibodies.
In schwannomas (and areas of schwannoma in hybrid tumors), immunoreactivity was diffuse and uniform compared to that in neurofibromas (and areas of neurofibroma in hybrid tumors), in which immunoreactivity was limited to only a proportion (40 to 60%) of the cells (Figs. 2, 3). The presence of neurofilament-positive axons in the neoplastic tissue was not restricted to neurofibromas (or areas of neurofibroma in hybrid tumors); axons were also detected in 9 schwannomas.
All the MPNSTs were positive for S100 protein, CNPase, or both, but the immunoreactivity was weak (+1) and limited to only a few cells in the tumor. Only 1 of 5 MPNSTs had neurofilament-positive axons entrapped in the neoplastic tissue. It was therefore difficult to divide the malignant tumors into schwannoma versus neurofibroma based on immunohistochemical characteristics.
Discussion
In humans, neurofibromas and schwannomas are often in specific anatomic locations—for example, schwannomas in the vestibular nerve or neurofibromas in the skin. 16 In accordance with those of other studies, 5,6,20,22,23 the tumors in this study were mainly found in the nerves of the heart, mediastinum, thoracic vertebrae, brachial plexus, and intercostal musculature. No difference was observed between schwannomas and neurofibromas in anatomic location. As with humans with neurofibromatosis, Sartin et al classified bovine PNSTs confined to the skin as neurofibromas, 31 and some authors described bovine PNSTs in the vestibulocochlear nerve or other cranial nerves. 21,28,37,40
In the present study, no PNSTs were found in the cranial nerves, and only 2 PNSTs, both neurofibromas, were found in the skin. Interestingly, the cows with MPNSTs were no older than 4.5 years, compared to the average age of 8.5 years in cows with benign PNSTs. Therefore, it seems unlikely that bovine MPNSTs arise from malignant transformation of benign PNSTs.
Distinction of those schwannomas (or areas of schwannoma in hybrid tumors) with a predominantly fascicular pattern from cellular neurofibromas was often difficult. Likewise, the neurofibromas (or areas of neurofibroma in hybrid tumors) with more loosely arranged cells resembled, in some cases, the Antoni B pattern in schwannomas. In these cases, immunohistochemistry was useful because the cells in schwannomas, in contrast to those of neurofibromas, were uniformly positive regardless of the pattern. Axons were commonly seen in neurofibroma but also in some schwannomas. Demonstration of neurofilament-positive axons alone is therefore not sufficient to differentiate neurofibromas and schwannomas. Similar results were found in a study on human schwannomas, in which axons identified by neurofilament immunohistochemistry were seen in more than one-third of the schwannomas. 25
Hybrid neurofibroma–schwannomas have been described in humans, 10 dogs, 33 cats, 34 and cattle. 38 In this study, 14 of 63 PNSTs had components of neurofibroma and schwannoma. The common occurrence of hybrid neurofibroma–schwannomas in cattle indicates that the 2 tumor types may be closely related. It also supports the hypothesis of Canfield, who suggested that neurofibromas resembled dysplastic tissue and that schwannomas may develop from neurofibromas. 6
Distinguishing benign from malignant PNSTs can be difficult because many PNSTs have ill-defined borders and areas of cellular pleomorphism. Some authors classified bovine PNSTs that had invaded adjacent tissue as malignant, 19 whereas others considered PNSTs with areas of necrosis, pleomorphic cells, and a high mitotic index as malignant. 24,40 The PNSTs classified as malignant in this study differed from the benign PNSTs by their invasive growth, in combination with widespread necrosis and hemorrhage as well as marked pleomorphism and nuclear atypia. In humans, the differentiation between benign and malignant PNSTs is based on the degree of pleomorphism and atypia, as well as the presence of necrosis and mitotic activity. 12 Most MPNSTs in human patients are high-grade invasive tumors with at least 4 mitotic figures per high-power field. 15 High mitotic activity was not a feature of the MPNSTs examined in this study, but the tumors differed substantially from the benign PNSTs by other features of malignancy.
Two of the MPNSTs were from a cow with metastases in the lungs and other organs, but PNSTs in lung are not always associated with malignancy. Misdorp reported that small cords of schwannomatous tissue around the bronchi were often found in animals with PNSTs, 20 and in the present study, a benign PNST was found in lung. Given that the cows with MPNSTs in this study did not have any clinical manifestations of disease at slaughter, it is impossible to say anything about the behavior of bovine MPNSTs or to differentiate benign from malignant PNSTs on a clinical basis.
S100 protein is the most widely used diagnostic marker for bovine schwannomas and neurofibromas. It has been used alone 2,13,29,36,38 and in combination with other markers such as glial fibrillary acid protein and neuron-specific enolase. 4,5,14,19,26,27,31,32,40
CNPase is an enzyme that is nearly restricted to oligodendrocytes and Schwann cells, 30,35 so it is more specific than S100 protein, which is present in many cell types. CNPase has not been tested on human or animal PNSTs before, but with positive immunolabeling of 61 of 63 (97%) PNSTs in this study, it proved to be a sensitive marker for bovine PNSTs. The immunolabeling for S100 protein was generally more intense than for CNPase. A plausible explanation is that the antibody to S100 protein is more sensitive and less affected by factors such as tissue preparation.
Conditions that reduce myelin in brain or nervous tissue have been shown to negatively affect the expression of CNPase. 17,18,39 Weak immunoreactivity for CNPase was seen in all MPNSTs and in some neurofibromas and schwannomas. Most of these had a preponderance of the fascicular pattern. Because the same tumors generally reacted intensely with antibodies against S100 protein, a possible explanation for the reduced CNPase reaction is that cells of the fascicular pattern tend to produce less myelin.
Human MPNSTs reportedly express S100 protein in 50 to 70% of cases, but only some of the cells are immunoreactive. 15 In studies of bovine MPNSTs, the tumors were strongly labeled for S100 protein. 19,24,40 This is in contrast to the results in the present study, in which the immunoreactivity for S100 protein was either absent or very weak. The discrepancy might be due to differences between this and other studies in the criteria used to classify bovine MPNSTs.
In conclusion, immunohistochemistry for the Schwann cell markers CNPase and S100 protein, in combination with antibodies against neurofilament, is useful to diagnose and classify bovine PNSTs. With those markers, bovine PNSTs can be divided into 4 subtypes that appear to be common in cattle, except for MPNSTs. Schwannomas, neurofibromas, and hybrid neurofibroma–schwannomas have different histologic and immunohistochemical characteristics, whereas their gross characteristics and biological behavior do not seem to differ. Still, the possibility to differentiate and subclassify the tumors is important to facilitate further comparative studies with PNST in humans and other animal species.
Footnotes
Acknowledgements
We thank E. C. Jansen, B. Andersen, and D. Brok for technical assistance and B. Kragh for critical reading of the manuscript.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
