Abstract
Feline lymphocytic cholangitis is a poorly characterized disease complex with respect to histologic lesions, immunophenotype, and etiopathogenesis. Seventy-eight cases of feline lymphocytic cholangitis (n = 51) and feline hepatic lymphoma (n = 27) were reviewed using standardized histopathology, immunophenotyping (B cell and T cell), polymerase chain reaction for T-cell receptor (TCR) gene rearrangement, and fluorescence in situ hybridization (FISH) for eubacteria. Five histopathologic features in cases of lymphocytic cholangitis assisted in its differentiation from hepatic lymphoma: bile duct targeting (n = 32, 62.7%), ductopenia (n = 9, 17.6%), peribiliary fibrosis (n = 37, 72.5%), portal B-cell aggregates (n = 36, 70.6%), and portal lipogranulomas (n = 38, 74.5%). The majority of lymphocytic cholangitis cases (n = 35, 68.6%) were T cell predominant; 15 (29.4%) had an equal mix of B cells and T cells, and 1 (1.9%) had a B cell–predominant infiltrate; 66.6% of hepatic lymphoma cases were T-cell lymphomas. TCR clonality results were unexpected, with 17.1% of cases of lymphocytic cholangitis having clonal or oligoclonal populations and with T-cell lymphomas having variable TCR clonality (63.6% clonal or oligoclonal, 36.3% polyclonal). The majority of lymphocytic cholangitis (n = 32 of 36, 88.8%) and all hepatic lymphoma cases had no detectable eubacteria using FISH. As demonstrated here, bile duct targeting, ductopenia, peribiliary fibrosis, portal B-cell aggregates, and portal lipogranulomas are lymphocytic cholangitis features that, along with polyclonal TCR (83%), help differentiate it from hepatic lymphoma. No strong evidence was found implicating in situ bacterial colonization as an etiopathogenesis of lymphocytic cholangitis.
Cholangitis and cholangiohepatitis are common acquired liver disorders of the domestic cat, second in prevalence to only the syndrome of hepatic lipidosis. 6,11 Cholangitis has been differentiated into four main subcategories: neutrophilic cholangitis, lymphocytic cholangitis, destructive cholangitis, and chronic cholangitis associated with liver fluke infestation. These subclassifications are based on the type of cellular infiltrate, degree of periportal fibrosis, presence of destructive lesions in the bile ductules, and evidence of fluke infestation (Opisthorchiidae). 32
The clinical differentiation of feline cholangitis/cholangiohepatitis is widely divergent, and subclassification is not possible without a liver biopsy. 10 Typically, lymphocytic cholangitis is slowly progressive, clinically silent during its initial stages, and usually chronically established at the time of liver biopsy and definitive diagnosis. 3,6 There is no confirmed age or breed predisposition for lymphocytic cholangitis, although one reference proposed a predisposition for longhair (Persian) cats. 23 A common association between inflammatory bowel disease, pancreatic inflammation, and lymphocytic cholangitis was first suggested in 1984 by Zawie and Garvey 34 and has since been confirmed by others. 33
Although lymphocytic cholangitis (with synonyms lymphocytic cholangiohepatitis, nonsuppurative cholangitis, nonsuppurative cholangiohepatitis, and lymphocytic portal hepatitis) is the most common subcategory of feline cholangitis, histologic features vary reflecting disease chronicity and, perhaps, immune-mediated cell targeting. Well-recognized features include small lymphocytes infiltrating the portal region, a variable degree of portal fibrosis, and biliary ductular or oval cell proliferation. 32 However, in some cats, lymphocytes seemingly target bile ductules and infiltrate into the biliary epithelium, 7,8 causing progressive ductopenia. 23 In others, expansion of an infiltrating mantle of lymphocytes confuses differentiation from a well-differentiated malignant lymphoma or lymphoproliferative disorder. 32 Progressive lymphocytic cholangitis/cholangiohepatitis has been characterized as a form of chronic lymphocytic cholangitis with the duct-destructive lesion. 7,8 Although it has been speculated that suppurative cholangitis/cholangiohepatitis precedes development of lymphocytic cholangitis/cholangiohepatitis, proof of this transition has never been documented or modeled in the cat.
Immunohistochemical characterization of infiltrating lymphocytes in feline lymphocytic cholangitis/cholangiohepatitis was described in 20 cats with a progressive disease syndrome. 7 Portal lymphocytes were predominantly T cells and infiltrated both the biliary ductular epithelium and the periportal hepatic parenchyma. A smaller yet consistent population of B lymphocytes was described that sometimes formed distinct aggregates or follicles. 7 Hepatic lymphoma, the most frequently observed hematopoietic neoplasm in feline livers, can be confused with lymphocytic cholangitis, particularly because it shows predilection for portal areas. 32 In such cases, immunohistochemical characterization of infiltrating lymphocytes and inspection for T-cell receptor (TCR) gene rearrangements might assist in differentiating inflammation from neoplasia. 26
To date, the etiopathogenesis of lymphocytic cholangitis remains speculative. Although an immune-mediated pathogenesis of this disease is suspected, 7,28 some authors have alternately proposed that the syndrome represents a chronic manifestation of acute neutrophilic cholangitis or ascending bacterial infection. 6,34 Evidence implicating a primary infectious cause is sparse, with two studies reporting the presence of Helicobacter DNA in a small subset of cholangitis/cholangiohepatitis-affected cats and control cats and a single study demonstrating modest zone 1 inflammation in cats with experimentally induced chronic Bartonella infections. 1,12,21 Evidence supporting an immune-mediated pathogenesis includes a predominance of CD3+ T lymphocytes that infiltrate the biliary ductular epithelium and periportal hepatic parenchyma, portal B-lymphocytic aggregates, and membranous and cytoplasmic major histocompatibility complex class II expression on the biliary ductular epithelium. 7 Thus, although evidence for an immune-mediated disorder is compelling, the etiopathogenesis remains enigmatic.
It is against this background that we sought to refine the histopathologic features of lymphocytic cholangitis, with a focus on the inflammatory infiltrate, apparent targeting of bile ductules, bile ductular proliferation, and portal fibrosis. We also characterized and compared the infiltrative lymphocyte populations in cats with advanced lymphocytic cholangitis, cats with modest portal inflammation, cats lacking inflammatory lesions, and cats with lymphoma, using immunophenotyping and polymerase chain reaction (PCR) detection of TCR gene rearrangement. Finally, we sought to investigate the potential role of active bacterial infection in lymphocytic cholangitis using fluorescence in situ hybridization (FISH).
Materials and Methods
Study Population and Tissue Samples
Liver samples from 78 cats were submitted to the Animal Health Diagnostic Center at Cornell University from 1991 to 2009 from veterinary practitioners, the Cornell University Hospital for Animals and three cases as second opinions from other pathology laboratories. Cases were selected from 256 liver biopsies identified using computer search strings (including hepatitis, cholangitis, cholangiohepatitis, cholestasis, and bile duct obstruction) after review of sections stained with hematoxylin and eosin (HE) and acquisition of tissue blocks. Cases were eliminated if histologic features were inconsistent with a diagnosis of lymphocytic cholangitis or portal lymphoma or if tissues were not available for immunohistochemistry. Of samples fulfilling our selection criteria, 62 were surgical wedge or laparoscopic biopsies and 14 were Tru-cut biopsies. Four cats had tissues collected at necropsy, and 33 cases had additional tissues submitted for histologic examination (n = 17 lymphocytic cholangitis, n = 16 lymphoma), including small intestine (n = 25), stomach (n = 7), pancreas (n = 6), gall bladder (n = 3), and mesenteric lymph node (n = 21). Selected cases demonstrated histologic features of lymphocytic cholangitis or hepatic lymphoma. We specifically included cases that displayed ambiguous lymphoproliferative features where the differentiation between lymphocytic cholangitis and hepatic lymphoma was difficult. Two cases had 2, consecutive biopsies submitted. Liver samples from 10 control cats lacking features of lymphocytic cholangitis or hepatic lymphoma were included as negative controls. Of these, 6 were normal, whereas 4 had other hepatic disorders, one each with hepatic lipidosis, chronic passive congestion, biliary cystadenoma, and feline infectious peritonitis with secondary pyogranulomatous hepatitis. Tissues had been fixed in 10% neutral buffered formalin, embedded in paraffin, and stained with HE in 4-μm sections for initial assessment. Sections were also stained with Masson’s trichrome and Gomori’s reticulin silver impregnation to objectively assess the extent and location of connective tissue deposition. Additional 4-μm-thick liver sections were mounted on charged microscope slides (Fisher Scientific) for leukocyte antigen and cytokeratin immunohistochemical staining. Two 25-μm-thick sections of liver tissue from 51 study cases (lymphocytic cholangitis, n = 35; lymphoma, n = 16) and 10 control cases were placed in sterile Eppendorf tubes for TCR clonality PCR. Five 4-μm-thick liver sections from 54 study cases (lymphocytic cholangitis, n = 36; lymphoma, n = 18) and 9 control cases were placed on positively charged slides for examination by FISH using eubacterial probes. Lack of adequate tissue samples restricted TCR clonality PCR and eubacterial probing in 27 and 24 study cases, respectively, and 1 control case for eubacterial probing.
Histologic Classification
All tissues from study and control cases were independently examined by two pathologists (A.W. and S.M.). The degree of inflammation was graded with the following scale:
Grade 0: No inflammation, defined by < 5 inflammatory cells per portal area, with most portal areas devoid of inflammatory cells.
Grade 1: Mild inflammation, defined by small numbers of inflammatory cells (> 5 but < 20) within the adventitia of the portal area, with inconsistent involvement of the portal triads.
Grade 2: Moderate inflammation, defined by intermediate numbers of inflammatory cells (> 20 but < 75) within the adventitia of the portal area, with most portal triads affected.
Grade 3: Severe inflammation, defined by large numbers of inflammatory cells (> 75) within the adventitia of portal triads and by diffuse involvement of portal triads (Figs. 1–3).
Cases were classified as lymphoma if there was a dense, homogeneous infiltration of lymphocytes that extended beyond the adventitial limits of the portal triads and had features of malignancy.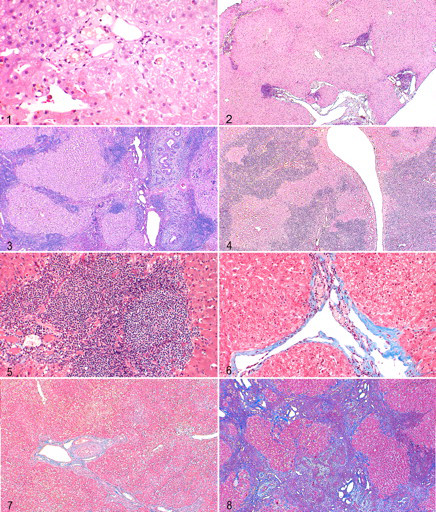
The degree of fibrosis was assessed using stained sections (HE, Masson’s trichrome, and Gomori reticulin) and graded as follows:
Grade 1: No fibrosis.
Grade 2: Mild fibrosis, defined as connective tissue (collagen and reticulin fibers) present in small amounts with patchy distribution within and around portal triads but no fibrous septa.
Grade 3: Moderate fibrosis, defined as diffuse moderate amounts of connective tissue within most portal triads, including presence of fibrous septa but no bridging.
Grade 4: Severe fibrosis, defined as large amounts of connective tissue within and bridging between portal triads (Figs. 5–8).
Grade 1: No change in bile ductule cross-sectional profile per portal area (1 to 2 bile ductule profiles).
Grade 2: Mild bile ductule proliferation (2 to 3 bile ductule profiles per portal triad).
Grade 3: Moderate bile ductule proliferation (4 to 6 bile ductule profiles per portal triad).
Grade 4: Severe bile ductule proliferation (> 7 bile ductule profiles per portal triad) (Figs. 9–12.).
Cholangiolar proliferation and ductopenia were assessed on stained sections (HE and cytokeratin immunohistochemistry). Regarding bile ductules, the following grades were assigned:
The following histopathologic features were recorded as present or absent: lymphoproliferation, ductopenia, peribiliary fibrosis, portal ductular peripheralization, apparent immunologic targeting of bile ductules or bile ductule destruction, portal lipogranulomas, portal B-cell aggregates, and bile duct obstruction. Lymphoproliferation was assessed in cases diagnosed as lymphocytic cholangitis and defined as exuberant lymphocytic proliferation that formed dense portal infiltrates and infiltrated beyond adventitial limits into the adjacent hepatic parenchyma (Fig. 4
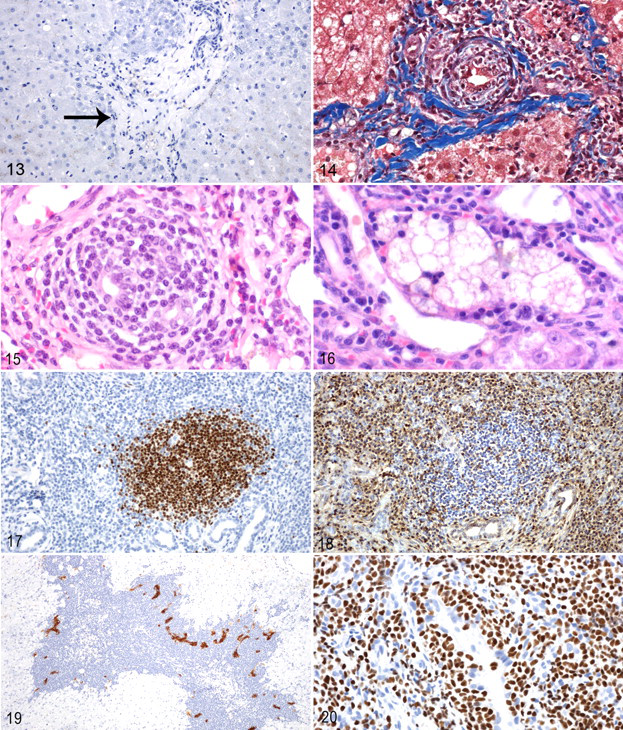
). These cases lacked lymphocytic cellular atypia, had polymorphous lymphocyte populations, and often had additional infiltrating leukocytes (plasma cells, Mott cells, macrophages); for these reasons, they were not diagnosed as lymphoma. Ductopenia was defined as zero bile ductule profiles per portal triad or remnant cytokeratin-positive epithelial cells not forming discrete bile ductules in greater than 30% of portal triads (Fig. 13
). Peribiliary fibrosis was defined as concentric laminations of at least 1 to 2 fibers that encircled a bile ductule (Fig. 14
). Portal ductular peripheralization was defined as a significant proportion of bile ductules and cytokeratin-positive cells at the intersection of portal and parenchymal structures (Fig. 19
.). Immunologic targeting of bile ductules or bile ductule destruction was defined by the presence of lymphocytes within or immediately adjacent to the bile ductule epithelium with concurrent evidence of bile ductule epithelial cell injury, necrosis, or attenuation (Fig. 15
). The presence of portal lipogranulomas was determined on HE sections (Fig. 16
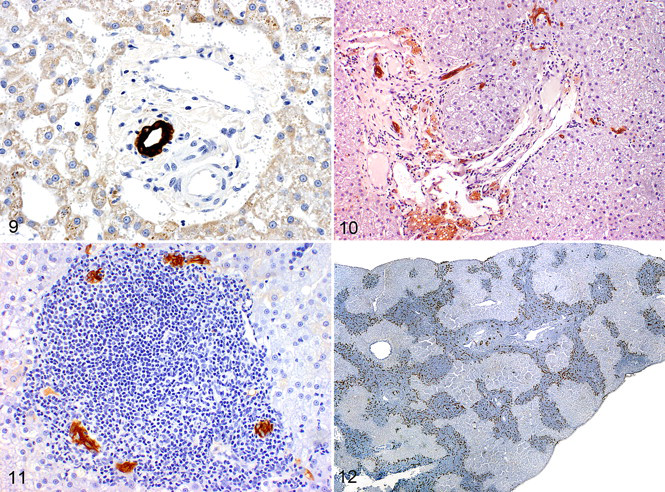
). The presence of B-cell follicles within the portal area was determined using immunohistochemistry for B-cell leukocyte antigens CD45RA B220, CD79, or Pax5 (Figs. 17, 18
). Histologic features of bile duct obstruction included dilated main ducts, evidence of bile ductule tortuosity, inspissated luminal bile, and moderate periductular edema.
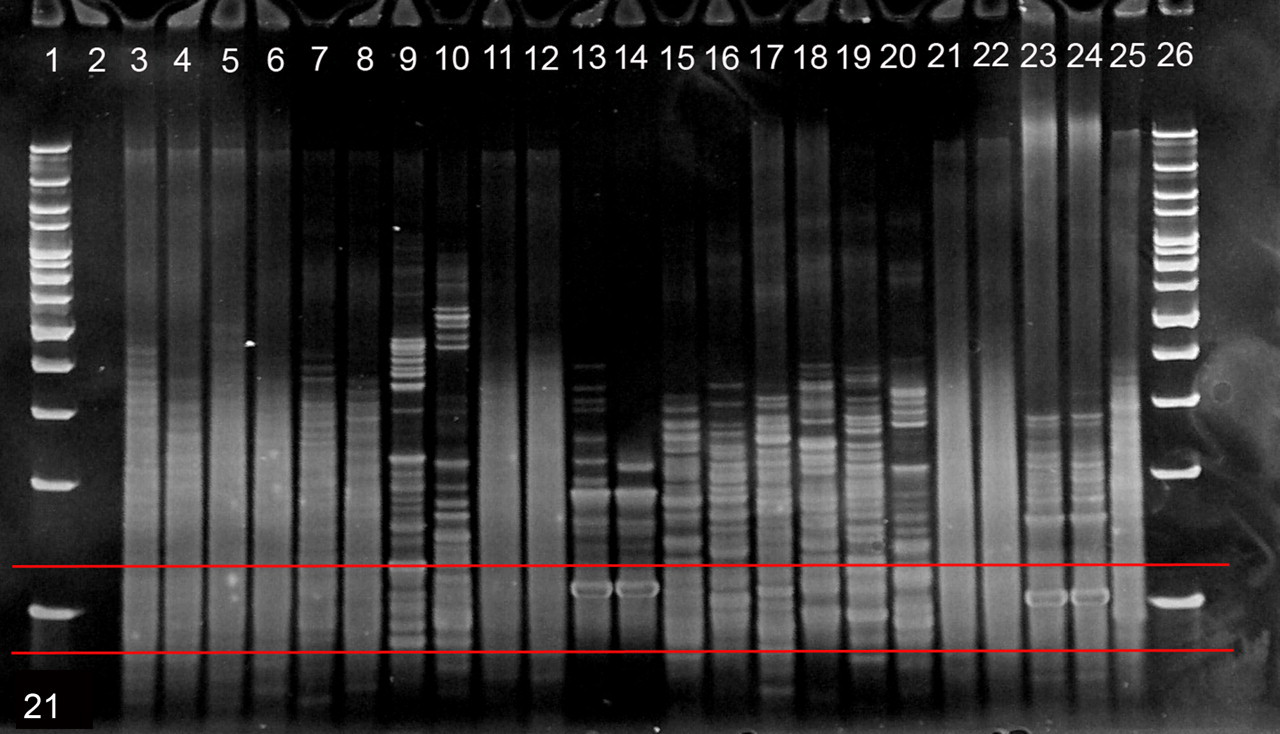
Polyacrylamide gel electrophoresis of denatured and reannealed T-cell receptor polymerase chain reaction amplicons. Red-bordered region depicts target region of 80 to 120 base pairs. Lanes 1, 26: 100–base pair DNA ladder. Lane 2: negative control. Lanes 3, 4: case No. 28 (liver), polyclonal. Lanes 5, 6: case No. 5A (liver), polyclonal. Lanes 7, 8: case No. 8 (liver), polyclonal. Lanes 9, 10: case No. 16 (liver), pseudoclonal. Lanes 11, 12: case No. 27 (liver), polyclonal. Lanes 13, 14: case No. 3 (liver), clonal. Lanes 15, 16: case No. 1 (liver), pseudoclonal. Lanes 17, 18: case No. 21 (liver), pseudoclonal. Lanes 19, 20: case No. 5B (liver), pseudoclonal. Lanes 21, 22: case No. 5B (spleen), polyclonal. Lanes 23, 24: case No. 6 (liver), clonal. Lane 25: polyclonal control.
For each leukocyte antigen immunohistochemical stain, the proportion of infiltrating leukocytes was estimated using the following scale, based on percentage of the total portal infiltrate with positive staining: Grade 1: 0 to 5% Grade 2: 6 to 25% Grade 3: 26 to 50% Grade 4: 51 to 75% Grade 5: 76 to 100%.
Immunohistochemistry
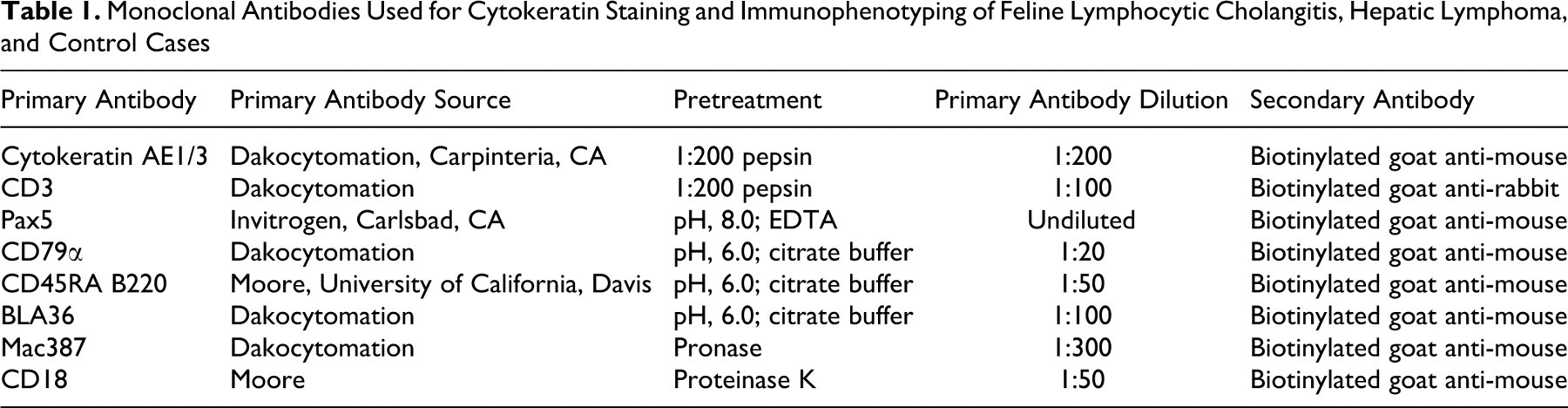
All study and control cases were examined immunohistochemically for the expression of common leukocyte antigens and cytokeratin AE1/3 (Table 1 ). All the cases were stained for CD3 (T-lymphocyte marker) and at least one B-lymphocyte marker (Pax5, CD45RA B220, BLA36, and CD79α), depending on antibody availability. Seventy-four cases were stained with Pax5, 76 for CD45RA B220, 14 for BLA 36, and 13 for CD79α. Seventy-six cases were stained with the myelomonocytic antigen Mac387, and one case was stained with CD18. The immunohistochemistry was performed using the Dako Autostainer Plus (Dakocytomation, Carpinteria, CA) using the streptavidin–biotin immunoperoxidase technique according to the manufacturer’s instructions (Zymed, San Francisco, CA) and previously described methods. 19,25 A duplicate of each section was incubated with normal mouse immunoglobulin G or normal goat immunoglobulin G for the same time as the primary antibody to be used as a negative control. Normal feline lymph nodes were used as positive controls for CD3, CD79a, CD45RA, CD18, BLA36, Pax5, and Mac387 staining. Within hepatic sections, the bile ductular epithelial cells were used as internal positive controls for cytokeratin staining.
Monoclonal Antibodies Used for Cytokeratin Staining and Immunophenotyping of Feline Lymphocytic Cholangitis, Hepatic Lymphoma, and Control Cases
Clonality Assessed Using PCR for the TCR
PCR for TCR gene rearrangement clonality was performed in duplicate in denaturing and nondenaturing conditions on tissues from 9 control cats, 35 lymphocytic cholangitis, and 16 cases of hepatic lymphoma, 11 of which were T-cell lymphomas on immunophenotyping. PCR for TCR gene rearrangement was performed as previously described by Moore et al 26 with PCR products separated by electrophoresis and bands in the 80– to 120–base pair range evaluated for clonality. The determination of a sample being clonal required the presence of the appropriately and same-sized 1 or 2 sharp bands in duplicate samples run side by side (monoallelic or biallelic rearrangement, respectively). Samples exhibiting 3 to 5 reproducible bands in duplicate analyses were classified as oligoclonal. Duplicate PCR reactions visualized on denatured and nondenatured gels were used to help distinguish true clonal samples from pseudoclonal samples. Pseudoclonal samples contained 1 or 2 bands of disparate size in duplicate analysis. The presence of a broad band, smear, or ladder of bands covering a range of product sizes was interpreted as polyclonal. Samples designated as pseudoclonal were omitted from interpretation.
FISH Using Eubacterial Probes
Tissue from all control cats, 36 lymphocytic cholangitis and 18 hepatic lymphoma, were evaluated for the presence of bacteria using FISH with eubacterial probe EUB338-cy3 and control probe N0N-EUB338-6FAM as previously described by Simpson et al. 30
Statistical Analysis
The normality for each data subset was tested using Kolmogorov–Smirnov statistic, histogram, and box plot. Because none of the parameters showed a Gaussian distribution, nonparametric tests were used. Differences in median age among cats with lymphocytic cholangitis, cats with hepatic lymphoma, and control cats and in graded histopathologic features (inflammation, fibrosis, and bile duct proliferation) and leukocyte antigen staining were analyzed using the Mann–Whitney U test. Differences in sex distribution between cases and controls, the histopathologic features graded as present or absent (lymphoproliferation, ductopenia, peribiliary fibrosis, peripheralization of ducts, apparent immunologic targeting of bile ductules or bile ductule destruction, portal lipogranulomas, and portal B-cell aggregate formation and concurrent bile duct obstruction), predominant immunophenotype (T vs B), clonality, and FISH results were analyzed by 2-by-2 tables (chi-square analysis) and Fischer exact tests. A 2-sided P value of < .05 was considered significant.
Results
Demographics
The median age of all cases (11.5 years; range, 0.5 to 19.0 years) and controls (8.0 years; range, 2 to 14 years) was not significantly different (P = .14). The median age of cats diagnosed with lymphocytic cholangitis (10.5 years; range, 1 to 19) and lymphoma (12.0 years; range, 0.5 to 17.0) was also not significantly different (P = .17). In addition, sex distribution between cases and controls was not significantly different. Among cases, there were 38 neutered females (48.7%), 33 neutered males (42.3%), 4 intact females (5.1%), 2 intact males (2.6%), and 1 cat of unknown sex (1.3%). Among controls, there were 2 neutered females (20%), 6 neutered males (60%), 1 intact female (10%), and 1 intact male (10%). Breed distribution among study cases included 60 domestic shorthair cats (76.9%), 12 domestic longhair cats (15.4%), 2 Maine Coon cats (2.6%), 1 Birman (1.3%), 1 Siamese (1.3%), 1 Manx (1.3%), and 1 cat of unknown breed (1.3%). Among control cats, there were 7 domestic shorthair cats (70%), 1 domestic longhair (10%), 1 Siamese cat (10%), and 1 Persian cat (10%). Concurrent diseases identified in cases included hyperthyroidism in 2 cases and one each of a stump pyometra, unspecified cardiac disease, hepatic abscess, urinary tract infection, megacolon, feline leukemia virus infection, and feline immunodeficiency virus infection. Lymphoma in sites other than the liver was diagnosed in 19 of 33 study cases (57.6%)—hepatic lymphoma (n = 16, 100%) and lymphocytic cholangitis (n = 3, 18.8%)—specifically in the small intestine (n = 17), mesenteric lymph nodes (n = 6), pancreas (n = 3), spleen (n = 1), and stomach (n = 3). In the 3 cats with lymphocytic cholangitis that had extrahepatic lymphoma, the hepatic lesions were considered to be inflammatory on the basis of a mixed lymphoid population (morphology and immunophenotype) in the portal area (n = 3) and polyclonal TCR clonality (n = 2). Concurrent inflammatory bowel disease was diagnosed in 5 of 10 cases of lymphocytic cholangitis and in zero cases of hepatic lymphoma where the small intestine was examined.
HE Histologic Features
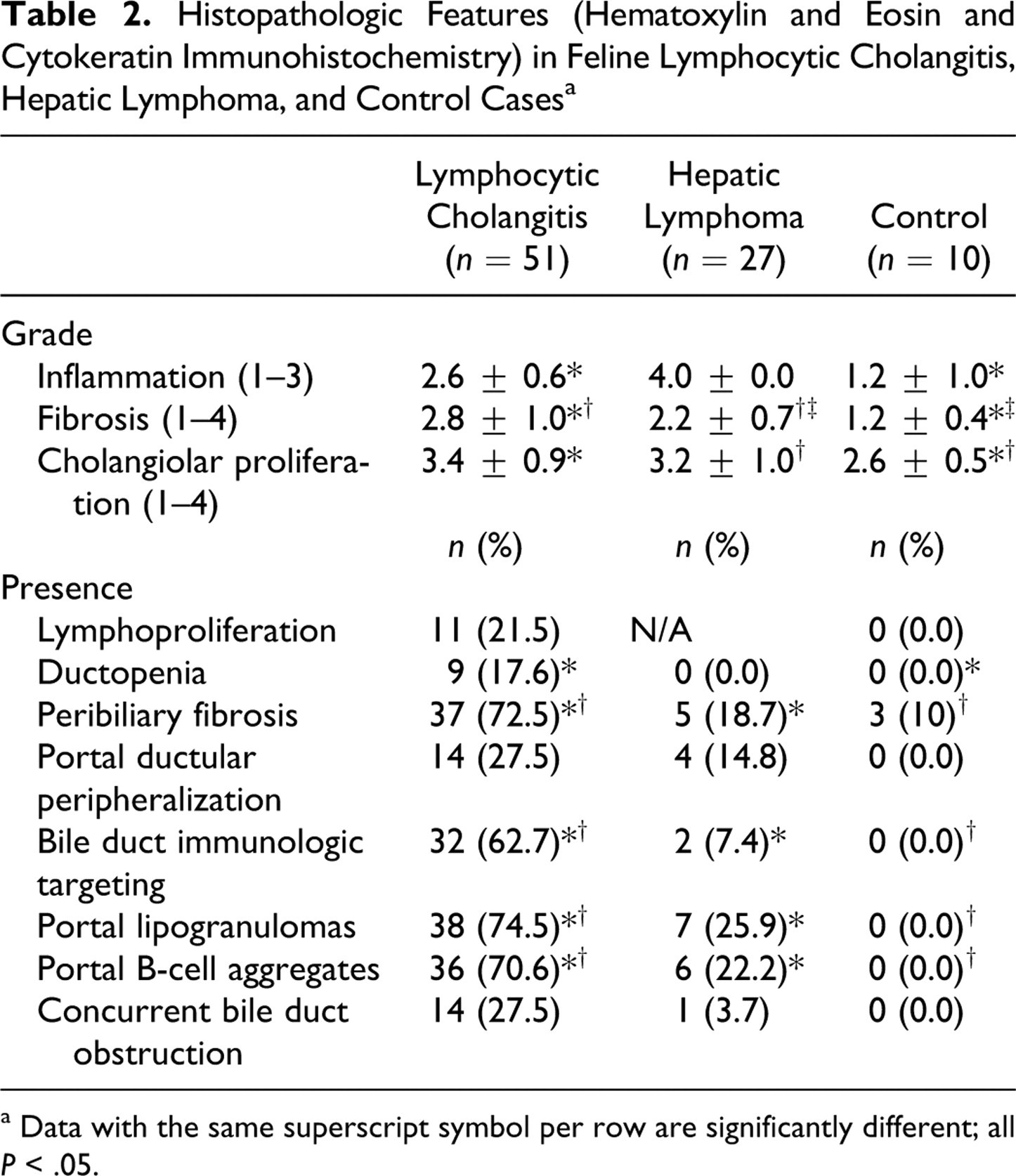
Of 78 cases, 51 were diagnosed with lymphocytic cholangitis and 27 as hepatic lymphoma based on light microscopic findings. Histopathologic grading and features are summarized in Table 2 . Cats with lymphocytic cholangitis had a significantly higher score for inflammation, compared with controls (P < .001) (Figs. 1–4). When multiple liver lobes were sampled, there was variability between lobes for the severity of inflammation. Cats with lymphocytic cholangitis and hepatic lymphoma received significantly higher scores for fibrosis, compared with controls (Figs. 5–8) (P < .001 and P = .001, respectively), and cats with lymphocytic cholangitis had significantly higher scores for fibrosis, compared with cats with hepatic lymphoma (P = .004). In cases of lymphocytic cholangitis with severe fibrosis, we often observed an expected nodularity of the hepatic parenchyma, reflecting regenerative activity and constrictive scarring. A moderate association for peribiliary fibrosis was noted for lymphocytic cholangitis when compared with control cases (P = .010), and a strong association was noted when lymphocytic cholangitis was compared with hepatic lymphoma cases (P < .001).
Histopathologic Features (Hematoxylin and Eosin and Cytokeratin Immunohistochemistry) in Feline Lymphocytic Cholangitis, Hepatic Lymphoma, and Control Cases a
a Data with the same superscript symbol per row are significantly different; all P < .05.
A significantly higher score for biliary hyperplasia was assigned to cats with lymphocytic cholangitis and hepatic lymphoma, compared with controls (P = .002 and P = .040, respectively) (Figs. 9–11). However, there was no significant difference in biliary hyperplasia scores between cats with lymphocytic cholangitis and hepatic lymphoma (P = .35). Control cats had, on average, a score of 2.6 ± 0.5 for portal bile ductile number, correlating to 2 to 6 bile ductules per portal region. Many study cats with substantial bile ductule proliferation had ductular peripheralization within the portal area, with cytokeratin-positive cells identified at the intersection of portal and parenchyma structures (Fig. 19). This finding was noted more frequently in cats with lymphocytic cholangitis (14 of 51, 27.5%) than hepatic lymphoma (4 of 27, 14.8%); however, there was no significant association of this feature with any of the 3 groups.
Ductopenia was identified in 9 of 51 cats (17.6%) with lymphocytic cholangitis (Fig. 13), a feature absent from cats with hepatic lymphoma and control cats. A significant association of this feature was noted for cats with lymphocytic cholangitis, when compared with cats with hepatic lymphoma (P = .02). Ductopenia was patchily distributed both within individual sections and between hepatic lobes and was notably associated with a diminished inflammatory infiltrate. Lipogranulomas were often identified within the portal adventitia of ductopenic portal triads (7 of 9, 77.7%). Cytokeratin-positive material was noted within the cytoplasm of macrophages in ductopenic triads of one cat. Of the cats with ductopenia (n = 9), 5 had moderate portal inflammation (55.5%); 4 had severe portal inflammation (44.4%); 7 had T cell–predominant inflammatory infiltrates (77.7%); 1 had B cell–predominant inflammatory infiltrates (11.1%); and 1 had mixed B cell and T cell inflammatory infiltrates (11.1%). Additional histologic features associated with ductopenia included evidence of duct targeting (66.7%) and peribiliary fibrosis (55.6%).
Lymphocytes targeting bile ductules (lymphocytic infiltration within or immediately adjacent to the biliary epithelium associated with concurrent bile ductule epithelial injury, necrosis, or attenuation) was strongly associated with the light microscopic diagnosis of lymphocytic cholangitis, when compared with control and hepatic lymphoma cases (P < .001 and P < .001, respectively). Of the cats with lymphocytic cholangitis with apparent bile duct targeting (n = 32), 22 had T cell–predominant portal infiltrates (68.8%), 9 had an approximately equal mix of infiltrating B cells and T cells (28.1%), and 1 had B cell–predominant portal infiltrates (3.1%).
Finding B-cell aggregates in portal regions was strongly associated with lymphocytic cholangitis when compared with the control and hepatic lymphoma cases (P < .001 and P < .001, respectively). B-cell aggregates did not form germinal centers in any cat. Finding lipogranulomas in portal regions was also strongly associated with lymphocytic cholangitis, when compared with control and hepatic lymphoma cases (P < .001 and P < .001, respectively). No difference in bile ductule targeting, portal B-cell aggregates or follicles, or portal lipogranulomas was noted between hepatic lymphoma and control tissues.
Concurrent bile duct obstruction (dilated main ducts, evidence of bile ductule tortuosity, inspissated luminal bile, and moderate periductular edema) was not a distinguishing feature of either lymphocytic cholangitis or hepatic lymphoma. Although cats with lymphocytic cholangitis commonly had notable periductular and portal fibrosis and moderate to severe inflammatory cell infiltration, overt canalicular cholestasis was rarely noted with ductopenia (1 of 9, 11.1%).
Features of lymphoproliferation (exuberant lymphocytic proliferation that formed dense portal infiltrates and infiltrated beyond adventitial limits into the adjacent hepatic parenchyma) were noted in 11 of 51 cases (21.5%) histologically classified as lymphocytic cholangitis (Fig. 4). These cases were not diagnosed with lymphoma, because the infiltrates lacked lymphocytic cellular atypia, had polymorphous lymphocyte populations, and often had additional infiltrating leukocytes (plasma cells, Mott cells, macrophages). Additional tissues were submitted for 5 of 11 lymphoproliferative cases, 2 of which (40%) had lymphoma diagnosed in either the small intestine (n = 1) and the mesenteric lymph node (n = 1), whereas 3 (60%) had no evidence of lymphoma elsewhere. The 2 lymphoproliferative cholangitis/cholangiohepatitis cases that had lymphoma in sites other than the liver had an equal mix of B cells and T cells infiltrating the portal area and had polyclonal TCR PCR products.
Leukocyte Antigen Immunohistochemistry
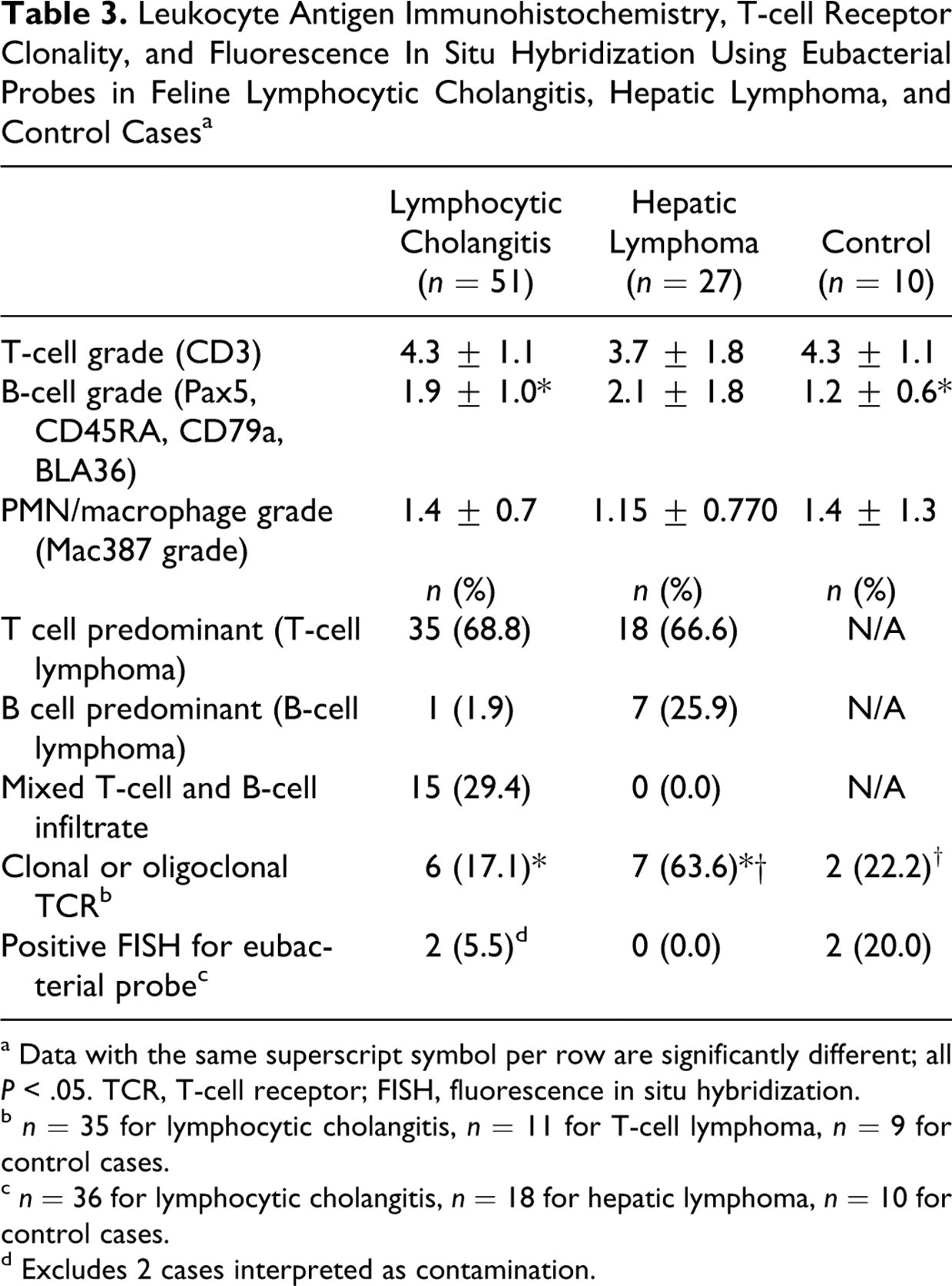
Immunohistochemical staining for leukocyte antigens (Table 3 ) confirmed that most cats with lymphocytic cholangitis (35 of 51, 68.6%) had T cell–predominant portal infiltrates. T cells constituted approximately 76 to 100% of the infiltrate in 29 of 35 cases (82.9%) and 51 to 75% of the infiltrate in 6 of 35 cases (17.1%). Of cats with T cell–predominant cholangitis/cholangiohepatitis (n = 35), 22 were graded with severe inflammation (66.7%), 10 with moderate (28.6%), and 3 with mild (8.6%) (Fig. 18). Of 51 cats with lymphocytic cholangitis, 15 (29.4%) had approximately equal proportions of infiltrating T cells and B cells, and 1 (1.9%) had a predominantly B-cell infiltrate. Of cats with a mixed B-cell and T-cell infiltrate (n = 15), 10 were graded with severe inflammation (66.7%), 4 with moderate (26.7%), and 1 with mild (6.7%). No significant difference was noted among the immunophenotypes (T cell predominant vs mixed B cell and T cell vs B cell predominant) and the severity of the portal inflammatory infiltrate (P = .96). Cats with lymphocytic cholangitis had small but significantly higher scores for B-cell infiltrates. No significant difference was noted among cats diagnosed with lymphocytic cholangitis, cats diagnosed with hepatic lymphoma, and control cats for proportions of infiltrating macrophages and polymorphonuclear cells, and, generally, these constituted a small proportion (0 to 5%) of the infiltrating population.
Leukocyte Antigen Immunohistochemistry, T-cell Receptor Clonality, and Fluorescence In Situ Hybridization Using Eubacterial Probes in Feline Lymphocytic Cholangitis, Hepatic Lymphoma, and Control Cases a
a Data with the same superscript symbol per row are significantly different; all P < .05. TCR, T-cell receptor; FISH, fluorescence in situ hybridization.
b n = 35 for lymphocytic cholangitis, n = 11 for T-cell lymphoma, n = 9 for control cases.
c n = 36 for lymphocytic cholangitis, n = 18 for hepatic lymphoma, n = 10 for control cases.
d Excludes 2 cases interpreted as contamination.
Among cats with hepatic lymphoma (n = 27), 18 were T-cell lymphomas (66.6%), 7 were B-cell lymphomas (25.9%) (Fig. 20 ), and 2 were non–B cell and non–T cell lymphomas (7.4%). Of the cases of lymphocytic cholangitis that displayed features of lymphoproliferation (n = 11), 4 had mixed T-lymphocyte and B-lymphocyte infiltrations (36.4%), 6 had T cell–predominant infiltrations (54.5%), and 1 had B-cell predominant infiltrations (9.1%). A lack of homogeneous immunophenotype in 4 lymphocytic cholangitis cases with lymphoproliferation was evidence against lymphoma. The 2 cases of lymphoproliferative lymphocytic cholangitis that had lymphoma in sites other than the liver had mixed T-lymphocyte and B-lymphocyte infiltrations.
TCR Clonality
PCR for TCR gene rearrangement clonality (Table 3) demonstrated a polyclonal product in 7 of 9 control cats (77.7%) and a pseudoclonal product in 2 of 9 control cats (22.2%). Of cats with lymphocytic cholangitis (n = 35), 29 (82.8%) yielded a polyclonal product (n = 24) or pseudoclonal product (n = 5), and 6 (17.1%) produced a clonal product (n = 5) or oligoclonal product (n = 1) (Fig. 21 ). Additional tissues from 5 of 6 cats with clonal or oligoclonal products showed no evidence of extrahepatic lymphoma (n = 3) and confirmed alimentary lymphoma (n = 2). Because of a lack of cellular atypia and the presence of a heterogeneous lymphocytic population, coupled with the lack of homogeneous immunophenotype, the liver lesions were diagnosed as lymphocytic cholangitis rather than lymphoma. Nevertheless, subtle infiltration of neoplastic lymphocytes cannot be discounted. Seven of eleven lymphocytic cholangitis cases that displayed features of lymphoproliferation had TCR PCR performed, and all 7 had polyclonal products. Of the 11 cases histologically diagnosed with hepatic T-cell lymphoma, 7 (63.6%) had a clonal TCR product (n = 5) or oligoclonal TCR product (n = 2). Four of eleven T-cell lymphoma cases (36.3%) produced a polyclonal (n = 2) or pseudoclonal (n = 2) TCR product. TCR clonality was performed on 3 cases of B-cell lymphoma and 2 cases of non–B cell, non–T cell lymphoma, and all produced a polyclonal TCR product. Cats with T-cell lymphoma had a significantly higher prevalence of clonal versus polyclonal TCR products, compared with cats with lymphocytic cholangitis (P = .006) and control cats (P = .02).
Eubacterial FISH
Eubacterial FISH (Table 3) detected bacterial organisms in 2 of 10 control cats. In 1 control cat with multiple biliary cystadenomas, 3 small clumps of bacteria were identified in the hepatic parenchyma. In another control cat with immunohistologically confirmed feline infectious peritonitis, a single sinusoidal clump of bacteria was identified. No cats diagnosed with hepatic lymphoma were FISH positive (0 of 18), and only 4 of 36 with lymphocytic cholangitis (11.1%) were FISH positive. The latter cats had organisms identified on biopsy section margins (wedge biopsy, n = 3; Tru-cut biopsy, n = 1), suggesting that these were contaminants. In 2 other cats with lymphocytic cholangitis (2 of 36, 5.5%), very low numbers of bacterial organisms were identified on the biliary epithelium (n = 1) or scattered between hepatocytes (n = 1). There were no significant differences in the number of FISH-positive cases between the groups. No organisms were identified with spiral morphology consistent with Helicobacter species.
Discussion
Lymphocytic inflammatory disease involving zone 1 in the cat remains an enigmatic syndrome. This study aimed to further characterize the lymphocytic cholangitis syndrome by defining histopathologic features, immunohistochemistry, and clonality of infiltrating lymphocytes and to determine whether eubacteria might play an etiopathogenic role. Ambiguous lymphoproliferative cases were deliberately selected for study inclusion to determine if immunohistochemistry and TCR clonality could help differentiate hepatic lymphoma from florid lymphocytic cholangitis. Definitively differentiating the syndromes of hepatic lymphoma and lymphocytic cholangitis may require antecedent and subsequent clinical data, not considered in this study. Age distinction did not help in clinically differentiating these disease processes. Cats with lymphocytic cholangitis reported herein were substantially older than cats in several previously published reports. 6,7,23
As in previous studies, cats with lymphocytic cholangitis had a high degree of fibrosis and bile ductular proliferation. 7,8,32 The degree of fibrosis in cases of lymphocytic cholangitis was highly variable among cats. Unexpectedly, the degree of fibrosis was also substantial in cats with hepatic lymphoma. Although a small but significantly higher degree of fibrosis was noted in cases of lymphocytic cholangitis compared with hepatic lymphoma, the difference was quantitatively small and thus unlikely to be diagnostically useful, with the exception that cats with hepatic lymphoma did not demonstrate severe bridging fibrosis. Recognizing peribiliary fibrosis in cats with hepatic lymphoma suggests either involvement of inflammatory mediators or cytokine release in antecedent inflammatory liver disease (ie, lymphocytic cholangitis). Hepatic fibroplasia encompasses a complex process primarily mediated by stellate cells in the space of Disse. Cytokines—specifically, platelet derived growth factor, tumor necrosis factor, interleukin-1, and transforming growth factor β—in conjunction with lipid peroxidation products produced by Kupffer cells, other inflammatory cells, hepatocytes, and bile duct epithelial cells, influence and drive the response. 9 Activation of stellate cells into a myofibroblastic phenotype is associated with enhanced formation and retention of excessive extracellular matrix. 9 Activated fibroblasts within zone 1 are the primary source of extracellular matrix deposition leading to peribiliary fibrosis. 9 Application of Masson’s trichrome allowed objective scoring of hepatic fibrosis that was not often appreciated on routine HE-stained sections. Concentric rings of peribiliary fibrosis were observed in a high percentage of cats with lymphocytic cholangitis, consistent with chronic bile duct damage. This feature was moderately associated with the diagnosis of lymphocytic cholangitis, as previously reported. 6,28 However, it is notable that we did observe peribiliary fibrosis in all groups of cats (lymphocytic cholangitis, hepatic lymphoma, and control cases); thus, we conclude that chronic asymptomatic development of peribiliary fibrosis may be a common manifestation in cats and not unique to lymphocytic cholangitis.
Finding a marked association of bile ductular hyperplasia with lymphocytic cholangitis is consistent with previous descriptions. 6,7,11,28,33 However, bile ductular hyperplasia was moderate to severe in cats with lymphocytic cholangitis, as well as in cats with hepatic lymphoma, such that no significant difference was found between groups based on this feature. Consequently, differentiation between cats with lymphocytic cholangitis and those with hepatic lymphoma or paraneoplastic effects (eg, increased biliary pressure, aberrations of the peribiliary vascular plexus, stimulation of cholinergic or dopaminergic nerves 22 ) was not possible with this morphologic feature. We were surprised to discover that control cats demonstrated cross sections of bile ductules ranging between 2 and 6 per portal region. Although no studies to date have quantified the average number of bile ductules in portal areas of cats, it has generally been assumed that normal cats have 1 to 2 bile ductule profiles per portal region.
Cats with either lymphocytic cholangitis or hepatic lymphoma may demonstrate peripheralization of bile ductules to the margins of the portal regions, suggesting a pathophysiologic response to bile ductule destruction or disappearance and induction of oval cell hyperplasia. 18,31 This histologic feature has not been described in cats with lymphocytic cholangitis, and it may serve as a marker of bile duct immunoinjury as lymphocytes target or “chase” bile ductules.
Bile duct targeting—as defined by lymphocytic infiltration within or immediately adjacent to the biliary epithelium with concurrent bile ductule epithelial swelling, necrosis, or attenuation—was detected in approximately 60% of lymphocytic cholangitis cases. This feature was significantly and strongly associated with lymphocytic cholangitis (when compared with control and hepatic lymphoma cases), and it appears to be diagnostically useful. This feature has been inconsistently recognized in other reports of lymphocytic cholangitis. 6 –8,11,23 In one report, bile ductule targeting occurred only in sections demonstrating suppurative inflammatory reaction, leading the authors to suggest ascending suppurative cholangitis as the underlying cause. 23 In the present study, a suppurative inflammatory process was observed in only 1 cat with bile ductule–targeting lesions, and this case had no observable bacteria on FISH.
Ductopenia, the absence of formed ductules in greater than 30% of portal areas, was unique to cats with lymphocytic cholangitis. Loss of bile ducts has been only briefly mentioned in a single prior report. 23 Ductopenia was often patchy in distribution within and between liver lobules, and ductopenic portal triads often had relatively reduced lymphocytic infiltrates, compared with intact portal triads. In humans, ductopenia characterizes a number of diverse syndromes, including primary biliary cirrhosis, vanishing bile duct syndrome associated with idiosyncratic drug reactions, primary sclerosing cholangitis, and a paraneoplastic phenomenon in Hodgkin lymphoma. 2,17 Common to these syndromes is that ductopenia reflects immune-mediated necroinflammatory bile duct destruction. 2 Cholestasis, a sequela of ductopenia, has been classically described to be associated with canalicular bile retention. However, we noted canalicular bile in only a single ductopenic cat, despite the fact that many of the cats are jaundiced at the time of liver biopsy acquisition (data not shown). Finding ductopenia in a subset of the lymphocytic cholangitis supports bile ductule destruction associated with an immune-mediated pathogenesis. Overall, our findings suggest that lymphocytic bile ductule targeting reflects an immune-mediated pathogenesis.
Finding aggregates of foamy macrophages (lipogranulomas) in portal regions was specific to lymphocytic cholangitis. Lipogranulomas, more commonly observed in liver tissue from older dogs compared with cats, represent a residual lesion marking cell death. As such, lipogranulomas within the adventitia of the portal triad may reflect cholangiolar destruction, especially in light of the fact that some of these harbor residual cytokeratin-positive cellular debris. We thus proffer that finding lipogranulomas within the confines of the portal triad will help discriminate lymphocytic cholangitis from hepatic lymphoma.
Our data corroborate previous findings that lymphocytic cholangitis is predominantly a T cell–mediated inflammatory disorder. 7 In approximately 70% of cats with lymphocytic cholangitis, T cells intimately encircle biliary ductules and infiltrate the biliary epithelium. In most cats, B cells were randomly dispersed among dense T-cell populations, with occasional formation of aggregates or early follicles. Similar B-cell aggregates have been described in cats with progressive lymphocytic cholangitis/cholangiohepatitis. 7 Although B-cell follicle formation is known to follow chronic immune stimulation, none of the aggregates identified in the present study formed germinal centers. Because this feature was strongly associated with the diagnosis of lymphocytic cholangitis, it may assist in discriminating lymphocytic cholangitis from hepatic lymphoma. However, its presence in approximately 20% of hepatic lymphoma cases means that it cannot be used as a single criterion to rule out a diagnosis of hepatic lymphoma. Approximately 30% of cats with lymphocytic cholangitis maintained a mixed B cell–T cell infiltrate, with approximately equal proportions of each cell type. Finding such a mixed but balanced population of lymphocytes may indicate differences in etiopathogenesis or disease stage relative to cats with T cell–predominant infiltrates. No significant difference was noted between the immunophenotype of the infiltrating lymphocytes (T cell predominant vs mixed T cell and B cell vs B cell predominant) and the severity of the portal inflammatory infiltrate. Continued study of cats with lymphocytic cholangitis will be necessary to more fully investigate the relevance of pleotrophic lymphocyte populations.
We confirmed that normal cats maintain a relatively small number of predominantly T lymphocytes within their portal triads, as previously reported. 7 Although these cells have not been definitively characterized, it is possible that they represent natural killer T cells (NKT cells), as present in human liver, 20 which harbors the largest population of NKT cells in the body. 20
Eleven cases classified as lymphocytic cholangitis had florid lymphocytic proliferations that we classified as lymphoproliferative on the basis of routine HE staining. Each case was ultimately classified as being predominantly inflammatory after observation of bile ductule targeting (n = 10) and portal B-cell aggregates or follicles (n = 8), clarification that the inflammatory infiltrate contained both B cells and T cells (n = 4), and discovery of polyclonal TCR (n = 7). Thus, an originally ambiguous diagnosis was definitively classified using adjunctive diagnostic features (immunohistochemistry and TCR clonality). Two lymphoproliferative lymphocytic cholangitis cases had lymphoma in extrahepatic sites. Both these cases had approximately equal proportions of T cells and B cells in their hepatic infiltrates, as well as polyclonal TCR products. We cannot discount that lymphoma had subtly integrated into a histologically inflammatory lesion.
We hypothesized that TCR clonality would definitively differentiate inflammatory versus neoplastic infiltrate in all cats. However, we discovered a subset of cats with apparent inflammatory disease (based on light microscopy) that had clonal or oligoclonal T-cell populations, and 4 cats with a histologic diagnosis of hepatic lymphoma had a polyclonal or pseudoclonal TCR product. Two hepatic lymphoma cats that had polyclonal TCR products had concurrent alimentary lymphoma. T-cell polyclonality can occur in lymphoma if rarely expressed Vγ or Jγ gene segments are expressed in neoplastic cells, if major chromosomal abnormalities exist, or if the neoplastic clone represents only a subset of the lymphocyte population within an infiltrating cell population. 26 Two oligoclonal cats with lymphocytic cholangitis had a confirmed alimentary T-cell lymphoma that may have subtly (histologically) integrated into hepatic tissue.
The protocol used for TCR PCR followed published methodologies applied to feline intestinal biopsies where polyclonal products were routinely documented. 26 Several considerations may explain our unexpected TCR findings in cases of lymphocytic cholangitis. First, a restricted diversity of responding inflammatory T cells in the liver could represent a clonal or oligoclonal population. This scenario has been confirmed in humans with Crohn disease in response to viral infections and in inflammatory reactions to certain neoplasias (particularly in aged individuals). 13,14,24 A pertinent example is that an oligoclonal CD8+ T-cell population has been characterized in human patients with chronic hepatitis C virus infection. 24 Second, the liver harbors a large number of NKT cells, and these cells have a restricted TCR diversity. 20 Similarly, because enteric intraepithelial T cells have oligoclonal TCR β-chain repertoires, it is possible that similar cells constitute the intraepithelial lymphocytes in bile ducts and that these dominate the inflammatory infiltrate in feline lymphocytic cholangitis. 16 Fourth, it is possible that cats with lymphocytic cholangitis may have clonal or oligoclonal T-cell populations that truly reflect an emerging lymphoma, as recognized in enteropathy-type intestinal T-cell lymphoma associated with refractory sprue in humans. 5
To investigate the potential that bacteria may instigate initial inflammation in feline lymphocytic cholangitis, we evaluated liver tissue from the cats using eubacterial FISH. We found that most cats (> 90%) with lymphocytic cholangitis had no detectable bacteria. However, 2 cats had small numbers of bacteria at the interface of biliary epithelium (n = 1) or scattered between hepatocytes (n = 1). One of these cats had a history of gall bladder dilation, but patency of the common bile duct was demonstrated. It is possible that the bacteria attached to the biliary epithelium represented an ascending infection secondary to intermittent or chronic bile stasis. Finding only exceptional cases with bacterial infection is similar to our previous study of Helicobacter species in cats with lymphocytic cholangitis 12 where the pathophysiologic significance of finding bacteria in the liver of a restricted subset of cats remains unclear. Overall, our findings do not support an active role of bacterial pathogens in lymphocytic cholangitis. However, we cannot discount an etiologic role of bacteria in initiating a portal inflammatory response in the absence of in situ colonization. It is also possible that bacterial components or products, rather than viable bacteria, are involved in the genesis of bile duct targeted injury. 15 Recent theories regarding the pathogenesis of inflammatory bowel disease argue that commensal enteric bacteria with high virulence stimulate an overly aggressive T-cell response, initiating a chronic inflammatory reaction. 27 To date, no cases of feline suppurative cholangitis/cholangiohepatitis have been documented to progress to lymphocytic cholangitis (S.C., unpublished). Evidence provided by the present study supports that an immune-mediated process is a major mechanism of feline lymphocytic cholangitis, and findings do not implicate a primary bacterial pathogenesis.
Regarding the terminology used throughout this article, lymphocytic cholangitis reflects nomenclature proposed to describe the feline syndrome involving nonsuppurative cholangitis/cholangiohepatitis. 32 Data presented herein justify modification of current terminology to more accurately reflect the histopathology, pathophysiology, and current understanding of this feline syndrome. Findings confirm that portal inflammation involves a variable mix of T lymphocytes and B lymphocytes, as well as macrophages, and, importantly, varying degrees of ductal immunotargeting and expansion of inflammatory lesions into the hepatic parenchyma, features not adequately reflected in the older terminology of lymphocytic cholangitis. Furthermore, subsets of lymphocytic infiltrates—some T cell predominant, some B cell predominant, and some with a mixed B-cell and T-cell population—suggest potential for etiopathogenic subtypes. Although the syndrome cause remains undetermined in the majority of affected cats, there remains uncertainty in the relationship between chronic neutrophilic cholangitis and lymphocytic cholangitis. We propose that nonsuppurative cholangitis–cholangiohepatitis better reflects pathologic processes characterized for this syndrome where cholangitis progresses as a continuum to cholangiohepatitis. As we continue to closely examine disease syndromes in domestic species, it is important to integrate emerging pathophysiologic features into proposed terminologies.
This study represents the largest peer-reviewed case series of feline lymphocytic cholangitis providing rigorous histologic and immunohistochemical characterization, lymphocyte clonality, and eubacterial probing. In addition to citing the typical features of portal lymphocytic infiltrations, biliary hyperplasia, peribiliary fibrosis, and portal-to-portal bridging fibrosis, we have characterized a progressive syndrome of bile duct targeting that can lead to generalized ductopenia. Through examination of biopsies collected from several liver lobes, we have documented a wide variation in inflammatory lesions, which demonstrates the need for the collection of liver biopsies from more than a single lobe to best characterize the histologic features. Furthermore, the variation of lesions within a single liver lobe argues for collection of wedge or laparoscopic biopsies, given the small size of needle samples and the inherent risks associated with automatic Tru-cut liver sampling in cats. 4,29 Our data demonstrate that bile duct targeting, ductopenia, peribiliary fibrosis, B-cell lymphoid aggregates, and lipogranulomas within portal regions are useful but imperfect features that may aid in distinguishing lymphocytic cholangitis from hepatic lymphoma on routine HE sections. We suggest a minimal staining panel to include routine HE, Masson’s trichrome, and immunohistochemistry for the identification of feline T cells and B cells, as well as biliary epithelial cytokeratin for best syndrome characterization. In this study, TCR clonality was useful as an adjunct diagnostic procedure in differentiating inflammatory and neoplastic hepatic lymphoid infiltrates (incorporating histopathologic and immunophenotypic data), but it was less useful as a single parameter. With wider application of the measures reported herein, we look forward to development of a more rigorous set of criteria for the differentiation of feline hepatic lymphoma or lymphoproliferative syndrome from florid lymphocytic cholangitis.
Footnotes
Acknowledgements
Special thanks are given to Dr Kent Hecker in the Faculty of Veterinary Medicine at the University of Calgary for his statistics advice. Our gratitude to the staff at the Cornell University Histology Laboratory—specifically, Marlene Nardi, Joy Cramer, and Casey Morgan for their exceptional work on the histologic staining and immunohistochemistry.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This work was funded by the Cornell University College of Veterinary Medicine Dean’s Fund for Clinical Excellence program and the Cornell Feline Health Center.
