Abstract
In contrast to dogs, the role of copper and iron accumulation in feline hepatic disease remains poorly characterized. Therefore, the objective of the current study was to compare the amount and distribution of copper and iron accumulation for different disease processes in feline liver biopsies. Liver biopsies (from 104 privately owned cats) were categorized by primary histopathologic lesion. Copper (by rubeanic acid) and iron (by Prussian blue) accumulation were graded by amounts (0–3) and location (centrilobular, midzonal, periportal, random). The Kruskal–Wallis test and Pearson chi-square test were used to assess differences in metal grade and location, respectively, between diagnostic categories. Histologic diagnoses were normal (
Introduction
Although copper and iron are important for normal cellular function, hepatic accumulation of excess copper and iron may contribute to ongoing hepatocyte loss, inflammation, and fibrosis as a result of oxidative damage. 16 Copper-associated hepatopathies may be divided into primary copper-associated hepatitis, due to inborn errors of copper metabolism and excretion, and hepatopathies that result in secondary copper accumulation. 16 Copper-associated hepatopathies are poorly characterized in cats. Primary copper-associated hepatitis has been reported in a 2-year-old Siamese cat 5 and a 2-year-old European Shorthair cat. 14 In both cases, copper accumulation was localized to the centrilobular region, and liver copper concentrations exceeded 2,000 ppm dry weight. The Siamese cat died acutely 4 days after liver biopsy. No other cause for death was identified on necropsy. 5 The European Shorthair cat was euthanized at the time of biopsy due to the severity of gross lesions and presence of ascites. 14 Primary copper-associated hepatitis has also been reported in a 2-year-old Abyssinian cat and 0.5-year-old Somali cat, though clinical findings and outcome were not reported. 9 Mild copper accumulation secondary to experimentally induced extrahepatic biliary obstruction has been reported in cats. 3 Although quantitative copper concentrations (mean of 269 μg/gm on a dry weight basis) were increased compared to baseline (mean of 67 μg/gm), rubeanic acid staining was negative, so the histologic distribution of copper accumulation could not be determined. Finally, periportal copper accumulation secondary to cholangiohepatitis has been noted anecdotally. 4
Iron-associated hepatopathies in dogs may occur secondary to chronic hepatic inflammation. 16 The role of excess hepatocellular iron in the progression of feline hepatopathies is currently unknown.
Successful treatment of metal-associated hepatopathies is dependent upon early diagnosis, requiring a heightened index of suspicion by the attending clinician and performance of special stains and quantitative metal analysis. The purpose of the current study, therefore, was to determine the prevalence of copper and iron accumulation in feline liver biopsies. The hypothesis for the present study was that copper and iron accumulation would differ in biopsies for cats with normal liver histology, inflammatory liver diseases, and noninflammatory hepatic disease processes. Objectives of the current study were to characterize histologic changes in feline liver biopsy samples using the World Small Animal Veterinary Association (WSAVA) liver standardization group guidelines, 24 determine the amount and distribution of copper and iron in these samples using semiquantitative grading schemes, and compare the amount and distribution of copper and iron accumulation for different hepatic disease processes.
Materials and methods
Sample collection
Feline liver biopsies submitted to the Pathobiology Service at the University of Tennessee College of Veterinary Medicine (Knoxville, Tennessee) between July 1, 2007, and June 30, 2009 were eligible for inclusion in the study. Cases were excluded if liver tissue was not present on hematoxylin and eosin–stained slides or if tissue blocks were not available for performance of special stains. Sections were obtained from tissue blocks and stained according to standard laboratory protocols with rubeanic acid and Prussian blue.11,20 Additionally, Masson trichrome staining was performed according to standard laboratory protocols on sections from samples positive for hepatocellular copper accumulation. All sections were 4 µm thick.
Histologic evaluation
Slides were independently reviewed by 2 board-certified anatomic pathologists (Newkirk, Reel) and categorized based on the predominant lesion present and according to published criteria. 24 Slides receiving discordant diagnoses were reviewed jointly to achieve consensus. The initial diagnostic categories used were: no significant findings, hepatocellular glycogen accumulation, hepatic lipidosis, cholangitis, portal hepatitis, hepatocellular carcinoma, cholangiocellular carcinoma, other carcinoma, lymphoma, biliary cyst, feline infectious peritonitis (FIP), peritonitis, portal vein hypoperfusion consistent with portosystemic shunt (PSS), and telangiectasia. Glycogen and lipid were differentiated based on histologic appearance. Glycogen accumulation was diagnosed based on the presence of lacey vacuoles that did not displace cell nuclei. Lipid accumulation was diagnosed based on the presence of clear, discrete vacuoles that, in severe cases, displaced cell nuclei. Final diagnostic groups included normal liver, congenital, infectious and/or inflammatory, neoplastic, and other liver pathology.
Grading of copper and iron
Slides stained with rubeanic acid and Prussian blue were examined (Newkirk) to determine the extent of copper and iron accumulation, respectively. The Barton semiquantitative histological grading system for iron 1 assesses the frequency of iron staining in each zone of the liver but does not address the amount of staining. This grading system was modified and simplified to reflect the total amount of staining and the zone in which most of the staining occurred. The amount and distribution of Prussian blue staining was graded separately in the hepatocytes and the sinusoids. Unlike the Barton grading system, staining in the connective tissue in portal areas was not assessed. Previously published copper grading systems7,21 graded the amount of copper staining on a scale of 0 (none) to 5 (abundant). This copper grading scheme was modified and simplified to match the iron grading system utilized. For hepatocellular carcinomas, staining was assessed in non-neoplastic hepatocytes only.
The amount of staining was graded on a scale of 0 to 3 (0 = no staining; 1 = mild staining; 2 = moderate staining; 3 = marked dense staining). The distribution of staining was categorized as centrilobular (CL), midzonal (MZ), periportal (PP), and random (R) based on the predominant pattern. Random distribution included those specimens where staining was diffuse and those specimens where there was not sufficient tissue to make an accurate assessment of zonality (3 tru-cut needle biopsies).
Statistical analysis
Differences in copper and iron accumulation between the WSAVA disease classification groups were assessed using the Kruskal–Wallis test. Means ± standard deviation (SD) were calculated for iron scores by diagnostic category. Means ± SD were not calculated for copper scores due to the small number of positive samples. Associations between disease classification groups and the locations of copper and iron occurrences were tested using the Pearson chi-square test. Significance for all tests was determined at values of
Results
Histologic samples
Meeting the inclusion criteria were 104 feline liver samples. Of these, adequate tissue was available for performance of all tests in 103 cases. One sample collected using a tru-cut biopsy needle had inadequate tissue available within the block for iron evaluation.
There were 12 samples with no significant findings on hematoxylin and eosin–stained sections. Sixteen samples contained neoplasia, including hepatocellular carcinoma (1), cholangiocellular carcinoma (5), other carcinoma (2), and lymphoma (9). Thirty-nine samples had evidence of inflammatory or infectious disease, including cholangitis (14), portal hepatitis (23), and FIP (2). There were 6 samples with evidence of congenital defects, including PSS (5) and telangiectasia (1). There were 32 samples with other changes on histologic evaluation, including hepatocellular glycogen accumulation (12), hepatic lipidosis (16), and biliary cysts (4). The total number of diagnoses was greater than the total number of samples because 2 samples had multiple diagnoses. One sample had cholangiocarcinoma with lymphoma, and 1 sample had lymphoma with biliary cysts. For Kruskal–Wallis analysis, the sample with lymphoma and biliary cysts was placed in the neoplasia group.
Special stains
Rubeanic acid stain uptake was absent in 92 samples (88.5%), 5 samples (4.4%) had grade 1 hepatocellular copper accumulation, 6 samples (5.7%) had grade 2 accumulation, and 1 sample (1.0%) had grade 3 accumulation. Histologic diagnoses (and location of copper staining) for positive samples were congenital (1 CL), infectious and/or inflammatory (7: 2 CL, 1 MZ, 2 PP, 2 R), neoplastic (2 PP), and other (2 CL). None of the samples had sinusoidal copper accumulation. Final diagnoses and special stain results for these samples are summarized in Table 1.
Final histologic diagnoses and results of special stain analysis for each sample with positive copper staining.*
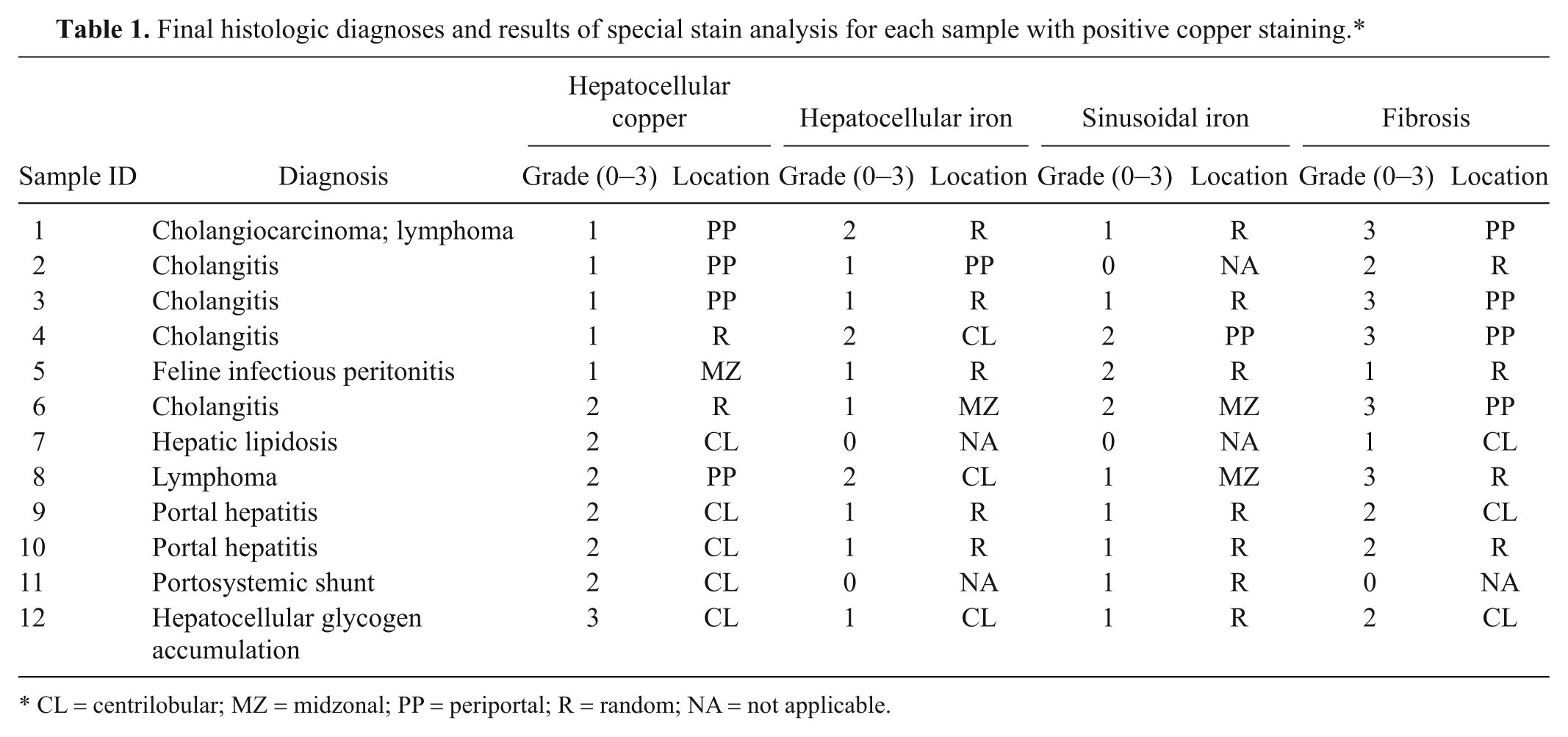
CL = centrilobular; MZ = midzonal; PP = periportal; R = random; NA = not applicable.
Hepatocellular uptake of Prussian blue stain was absent in 17 samples (16.3%), and 1 sample could not be evaluated due to tissue quality. There were 38 samples (36.5%) with grade 1 iron accumulation, 40 samples (38.5%) had grade 2 accumulation, and 8 samples (7.7%) had grade 3 accumulation. Hepatocellular iron distribution was CL in 38 samples (36.5%), MZ in 13 samples (12.5%), PP in 2 samples (2%), and R in 33 samples (31.7%). Iron scores and distribution by diagnostic category are summarized in Table 2. There were no significant differences in the amount of hepatocellular iron accumulation for the different disease categories, and no association between the location of iron accumulation and disease category was detected.
Mean and standard deviation iron scores and distribution by diagnostic category for 104 feline hepatic biopsies.*
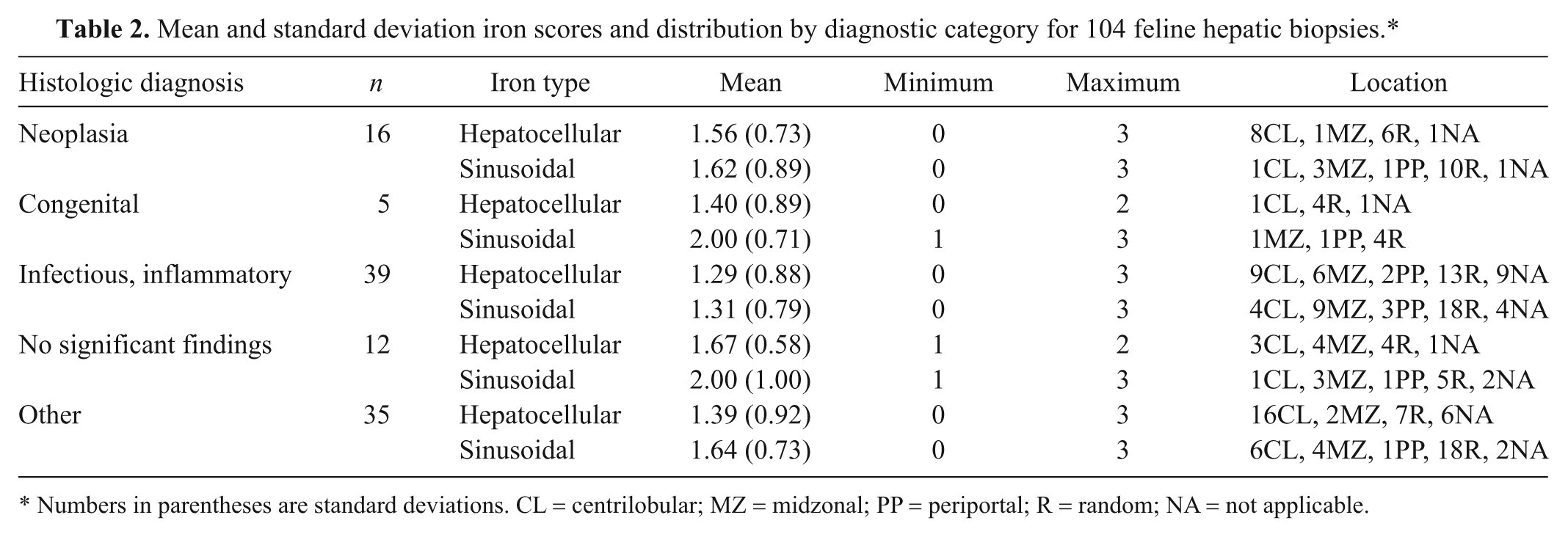
Numbers in parentheses are standard deviations. CL = centrilobular; MZ = midzonal; PP = periportal; R = random; NA = not applicable.
Sinusoidal accumulation of iron was absent in 9 samples (8.7%), and 1 sample could not be evaluated due to tissue quality. Grade 1 accumulation was present in 44 samples (42.3%), grade 2 was present in 39 samples (37.5%), and grade 3 was present in 11 samples (10.6%). Sinusoidal iron distribution was CL in 12 samples (11.5%), MZ in 20 samples (19.2%), PP in 7 samples (6.7%), and R in 55 samples (52.9%). The amount of sinusoidal iron accumulation did not differ significantly between the different disease categories. Furthermore, the location of iron accumulation and type of disease were not associated.
Of the 12 samples with histologically normal livers, none were positive for copper by rubeanic acid. Hepatocellular iron accumulation was present in 11 out of the 12 samples. Accumulation was grade 1 in 5 samples, grade 2 in 5 samples, and grade 3 in 1 sample. Sinusoidal iron accumulation was present in 10 samples. Six samples had grade 1 sinusoidal iron accumulation, and 4 samples had grade 2 accumulation.
Discussion
In the current study, copper accumulation was uncommon (11.5%) but less rare than anticipated based on the limited data available from other reports.3-5,9,14 Grade 2 to 3 hepatocellular copper accumulation was present in 6.7% of feline hepatic biopsies evaluated, primarily with a centrilobular pattern of distribution. Hepatocellular iron accumulation occurred commonly (82.7%), and grade 2 and 3 accumulation was present in 50% (6/12) of histologically normal livers. This suggests that higher hepatic concentrations of iron may be normal for domestic cats compared with dogs, though further evaluation is necessary to conclude this definitively. No associations were identified between histologic diagnosis category and the grade or distribution of copper or iron accumulation.
Copper-associated hepatopathies may be divided into primary copper-associated hepatitis, due to inborn errors of metabolism and copper excretion, and hepatopathies that result in secondary copper accumulation. 16 Genetic bases for primary copper-associated hepatitis have been determined for the Bedlington Terrier 10 and the Labrador Retriever. 6 Copper-associated hepatopathies have also been described in West Highland White Terriers, Skye Terriers, Dalmatians, and Doberman Pinschers, though genetic bases and causality remain undetermined for these breeds.12,22 In dogs with primary copper-associated hepatitis, copper accumulation is primarily located in hepatocytes and Kupffer cells in the centrilobular zone. Bedlington and West Highland White Terriers are relatively resistant to the effects of copper and rarely develop inflammation until copper concentrations are extraordinarily high (1,000−2,000 ppm dry weight), while in Doberman Pinschers, copper concentrations as low as 600 ppm are associated with histologic changes. 13
Feline copper-associated hepatitis was first reported in a 2-year-old female spayed Siamese cat that presented with nonspecific signs of malaise and increased liver enzymes. 5 Hepatic work-up was not pursued, and the cat re-presented 39 days after initial examination in hepatic failure. The patient died acutely a few days after liver biopsy and before treatment could be initiated. In a second case report, a 2-year-old male neutered European Shorthair cat presented for nonspecific signs of fever and hyporexia, was diagnosed with cirrhosis at exploratory laparotomy, and then euthanized prior to final diagnosis. 14 In both cases, necropsies were performed, and copper accumulation was found to be localized to the centrilobular region, with liver copper concentrations exceeding 2,000 ppm dry weight. Reference intervals for feline hepatic copper concentration for the laboratories used ranged from 26 to 188 ppm dry weight.5,14 Copper-associated hepatitis has also been reported in a 2-year-old male intact Abyssinian cat and a 0.5-year-old male intact Somali cat, though clinical findings and outcome were not reported. 9
Prognosis for dogs with primary copper-associated hepatopathies varies with stage of disease at diagnosis; dogs diagnosed while still asymptomatic have a much better outcome. It seems reasonable that stage of disease at diagnosis and severity of copper accumulation would similarly impact on prognosis for cats. One of the authors (Whittemore) has documented copper-associated hepatitis in 2 Sphinx cats with chronic malaise, hyporexia, and marked increases in alanine aminotransferase activities. Both had centrilobular copper accumulation with associated inflammation and fibrosis on hepatic biopsy. Both cats were less affected than cases reported in the literature, based on age at diagnosis (3 and 4 years, respectively) and quantitative copper concentrations (436 and 548 ppm dry weight basis, respectively). They were clinically normalized with medical management, and repeat biopsies in 1 cat documented resolution of inflammation with plateau in fibrosis and lobule collapse.
Hepatic copper accumulation may also occur in dogs secondary to ongoing hepatic inflammation and cholestasis.17,19 Increased amounts of copper were found in 29 out of 101 dogs with primary hepatitis, suggesting that accumulation of hepatic copper may be more common than previously thought. 15 With secondary copper accumulation, hepatocellular copper is primarily localized to the periportal region. Treatment is focused on correction of the primary hepatopathy and nonspecific reduction in copper and iron absorption through dietary modification and zinc administration.
In the current study, hepatocellular copper accumulation was documented in 12 out of 104 samples. Periportal or random accumulation was noted in 6 samples, 4 of which were diagnosed with cholangitis. One sample, diagnosed with FIP, had midzonal accumulation. Centrilobular copper accumulation was noted in 5 samples, all of which had grade 2 or 3 accumulation. Histologic diagnoses for these samples were variable, though centrilobular inflammation was not noted. Fibrosis varied in severity for samples with copper accumulation but had a similar pattern of distribution based on Masson trichrome staining, suggesting that copper-associated hepatocellular damage may occur without evidence of significant histologic inflammation in cats. In Doberman Pinschers with subclinical copper-associated hepatitis, the predominant histologic findings were centrilobular copper-laden hepatocytes with apoptotic hepatocytes; inflammation in these cases was slight. 12
Iron homeostasis is primarily achieved through regulation of intestinal absorption. 23 Absorption of non-heme iron, found in plant sources, is influenced by a variety of dietary factors and iron status in the body. In contrast, absorption of heme iron, found in hemoglobin and myoglobin, is not influenced by body stores; absorption is enhanced in the presence of dietary meat. 23 Once absorbed, iron is stored in hepatocytes in a protein-bound ferric state that has minimal oxidative impact. 2 Excess hepatocellular iron overwhelms this process, generating free radicals and resulting in oxidative damage.
Iron-associated hepatopathies in dogs usually occur secondary to chronic hepatic inflammation. 16 Increased hepatic iron concentrations have been reported following experimental induction of liver disease using orally administered carbon tetrachloride. 18 Hepatocellular iron accumulation greater than or equal to grade 2 is associated with disease progression in dogs. 19 In the current study, grade 2 and 3 iron accumulation was present in 38.5% and 7.7% of samples, respectively. Additionally, grade 2 or 3 accumulation was present in half of the histologically normal liver samples. This suggests that feline hepatocytes may either be more resistant to the oxidative effects of excess iron or may have a greater capacity for iron storage than canine hepatocytes, perhaps due to the cat’s status as an obligate carnivore.
There were a number of limitations to the present study. The indications for hepatic biopsy, final clinical diagnoses, and outcome were unknown for the majority of cases. Because liver biopsies were submitted from a number of practices, biopsy technique, sample quality, the number and location of lobes sampled, and additional testing were variable. Additionally, quantitative metal analyses were not available for most samples. For optimal case evaluation, both quantitative and semiquantitative assessment of metals should be performed in cases where metal accumulation is a concern. To prevent bias toward an association, histologic diagnoses were based strictly on interpretation of hematoxylin and eosin–stained sections, independent of special staining results. This may have altered the histologic diagnoses for some cases. Finally, failure to identify an association between histologic diagnosis and copper accumulation may reflect type 2 error given the small number of samples with copper accumulation.
In conclusion, out of 104 feline liver samples, hepatocellular and sinusoidal iron accumulation was quite common and not associated with histologic diagnosis. Presence of grade 2 to 3 hepatocellular iron in half of the histologically normal liver biopsies suggests that iron accumulation is unlikely to be associated with disease progression in cats. Hepatocellular copper accumulation was more common in cats than previously reported, had a similar pattern of distribution to fibrotic changes, and was not present in histologically normal liver biopsies. Further evaluation of hepatocellular copper accumulation in cats with known clinical signs, biochemical results, quantitative metal analyses, treatments, and outcomes may be warranted.
Footnotes
Acknowledgements
Preliminary results were presented as a research abstract at the American College of Veterinary Internal Medicine Annual Meeting in Denver, Colorado, 2011.
a.
SAS/STAT software, Version 9.2, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by a grant from the Companion Animal Fund, University of Tennessee College of Veterinary Medicine.
