Abstract
Gammaretroviral vectors are an efficient means to effect gene therapy. However, genotoxicity from insertion at nonrandom sites can confer a competitive advantage to transduced cells, resulting in clonal proliferation or neoplasia. Six pig-tailed macaques (Macaca nemestrina) underwent total body irradiation and reconstitution with autologous stem cells genetically modified by a gammaretroviral vector overexpressing HOXB4. Two animals were euthanized owing to irradiation- or transplantation-associated toxicity, whereas the other 4 had successful reconstitution. Of the 4 macaques with successful reconstitution, 1 has no long-term follow-up information; 1 was euthanized owing to infection with simian varicella virus infection 18 months post–total body irradiation; and the 2 others are described herein as case Nos. 1 and 2. After being stable for 3 years, case No. 1 developed pancytopenia and petechiation, and after 2 years of stability case No. 2 developed anemia and thrombocytopenia. Despite therapy, the animals deteriorated and were euthanized. Gross findings included emaciation; case No. 1 also had hemorrhage, peritonitis, and cholecystitis. Histologically, bone marrow was hypercellular with predominately blast cells of all hematopoietic lineages, though with myeloid predominance, and with maturation arrest and blast cell dysplasia (myelodysplasia). Myelodysplasia was likely from a combination of insertional mutagenesis by the retroviral vector and overexpression of HOXB4. Consequences of myelodysplasia included the blood dyscrasias and, in case No. 1, hemorrhage, bacterial cholecystitis, hepatitis, and peritonitis.
Gene therapy holds tremendous promise for therapy of a variety of diseases, and retroviral vectors are one of the most efficient means to effect such treatments1–3,6. Gammaretroviral vectors in particular have demonstrated efficacy to cure genetic disease such as SCID (severe combined immunodeficiency) and chronic granulomatous disease1–3,6. Despite early successes, a significant percentage of patients treated with retroviral vector gene therapy and cured of SCID subsequently developed leukemia owing to retroviral-induced insertional mutagenesis. Further investigations into possible genotoxicity of retroviral vectors suggested that insertion of the retroviral vector at specific, nonrandom sites may confer a competitive advantage to transduced cells, which can eventually result in clonal proliferation or neoplasia.1–3,5,7,10
As part of a cohort of 6 pig-tailed macaques (Macaca nemestrina), 2 juvenile male pig-tailed macaques (case Nos. 1 and 2) were total body irradiated with 800 cGy and then transplanted with autologous stem cells that had been genetically modified by a gammaretroviral vector expressing HOXB4 (homeobox protein B-4), as previously described. (Case No. 1 was animal K03290 and case No. 2, animal K00339, in Zhang et al.)
HOXB4 is a homeobox gene that encodes a DNA-binding protein whose overexpression can expand hematopoietic stem cells. As such, HOXB4 is a candidate for therapeutic hematopoietic stem cell expansion. Overexpression of HOXB4 was documented in case Nos. 1 and 2, indicating successful insertion. 9 Of the 6 macaques in the above-described cohort, 2 were euthanized because of irradiation- or transplantation-associated toxicity, whereas the other 4 had successful reconstitution. Of the 4 macaques with successful reconstitution, 1 has no long-term follow-up information; 1 was euthanized owing to infection with simian varicella virus infection 18 months post–total body irradiation; 4 and the 2 others are described herein.
Case No. 1 was clinically stable for 3 years, at which point a routine blood collection identified anemia and thrombocytopenia, which progressed to neutropenia and eventually pancytopenia. Case No. 2 was clinically stable for 2 years, after which routine blood collection identified anemia and thrombocytopenia. Therapy over a number of weeks in both cases included numerous plasma-rich and whole blood transfusions, broad-spectrum antibiotics, and nutritional support. The animals did not respond to therapy, and euthanasia was elected.
Prominent gross findings included poor body condition; case No. 1 also had multicentric hemorrhage, including the gastrointestinal tract and abdomen (Fig. 1), fibrinous to fibrous peritonitis, and hepatomegaly with fibrosing cholecystitis.
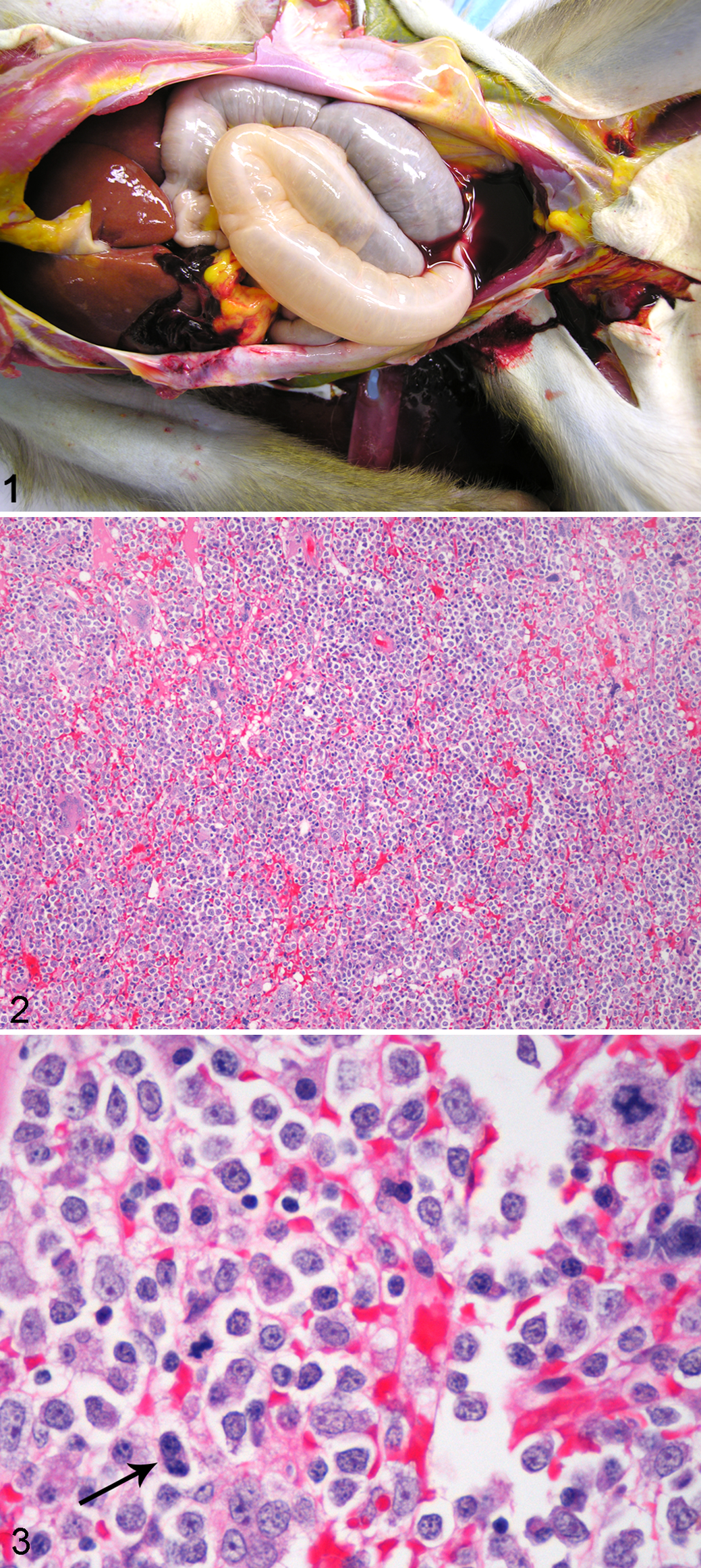
Tissue and organ samples were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 3 to 5 μm, and stained routinely with hematoxylin and eosin. Histology in both cases identified highly cellular bone marrow composed of predominately immature hematopoietic cells of all lineages, though with a predominance of myeloid blast cells (Figs. 2, 3). Blast cells sometimes exhibited irregular, large nuclei with scattered irregular mitotic figures, and occasional binucleated cells (Fig. 3). Maturation arrest was present in all lineages; however, rare maturation was seen in the erythroid lineage. Additionally, there was multifocal single-cell necrosis/apoptosis. In case No. 1, other prominent histologic findings included transmural chronic active cholecystitis associated with Gram-positive cocci, secondary chronic active hepatitis and peritonitis, multicentric hemorrhage, and systemic lymphoid depletion. Both cases also had diffuse testicular atrophy. Finally, case No. 2 had extramedullary hematopoiesis present in numerous tissues and organs, including lymph nodes, spleen, thymus, liver, lungs, and adrenal glands.
Changes in the bone marrow; hyperplasia with maturation arrest and cellular dysplasia; are best classified as myelodysplasia. 8 Myelodysplasia can progress to leukemia, 8 which may have occurred if these animals were not euthanized. Indeed, case No. 2 was previously reported as developing leukemia based on in vitro antemortem analysis of bone marrow aspirates. 10 We believe that this animal is now better classified as developing myelodysplasia based on evaluation of all findings, including histologic examination of bone marrow. Myelodysplasia resulted in the described blood dyscrasias, which led to thrombocytopenia, anemia, and in case No. 1, leukopenia. In case No. 1, consequences of the blood dyscrasias resulted in multicentric hemorrhage and immunosuppression with development of opportunistic bacterial cholecystitis, hepatitis, and peritonitis. In both cases, testicular atrophy were chronic consequences of total body irradiation.
In case No. 2, previously published data on stem cell analyses from bone marrow aspirates, obtained antemortem during the clinical phase of demise and evaluated in vitro, revealed, in addition to overexpression of HOXB4, dysregulated expression of oncogenes and a block in myeloid differentiation. 10 Additionally, knockdown of HOXB4 in a leukemic canine cell line that was also overexpressing HOXB4 restored myeloid differentiation in vitro, suggesting that overexpression of HOXB4 was critical to induction of the blood dyscrasias. 10 In case Nos. 1 and 2, multiple integration sites were identified on in vitro evaluation of marrow stem cells. 10 These findings, in combination with the experimental history, clinicopathologic changes, and histologic changes, suggest that myelodysplasia in both cases was due to a combination of insertional mutagenesis from the retroviral vector and resultant overexpression of HOXB4. Finally, in the 4 macaques that had successful reconstitution with marrow cells genetically modified with a gammaretroviral vector to overexpress HOXB4, 9 2 animals (case Nos. 1 and 2) developed myelodysplasia.
Experience with animal models and humans undergoing retroviral-mediated gene therapy indicates that integration sites are nonrandom; yet, in most cases, the integration event itself is a neutral event. However, nonrandom integration can affect gene expression adversely, which results in a risk—namely, that altered gene expression, with or without subsequent mutations in transduced cells, can result in clonal expansion or neoplasia. This risk, of course, is significantly increased if the therapeutic gene incorporated in the viral vector can give a growth advantage to transduced cells, as with the gene-encoding HOXB4. As such, the design of retroviral vectors and therapeutic genes is critical to minimize these risks, and the use of animal models is crucial to evaluate the safety of gene therapy before human trials. Despite the adverse consequences of retroviral-mediated gene therapy as described in this report, the future of gene therapy still holds promise as methodologies continue to evolve to identify and ameliorate risks of nonrandom insertions and specific therapeutic genes.
Footnotes
Acknowledgements
We wish to thank Mac Durning, Trevor Pierce, and the veterinary and husbandry staff of the Washington National Primate Research Center for providing excellent technical support.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
This work was supported by the Washington National Primate Research Center (NIH/NCRR grant No. P51 RR000166) and the Fred Hutchinson Cancer Research Center (NIH grants Nos. HL53750, HL84345, DK56465, and DK47754).
