Abstract
A 4.5-year-old spayed female Great Pyrenees with hypothyroidism and hypoadrenocorticism had a slightly enlarged pituitary gland and bilaterally atrophic adrenal and thyroid glands. Lymphocytic adenohypophysitis and adrenalitis were found in which B lymphocytes and plasma cells dominated the adenohypophysitis but T cells dominated the adrenalitis. The thyroid gland had extensive follicular atrophy and collapse. The combination of primary hypothyroidism and hypoadrenocorticism resembles type II autoimmune polyendocrine syndrome or Schmidt syndrome in humans. Adenohypophysitis is rare in dogs and not reported in polyendocrine disease in animals.
Keywords
Polyendocrine disease is rare in dogs. Some cases resemble human type II autoimmune polyendocrine syndrome, or Schmidt syndrome, 3,7,10 a condition with 2 of the 3 major endocrine diseases (hypoadrenocorticism, hypothyroidism, and type I diabetes mellitus). 1,5 Adenohypophysitis is a rare autoimmune disease of humans 12 and is documented in rare cases of Schmidt syndrome. 9,11 There is only 1 report of adenohypophysitis in a dog with concurrent sebaceous adenitis but no other endocrine disorder. 8 In this report, we describe adenohypophysitis in a dog with a polyendocrine disease that resembled Schmidt syndrome.
History
A 4.5-year-old spayed female Great Pyrenees was presented to the Ontario Veterinary College Teaching Hospital for progressive weakness over 2 months. The dog had marked muscle atrophy of the hind limbs. Neurological examination revealed normal findings, and the result of the acetylcholine receptor antibody test was negative, ruling out myasthenia gravis. The pre- and postadrenocorticotrophic hormone stimulation cortisol levels were low (< 28 nmol/liter; reference interval [RI], 30–300 nmol/liter). Both thyroxine (T4) and free T4 were low (T4 < 13 nmol/liter; RI, 13–50 and fT4 = 3.2 pmol/liter; RI, 8–40), but thyroid-stimulating hormone concentration was normal (0.25 ng/ml; RI, 0.05–0.75). There was moderate normocytic normochromic nonregenerative anemia—hematocrit, 0.27 liter/liter (RI, 0.39–0.55); reticulocyte count, 35.6 × 109/liter (RI, 55–85)—and a neutrophilia with a left shift: neutrophils, 29.8 × 109/liter (RI, 2.9–10.6) and bands, 0.69 × 109/liter (RI, 0.0–0.3). The serum concentrations of sodium (148 mmol/liter; RI, 140–154) and potassium (4.4 mmol/liter; RI, 3.8–5.4) and the sodium:potassium ratio (Na:K, 34; RI, 29–37) were within normal limits. Based on these findings, the diagnosis was concurrent glucocorticoid-deficient hypoadrenocorticism and hypothyroidism. Because there was no clinical response to supplementation with prednisone and levothyroxine, the dog was euthanized.
Necropsy Findings
At necropsy, the adrenal glands were small (1.4 × 0.3 × 0.3 cm) and gray, with thin cortices and a 1:2 corticomedullary ratio (bilateral adrenocortical atrophy). Both lobes of the thyroid gland were small (2.0 × 0.75 × 0.3 cm) and light yellow (bilateral atrophy). The pituitary gland was slightly enlarged (0.5 × 0.4 × 0.4 cm). Other lesions included chronic degenerative joint disease of the stifles and aspiration pneumonia.
Histologic and Immunohistochemical Methods
Tissues were fixed in 10% neutral buffered formalin, routinely processed, and stained with hematoxylin and eosin (HE). Sections of the pituitary, adrenal, and thyroid glands were evaluated immunohistochemically to detect CD18 (cells of bone marrow origin), CD3 (T cells), and CD79a (B cells). Tissues from healthy adult dogs were used as controls. Polyclonal rabbit anti-human CD3 and monoclonal mouse anti-human CD79a were from DakoCytomation (Mississauga, Ontario, Canada), monoclonal mouse anti-dog CD18 (clone CA1.E9) was from the Leukocyte Antigen Biology Laboratory (University of California, Davis, California). Primary antibodies were omitted for negative controls.
Histologic Findings
Individual and clustered lymphocytes and plasma cells were scattered throughout the parenchyma of the pars intermedia and pars distalis of the anterior pituitary gland. Prominent perivascular lymphoid cell aggregates were at the periphery of the gland (Figs. 1, 2). Rare band neutrophils were identified (interpreted as migrating immature neutrophils). No cell death or fibrosis was seen. There were no lesions in the brain. The microglia and lymphocytes within the pituitary gland were CD18 positive. Most lymphocytes (70%) were CD79a-positive B cells, whereas fewer (30%) were CD3-positive T cells. B cells especially outnumbered T cells in the peripheral lymphoid cell aggregates (Figs. 3, 4). In healthy control dogs, individual CD18-positive microglia were present, but no CD3- or CD79a-positive cells were detected.
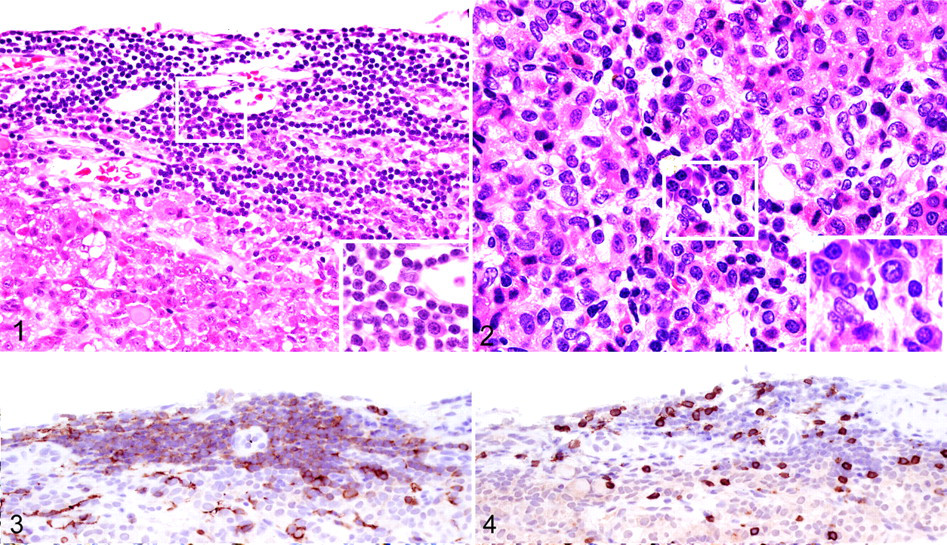
In the adrenal glands, the zonae fasciculata and reticularis were collapsed and almost completely replaced by numerous lymphocytes, plasma cells, and macrophages that contained fine granular golden-yellow pigment (interpreted as ceroid-lipofuscin). Epithelial cells of the zona glomerulosa were vacuolated. Most of the lymphocytes were T cells; B cells were rare.
In the thyroid gland, most follicles were small and irregular, with scant basophilic granular colloid. Rare follicles were enlarged up to 300 μm in diameter, distended by colloid, and lined by flattened epithelium (consistent with inactivity). The stroma was prominent, indicating follicular atrophy and collapse; 10% of the parenchyma was replaced by adipose tissue. No inflammatory cells were seen.
Other lesions included hepatic glycogenosis (attributed to prednisone therapy), necrosuppurative bronchopneumonia (consistent with aspiration and probably causing the neutrophilia), mild left atrioventricular valvular endocardiosis, and erythroid hypoplasia of the bone marrow (consistent with the nonregenerative anemia considered secondary to hypothyroidism and/or hypoadrenocorticism).
Discussion
Lymphocytes and plasma cells are not normally found in the pituitary gland. The lack of lesions in the rest of the brain indicates a primary inflammation of the pituitary gland and not part of encephalitis. We found 1 report of canine adenohypophysitis, involving a dog that had concurrent atrophic sebaceous adenitis but no other endocrine lesion. 8 The adenohypophysitis was also lymphocytic and associated with loss of pituitary gland parenchyma, of which the functional significance was not indicated. 8 Our case of adenohypophysitis was mild and may not have been functionally significant, owing to the presence of normal pituitary parenchyma and the results of the hormone assays. The many plasma cells and B lymphocytes may represent an antibody-mediated immunity against secreted products. However, serum concentration of thyroid-stimulating hormone, a major autoantigen in a rat model of autoimmune adenohypophysitis, 13 was within normal limits.
Primary hypophysitis in humans has 3 distinct histopathological subtypes: lymphocytic, granulomatous, and xanthomatous. 6 Lymphocytic adenohypophysitis eventually destroys the gland; disrupts the adrenal, gonadal, and thyroidal axes; and causes diabetes insipidus. 6 The condition is rare, with only about 100 cases reported, mostly in young women during late pregnancy or in the postpartum period. 12 It is also found with other immune-mediated diseases, including Hashimoto thyroiditis and adrenalitis. 6 In our case, noninflammatory thyroid atrophy, low serum thyroxine concentration, and normal serum thyroid-stimulating hormone concentration indicate an atrophic form of primary hypothyroidism, rather than one secondary to pituitary dysfunction.
The lymphocytic adrenalitis with sparing of the zona glomerulosa is consistent with primary glucocorticoid-deficient hypoadrenocorticism. All layers of the adrenal cortex are normally affected in the putatively immune-mediated canine primary hypoadrenocorticism. 4 The sparing of the zona glomerulosa in the present case is atypical and usually seen in secondary hypoadrenocortism caused by destruction of the pituitary gland or chronic glucocorticoid therapy. The presence of many T cells suggests an immune-mediated pathogenesis. Glucocorticoid-deficient hypoadrenocorticism has been observed in some cases of canine polyendocrine disease, 3,7 and lymphocytic adrenalitis with destruction of the zona fasciculata and reticularis was present in 1 case. 7 Autoimmune hypoadrenocorticism is the most common cause of primary adrenal gland failure in humans in the Western world, and patients often develop other autoimmune disorders, including Schmidt syndrome. 5
Autoimmune polyendocrine syndrome is diagnosed when there are multiple inflammatory and atrophic lesions of the endocrine glands. 1,5 The adrenalitis and thyroid gland atrophy seen in this case are like autoimmune polyendocrine syndrome type II or Schmidt-like syndrome in humans, a condition with at least 2 of the 3 major autoimmune endocrine diseases (hypoadrenocorticism, hypothyroidism, and type I diabetes mellitus). 1,5 Other endocrine and nonendocrine disorders, including hypophysitis, are documented in rare cases of Schmidt syndrome in humans 9,11 but not in canine polyendocrine disease. 3,7,10 In humans, at least 7 autoimmune polyendocrine syndromes are recognized with different combinations of affected endocrine glands. 1,5 The diagnosis is often based on detection of autoantibodies against the affected endocrine glands. 5 Autoantibodies have been demonstrated in some cases of canine polyendocrine disease, 2 but the results are variable. 2,7 No tests for autoantibodies to endocrine glands were done in our case. The polyendocrine lymphocytic inflammatory and/or atrophic lesions, however, are consistent with an autoimmune pathogenesis.
To our knowledge, this is the first report of adenohypophysitis with polyendocrine disease in a dog. Additional cases of adenohypophysitis might be found if the pituitary gland is routinely examined in dogs with endocrine disorders.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
