Abstract
Inflammatory changes associated with periarticular pure gold bead implants were studied in dogs involved in a clinical trial investigating motor dysfunction and chronic pain owing to hip joint dysplasia and osteoarthritis. Gold beads were percutaneously implanted via a needle into different locations surrounding the greater trochanter of the femur. Nine dogs with implants were necropsied. In all examined animals, characteristic histologic lesions were observed in the tissue surrounding the gold implants—namely, a fibrous capsule composed of concentric fibroblasts intermixed with a variable number of inflammatory cells and a paucicellular innermost layer of collagen with a few fibrocyte-like cells in empty lacunae. Lymphocytes dominated the inflammatory infiltrate, with rarely observed macrophages present in close proximity to the implant site. No giant cells were observed. Immunohistochemistry showed mixed populations of lymphocytes, both CD3 positive (T cells) and CD79a positive (B cells), which in some cases formed lymphoid follicles. Diffuse inflammatory changes were present to a minor extent in the perimysium and surrounding fascia. The inflammation observed in dogs is similar to that observed with gold implants in humans. It is possible that the clinically beneficial effect of gold beads for chronic osteoarthritis depends on sustained localized inflammation with localized release of soluble mediators. The encapsulation of the implant by a paucicellular and poorly vascularized fibrous capsule may help prevent an exaggerated inflammatory reaction by sequestering the gold bead from the surrounding tissue.
The parenteral therapeutic use of gold salts (chrysotherapy) is well known in human medicine and has been used in cases of rheumatoid arthritis. 23 The mechanisms behind the anti-inflammatory effect of monovalent gold salts and its metabolites, such as Au3+, Au(CN)2 –, and metallic gold, is incompletely understood. 5 One proposed mechanism of action for Au+ is due to binding with thiolate and thioether ligands on proteins. 5 This is thought to inhibit presentation of complexed antigen to T cells, activity of certain transcription factors, and enzymes such as protein tyrosine phosphatase in T cells, protein kinase C in neutrophils and T cells, and proteins important for B cell differentiation. Au(CN)2 – is an effective inhibitor of the respiratory burst of neutrophils and monocytes; it also inhibits the proliferation of lymphocytes. 5
A more recent therapeutic application of gold is the implantation of gold beads directly into a painful anatomic region, such as the periarticular tissue of arthritic joints. This technique is derived from acupuncture and has been used in veterinary medicine for the past decade, largely as a treatment in canine hip dysplasia. 10 A recent double-blind, placebo-controlled clinical trial performed by Jæger et al showed that the performance of dogs with hip joint osteoarthritis significantly improved after receiving gold bead implants. 13
Although it is widely accepted that gold may have an anti-inflammatory effect, it may cause hypersensitivity and tissue inflammation.7,14,25 In vivo and in vitro studies have shown that metallic gold implants release gold ions in tissues and cell cultures as a result of interaction with oxidative agents most likely provided by macrophages.4,18 Some believe that the pro-inflammatory properties of gold can be attributed to ionized gold and its metabolites.4,5
The purpose of the present article is to investigate the tissue inflammatory response surrounding metallic gold beads implanted into dogs as a treatment for spontaneous osteoarthritis.
Material and Methods
Animals
From 2000 to 2004, 9 dogs into which gold beads had been implanted were subjected to postmortem examination (dog Nos. 1–9). All dogs had been part of a double-blind, placebo-controlled clinical trial involving 80 dogs with clinical and radiographic signs of hip joint dysplasia and osteoarthritis. In the clinical trial, the dogs received either gold bead implants (n = 38) or no implants (n = 42) 13 to evaluate the pain-relieving effect of gold bead implantation. The dogs included in this report (n = 9) were euthanized at the owners' request owing to unrelated disease (dog Nos. 1–4, 6, 7) or insufficient pain-relieving effect (dog Nos. 5, 8, 9). Twenty-nine of the treated dogs and all 42 of the control dogs from the clinical trial were unavailable for postmortem examination. Six unrelated dogs received for routine postmortem examination at the Department of Basic Sciences and Aquatic Medicine, Norwegian School of Veterinary Science, were included as controls for the current study (dog Nos. 10–15). Three control dogs (dog Nos. 10–12) had a clinical history of hip joint osteoarthritis, whereas the other 3 (dog Nos. 13–15) had no history of clinical hip joint problems. Table 1 presents details on age, gender, and breed of the dogs.
Dogs Used in the Study With Details on Main Findings of the Postmortem Examination
a Gold beads implanted around only the right hip joint, and only the right side was sampled for histology.
b Dogs necropsied as controls in the histopathologic investigation; not included in clinical trial.
Gold Bead Implantation
The gold bead implantation procedure has been described. 13 Briefly, small 24-karat gold beads (purity: 99.997% metallic Au, 2 mm length, 1 mm in diameter, mean weight of beads = 0.036 g; AS Nogusra, Oslo, Norway) were implanted bilaterally (unilaterally in dog no. 2) in 3 to 5 different acupuncture positions surrounding the greater trochanter of the femur. Before each implantation, the 24-karat gold beads were packed and autoclave sterilized along with a stiletto.
Implantation was performed with the dogs under general anesthesia, and the beads were inserted through the skin with long stainless steel needles (Hypojet 60 mm × 2.0 mm, 14 G × 2.5 in.) after standard surgical preparation of the skin. With the dog in lateral recumbency, the needles were inserted in a clockwise position at 3, 5, 7, 9, and 12 o’clock with the trochanter major at the center. At each position, needles were placed obliquely toward the joint capsule of the femoral head with fluoroscopic guidance. When all 5 needles were in place, 2 gold beads were dropped into each needle with a forceps and a funnel made from a 2-ml syringe. A stiletto with the same length as the needle pushed the gold beads just outside the needle tip to leave them close to the joint capsule.
Histology
Several tissue sections containing gold beads were collected from the joint capsules bilaterally. For comparison, additional tissue sections that did not contain gold beads were collected from adjacent areas, approximately 10 mm from the sites of the gold bead–containing samples. Tissue samples were collected from the heart, kidney, liver, and lung from 7 of the 9 treated dogs and from all 6 control dogs (dog Nos. 10–15). Tissue samples were also collected from one or both ileofemoral lymph nodes from dog Nos. 1–3, 5, 7, 12, and 13, as well as sections of periarticular muscle and connective tissue unilaterally from each control dog (dog Nos. 10–15). Further tissue collection was performed if macroscopic lesions unrelated to hip joint osteoarthritis were present.
Tissues were fixed in 10% buffered formalin. Gold beads were excised from the samples with a razor blade before being embedded in paraffin wax. Sections (5 μm) were cut and stained with hematoxylin and eosin. In addition, special stains—including Elastic van Gieson (ElvG), Martius scarlet blue (MSB), and periodic acid–Schiff (PAS)—were performed to examine the capsule for the presence of elastic fibers and collagen, fibrin, and glycoproteins, respectively. Berlin blue staining for hemosiderin was performed to evaluate draining of hemorrhage into the ileofemoral lymph node.
Immunohistochemistry
The primary antibodies used for labeling T cells and B cells were a polyclonal rabbit anti-human CD3 (DakoCytomation, Glostrup, Denmark) and a monoclonal mouse anti-human CD79αcy (HM57) (DakoCytomation), respectively. Immunolabeling was performed with the Envision+ kit (DakoCytomation). Sections (5 μm) of the formalin-fixed, paraffin-embedded tissue were collected on poly-L-lysine-coated slides. Slides were dried for 30 minutes at 58°C, followed by deparaffination in xylene at 58°C. The tissue was rehydrated in a series of decreasing concentrations of ethanol and kept overnight in distilled water. For better antigen retrieval, the sections were heated at 92°C for 10 minutes in Tris–EDTA buffer (pH, 9.1). Sections were then chilled in distilled water at 4°C. Sections were incubated in 3% H2O2 in methanol to inhibit endogenous peroxidase, followed by washing three times in Tris-buffered saline (TBS). To block nonspecific binding, the sections were incubated for 20 minutes in normal goat serum diluted 1:50 in 5% bovine serum albumin (BSA) in TBS. The blocking solution was tapped off before application of the primary antibodies. The primary antibodies were diluted 1:50 (CD79αcy) and 1:400 (CD3) in 1% BSA/TBS and incubated for 1 hour at 20°C. The sections were subsequently washed three times in TBS. Horseradish peroxidase–labeled, polymer-conjugated secondary antibodies from the Envision+ kit (goat anti-rabbit or goat anti-mouse) were diluted 1:50 in 1% BSA/TBS and incubated for 30 minutes. Labeling was visualized with AEC. Sections were counterstained with hematoxylin for 1 minute. For both antibodies, a section containing sheep Peyer’s patches was used as a positive control. Each run included a negative control in which the primary antibody was replaced by 1% BSA/TBS. Immunohistochemical evaluation included two different tissue sections with implant-related lesions from treated dogs and one tissue sample from each control dog.
Results
Necropsy
At necropsy, all dogs treated with gold beads had severe bilateral (dog Nos. 1, 3–9) or unilateral (dog No. 2) hip joint osteoarthritis. Hip joint osteoarthritis was characterized by a shallow acetabulum, moderate to severe thickening of the joint capsule, moderate to severe flattening of the femoral head, severe destruction of articular cartilage, osteophyte formation at the insertion of the joint capsule, and bilateral (dog Nos. 1, 3–9) or unilateral (dog No. 2) destruction of the teres ligament. Gold beads were found in periarticular adipose tissue, connective tissue, and muscle tissue. In muscle tissue, the beads were usually located immediately underneath the epimysium. The beads were encapsulated by a gray/white semitransparent tissue, 0.5 to 1.0 mm thick. The control dogs had no (dog Nos. 13, 15) or moderate (dog Nos. 10–12, 14) hip joint osteoarthritis. When present, the hip joint osteoarthritis in control dogs was characterized by osteophyte formation at the periphery of the articular surface of the femoral head. Any additional diagnoses are given in Table 1.
Histology
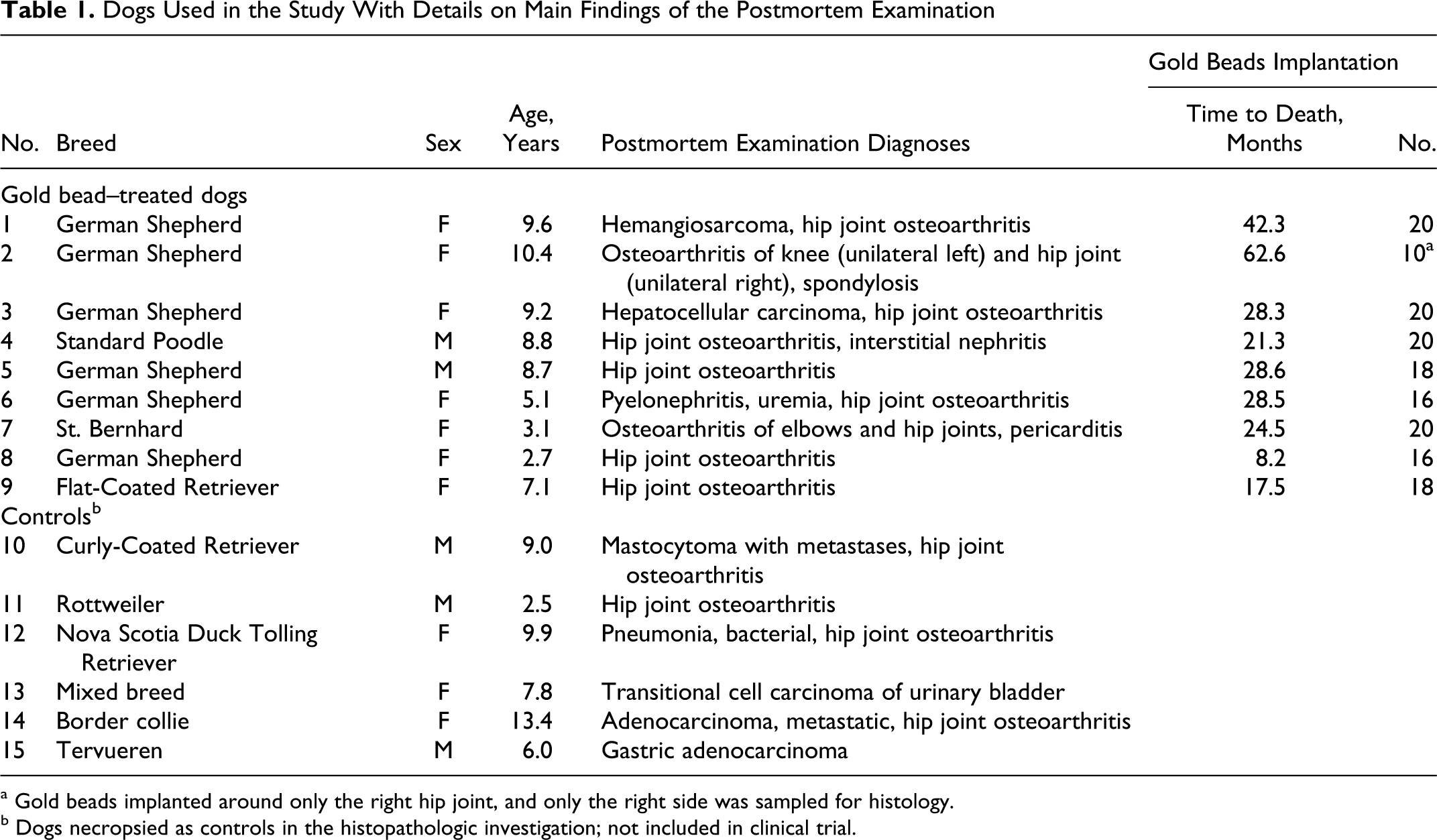
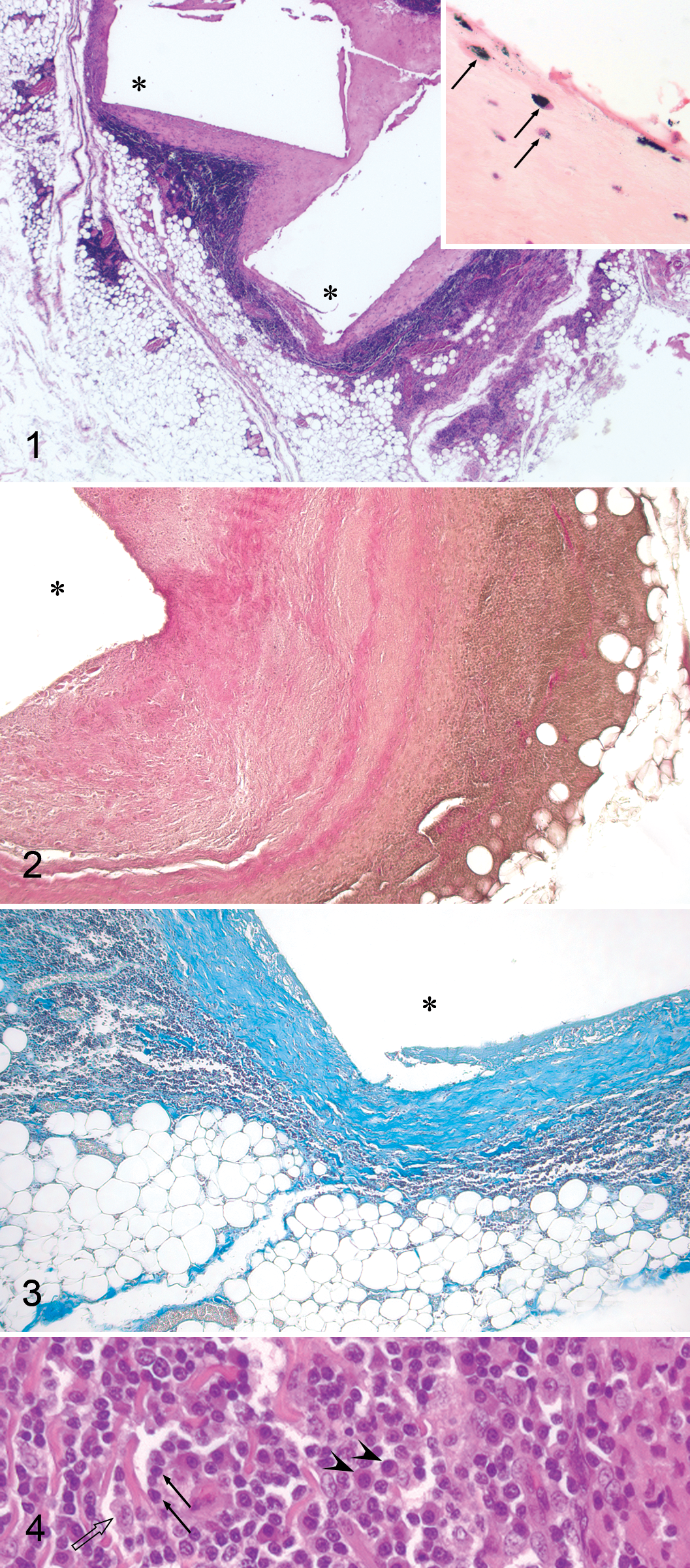
Cavities representing the site of gold beads were found in histologic sections from all tissues collected from implantation sites (Fig. 1). Cavities were outlined by a thick capsule (200–800 μm), and in all treated dogs (except dog Nos. 8 and 9), the innermost layer contained homogeneous, hyaline, slightly fibrillar eosinophilic material with few cells and no vessels (Fig. 1 inset). This eosinophilic extracellular material gave a weak collagen reaction in ElvG staining (Fig. 2) and a blue MSB positivity (Fig. 3). In ElvG-stained capsules with medium to low thickness, the material was diffusely pink. The thickest capsules had one or more layers of pink-staining collagenous material separated by irregular layers of amorphous to homogeneous, faintly stained pink to brownish material with a few indistinct pink fibrils (Fig. 2). No PAS-positive material was seen in the capsule.
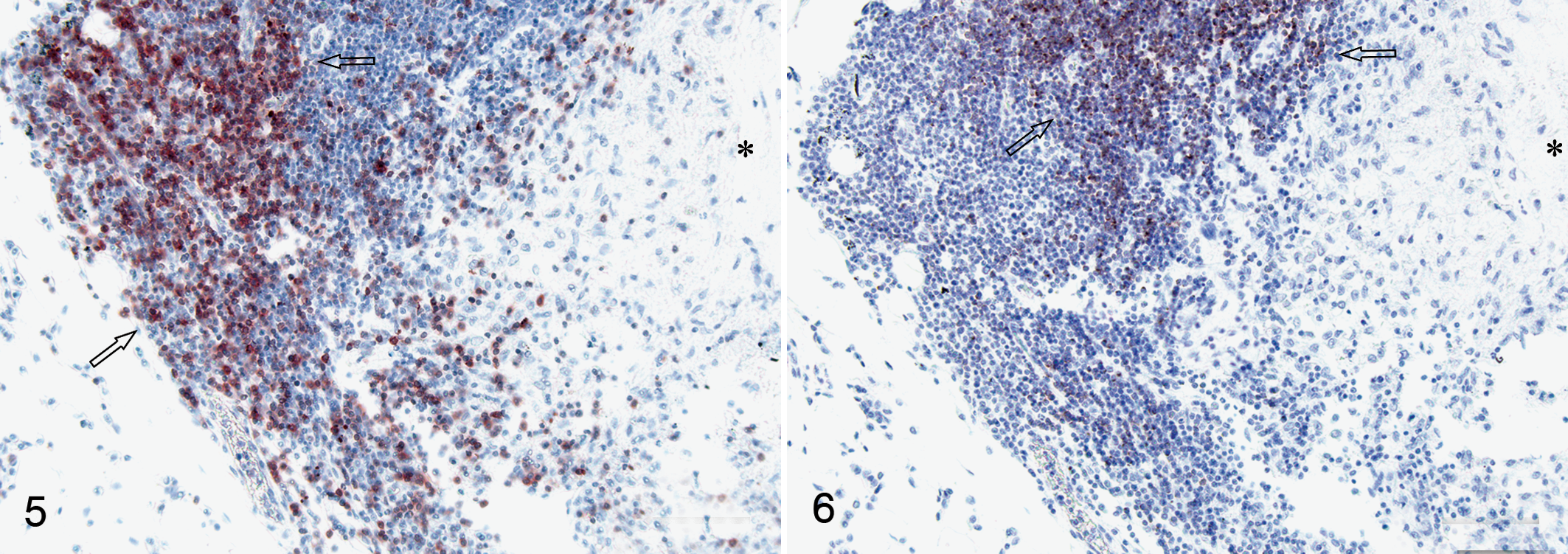
Periarticular tissue; dog No. 1. Labeled CD3+ cells in the pericapsular inflammatory infiltrate. Area with focally increased density of CD3+ cells are indicated (open arrows). Area of tissue closest to the gold implant is indicated by asterisk. Envision+ kit. Hematoxylin counterstain.
In some tissue samples, scattered fibrocyte-like cells were seen at the inner margin of the capsule; but in most samples, the inner margin of the capsule was paucicellular and only had rare cells with absent cellular details contained in lacuna-like spaces. A consistent observation was the presence of a layer of scattered fibrocyte-like cells at the outer margin of the paucicellular eosinophilic capsule. Bordering this layer and embedded in similar collagenous material was a 100- to 120-μm-thick layer of concentrically arranged fibroblasts containing a few macrophages and lymphocytes and surrounded by densely cellular infiltrates dominated by lymphocytes with fewer plasma cells and macrophages (Fig. 4) and scattered neutrophils and eosinophils. Giant cells were not observed. There was a significant variation in the magnitude of the inflammatory infiltrate (Figs. 2, 3), and the dense infiltration of lymphocytes usually created a layer 100 to 200 μm thick, but sometimes the layer extended for more than 1,000 μm from the layer rich in fibroblast-like cells. Lymphoplasmacytic inflammation was observed multifocally and perivascularly in adipose tissue, skeletal muscle, fascia, and occasionally, nerves farther from the gold bead cavity. In most samples, muscle fibers directly adjacent to implants showed atrophy and were replaced by adipose and fibrous tissue.
Dog No. 8, which had the shortest exposure to implants, did not have a layer of homogeneous eosinophilic material close to the implants, and the capsules were only 70 to 90 μm thick. Surrounding the capsules, dense lymphocytic infiltrates formed a layer approximately 400 to 500 μm thick. In tissue samples from dog No. 9, the capsules consisted of only a 40- to 50-μm layer of fibrous tissue surrounded by scattered lymphocytes. Some focal aggregates of lymphocytes, up to 30 μm in diameter, were observed.
In all available samples from the ileofemoral lymph nodes, macrophages containing hemosiderin were seen—a change that was similar in treated animals and controls. The histologic examination of heart, kidney, liver, and lung showed no consistent changes that could be attributed to the gold implants (data not shown). Periarticular skeletal muscle and fibrous tissue from control dogs did not contain the inflammatory lesions observed in the treated dogs. However, mild focal lymphohistiocytic infiltrates were found associated with random muscle fiber loss and degeneration in dog Nos. 10, 13, and 14. Similar changes were present in the samples that did not contain gold implants collected from the treated dogs.
Immunohistochemistry
Immunohistochemistry showed that CD3+ and CD79αcy+ lymphocytes were both present in the infiltrates associated with the implants and that the two cell populations were present in approximately equal numbers (Figs. 5, 6 ). The two cell populations tended to occupy separate parts of the inflammatory focus (Figs. 5, 6), showing an organization suggestive of lymphoid follicle formation. Scattered CD3+ cells were present within the areas of CD79αcy+ cells (Fig. 5). Scattered CD3+ cells, along with fewer CD79αcy+ cells, were also present in the soft tissue surrounding the capsule.
Discussion
This is the first work to describe long-term inflammatory cell responses after clinical use of gold bead implantation in dogs. The results show that gold beads caused chronic local inflammatory changes in all the treated dogs. Twenty-four-karat gold is generally considered to be inert and stable.11,21 However, it has been known for many years that the systemic administration of soluble gold salts may induce adverse immunopathologic reactions. The trivalent gold ion is a reactive intermediate metabolite formed by in vivo oxidation of Au+, and it is the metabolite most strongly associated with adverse reactions to gold salts. 5 The generation of Au+3 most likely takes place in phagocytic lysosomes owing to myeloperoxidases and other lysosomal enzymes. 5 Some adverse immune reactions are elicited by T-cell sensitization to the Au3+ ion itself. 5 Au3+ readily reacts with proteins and, possibly, lysosomal proteins causing permanent protein denaturation, which might sensitize T cells. 5 In mice, gold-induced autoimmunity is probably T cell dependent, but the underlying mechanism has not been established.8,9
The most common problems encountered in humans subjected to chrysotherapy are contact urticaria and glomerulonephritis, which are mediated via type I and type III hypersensitivity, respectively. 23 Furthermore, exposure to metallic gold in diverse alloys represents a risk of inducing immunopathology. In humans, hypersensitivity is known to occur through epicutaneous exposure to gold alloys and, in particular, when the alloys are kept in close contact with dermal tissue, as with piercings. 11
Adverse effects of metallic gold and gold alloys are clinically observed in human medical and dental practices.1,24 Twenty-four karat gold is considered unsuitable for some metal implant purposes because of its pliability. Therefore, few reports have addressed the biocompatibility of 24-karat gold but more commonly focused on high gold alloys and other clinically relevant alloys. 15 The precious and semiprecious metals currently used in dentistry include gold, palladium, and silver, whereas titanium is commonly used in orthopaedic implants. Nonprecious metals such as nickel, chromium, cobalt, and molybdenum are commonly included in alloys to increase resistance to corrosion, wear, and tear and to improve mechanical properties.6,19
Experiments in rats have shown that histologic changes around metal implants generally include edema, connective tissue proliferation, hyalinization, capillary proliferation, epithelial proliferation, plasma cell infiltration, lymphocyte infiltration, and giant cell infiltration. 15 In general, nonprecious alloys produce severe tissue reactions whereas precious metals produce mild reactions. 15
The inflammatory changes observed in the treated dogs in the present study were dominated by lymphocyte infiltration, which is in agreement with studies using gold implants in humans.1,25 A typical foreign body reaction with granulomatous inflammation and multinucleated giant cells was not observed in the treated dogs. This is in accordance with studies involving gold and high gold alloys in humans, rats, and dogs in which granulomatous reactions are rarely reported.1,25,26,30 Conversely, nonprecious metals and alloys of low gold content are reported to cause granulomatous inflammation in guinea pigs and rats.2,16
T lymphocytes and B lymphocytes were both prominent in the inflammatory infiltrates in the dogs. In contrast, two studies describing chronic inflammatory reactions to gold in humans found a clear predominance of either T cells or B cells.12,25 An interesting observation in the present study was the tendency of T cells and B cells to occupy separate regions in the lymphocytic infiltrates, resulting in some resemblance to lymphoid follicles. The de novo formation of lymphoid follicles at sites of chronic inflammation has been described17,27,29,31 and has been reported in lesions associated with inflammation caused by metallic gold in humans.12,22
The eosinophilic material found in the surrounding capsule closest to the implants most likely represents fibrous tissue. The initial host response to metal implants is formation of surrounding granulation tissue, which usually matures into a capsule that is largely collagenous but may resemble fibrocartilage if the implant is in relative motion and is in contact with bone or articular tissue. 3 Collagenous capsules around gold implants have been described in humans and dogs.1,30 The encapsulation of the gold beads by a paucicellular and poorly vascularized fibrous capsule probably helps impede the inflammatory reaction by isolating the metal implant from the surrounding tissue.
The homogeneous to amorphous material that stained weakly with ElvG and was present closest to the implants needs further examination, as well as do the interposing typical collagen fibers in the capsule. Bair et al reported an amorphous eosinophilic coagulum with small numbers of inflammatory cells on the tissue–implant interface, which may correspond to what we describe. 1 The material may represent degraded collagen.
There was no evidence of systemic adverse effects to the gold implants in the limited subset of tissue sampled. However, owing to the small number of animals and tissues examined, it is premature to draw a broad conclusion. Systemic effects or lesions at sites other than that of implantation have not been described in human studies. 11
The selection of 24-karat gold for the clinical trial was based on the notion that the acupuncture implants should be well tolerated by the host to avoid rejection and sequestration; 13 however, the assumption of the general inertness of metallic gold has been challenged by observations in medicine and odontology for some time and more recently in organic chemistry.11,21 Our findings show that all dogs receiving 24-karat gold developed a local inflammatory reaction around the implants. In this respect, our results differ from previous results in humans and experimental animals. Findings from human studies suggest that inflammatory reactions to gold implants occur in a limited number of patients; 28 yet, these human studies could be biased by the primary availability of specimens from cases where the implants had to be removed owing to complications. 25 Thus, little is known about the patients that do not have adverse clinical symptoms after implantation. In rats, the tissue reaction to gold has been reported to be not significantly different from that seen in controls. 15 Moreover, the duration of some animal experiments are quite short (2–3 months) compared with the period that some of the dogs in our study carried gold implants, which ranged from 8 months to 5 years.4,15,20 The strong inflammatory reaction in the treated dogs in the present study compared with that reported from some previous animal experiments could be due to a longer period of contact with gold. That dog Nos. 8 and 9, with the shortest exposure to gold implants, had the thinnest capsules and the mildest lymphocytic infiltrate is in agreement with this view.
The mechanism behind the clinically observed pain-relieving effect of multiple periarticular gold beads is unclear. 13 Both the direct impinging of the beads on the surrounding nerves, muscle, and fibrous tissue and the induced inflammation might mediate release of soluble factors that modulate the dogs' perception of pain. The role of inflammation in the pain-relieving effect of gold implants warrants further investigation. The findings of the present study suggest that this role could be addressed in studies based on implant material known to cause more inflammation than that of 24-karat gold.
Although inflammation was evident around the implants, the severity of the lesions was minor compared with the hip joint osteoarthritis present in all treated dogs. The lesions surrounding the implants must therefore be considered of secondary importance to the overall pathology of the hip region. The current general understanding of the biocompatibility of metal implants still advocates that gold of high purity should be the preferred material in procedures such as those used in this experiment.
Footnotes
Acknowledgements
We acknowledge Inger Rudshaug for skilful technical assistance with the immunohistochemistry.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The work was supported in part by grant Nos. NFR 123873/320 and NFR 141822/320 by the Research Council of Norway, Dyreidentitet AS, and the Norwegian School of Veterinary Science.
