Abstract
Spontaneous morbidity primarily affecting female breeders in 3 independent breeding colonies of NSG (NOD.Cg-Prkdcscid I12rgtm1Wjl /SzJ) mice prompted an investigation to uncover the cause of disease. Necropsies were performed on 264 (157 female and 107 male) spontaneously sick, experimentally unmanipulated NSG mice. In sum, 42 mice (15.9%) had acute or chronic renal inflammatory lesions, of which 12 had concurrent histologic evidence of an ascending urinary tract infection. From 94 kidneys cultured for bacterial organisms, 23 (24.5%) grew Enterococcus sp and 19 (20.2%) grew Klebsiella oxytoca. Female mice were twice more likely than males to present with nephritis. These findings indicate that bacterial nephritis is a major contributor to morbidity in the NSG strain.
NSG (NOD.Cg-Prkdcscid I12rgtm1Wjl /SzJ) mice carry several mutations that affect the immune system and render them severely immunodeficient. 1,8,9,16,17 Mutant mice combine the features of the NOD/ShiLtJ background, the severe combined immune deficiency mutation (scid), and an interleukin-2 (IL-2) receptor γ chain deficiency. NOD mice lack hemolytic complement because of a 2–base pair deletion in the C5 structural gene, and they carry a unique MHC haplotype, which leads to defective natural killer cell function and defects in the immunoregulation of antigen-presenting cells. 8,14,15 Homozygosity for the scid mutation results in a double-stranded DNA repair defect and a defect in the rearrangement of genes that code for antigen-specific receptors on lymphocytes. 4,5 As a result, scid mice lack detectable immunoglobulin M (IgM), IgG1, IgG2a, IgG2b, IgG3, and IgA. The IL-2 receptor γ chain, also known as the common cytokine receptor gamma chain, is shared with receptors for IL-4, IL-7, IL-9, IL-15, and IL-21. The null mutation in the γ chain of the IL-2 receptor leads to deficiencies in cytokine signaling and failure of clonal lymphocyte expansion. 1,9 All these mutations are combined in the NSG strain. As a result, NSG mice lack mature T cells and B cells, NK cells, and hemolytic complement, and they are deficient in cytokine signaling. Mice of the NSG strain are unable to mount an effective adaptive immune response to foreign organisms or cells and are therefore considered a superior, long-lived model suitable for studies employing xenotransplantation strategies. 13 In recent years, the NSG strain has become a popular tool in research. It has been engrafted with many normal and malignant human cell populations and tissues, resulting in “humanized mice,” given that the various types of human cells will retain their same tissue functions. The strain readily supports engraftment of human CD34+ hematopoietic stem cells, peripheral blood mononuclear cells, and human embryonic stem cells. 1,9,11,17 Morbidity and mortality of severely immunodeficient mice within breeding colonies occur in spite of diligent health-monitoring programs and in colonies maintained behind barriers known to exclude mouse-adapted pathogens. 12 Sporadic morbidity was seen at The Jackson Laboratory (TJL) NSG breeding colonies. In addition, we were contacted by 2 independent academic institutions maintaining NSG breeding colonies, both of which reported sporadic morbidity and mortality in their NSG breeders, primarily affecting breeding females older than 200 days. This article describes the pathologic findings of the clinical investigation that ensued.
Materials and Methods
Sick NSG mice were examined from 3 breeding colonies maintained at TJL, either in Bar Harbor, Maine, or Sacramento, California. Mice were housed in a positive, individually ventilated rack, and the bedding used in the mouse boxes was either white pine shavings (Crobb Box Co, Hancock, Maine), in the Bar Harbor colonies, or Beta Chip (Nepco, Warrensburg, NY), in the Sacramento colony. Owing to their severe immunodeficiency, production colonies of NSG mice at TJL are housed behind a maximum-level barrier. Their health is routinely monitored with immunocompetent sentinel mice through the implementation of statistically valid sampling procedures and recognized serological and molecular testing protocols. The maximum-level barrier excludes a defined list of viral, bacterial, fungal, and protozoal organisms: ectromelia virus, Theiler mouse encephalomyelitis virus, Hantaan virus, K virus, lactic dehydrogenase elevating virus, lymphocytic choriomeningitis virus, mouse adenovirus, mouse cytomegalovirus, mouse hepatitis virus, mouse minute virus, mouse norovirus, mouse parvovirus, mouse thymic virus, pneumonia virus of mice, polyoma virus, reovirus 3, rotavirus, Sendai virus, Bordetella bronchiseptica, cilia-associated respiratory bacillus, Citrobacter freundii, Clostridium piliforme, Corynebacterium bovis, Helicobacter spp, Klebsiella pneumonia, Mycoplasma pulmonis, Pasteurella pneumotropica, Pneumocystis murina, Pseudomonas spp, Salmonella spp, Staphylococcus aureus, Streptobacillus moniliformis, Streptococcus spp, Encephalitozoon cuniculi, Trichomonas, Giardia, Spironucleus. In addition, all fleas, lice, mites, pinworms, round worms, and tapeworms are excluded. “Sick” mice were submitted to TJL diagnostic service in Bar Harbor, Maine, or to the Comparative Pathology Laboratory (CPL) at the University of California at Davis for necropsy between June 2007 and May 2009. A total of 226 mice (135 females and 91 males) were submitted, ranging in age from 23 to 537 days. Criteria for submission were clinical assessment of a sick mouse, including any of the following signs: hunched posture, dehydration, ruffled hair coat, weight loss, and/or the presence of a grossly visible tumor. From all 3 breeding colonies at TJL, less than 1% of breeding animals were found to be sick and were submitted for pathology evaluation. Complete necropsy was performed on all mice, and microbiologic cultures were performed on selected tissues presenting gross lesions. Organ samples were cultured at TJL on Columbia agar with 5% defibrinated sheep blood (Northeast Laboratory Services, Waterville, ME) or at CPL on Tryptic Soy Agar with 5% sheep’s blood (Hardy Diagnostics, Santa Maria, CA). Additional tissue samples were cultured at TJL on MacConkey agar (Difco, Sparks, MD) for 24 hours at 37°C (directly or after enrichment in tryptose phosphate at 37°C for up to 1 week) or at CPL on MacConkey agar (Hardy Diagnostics, Santa Maria, CA) for up to 72 hours at 37°C. Plates were processed every 24 hours with biochemical methods for identification of bacterial colonies. For specimens requiring more extensive identification, Analytical Profile Index testing system was used (BioMerieux, Marcy l’Etoile, France). Tissues collected for histology were fixed in Tellyesniczky/Fekete fixative (100 ml 70% ethanol, 5 ml 37 to 40% formalin, 5 ml glacial acetic acid) or 10% buffered formalin. Tissues were paraffin embedded and stained with hematoxylin and eosin, and selected slides were stained with Brown and Brenn, a tissue gram stain. In addition, fixed tissues or glass slides representing 38 sick NSG mice (22 females and 16 males) from 2 independent academic institutions maintaining NSG breeding colonies were sent to us and included in this study.
Results
Most sick NSG mice presented with hunched postures and scruffy hair coats but were otherwise alert and responsive. Nineteen mice appeared normal and 8 mice were submitted moribund in lateral recumbency. Gross necropsy revealed mottling of the kidneys in 36 (13.6%) mice. Furthermore, 42 mice (15.9%) had histologic evidence of acute or chronic renal inflammatory lesions, of which 11 (26.2%) had bilateral renal lesions (Table 1 ). Overall, 34 mice had chronic lesions and 8 had acute lesions. Chronic lesions were characterized by multifocal interstitial fibrosis with tubular degeneration and loss. No inflammatory cells were associated with these areas in any of the mice. In addition, kidneys presenting chronic lesions often had radiating segmental fibrosis, suggesting chronic infarcts; however, no thrombi were evident within the vasculature. Acute lesions were characterized by bacterial colonies scattered throughout the renal interstitium and within the distal and proximal tubules but rarely within tubular epithelial cells. Bacterial colonies were accompanied by minute numbers of neutrophils (Fig. 1 ). No lymphocytic or plasmacytic infiltrates were present in any of the affected kidneys. Twelve mice with renal lesions had an ascending urinary tract infection, with few neutrophils scattered along the lower urinary tract and male accessory reproductive glands. Inflammation was minimal but included cystitis, prostatitis, and seminal vesiculitis. Three of these mice had bacteria colonizing their lower urinary tracts, morphologically similar to the bacteria found in their kidneys. All other mice with nephritis had no lesions in the lower urinary tract, as well as no lesions indicating an alternate route of bacterial infection. Seven mice with nephritis had concurrent histologic evidence of bacteremia, with bacterial colonies found in the heart (Fig. 2 ) and/or liver and lungs. In all tissues, bacterial colonies appeared to proliferate undisturbed within the parenchyma and did not seem to provoke an adaptive immune response (Fig. 3 ). Renal microbiologic cultures were performed on 94 mice, of which 23 (24.5%) and 19 (20.2%) had positive growth of Enterococcus sp and Klebsiella oxytoca, respectively. All kidneys with positive bacterial cultures had a corresponding histologic renal lesion, and Brown and Brenn stains performed on selected sections were consistent with bacterial culture results. Forty-eight cultures had negative growth and 4 had mixed bacterial growth. Female mice were twice more likely than male mice to develop nephritis (19.7% and 10.0%, respectively). However, the mean age of presentation with nephritis was almost identical for males and females (210.0 and 217.2 days, respectively). Tumor profiles (unpublished data) included mammary adenocarcinomas, osteosarcomas, and lymphomas at frequencies and age of onset compatible with other NOD-derived strains. 15
Incidence of renal and lower urinary tract bacterial infections in NSG mice and renal culture results.
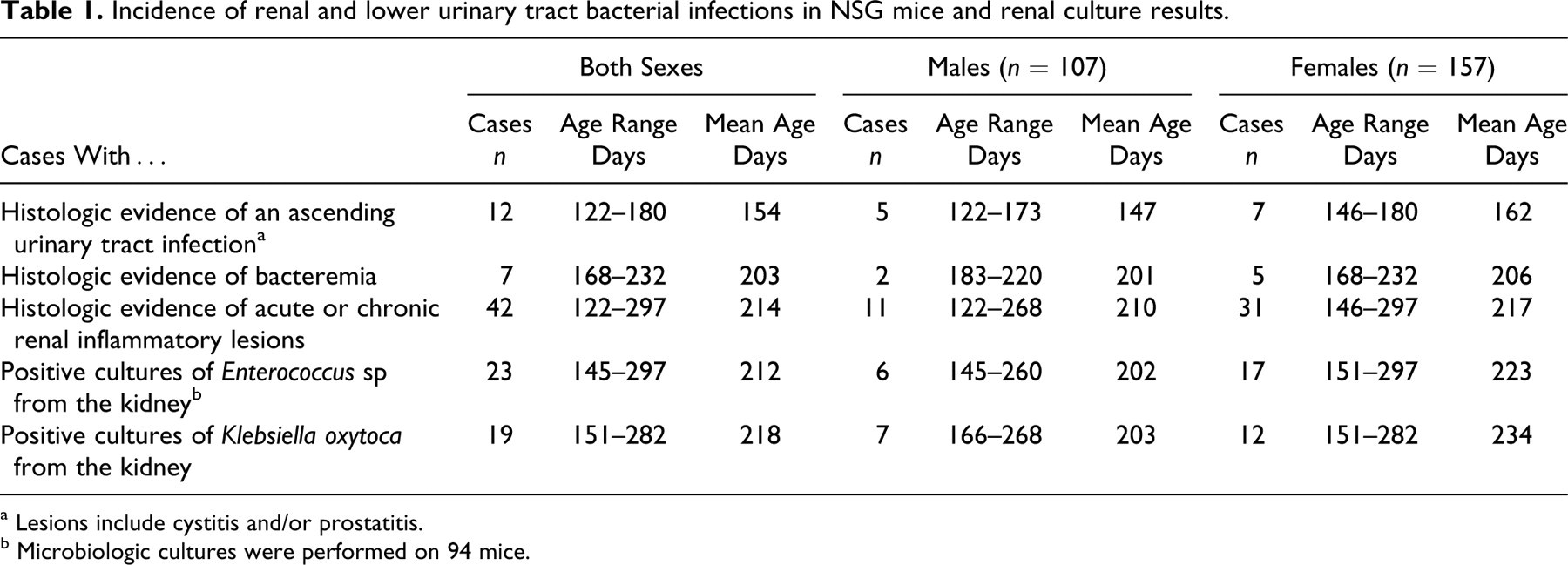
a Lesions include cystitis and/or prostatitis.
b Microbiologic cultures were performed on 94 mice.
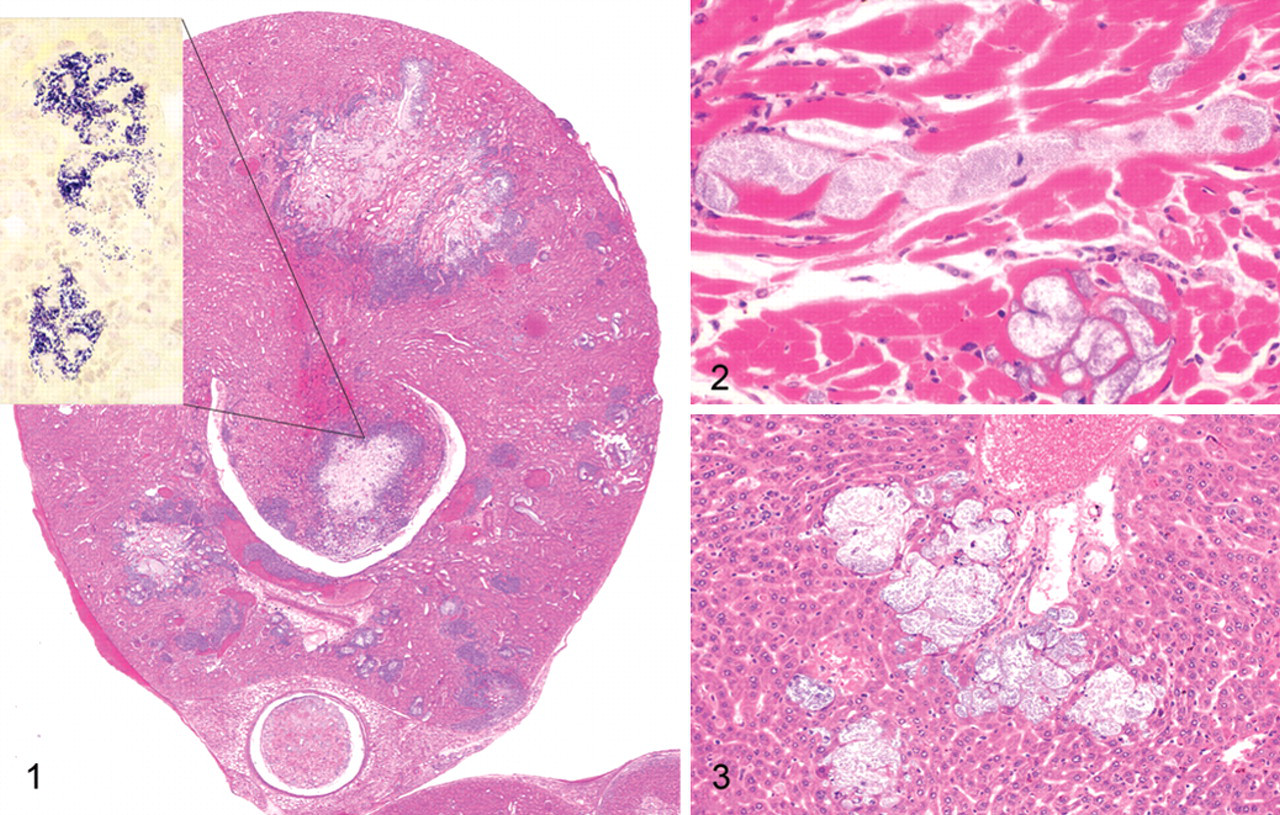
Kidney; NSG mouse No. 1. Acute multifocal parenchymal necrosis. HE. Inset: Renal papilla, clusters of cocci bacteria. Brown and Brenn.
Discussion
Lesions were caused by 2 bacterial organisms and were most consistent with an ascending urinary tract bacterial infection. Out of 42 mice with nephritis, 12 mice (28.6%) had a concurrent lower urinary tract infection, with 3 mice having bacterial colonies along their lower urinary tract morphologically similar to the bacteria found in their kidneys. From our experience with the NSG strain, skin wounds and the molar gingival sulcus often serve as port of entry for bacteria, which will disseminate to other organs. In all 42 mice diagnosed with nephritis, no lesions were noted in the gingiva or skin. These findings support the hypothesis that opportunistic bacteria colonized the lower urinary tract and ascended to the kidneys leading to chronic bacterial nephritis. K oxytoca and Enterococcus organisms are not considered primary mouse pathogens, and both organisms are often isolated from the gastrointestinal tract of healthy immunocompetent mice. 3 Enterococci are facultative anaerobic gram-positive cocci and are viewed as a commensal organism in the intestines of mice. Enterococci are frequently cultured from the intestines of mice of many inbred strains at all health status levels at TJL. However, there is a single report in which enterococci were isolated from lung abscesses in a mouse model of X-linked chronic granulomatous disease. 2 The absence of hemolytic complement is likely to play a critical role in the increased susceptibility of NSG mice to enterococci because it mediates neutrophil killing of enterococci in immunocompetent mice. 8 K oxytoca is a gram-negative rod-shaped bacteria. Although occasionally isolated from mice ceca and feces, the organism is not a permanent resident of the murine intestinal tract, and it could be eliminated from a colony by implementing an eradication program, as is currently in place in rooms housing colonies of NSG mice at TJL. K oxytoca is recognized as an opportunistic pathogen of mice that can naturally infect them and lead to clinical disease. 3 In immunocompetent mice, morbidity caused by K oxytoca is sporadic and associated with urinary and genital tract infections, otitis media, pneumonia, and abscesses. 3 Infections with K oxytoca leading to clinical disease have been reported in immunodeficient strains of mice, such as the C3H/HeJ strain that carries the Tlr4(Lps-d) allele. 10 These mice are insensitive to lipopolysaccharide signaling by the Toll-like receptor and to the activation of the innate immune system. In addition, the inflammatory mechanisms induced by lipopolysaccharide, as well as several genetic factors of both the host and the bacteria, were found to be essential for resistance to experimental pyelonephritis in mice. 10,18,19 NSG mice harbor several mutations that severely impair their innate and adaptive immune responses. At TJL, NSG mice are bred behind stringent barriers designed to exclude specific murine pathogens and several opportunistic organisms. The elimination of K oxytoca from NSG breeding colonies previously maintained at TJL was successful, and routine culturing of ceca and fecal material has indicated that all breeding colonies at TJL are now free of K oxytoca (unpublished data). The eradication of K oxytoca has led to a reduction in the incidence of nephritis and urinary tract infections in this strain. Since the complete exclusion of K oxytoca from the production colonies, Enterococcus is the sole organism cultured from kidneys of sick NSG mice. This finding suggests that the immune deficiency of the NSG strain is so severe that any nonpathogenic organism, even an intestinal commensal, could cause clinical disease.
Three breeding colonies of NSG mice were housed in 3 separate buildings at TJL in Bar Harbor and Sacramento. Note that morbidity was sporadic and that less than 1% of all mice older than 200 days were recognized as sick and sent for a diagnostic necropsy. Out of the 3 NSG colonies, 1 colony had significantly lower numbers of sick NSG mice, as well as better reproductive performance (L. D. Shultz, personal communication). This colony, like the other 2 at TJL, is subjected to stringent husbandry procedures and is monitored by the same comprehensive health monitoring program. However, 2 significant differences were noted among the 3 colonies. First, entry into this colony is restricted, and access is limited to 2 designated and highly trained caretakers. Second, change of bedding occurs weekly in this colony, whereas the other 2 colonies have a “check-change” program (ie, bedding is changed as needed) or a biweekly cage-changing program. Because all bedding and cages are autoclaved, this difference suggest the necessity of a minimal inoculating dose of opportunistic bacteria to induce morbidity in NSG mice; perhaps frequent changes of sterile bedding might reduce infection rates. In this study, sick individual NSG mice were often part of a breeding unit and were cohoused with unaffected mice. Some of the mice were housed as breeding pairs and others as trio matings. Sick mice submissions were sporadic, and often only a single mouse per box showed clinical signs of disease. When their cage mates were examined, they were frequently found to be normal. The basis for this disparity remains to be elucidated. Female mice were twice more likely to develop nephritis. If the route of infection was indeed ascending, then this finding is probably associated with anatomic, hormonal, and behavioral factors and is compatible with previous studies performed in animals and humans. 6-7 In summary, severely immunocompromised strains such as the NSG mouse are susceptible to infections by bacteria considered to be nonpathogenic in immunocompetent mice. K oxytoca and Enterococcus sp can both cause disease and should be considered pathogens in severely immunodeficient strains. In view of the enhanced susceptibility of these mice to infections by opportunistic organisms, it is essential that NSG mice be housed under optimal conditions, which include the eradication of opportunistic bacteria, the implementation of rigorous management practices, and the maintenance of a stringent barrier system.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was supported by the following grants (to LDS ) National Institutes of Health grants DK072473, CA34196, and AI46629, The Biology of the Beta Cell Consortium, The Juvenile Diabetes Foundation International, and the Helmsley Foundation.
