Abstract
Anthrax is a lethal disease caused by the bacterium Bacillus anthracis. There are three principal forms of the disease in humans—cutaneous, gastrointestinal, and inhalational—depending on the route of exposure. Of these, inhalational anthrax is the most dangerous; it is rapidly fatal; and it has been used as a deadly biological warfare agent in the last decade. Suitable animal models of inhalational anthrax have been utilized to study pathogenesis of disease, investigate bacterial characteristics such as virulence, and test effectiveness of vaccines and therapeutics. To date, mice, guinea pigs, rabbits, and nonhuman primates are the principal animal species used to study inhalational anthrax. Mice are valuable in studying early pathogenesis and bacterial characteristics. Few pathologic changes occur in the mouse models but may include marked bacteremia and lymphocyte destruction in the spleen and mediastinal lymph nodes. Rabbits and guinea pigs rapidly develop fulminate systemic disease, and pathologic findings often include necrotizing lymphadenitis; splenitis; pneumonia; vasculitis; and hemorrhage, congestion, and edema in multiple tissues. Nonhuman primates consistently develop the full range of classic lesions of human inhalational anthrax, including meningitis; lymphadenitis; splenitis; mediastinitis; pneumonia; vasculitis; and hemorrhage, congestion, and edema in multiple tissues. This review focuses on basic characteristics of the bacterium and its products, key aspects of pathogenesis, and the pathologic changes commonly observed in each animal model species.
Anthrax is a dangerous bacterial infection of livestock and humans caused by the gram-positive spore-forming bacterium Bacillus anthracis. The spores may remain dormant yet viable in the environment for decades. It is also of great concern as a biological weapon and is categorized by the Centers for Disease Control as a category A biological threat agent. Human anthrax is often classified as cutaneous, inhalational, or gastrointestinal, according to the route of bacterial infection. All forms may be lethal in humans and animals. Inhalational anthrax is of particular concern because mortality rates of untreated human cases may approach 100%, compared with 10 to 20% for the cutaneous form. Human cases studied to date have involved natural infections, such as occupational exposure (woolsorter’s disease) 2,3,11 as well as cases arising from either accidental or intentional release of B anthracis. 5,24,25 In the United States in 2001 there was intentional release of this agent in a series of bioterrorism attacks, and in 1979 in Sverdlovsk (of the former Soviet Union) there was accidental release of an aerosolized infective cloud of B anthracis. Information gathered from dozens of patients and victims from these events has led to a greater understanding of the human disease and appropriate medical treatment for improved patient survival. 1,10,36 Currently, two Food and Drug Administration–approved anthrax vaccines are available and used for at-risk individuals. Ongoing studies are aimed at (1) developing improved vaccines that provide increased immune protection 26,49,52,57 and (2) testing the effectiveness of combination therapies, such as vaccination with postexposure antibiotics. 62 Approved antibiotics are also available to treat human anthrax; however, their success in treating the disease is partially based on early diagnosis and immediate initiation of treatment following exposure, drug choice and dose, and duration of therapy.
Inhalational anthrax has a rapid disease course, and death may occur in humans and animals within a few days of exposure, if left untreated. 17 Inhaled spores enter the respiratory system, including the upper respiratory tract, lungs, terminal airways, and alveoli, and may be taken up by phagocytic cells (alveolar macrophages and dendritic cells) 12,13 in which they have been shown to germinate. 14,37,55,65 Infected phagocytic cells move into the lymphatic vessels and are taken to the regional lymph nodes, carrying spores and germinated bacilli. 53 Prolonged antibiotic treatment in human patients with inhalational anthrax is necessary because spores not taken up by phagocytic cells within the lungs can remain dormant there and thus impervious to antimicrobial agents for long periods or until some event induces them to germinate into vegetative bacilli. 15,16,27,39,40,51
To clarify, the lungs have been shown to be important in the initial pathogenic events establishing systemwide infection; however, inhalational anthrax is generally not considered a primary pulmonary disease. 6 In most animal models, pneumonia may be noted and develop for several reasons. The pneumonia pattern typically present is that of hematogenous origin (interstitial pneumonia), as opposed to that of inhaled bacteria (bronchopneumonia). 28,67 Some factors contributing to the presence of acute interstitial pneumonia noted in humans and animals with inhalational anthrax may include effects of B anthracis and its exotoxins on lung tissues, marked congestion and edema, disrupted blood supply, and preexisting subclinical pulmonary pathology.
When phagocytic cells carrying B anthracis reach the regional lymph nodes, the bacteria are released from dying cells, multiply rapidly, and enter the bloodstream, giving rise to systemic bacteremia and widespread dissemination of bacilli. Once bacteremia is established, any organ or tissue may become infected by B anthracis, particularly if preexisting pathology, injury, or inflammation is present. 29,30
Despite its low or sporadic natural occurrence, modern research efforts have nonetheless focused on inhalational anthrax because it is one of the most significant biological threat agents that the world faces today. It is the most aggressive and lethal form of the disease, and it requires immediate and prolonged medical countermeasures for patient survival. 62,63 Aggressive combination antimicrobial therapy that is begun during the initial phase of the disease with intense supportive care, including drainage of pleural effusions, has significantly improved survival rates in patients in the past decade. 41 Understanding the pathogenesis of human inhalational anthrax and development of appropriate medical countermeasures are made possible by modern research efforts using appropriate animal models. To date, the inhalational anthrax animal models include mice, guinea pigs (GPs), rabbits, and nonhuman primates (NHPs). This review focuses on the pathology of inhalational anthrax in these animal models.
Bacterium
Naturally occurring B anthracis expresses primary virulence factors encoded on 2 extrachromosomal DNA molecules (plasmids): pXO1 and pXO2. The pXO1 plasmid carries genes that encode the protein components of the exotoxins lethal toxin (LT) and edema toxin (ET). Lethal factor is a zinc metalloprotease, and edema factor is a calmodulin-dependent adenylate cyclase. When the proteins lethal factor and edema factor individually bind with another pX01-encoded toxin component—protective antigen (PA)—LT and ET are respectively produced. Specifically, a pore is formed in the cell membrane by the PA component, and expression of the toxic moieties of LT and ET occurs primarily within cells. 59 Some of the local and distant effects of LT and ET include induction of apoptosis in dendritic cells, inhibition of T- and B-lymphocyte functions, induction of endothelial cell apoptosis causing vascular leakage, and inhibition of platelet aggregation. 20,21,59 The pXO2 plasmid codes for genes capA, capB, and capC that form the bacilli’s antiphagocytic polyglutamyl capsule. 19 Expression of these major virulence factors is complex and at least in part affected by host tissue environment, such as temperature, carbon dioxide concentration, and composition of blood serum. Additionally, the pathologic changes noted in tissues infected with virulent B anthracis are likely due to a combination of destructive effects of the bacteria, exotoxins, and host responses to bacilli and exotoxins. 48 Susceptibility to the bacteria and its exotoxins is variable among animal species, and some animals that are resistant to infection with B anthracis may be susceptible to the effects of LT and ET and vice versa. 43
Attenuated strains of B anthracis lacking one or both plasmids have been developed for use as vaccines and can be studied in standard laboratories (biosafety level 2). These include but are not limited to the Sterne strain (lacking pXO2) and the Pasteur strain (lacking pXO1).
Mouse Models
Differences in host susceptibility to virulent B anthracis were first characterized among various mouse strains. 38,47,64 There are highly resistant strains of mice, such as the BALB/c strain, and highly susceptible mice, such as the A/J and DBA/2J strains. Those mice with intermediate susceptibility include C57BL/6. 38,47,64,65 The A/J mouse is also susceptible to attenuated forms of B anthracis. 36,47,64,65 Some mouse strains have been shown to display variability in sensitivity to LT and ET per se. Susceptibility of a mouse strain to LT does not necessarily correlate with its susceptibility to B anthracis infection. 58,64,65 The A/J mouse that is challenged with an attenuated B anthracis (Sterne strain) provides an appealing anthrax animal model because (1) there are no enhanced biocontainment requirements and (2) studies may be conducted under biosafety level 2 conditions. 18 This strain expresses the virulence factors LT, ET, and PA. In contrast, research with virulent strains of B anthracis, such as the Ames strain, must be conducted in biosafety level 3 containment facilities. In addition, most studies using the murine anthrax models have utilized differing routes of exposure, including subcutaneous, intraperitoneal, intranasal, and intratracheal. 47 Although such studies are economical, practical, and safe, it is difficult to fully and confidently extrapolate all data from them and liken them to an aerosol challenge model with a virulent strain of B anthracis. Only recently have true murine inhalational models been described in the literature. 16,38,40,46 Even fewer reports have described mice aerosolized with fully virulent B anthracis. 16,38 Currently, there are two mouse models used to understand a number of aspects of the pathogenesis of aerosolized anthrax. These involve A/J mice aerosol challenged with the Sterne strain (A/J-aeroSterne) and BALB/c mice aerosol challenged with the Ames strain (BALB/c-aeroAmes).
A/J-aeroSterne mice aerosol challenged with doses of approximately 20 times median lethal dose (4 × 106 colony-forming units [cfu]) die between 4 and 6 days postexposure. This is similar to the time to death reported for A/J mice challenged with Sterne strain administered subcutaneously or intraperitoneally. Host cell phagocytosis of the spores and their subsequent germination, multiplication, and dissemination occur rapidly after aerosol exposure. A recent study suggested that phagocytosis of the spores in the lungs and their translocation to regional lymph nodes with subsequent outgrowth, multiplication, and spread of bacilli to the liver and spleen can occur within 24 hours of aerosol exposure. 46 The nares and nasal-associated lymphoid tissue have been shown to be an additional site for initial spore uptake, germination, multiplication, and rapid dissemination in mice. 31 –33 Histologically, A/J mice may display severe bacteremia, with bacilli frequently noted within blood vessels of the lung and liver. However, pathologic changes in this model are of limited degree. Splenic lesions in A/J mice may include minimal lymphocyte destruction, white pulp depletion, and, rarely, hemorrhage. The brain, adrenal gland, gastrointestinal tract, kidney, urinary bladder, and sex organs are generally unchanged in this model. Classic pathologic changes of human inhalational anthrax—such as marked congestion, edema, fibrin, hemorrhage, and necrosis in multiple tissues; marked lymphocytolysis with lymphoid depletion in multiple lymphoid tissues; and vasculitis—are not well represented in the A/J-aeroSterne model. In this regard, whereas the Sterne strain expresses LT, ET, and PA, it lacks the antiphagocytic polypeptide capsule, which may account for some of these findings. Additionally, mice are unique among the animal models of anthrax because strains of B anthracis that lack pX01 but retain pX02 (are encapsulated but produce no toxins) are nearly fully virulent and lethal in mice, whereas they are highly attenuated in other animals. The reasons for this are not clear. 40,47,66
Mouse strain susceptibility differences are also evident when challenged by aerosol with virulent B anthracis (Ames strain). Variations in median lethal dose and time to death postexposure are described for several mouse strains (A/J, C57BL, BALB/c, and Swiss Webster). 38 Not surprisingly, the A/J mouse is the most susceptible, with the shortest time to death: 2.2 days. The BALB/c mouse is the least susceptible, with a time to death of 3.3 days. This slightly longer time to death makes the BALB/c appealing because of the larger window of time available to test medical countermeasures. This strain was further evaluated, and pathologic changes in tissues of BALB/c mice included a marked bacteremia, with bacilli most notably evident in the alveolar septal capillaries, renal glomerular capillaries, and hepatic sinusoids. Mediastinitis may be present, as characterized by the presence of polymorphonuclear leukocytes (PMNs), fewer macrophages, and lymphocytes. Lymphocyte destruction may be present, with lymphoid depletion of the white pulp of the spleen and with infiltrates of PMNs in the red pulp. Hepatic degeneration and necrosis (ischemia) associated with clusters of bacilli plugging the sinusoids occur rarely. 38 Mouse models have been valuable in determining early pathogenesis, how the agent establishes infection in the host, and characteristics of the spore and vegetative bacilli.
Rat and Hamster Models
Laboratory rats, such as the Fisher 344, Sprague Dawley, Brown Norway, as well as other inbred strains of rats, have been used as models to study B anthracis exotoxins LT and ET. 7,8 The Fisher 344 rat is highly sensitive to LT and will succumb in less than 1 hour following intravenous injection. This sensitivity to the toxin makes it an ideal model for antitoxin studies yet is beyond the scope of this review. The rat has been an important animal model for the understanding of anthrax exotoxins but has not been developed as an animal model of inhalational anthrax.
Similarly, there are scattered reports of B anthracis testing in Syrian hamsters challenged by the subcutaneous or intraperitoneal routes, although they have not been developed as an animal model of inhalational anthrax. 23,57
GP Model
Hartley GPs (Cavia porcellus) have been widely utilized in the last 4 decades in anthrax vaccine studies, with limited success. 22,44,45 Survival in this model is variable following vaccination and challenge with virulent B anthracis (Ames strain), and the responses in this model may not be good predictors of vaccine immune responses in other species. 22,67 The GPs' innate sensitivity to certain antibiotics limits their use in some antibiotic therapeutic research.
There are few published descriptions of the pathologic changes in GPs challenged with aerosolized anthrax. Two articles published in the 1950s were restricted to descriptions of the lungs and kidney yet greatly enhanced our understanding of host infection following inhalation of spores in all species. 53,54 These experiments detailed the primary pathological events within the lungs of Hartley GPs following intratracheal exposure to attenuated strains of B anthracis (M36) and aerosol with Bacillus subtilis, a hay or grass bacillus. Subsequent to challenge, spores were primarily found within pulmonary sessile septal cells (eg., dendritic cells) and within alveolar macrophages at 35 minutes (intratracheal) and 60 minutes (aerosol) postexposure. The septal dendritic cells were described as “rounding up,” detaching, and moving into lymphatic vessels following uptake of spores. A rapid and transient increase in interstitial PMNs was noted. Intracellular and free spores and bacilli were found in the tracheobronchial lymph node by 4 hours (intratracheal) and 5 hours (aerosol) postexposure. Once within a phagocytic cell, the spore’s staining characteristics were altered, presumably because of germination, but vegetative bacilli were found in only the regional lymph nodes.
Renal tubular necrosis and hemorrhage have been described in only GPs and rabbits. 53,54,67 The pathogenesis of renal tubular lesions in these models is unknown. It has been postulated that terminal hypovolemic, septic, or cardiogenic shock may be the cause of renal anoxia and subsequent ischemic necrosis of tubular epithelial cells in the GP. Additionally, direct and indirect effects of exotoxins, inflammatory mediators, and disseminated intravascular coagulation have been suggested to contribute to renal tubular necrosis in the GP. 53 The pathogenesis of renal tubular lesions in the GP and rabbit clearly warrants further investigation.
Little else describing pathologic changes following aerosol exposure to anthrax has been documented in GPs. In a recent study at our institute, a group of 26 male and female adult Hartley GPs were challenged with 5.2 × 101 to 1.92 × 106 cfu of aerosolized virulent B anthracis (Ames strain) and euthanatized in a serial manner at 6, 24, and 48 hours postexposure (M. L. M. Pitt and N. A. Twenhafel, unpublished data). GPs were aerosol challenged in a whole-body chamber. Full necropsies were performed and tissues taken for histologic evaluation (Table 1 ). At 6 hours postexposure, 6 animals were euthanatized, and no gross lesions were noted. However, histologic changes in the lungs at this time already included pulmonary edema, congestion, alveolar histiocytosis, and low numbers of perivascular PMNs. There was also marked congestion and edema in the nasal turbinates and the mandibular and mediastinal lymph nodes, as well as mild germinal center lymphocytolysis within splenic corpuscles. Scanning and transmission electron microscopy (SEM and TEM, respectively) was performed on lung tissue in one of these cases to confirm that the described changes were due to the presence B anthracis. Bacilli were identified by SEM (Fig. 1) and TEM within pulmonary interstitial blood vessels and free within alveoli. This compelling evidence suggested that the initial pathogenic events—including phagocytosis, transport to regional lymph node, germination, multiplication, and bacteremia—occurred in this GP by 6 hours postexposure.
Pathologic Findings of 26 Male and Female Hartley Guinea Pigs Challenged With Bacillus anthracis and Serially Euthanatized a

a Aerosolized B anthracis (Ames strain) spores (5.2 × 101 to 1.92 × 106 colony-forming units). Number of guinea pigs affected in parentheses.
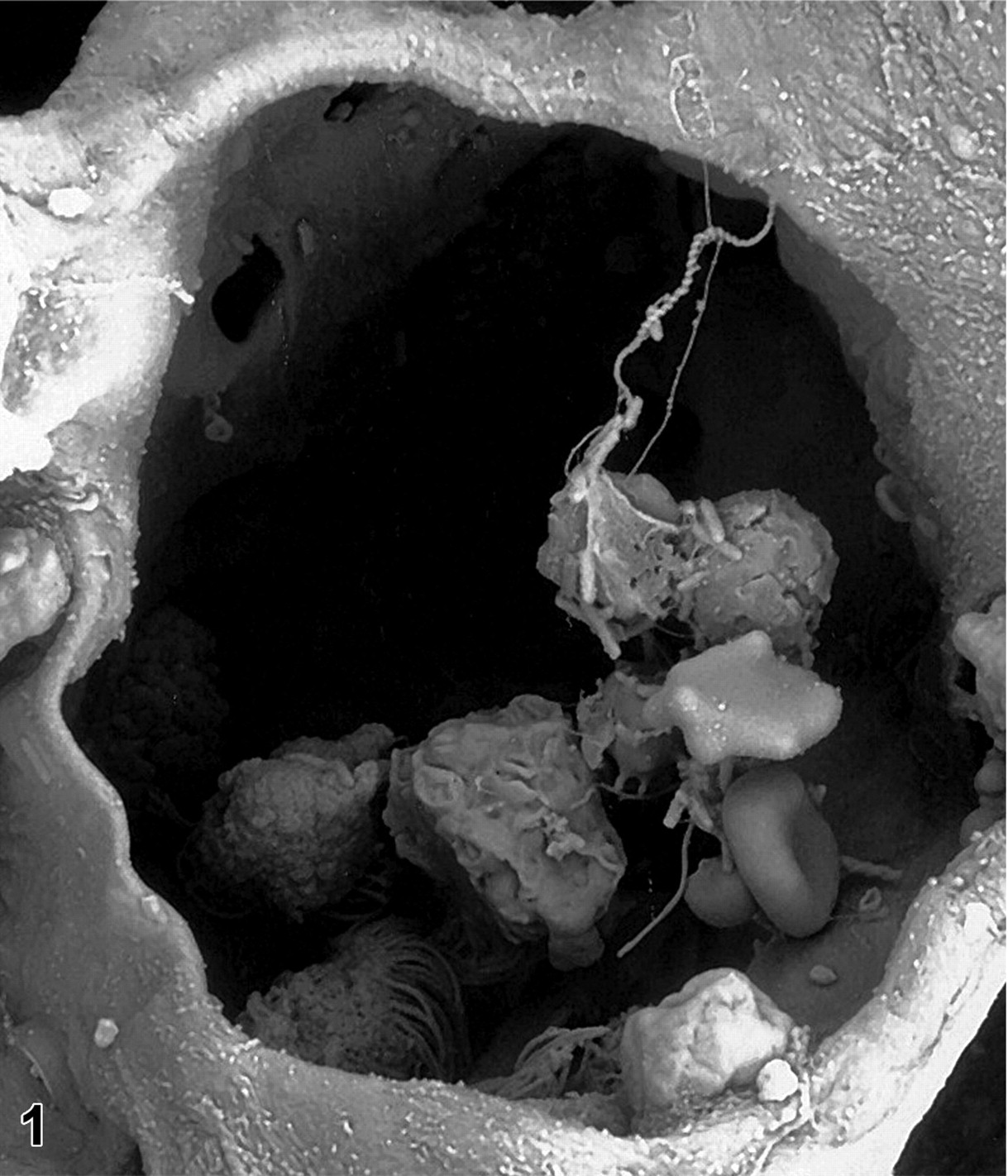
Lung; female Hartley guinea pig. Scanning electron micrograph of lung of a guinea pig aerosolized with a lethal dose of anthrax spores and euthanatized 6 hours postexposure. Within the pulmonary alveolus there are 2 red blood cells; one is concave and the other is unusually shaped and flattened. There are also multiple alveolar macrophages with irregularly textured cell surfaces. Some of the alveolar macrophages have many cytoplasmic extensions resembling cords that extend from the macrophage cell surface to the alveolar wall. Several visible bacilli have a classic blunt end, or boxcar shape. The bacilli are attached to the macrophage cell surfaces and are seen intertwined in ropes of twisted protein fluid (most likely, edema or surfactant).
The remaining GPs were evaluated at 24 and 48 hours postexposure. Noncollapsing lungs (edema) with moderate to marked red mottling (congestion and/or hemorrhage) was grossly seen in most animals. Histologic changes included pulmonary congestion, edema, and alveolar histiocytosis; interstitial pneumonia; and draining inflammation in mediastinal and mandibular lymph nodes. Splenic lesions were more pronounced from earlier time points, with moderate germinal center lymphocytolysis, increased congestion most notably in the marginal zone with fibrin, mild to moderate PMN inflammation in the red pulp, and diffuse moderate necrotizing splenitis. Other changes included moderate congestion and edema of the meninges, gastrointestinal congestion, edema of the nasal turbinates, ulcerative and suppurative rhinitis, acute tracheitis, and adrenal gland hemorrhage. Bacilli were histologically evident by 48 hours and found within blood vessels of the spleen, lungs, mediastinal lymph node, meninges, and renal glomeruli.
There was widespread systemic disease by 48 hours postexposure, indicating that the GP is exquisitely sensitive to aerosol challenge with virulent B anthracis. Additionally, the SEM and TEM evidence found within the lung of an animal at 6 hours postexposure suggests that the initial uptake of spores and subsequent pathogenic events leading to bacteremia and pulmonary changes occurred rapidly in the GP. The consistent finding of congestion within the nares and subsequent rhinitis, coupled with draining inflammation noted in the mandibular lymph nodes, suggests that the nasal tissues, with their local phagocytic cells and regional lymph node, may play an important role in the pathogenesis of inhalational anthrax in the GP and should be further evaluated.
Rabbit Model
The pathology of inhalational anthrax in New Zealand white (NZW) rabbits (Oryctolagus cuniculus) is well documented, and this model has been extensively used in aerosolized anthrax research. 67 Rabbits are aerosol challenged in a muzzle-only mask. Similar to GPs, NZW rabbits are extremely susceptible to lethal infection with B anthracis; they have an innate sensitivity to certain antibiotics, which may limit their use in therapeutic studies. 67 NZW rabbits aerosolized with 8.34 × 104 to 1.03 × 107 cfu of virulent B anthracis (Ames strain) succumb within 2 to 4 days postexposure.
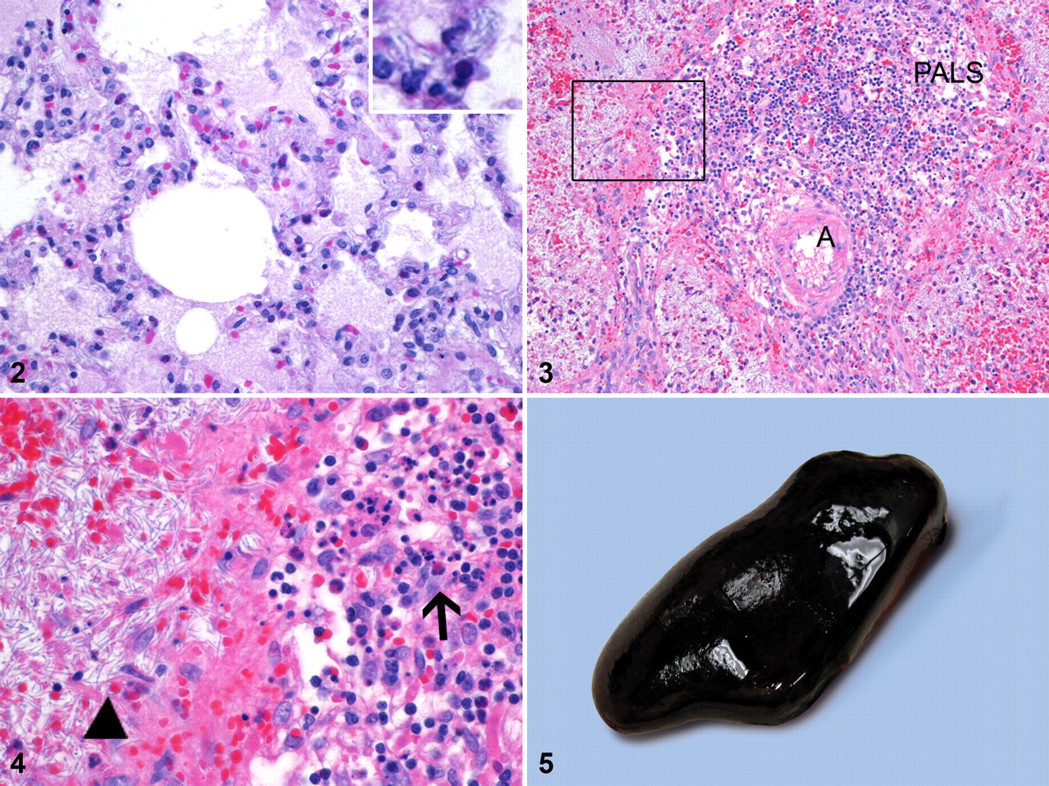
Primary gross lesions in NZW rabbits may include epistaxis, splenomegaly, adrenal gland hemorrhage, mandibular lymph node hemorrhage, and edema of the lungs. 67 Significant histologic findings include interstitial pneumonia (Fig. 2), splenitis, and lymphadenitis. There is often pronounced destruction of lymphoid tissues—most notably, the spleen (Fig. 3); mediastinal, mandibular, and mesenteric lymph nodes; and the Peyer patches of the small intestine and sacculus rotundus. Lymphoid depletion in multiple lymphoid tissues is often accompanied by hemorrhage, necrosis, inflammation, and myriad bacilli (Fig. 4). Additional histologic changes in the NZW rabbit model include adrenal gland and ovarian hemorrhage, systemic bacteremia, and necrotizing or fibrinoid vasculitis. 67 Other less commonly noted histologic changes include necrosis and hemorrhage within the gastrointestinal tract, including the stomach, sacculus rotundus, and cecal appendix; necrosis and hemorrhage in the renal tubules; and congestion or hemorrhage within the meninges, brain, adrenal glands, and ovaries. Investigators conducting therapeutic studies should consider the animal’s sensitivity to the therapeutic agent, immune response to vaccinations, and susceptibility to B anthracis and LT and ET.
NHP Models
For the past 60 years, NHPs have been extensively used to study inhalational anthrax. 2,28 –30,60,61 The rhesus macaque (Macaca mulatta) was used almost exclusively in research from the 1990s until the turn of the century. Within the past 10 years, likely owing to the limited availability and expense of rhesus macaques, there has been resurgence in the use of cynomolgus macaques (Macaca fascicularis) and the introduction and use of a new species, the African green monkey (Chlorocebus aethiops). These three NHP species have been well characterized in the literature and proven to be valuable animal models of inhalational anthrax. 28 -30,60,61
NHP models display similar clinical and pathologic changes when exposed to lethal doses of aerosolized virulent strains of B anthracis. Adult and juvenile animals, as well as male and female animals, have been used in various studies and have been shown to respond similarly when challenged with doses ranging from 2 × 104 to 5 × 1010 cfu of virulent aerosolized anthrax. 28 –30,60,61 NHPs are aerosol challenged in a head-only chamber (from the neck up). The time to death has been reported to range from 3 to 20 days postexposure in rhesus macaques, 28 –30 from 2 to 10 days postexposure in cynomolgus macaques, 61 and from 3 to 25 days postexposure in African green monkeys. 60 Animals often die acutely without clinical signs of disease. 28 –30,60,61
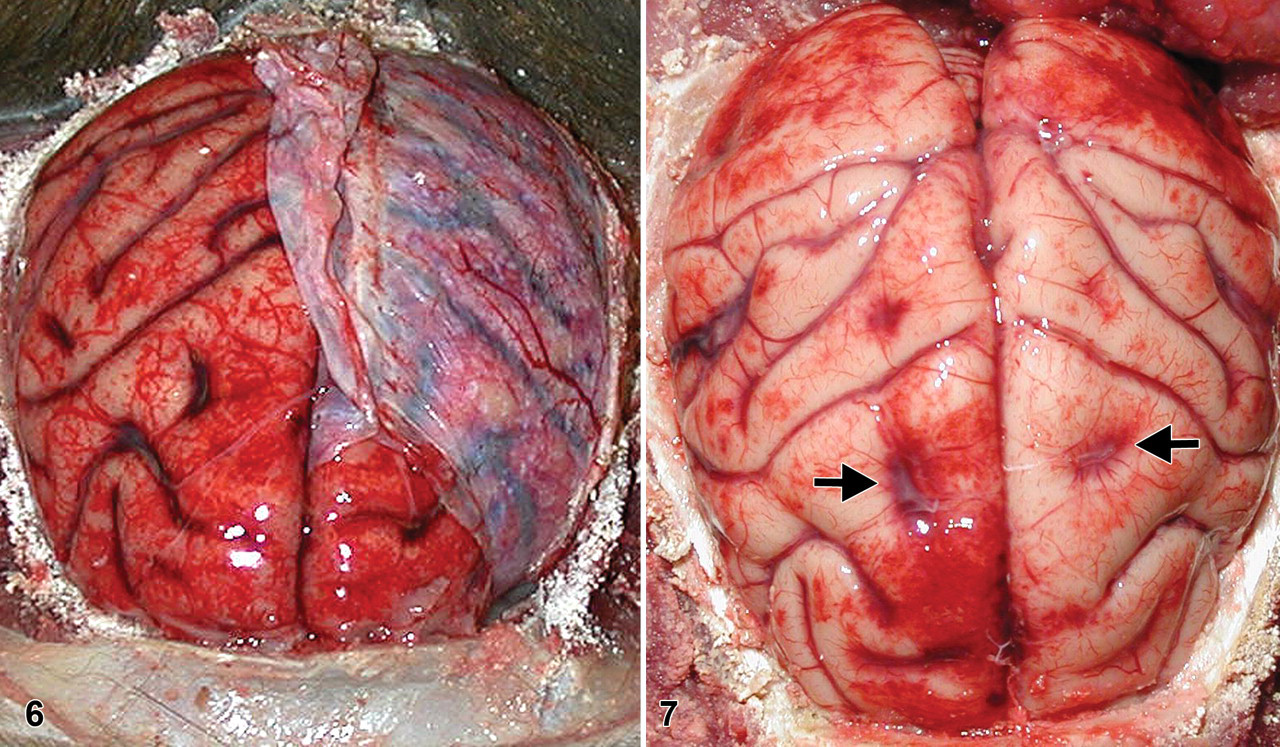
Gross necropsy findings in these NHP species after aerosol challenge with virulent B anthracis include edema, congestion, hemorrhage, and necrosis in the lungs mediastinum, meninges, lymph nodes (tracheobronchial, mediastinal, mesenteric, axillary, and inguinal), adrenal glands, gastrointestinal tract, and urogenital organs. Splenic changes may include overall enlargement with rounded edges, giving it a plump appearance (Fig. 5), a dark red to black color, and a friable parenchymal consistency on cut surface (historically referred to as blackberry jam). Meningitis is a frequently reported gross finding and typically characterized as being red or hemorrhagic (“cardinal’s cap”) (Figs. 6, 7) or milky and opaque. Pleural serosanguineous effusion, pericardial effusion, and widened mediastinum have frequently been described in the African green monkey. 60
Histologic changes commonly present in multiple organs and tissues in these models include congestion, edema, fibrin, hemorrhage, necrosis, parenchymal loss, infiltrates of acute inflammatory cells (PMNs), and vasculitis. These changes affect the lungs, mediastinum, lymph nodes (tracheobronchial, mediastinal, mesenteric, axillary, and inguinal), spleen, meninges, brain, adrenal glands, mesentery, liver, gastrointestinal tract, and urogenital organs. Bacilli may be present within blood vessels (bacteremia) or extravascular within organ parenchyma and various tissues.
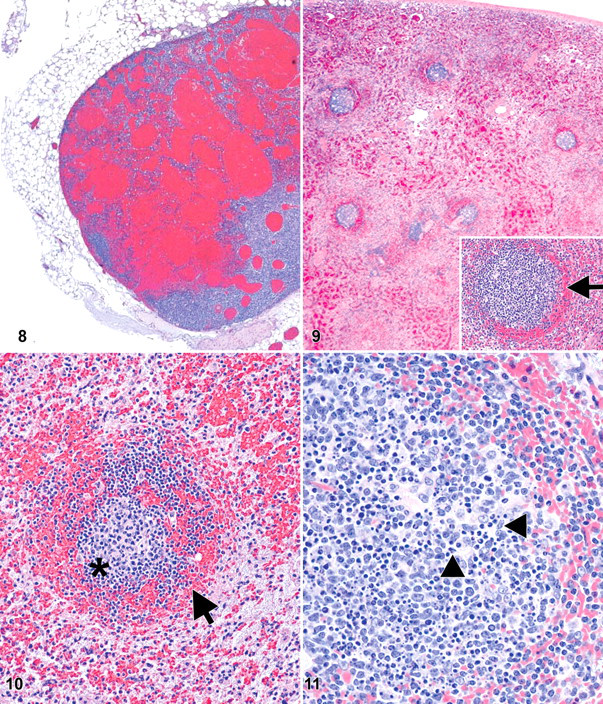
The mediastinal lymph nodes—specifically, the tracheobronchial lymph node—and the spleen are key lymphoid tissues consistently affected in all NHP species, as with inhalational anthrax in mice, GPs, and rabbits. 38,54,67 Necrotizing or necrohemorrhagic lymphadenitis (Fig. 8 ) and splenitis with marked destruction of lymphocytes (lymphocytolysis) and generalized lymphoid depletion are hallmark lesions consistently present in this disease. The B lymphocytes within the germinal centers are destroyed, and there is progressive reduction of small lymphocytes of the mantle zone, resulting in the loss of follicular/corpuscular structure. The collapse of B-cell zones with concurrent loss of T-cell zones results in generalized lymphoid depletion of the organ. 28 –30,60,61 A classic microscopic appearance of the spleen is generalized paucity of white pulp, marked lymphocytolysis within the germinal centers of corpuscles (Fig. 11) and periarteriolar lymphoid sheaths, loss of small lymphocytes within mantle zones, marginal zone congestion/hemorrhage (Figs. 9, 10), red pulp necrosis and congestion, and increased PMNs within the red pulp. Interestingly, when bacilli are evident in the spleen, they are often restricted to the red pulp. The lymphoid tissues most frequently altered by some or all of these changes in NHPs include mediastinal lymph nodes (most notably, the tracheobronchial), the spleen, other lymph nodes (mandibular, axillary, mesenteric, and inguinal), and gut-associated lymphoid tissue.
The mechanisms of lymphoid destruction are not completely understood; however, unregulated (so-called bystander) apoptosis of lymphocytes has been described in humans and animals with sepsis. 50,56 Initial destruction of lymphocytes in germinal centers rapidly gives way to widespread lymphoid depletion as the organ becomes increasingly compromised by bacterial proliferation, host response to infection (ie, cytokines and chemokines), circulatory disturbances, and effects of the bacterium’s exotoxins.
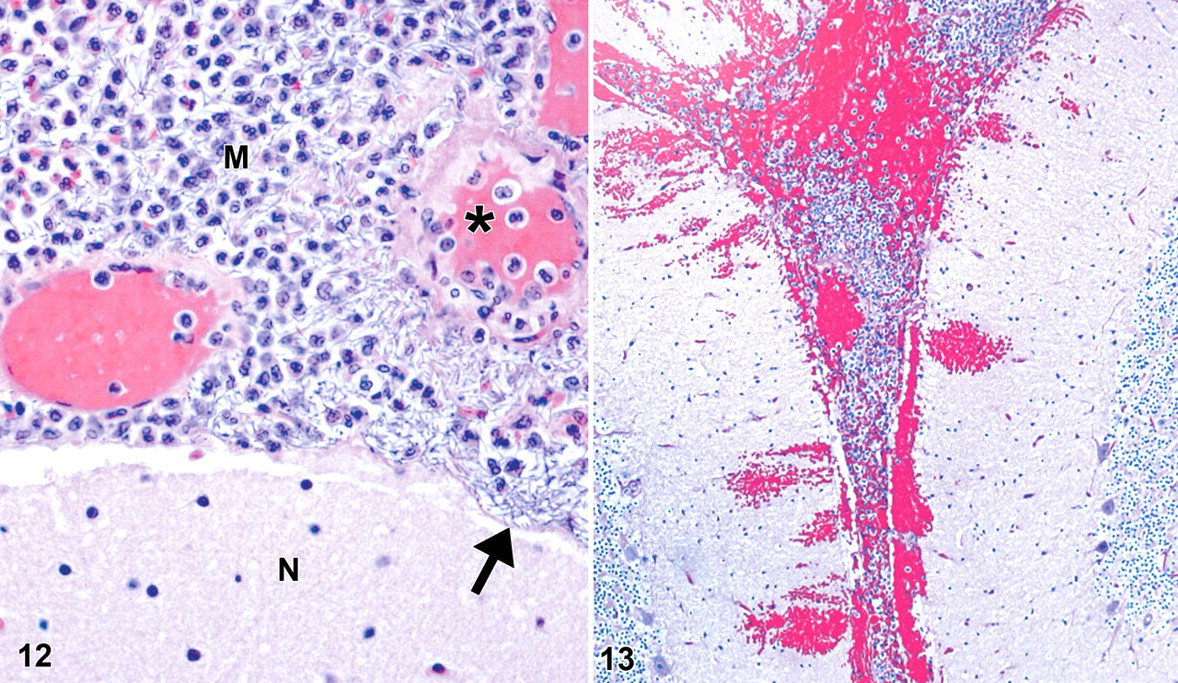
Meningitis is commonly reported in rhesus macaques, cynomolgus macaques, and African green monkeys and documented as early as 3 to 5 days postexposure. 9,28 –30,60,61 The bacterium’s ability to cause severe meningitis in humans and NHPs is not well characterized. Bacteria, PMNs, hemorrhage, and fibrin can markedly expand the meninges—specifically, the arachnoid—yet do not typically cross the pia mater into the brain (Fig. 12). The pia mater usually remains intact until the infection becomes so severe that the pia mater is disrupted and there is local extension of infection into the neuropil (Fig. 13). Fibrinoid or necrotizing vasculitis is frequently present in the blood vessels of the arachnoid (Fig. 12). These vessels may be highly susceptible to the effects of bacilli and exotoxins, leading to increased vascular permeability and thereby allowing the bacteria to escape from blood vessels and invade the tissue. Additionally, the leptomeninges may provide an ideal growth environment for the bacilli because of tissue-specific O2 and CO2 tension, pH, temperature, rich blood supply, and the presence of cerebrospinal fluid. Continued research is needed to clarify the pathogenesis of anthrax meningitis, and these studies are restricted to using NHP models.
A recent report described the pathologic findings in a group of common marmosets (Callithrix jacchus) challenged with lethal aerosol doses of virulent B anthracis (Ames strain), 1.47 × 103 cfu. 42 Six animals succumbed (50% of challenge group) between 2 and 4 days postexposure. Gross findings included pulmonary hemorrhage (3 of 6), splenomegaly (2 of 6), mediastinal (tracheobronchial) lymph node hemorrhage (1 of 6), and hepatomegaly (1 of 6). Histologic features in the marmosets included marked bacteremia in multiple tissues (including meninges and brain), fibrin deposition in multiple tissues, splenitis with lymphoid depletion, interstitial pneumonia, renal congestion and tubular necrosis, and hepatic congestion. The reported low incidence of classic lesions within the lymph nodes and spleen, plus the absence of meningitis, suggests that the common marmoset model may not mimic the human inhalational anthrax as closely as the rhesus macaque, cynomolgus macaque, and African green monkey. Nonetheless, other features of this model, with factors such as size and availability of these monkeys, may make them useful for future research.
Summary
Inhalational anthrax in humans has a reported incubation period of 4 to 6 days from exposure to initial clinical symptoms and death. 1 Clinical symptoms include fever and chills, sweats, fatigue and malaise, cough, dyspnea, and nausea and vomiting. Hallmark human lesions include abnormal chest radiographs showing a widened mediastinum, moderate to marked pleural effusion, pericardial effusion, and meningitis. 1,4,24 Computed tomography in patients frequently shows mediastinal lymphadenopathy. 36,41 Key histologic findings included pneumonia; splenic lymphoid depletion; meningitis; hepatic, gastrointestinal, and urogenital hemorrhage and/or inflammation; and anthrax bacteremia and toxemia. 1,35,41 These are the important features of human inhalational anthrax to which the animal models are compared. 34
Continuing efforts to provide improved medical countermeasures against inhalational anthrax, such as vaccination and antibiotic therapy, remain an active area of research, and these studies require suitable animal models. Although the NHPs—specifically, the rhesus macaque—have long been considered the gold standard of inhalational anthrax animal models, there is no perfect animal model for this disease. Proponents of inhalational anthrax in multiple species have provided information on various aspects of the bacterium and disease process in mammals. There is utility in the differences among models. For example, the anatomic configuration of the nasal and oral cavities of humans and NHPs permit nasal and oronasal breathing; however, mice, rats, hamsters, GPs, and rabbits are obligate nose breathers owing to the close apposition of the epiglottis and soft palate. These anatomic differences in species are of special note because aerosolized anthrax spores may be more readily deposited in the upper respiratory tract in obligate nose breathers. Histologic changes noted in the aerosol model of GPs challenged with virulent B anthracis (Ames strain; M. L. M. Pitt and N. A. Twenhafel, unpublished data) suggest that the nasal turbinates with associated regional lymphoid tissues may be one of the initial sites of deposition and retention of anthrax spores, as well as germination and multiplication of bacilli. Recent studies have also shown the nares and nasal-associated lymphoid tissue to be important primary sites of infection in mice. 31 –33 It is unclear what consequence, if any, spore deposition and phagocytosis in the nares and upper respiratory tract may have as opposed to the lower respiratory tract on the course of disease, thus warranting continued investigation.
Mouse models have been important in identifying early pathogenic events leading to systemic infection and in defining essential features of the bacterium. Despite enormous progress in these areas, much about the initial pathogenic steps of inhalational anthrax remains unknown. Continued work on identifying anatomic sites of spore uptake, germination, multiplication, and dissemination is most practical in mouse models and is an area of intense interest and debate.
Rabbits and GPs are valuable models that develop many lesions found in human inhalational anthrax, except meningitis. These species are highly sensitive to infection with B anthracis and have been used in preliminary vaccine and therapeutic trials. It is generally more time- and cost-efficient to eliminate potential drug and vaccine candidates early in their development; therefore, initial trials will often challenge with the most sensitive animal models, such as rabbits and GPs, to evaluate product effectiveness. Rabbits and GPs are the only two models shown to develop renal tubular lesions. The pathogenesis of these lesions has not been addressed and should be further evaluated. Rabbits and GPs are innately sensitive to certain antibiotics that restrict their use in some therapeutic studies.
NHPs—specifically, rhesus macaques, cynomolgus macaques, and African green monkeys—mimic human disease and they do so consistently. They are frequently used in final drug and vaccine efficacy trials. The rhesus macaque, cynomolgus macaque, and African green monkey are the most commonly utilized NHP species in modern research. Clearly, much can be learned from research with these models. One such example would be studies of anthrax meningitis, which can be conducted in only the NHP model. Continued research is needed to clarify why this tissue is susceptible to infection and to provide insight into ways to prevent meningitis from developing. There are drawbacks to using NHPs in research—namely, these studies are expensive, labor intensive, and time-consuming, potentially taking years to complete.
Future research of inhalational anthrax is aimed at attaining a more complete understanding of the pathogenesis of disease, specifically how B anthracis spores, vegetative bacilli, and exotoxins are able to evade host immune systems and rapidly cause lethal infection in multiple species. Additionally, development of improved medical countermeasures will continue to require testing in animal models. These investigations will need information gathered from multiple animal species to meet the Food and Drug Administration’s animal rule. As such, this review focuses on the pathology of the inhalational anthrax animal models to assist the pathologist in evaluating studies utilizing these models.
Footnotes
Acknowledgements
Research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals and so adheres to principles stated in the Guide for the Care and Use of Laboratory Animals (National Research Council, 1996 [27a]). The facility where this research was conducted is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the US Army or the Department of Defense.
I would like to extend special thanks to Dr Ludmila Asher, Dr Chris Cote, Dr Dave Fritz, Dr Louise Pitt, COL Keith Steele, Dr Susan Welkos, and COL Gary Zaucha for their critical review of this article and images. I would also like to thank members of the Pathology Division at the US Army Medical Research Institute of Infectious Diseases for their technical expertise and dedicated support: S. Akers, J. Brubaker, D. Carter, N. Davis, P. Fogle, A. Grove, J. Jakubec, M. Kennedy, G. Krietz, K. Kuehl, K. McKinney, C. Mech, S. Mierzwa, F. Nugent, R. Reyes, and B. Wilke. I thank Mr William Discher for visual support.
The author declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author received no financial support for the research and/or authorship of this article.
