Abstract
Membrane transport processes, at both the plasma membranes and intracellular membranes, play critical roles in renal function and are a determining factor in the susceptibility of renal epithelial cells to blood-borne drugs and toxic chemicals. Proximal tubular epithelial cells possess a large array of transport proteins for organic anions, organic cations, and peptides on both basolateral and brush-border plasma membranes. Although these transporters function in excretion of waste products and reabsorption of nutrients, they also play a role in the susceptibility of the kidneys to drugs and other toxicants in the blood. The proximal tubules are typically the primary target cells because they are the first epithelial cell population exposed to such chemicals in either the renal plasma or glomerular filtrate and because of their large array of membrane transporters. Besides transport across the basolateral and brush-border plasma membranes, transport across intracellular membranes such as the mitochondrial inner membrane is a critical determinant of metabolite distribution. To illustrate the function of these transporters, carrier-mediated processes for transport of the tripeptide and antioxidant glutathione across the basolateral, brush-border, and mitochondrial inner membranes of the renal proximal tubule are reviewed. Studies are summarized that have identified the involvement of specific carrier proteins and characterized the role of these transporters in glutathione metabolism and turnover, susceptibility of the proximal tubules to oxidative and other stresses, and modulation in disease and other pathological processes.
Keywords
Membrane transport processes play critical roles in numerous physiological and pharmacological processes, including maintenance of cellular ion gradients, regulation of metabolite distribution, drug accumulation in target organs, and drug excretion. Membrane transport processes are central to the functions of the kidneys, and in particular of the proximal tubular epithelial cells, in ion and metabolite homeostasis, nutrient reabsorption, excretion of waste products, and drug distribution and metabolism. When considering exposure of the kidneys to drugs and potentially toxic chemicals, however, these basic physiological functions take on a new dimension because they are major contributory factors to the sensitivity of the kidneys to numerous blood-borne toxic chemicals.
There are 4 key reasons the kidneys are frequent and sensitive targets for blood-borne drugs and toxic chemicals. First, the kidneys receive an inordinately high rate of blood flow in relationship to their weight (ie, 25% of cardiac output compared with 1–2% of body weight). Second, the process of glomerular filtration delivers all substances below a cut-off molecular weight in the plasma to the tubular lumen. Hence, whereas proteins below a molecular weight of 20 kDa freely pass through the glomerular basement membrane, those proteins as large as albumin (Mr = 65 kDa) and those that have negative charges are not normally filtered. Third, renal tubular epithelial cells, particularly those of the proximal tubule, contain a large array of plasma membrane transporters that can result in intracellular accumulation of blood-borne chemicals to much higher concentrations in the epithelial cells than were in the blood. Fourth, bioactivation enzymes exist within renal epithelial cells, particularly those of the proximal tubules, that can metabolize many drugs and other chemicals to reactive and toxic forms. 44,47,48 The potential fates of drugs to which the kidneys are exposed are summarized in Fig. 1 . Central to the unique physiology and responses of the kidneys is the function of membrane transport proteins. Not only do membrane transporters play a quantitatively significant role in renal drug handling and disposition, but they are also important sites for drug–drug interactions and potential therapeutic target sites.
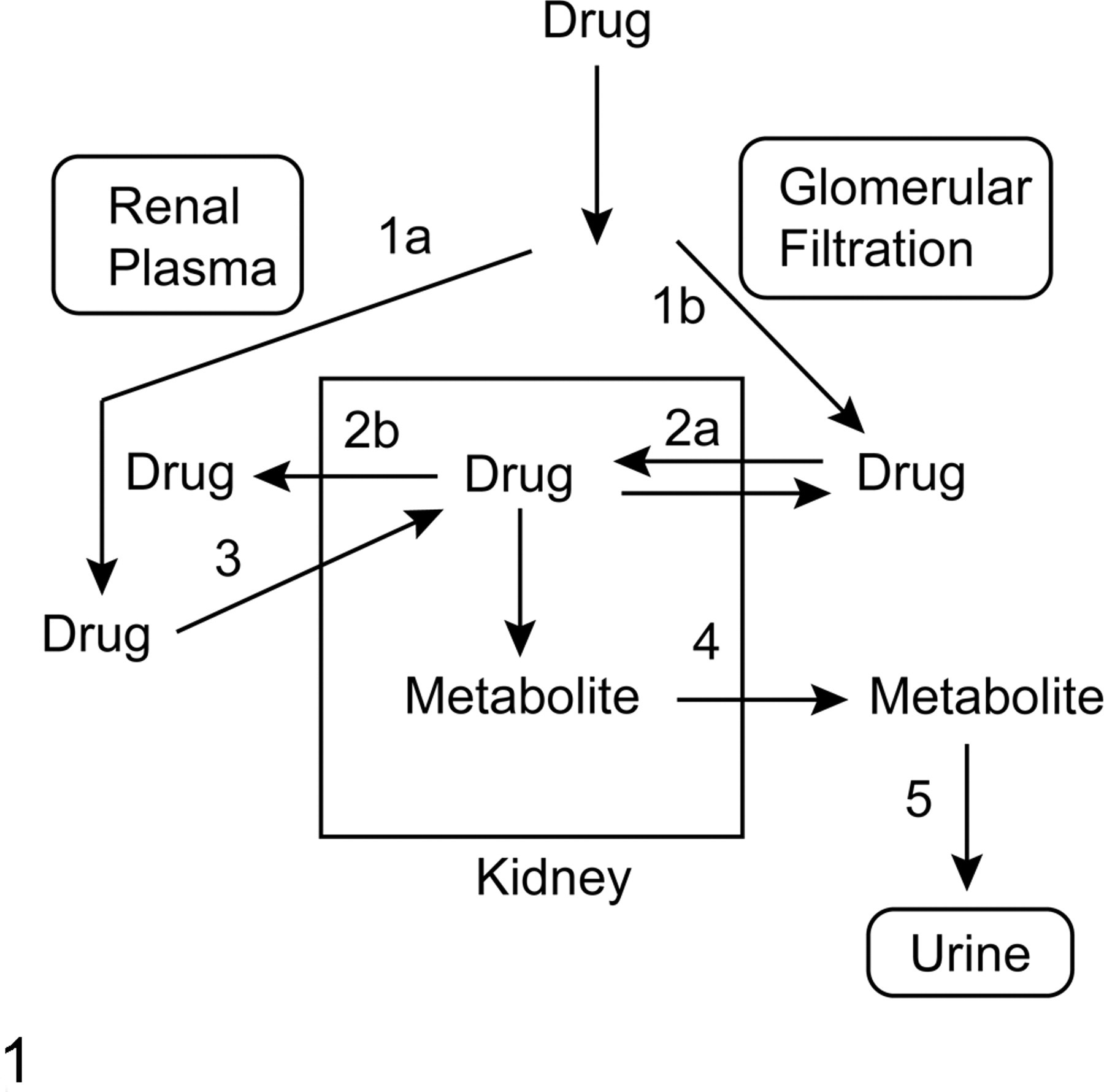
General scheme showing the roles of membrane transport in the renal handling of drugs. Kidneys are exposed to blood-borne drugs by either their presence in the renal periplasmic space (pathway 1a) or glomerular filtration (pathway 1b). Drugs in the tubular lumen may be absorbed or secreted by transport across the brush-border plasma membrane (pathway 2a) and then subsequently reabsorbed into the blood by efflux across the basolateral plasma membrane (pathway 2b). Alternatively, drug in the renal plasma may be absorbed into the proximal tubular cell by uptake across the basolateral plasma membrane (pathway 3) and metabolized inside the cell, and then the metabolite can be transported across the brush-border plasma membrane (pathway 4) for excretion into the urine (pathway 5).
Many drugs or environmental chemicals (or their metabolites) that undergo renal absorption or excretion are either organic anions or organic cations. Because charged molecules do not readily cross biological membranes by diffusion, specific carrier proteins have evolved for their transport across both plasma membranes and intracellular membranes. To illustrate the critical functions of carrier proteins in the transport of chemicals across renal membranes, this review will focus on the pathways for transport of the tripeptide and important antioxidant glutathione (GSH) across renal plasma membranes, both basolateral plasma membrane (BLM) and brush-border plasma membrane (BBM), and the mitochondrial inner membrane (MIM) as an example of a key intracellular membrane. Although GSH, being composed of 3 amino acids, contains both negative and positive charges at physiological pH, it has a net negative charge and is, therefore, treated as an organic anion. Accordingly, the major organic anion carriers on the BLM, BBM, and MIM will first be summarized briefly. The remainder of the review will focus on pathways for renal membrane transport of GSH, focusing on energetics, identification of carriers involved, the relationships between transport and cellular energy and redox status, and the toxicological and pathological roles of these transport pathways.
Overview of Pathways for Renal Membrane Transport of Organic Anions
A number of reviews over the past decade have summarized organic anion transporter families that are expressed in the kidneys and elsewhere, 11,16,30,32,41,63,67,80,84,93 and these publications can be referenced for more detailed information. This section will briefly summarize some of the more thoroughly studied carrier proteins that are most relevant for understanding renal function in drug reabsorption and excretion. There are 3 major families of carrier proteins on plasma membranes of renal proximal tubule that mediate uptake or efflux of organic anions. These include the organic anion transporters (Oats; Slc22a gene family), the organic anion transporting polypeptide carriers (Oatps; Slc01 gene family), and the multidrug resistance proteins (Mrps; Abcc gene family). Two members of the sodium-sulfate/carboxylate cotransporter family (Slc13a gene family) are also involved in the transport of key organic anions across the renal BLM and BBM.
Functions of the major carriers for organic anions on the BLM are schematically summarized in Fig. 2 . Numbers aligned with carriers represent distinct transport mechanisms. The (Na++K+)-stimulated ATPase, which is considered a primary active transporter, generates the Na+ ion gradient that is used by secondary active transporters, such as the sodium–dicarboxylate 3 (NaC3; formerly designated as NaC2) transporter, to couple uptake of an anionic species to that of Na+ ions down its electrochemical gradient. Two members of the Oat family (Oat1 and Oat3) are considered tertiary active transporters, because they couple the outwardly directed gradient for 2-oxoglutarate (2-OG) with organic anion uptake. Both carriers have a broad substrate specificity, although p-aminohippurate (PAH) and estrone 3-sulfate are considered prototypical substrates for Oat1 and Oat3, respectively. Of the Slc22a-family transporters for organic anions, Oat1 is the predominantly expressed carrier on the BLM of rodent proximal tubular cells, 60 whereas Oat3 is more highly expressed in human proximal tubular cells. 57 Another important difference between human and rodent proximal tubular cells is that OAT2 is expressed on the BLM only in the former species; this carrier mediates the uptake of organic anions from renal plasma/interstitial fluid into the cytoplasm, apparently without coupling or exchange with another anion or cation. Finally, 2 isoforms of the Mrps, Mrp5 and Mrp6, are expressed on the BLM and function as primary active transporters in the efflux of organic anions.
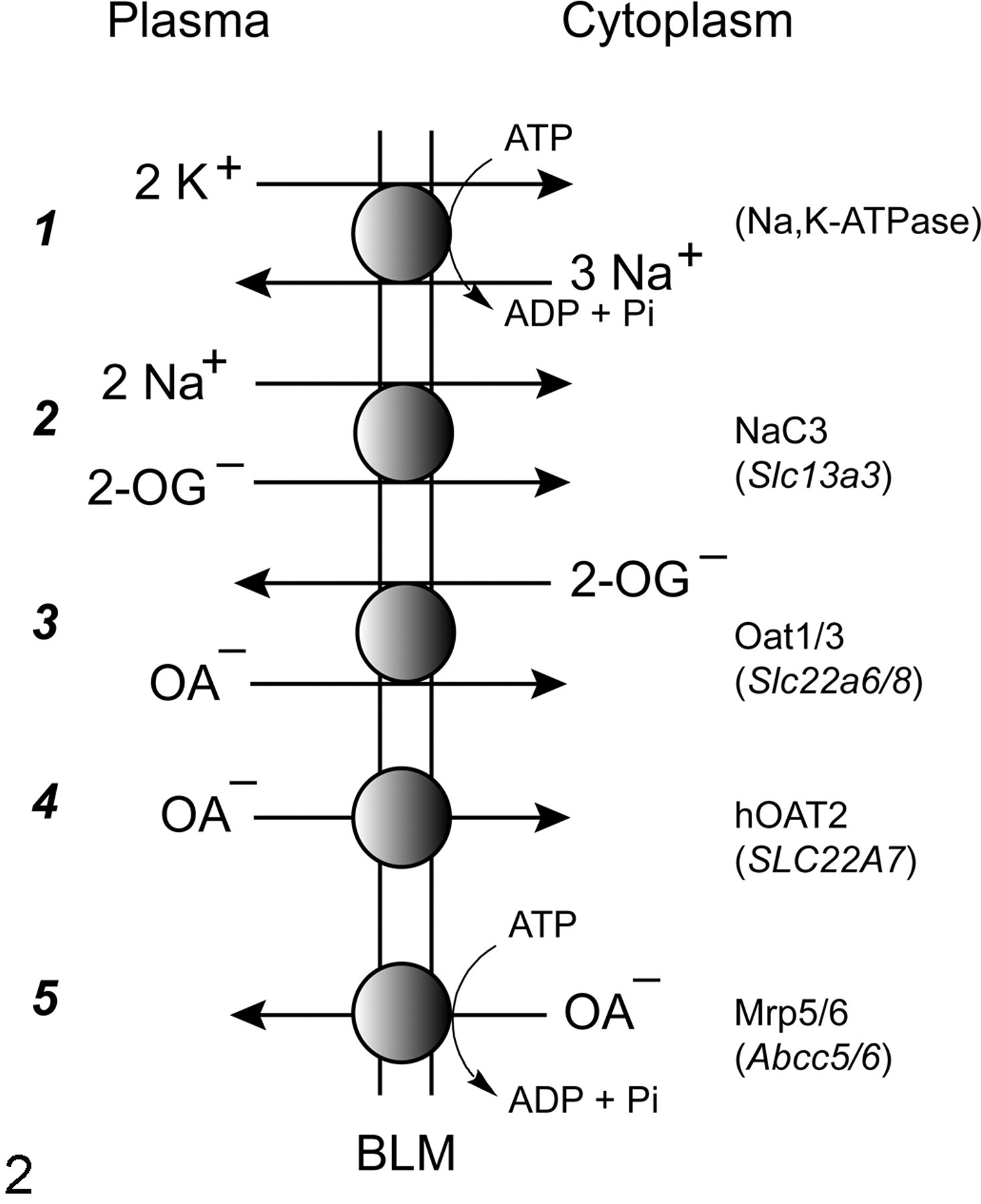
Key carriers for transport of organic anions (OA–) across the renal basolateral plasma membrane (BLM).
Functions of the major carriers for organic anions on the BBM are schematically summarized in Fig. 3 . As in the previous figure, numbers aligned with carriers represent distinct transport mechanisms and follow the sequence in Fig. 2. Oatp1a1 is highly expressed 57,60 and catalyzes the uptake of organic anions from the tubular lumen in exchange for GSH. Although uptake of organic anions at the renal BBM may be important for understanding reabsorption, processes mediating efflux across this membrane are critical as the terminal step in excretion into the urine. hOAT4, which is an OAT carrier protein that is unique to humans, may function as an exchanger or a facilitative carrier. Two variants of another Slc01 family member that are expressed only in rat kidney (Oatp1a3_v1/2) and 2 isoforms of Mrp (Mrp2/4) also play major roles in organic anion efflux into the tubular lumen.
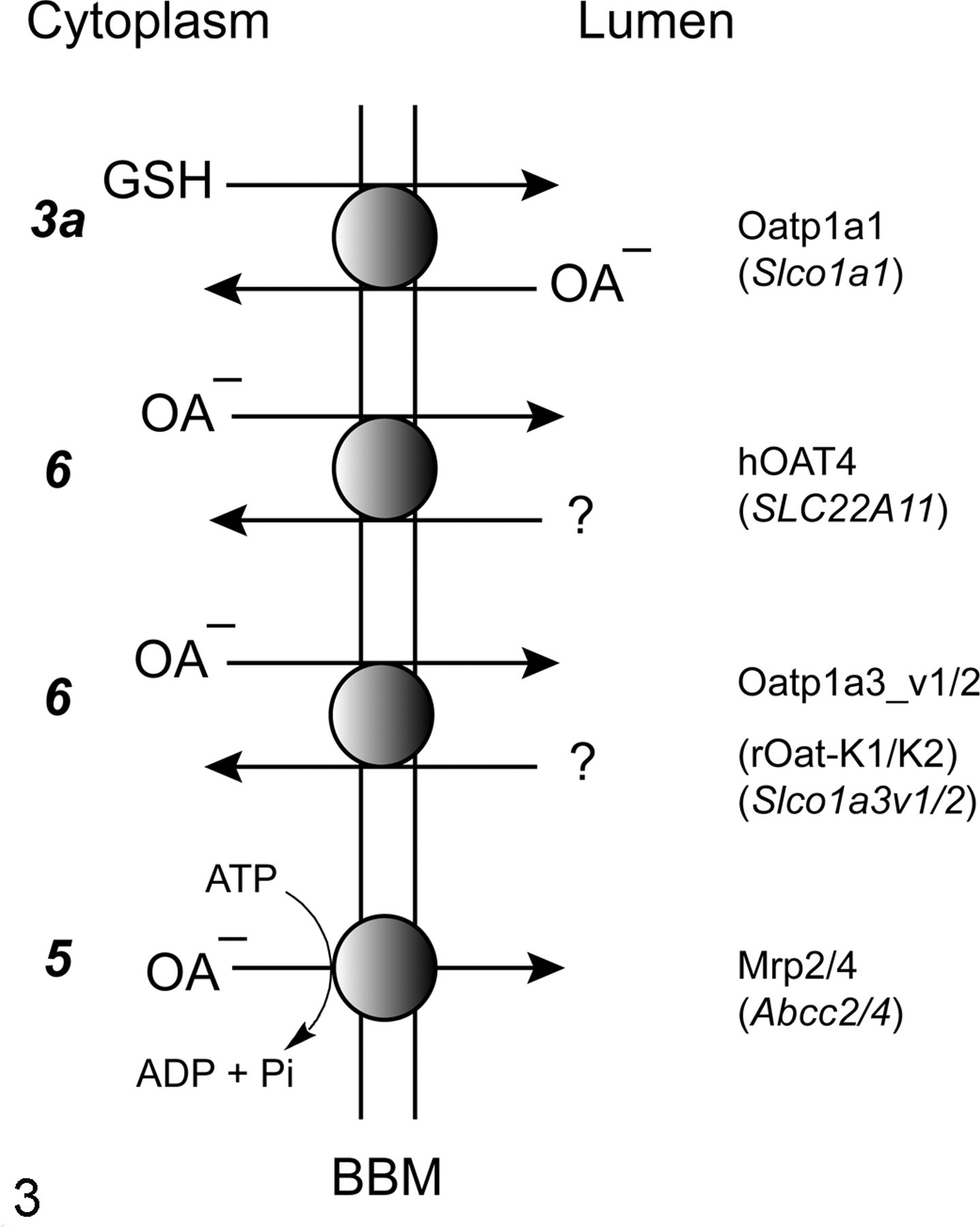
Key carriers for transport of organic anions (OA–) across the renal brush-border plasma membrane (BBM).
The MIM is an important intracellular membrane in renal cells at which transport of organic anions is critical to function. Because of compartmentation between cytoplasm and mitochondria for the various steps in intermediary metabolism of carbohydrates and many amino acids, transport of organic anions across the MIM is a regulatory step in cellular metabolism and energetics. 37,40,72,73,82,87 As shown in Fig. 4 , the various organic anion transporters, with the exception of the monocarboxylate transporter (gene family Slc16a), belong to the Slc25a gene family, of which there are 29 gene products and 3 pseudogenes known in humans. Unlike the organic anion carriers of renal plasma membranes, the mitochondrial carriers have a high degree of substrate specificity, although a range of substrates exist for some of the carriers (see below). The carriers exist in virtually all mitochondria, although there are some tissue and cell type–specific patterns of expression. Substrates for these carriers include adenine nucleotides; inorganic phosphate; mono-, di-, and tricarboxylates, including many citric acid cycle intermediates and glycolytic oxidation products such as pyruvate and lactate; and various amino acids, including glutamate and aspartate. Some of the carriers are electroneutral exchangers (ie, no net transfer of charge across the MIM), such as the dicarboxylate carrier (DIC; Slc25a10), which exchanges 2-OG for other dicarboxylates, and the 2-oxoglutarate carrier (OGC; Slc25a11), which exchanges 2-OG for inorganic phosphate, whereas others are electrogenic exchangers (ie, net transfer of charge across the MIM), such as the adenine nucleotide translocases (ANT1/2; Slc25a4/5).
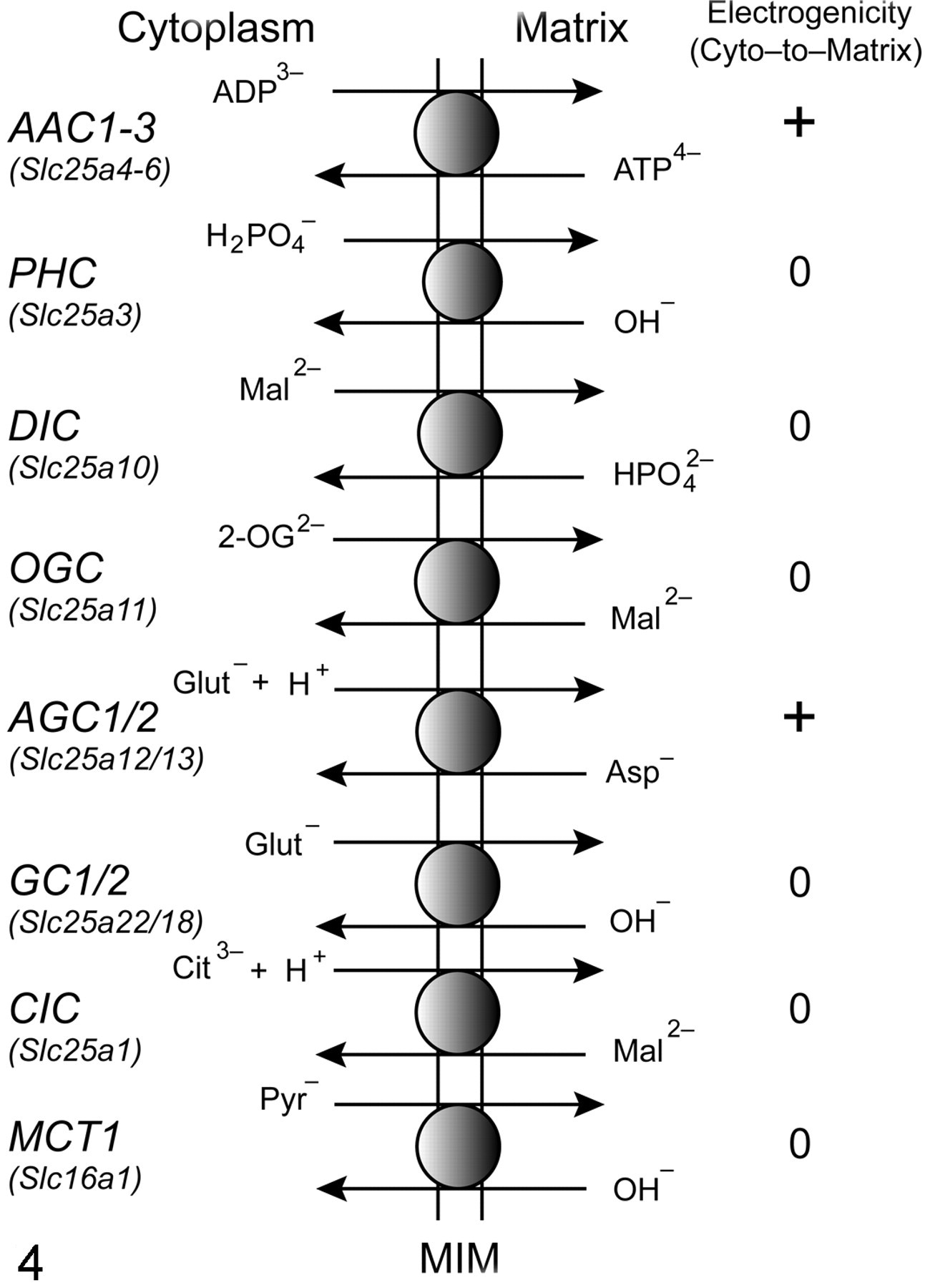
Major organic anion transporters of the renal mitochondrial inner membrane (MIM). AAC1-3, isoforms of the adenine nucleotide translocase that mediate electrogenic exchange of matrix adenosine triphosphate (ATP) for cytoplasmic adenosine diphosphate (ADP); PHC, inorganic phosphate carrier, which mediates electroneutral exchange of monovalent inorganic phosphate for hydroxyl ion; DIC, dicarboxylate carrier, which mediates electroneutral exchange of dicarboxylates such as malate (Mal2–) for divalent inorganic phosphate; OGC, 2-oxoglutarate carrier, which mediates electroneutral exchange of 2-oxoglutarate (2-OG) for other dicarboxylates such as Mal2–; AGC1/2, isoforms of the glutamate–aspartate exchanger, which mediate electrogenic exchange of glutamate (Glut–) and a proton for aspartate (Asp–); GC1/2, isoforms of the glutamate hydroxide exchanger, which mediate electroneutral exchange of Glut– for hydroxyl ion; CIC, citrate or tricarboxylate carrier, which mediates electroneutral exchange of trivalent citrate (Cit3–) and a proton for dicarboxylates such as Mal2–; MCT1, monocarboxylate carrier, which mediates electroneutral exchange of monocarboxylates such as pyruvate (Pyr–) for hydroxyl ion. + = electrogenic; 0 = electroneutral.
Principles of GSH Homeostasis and Role of Plasma Membrane Transport
GSH is a tripeptide composed of the amino acids L-glutamate, L-cysteine, and glycine. Unlike most proteins and peptides, the L-glutamyl and L-cysteinyl residues are linked through the γ-carboxyl group of L-glutamate, making the isopeptide bond resistant to most proteases. GSH plays numerous roles in cells, including maintenance of thiol–disulfide status of proteins, as a substrate and cofactor in various drug metabolism reactions, and regulation of cell growth, proliferation, and stress response. A recent review by Ballatori and colleagues 7 highlights the numerous associations between alterations in cellular GSH homeostasis and several pathological states and the interface between GSH and certain signaling pathways that are key underlying regulators of cellular responses to many stresses. These alterations include genetic defects in enzymes of GSH metabolism, upregulation or downregulation of GSH synthesis, and changes in membrane transporters that play roles in cellular and tissue distribution of GSH.
Unlike most peptides and proteins, GSH is not synthesized on ribosomes but is synthesized in the cytoplasm of most cells by the sequential action of 2 adenosine triphosphate (ATP)–dependent enzymes, the glutamate–cysteine ligase (GCL; also called γ-glutamylcysteine synthetase; EC 6.3.2.2) and GSH synthetase (GS; EC 6.3.2.3). As with many highly regulated compounds, GSH degradation occurs by a pathway separate from that of its synthesis and occurs predominantly in the renal proximal tubular cells. 48 As shown in Fig. 5 , degradation of luminal GSH involves initial cleavage of the γ-glutamyl peptide bond between the L-glutamyl and L-cysteinyl residues by either a hydrolysis or transpeptidation reaction (reaction 3), which is catalyzed by γ-glutamyltransferase (GGT; EC 2.3.2.2), followed by hydrolysis of the peptide bond between the L-cysteinyl and glycyl residues (reaction 4), which is catalyzed by dipeptidase (DP) activity. These degradation reactions are followed by cellular uptake of the constituent amino acids by BBM amino acid transporters (reactions 5a, 5b, and 5c). These amino acids are salvaged for protein synthesis (particularly L-cysteine, which is limiting) or for intracellular resynthesis of GSH. Whereas GSH synthesis occurs intracellularly in the cytoplasm, degradation occurs extracellularly because both GGT and DP are integral membrane proteins whose active sites face the external side of those cells in which the protein is found. Another major difference between synthesis and degradation is that whereas GCL and GS are found fairly ubiquitously in the cytoplasm of mammalian cells, GGT is selectively localized predominantly on the BBM of several epithelial cells, including those of the renal proximal tubule, jejunal epithelium, biliary epithelium, and choroid plexus.
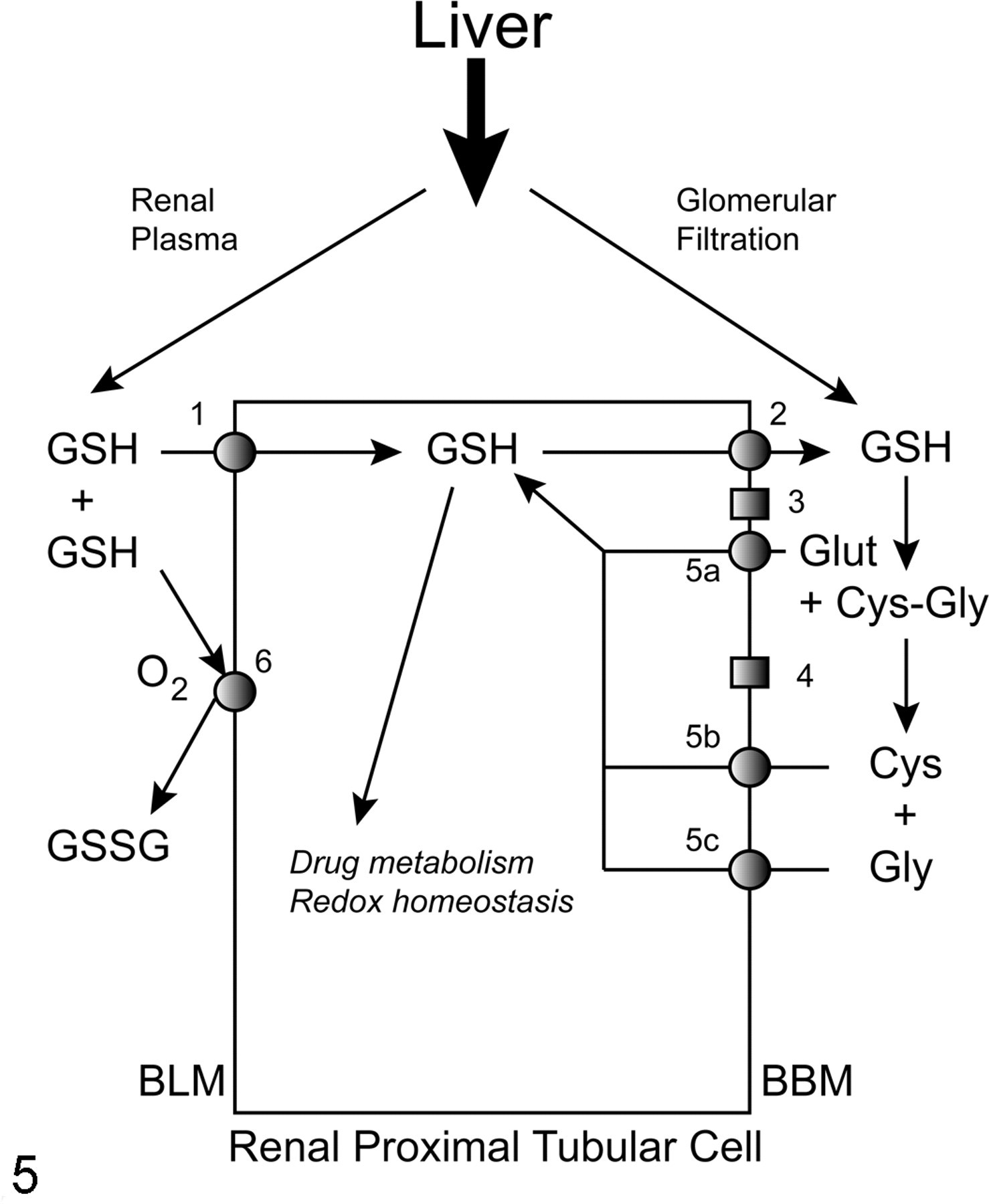
Role of the kidneys in interorgan metabolism of glutathione (GSH). The liver is the primary source of extracellular GSH, to which the kidneys become exposed by either glomerular filtration or in the renal plasma. At the basolateral plasma membrane (BLM), GSH either is transported into the proximal tubular cell by various uptake carriers (pathway 1) or can be oxidized to glutathione disulfide (GSSG) by thiol oxidase (pathway 6). At the brush-border plasma membrane (BBM), luminal GSH is derived from either glomerular filtration or efflux (pathway 2). In the tubular lumen, GSH is successively degraded to its constituent amino acids (L-glutamate [Glut], L-cysteine [Cys], glycine [Gly]) via intermediate formation of the dipeptide L-cysteinylglycine (Cys-Gly), as catalyzed by γ-glutamyltransferase (GGT; pathway 3) and a dipeptidase activity (DP; pathway 4). The constituent amino acids can be reabsorbed into the proximal tubular cell by various amino acid transporters (pathways 5a, 5b, 5c).
Another source of GSH for the kidneys, besides glomerular filtration and intracellular synthesis, is the GSH present in renal plasma that does not undergo glomerular filtration. This pool of GSH can have 2 fates: transport across the BLM into the renal proximal tubular cell by multiple carriers (see below; Fig. 5, reaction 1) or oxidation to glutathione disulfide (GSSG) (Fig. 5, reaction 6), which is catalyzed by a copper-containing, integral membrane enzyme called thiol oxidase (EC 1.8.3.2). 50,54 The precise physiological function of this enzyme and its importance in overall GSH homeostasis are unclear at present. It is clear, however, that whereas many intracellular enzymes and proteins that have cysteinyl residues that are critical for their function require the sulfhydryl groups to be in the reduced form for optimal activity, most membrane and extracellular enzymes and proteins with critical cysteinyl residues require the sulfhydryl groups to be either oxidized or in a disulfide linkage. Hence, it is reasonable to assume that this oxidase activity plays some role in determining the redox state in the extracellular milieu.
Whereas GSH synthesis occurs in most cells, including the renal proximal tubules, the liver has the highest synthetic capacity for GSH and, via efflux across both the canalicular and sinusoidal plasma membranes, is the primary source of extracellular GSH. Because the GSH molecule is resistant to breakdown by proteases, it can translocate as the intact tripeptide from the liver to the kidneys through the plasma. Accordingly, the concept of “interorgan metabolism” was conceived for GSH in the late 1970s to explain the apparent release of large amounts of GSH from the liver and its removal by the kidneys and other organs and was later shown to be a highly regulated process involving hormonal and other signals. 4,6,9,21,24,25,28,31,65,70,71,75,85,86,91 These processes also function in an analogous manner in the interorgan metabolism of GSH S-conjugates and are responsible for their selective and potent nephrotoxicity. 55 Hence, the differential subcellular localization and tissue distribution of membrane transporters and GGT determine the directional flux of GSH and GSH S-conjugates from liver to kidney. 9
GSH Transport Across Renal BLM: Physiological and Toxicological Aspects
From the above discussion of interorgan metabolism, it is clear that whereas the liver is the primary source of plasma GSH, the kidneys are the major sites for removal and turnover of GSH. Such removal can occur by 2 mechanisms: glomerular filtration and transport across the BLM. During a single pass through the renal circulation, 80% of the plasma GSH is extracted by the kidneys; because glomerular filtration only accounts for only ~30% extraction, this means that more than half of the GSH that is extracted by the kidneys occurs by a basolateral mechanism. 3,25,28 The dogma in the literature in the 1970s was that intact GSH was not taken up via carrier-mediated transport by cells of renal or any other tissue origin and that any apparent uptake was actually due to degradation of GSH, uptake of the constituent amino acids, and intracellular resynthesis of GSH. 1,5,35 Despite a large and varied database of studies in multiple experimental models (ie, in vivo studies in rats and in vitro studies in isolated perfused rat kidneys, isolated proximal tubular cells, isolated perfused proximal tubules, and isolated BLM vesicles) and from multiple laboratories, even a recent review 8 continued to dispute the occurrence and physiological significance of carrier-mediated uptake of intact GSH into the renal proximal tubular cell. This issue and the evidence in support of BLM GSH transport have recently been discussed in some detail. 45,49
Overwhelming evidence demonstrated uptake of GSH at the renal BLM by both Na+-dependent and Na+-independent processes. 51,52 Moreover, studies of BLM GSH transport in the presence of selective inhibitors and substrates for known carriers and investigations of cation, ATP, and membrane potential dependence of transport suggested the potential function of at least 3 transport proteins. 52,56,89 As illustrated in Fig. 6 , these specificity, energy, and cation dependence studies were consistent with the uptake of GSH by 2 mechanisms: The first proposed mechanism involves coupling of GSH uptake to that of Na+ ions by action of the NaC3 carrier. The second proposed mechanism involves the exchange of GSH with 2-OG by action of Oat1 and/or Oat3. The energetics underlying the transport processes are also summarized in the figure, showing that although there is a large electrochemical gradient that disfavors GSH uptake (ie, ≥ 250-fold concentration gradient from cytoplasm to plasma and an inside-negative membrane potential), coupling of GSH uptake with Na+ ions or exchange with 2-OG can readily account for the ability to transport against this gradient.
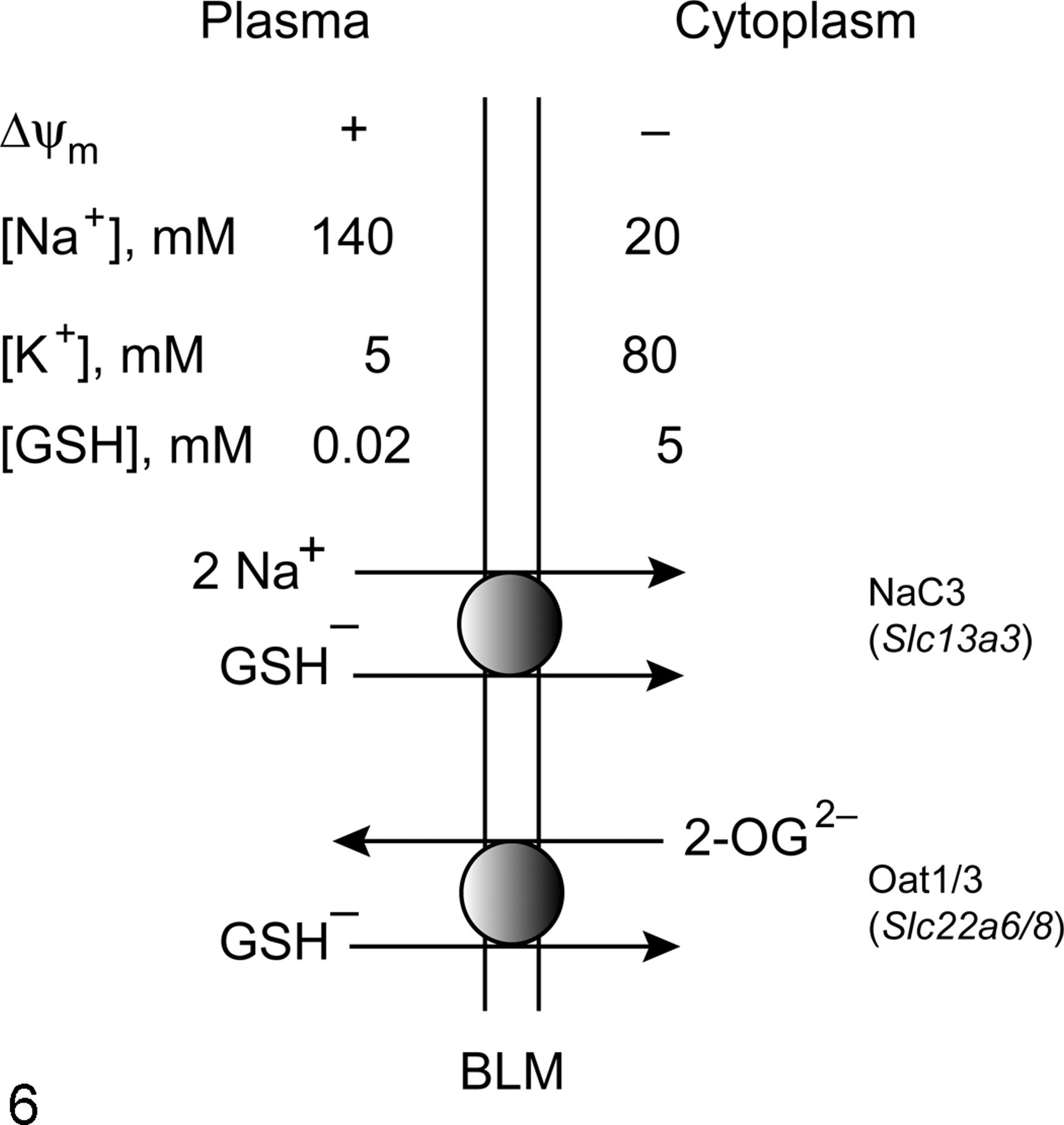
Energetics of glutathione (GSH) transport across the renal basolateral plasma membrane (BLM). GSH is transported as an organic anion either by cotransport via the sodium–dicarboxylate 3 carrier (NaC3) or in exchange for 2-oxoglutarate (2-OG) via either organic anion transporter 1 or 3 (Oat1/3). Driving forces that influence GSH uptake across the BLM include the inwardly directed positive membrane potential (ΔΨm), the transmembrane gradients for Na+ and K+ ions, and the outwardly directed GSH-concentration gradient.
Regarding identification of the carriers responsible for mediating uptake of GSH across the renal BLM, the indirect evidence in support of the 3 carriers illustrated in Fig. 6 included coupling of a portion of the total uptake to that of 2 Na+ per molecule of GSH (NaC3), stimulation of uptake by an inwardly directed negative membrane potential (NaC3), inhibition by probenecid and PAH (Oat1/3), and inhibition by dimethylsuccinate (NaC3, Oat1/3). Direct support for the function of a specific carrier was recently described for Oat3. 60 In that study, we demonstrated that bacterially expressed and reconstituted Oat3 transported GSH in exchange for both PAH and 2-OG and PAH and 2-OG in exchange for GSH.
As noted above and discussed in recent reviews, 45,49 there is uncertainty regarding the precise physiological function of GSH uptake across the renal BLM. Although it has been argued that there is no compelling rationale for BLM uptake to contribute significantly to overall GSH turnover under normal, physiological conditions, 8 consideration of several factors leads us to the overall conclusion that although BLM uptake is but one component of a multicomponent process for renal extraction and turnover of GSH, it does contribute significantly to the process. First, of the 80% of the plasma GSH that is extracted by the kidneys during a single pass through the renal circulation, nearly two thirds of this occurs by a basolateral mechanism that is independent of both renal GGT activity and glomerular filtration. Second, although plasma GSH concentrations are typically 5 to 20 μM whereas renal cellular GSH concentrations are 1 to 5 mM, 3,53 the high rate of renal blood flow relative to renal mass ensures that delivery of GSH to the kidneys via the basolateral route will be quantitatively significant. Other potential functions for BLM GSH uptake, such as function as a carrier for L-cysteine or salvage of γ-glutamyl peptides, may also be important and are discussed elsewhere. 49
A more obvious rationale for function of BLM GSH uptake has been generated for toxicological or pathological conditions in which oxidative stress and depleted intracellular concentrations of GSH are present. Hence, pharmacological modulation of BLM GSH transport in oxidatively stressed renal proximal tubular cells markedly protects these cells from injury. 29,56 The use of inhibitors of the putative BLM transporters demonstrated that the protection was dependent on GSH uptake.
GSH Transport Across Renal BBM: Physiological Role in GSH Turnover
In contrast with GSH transport across the BLM, the critical physiological importance of GSH transport across the BBM of renal proximal tubule has long been appreciated. 26,34,83 Rather than uptake occurring, transport of GSH across the renal BBM is primarily if not entirely efflux from the cell into the tubular lumen. Pathways for GSH transport across the renal BBM are illustrated in Fig. 7 . Also unlike the situation at the BLM, transport of GSH across the BBM occurs with strong and favorable driving forces; these driving forces include the outwardly directed GSH electrochemical potential and the extremely high GGT activity on the BBM that would remove virtually all the GSH with which its active site comes in contact. Although these carrier-mediated pathways are theoretically reversible and could mediate uptake of GSH, this would likely only occur under extreme, nonphysiological conditions, such as the presence of greater than millimolar concentrations of GSH in the lumen or inhibited or defective GGT. Nonetheless, experiments showing that inhibition of renal GGT activity in mice produces a profound glutathionuria provide evidence both for the importance of BBM efflux and GGT activity in GSH turnover and for the improbability that GSH uptake across the BBM would occur, even under fairly extreme, nonphysiological conditions. 26
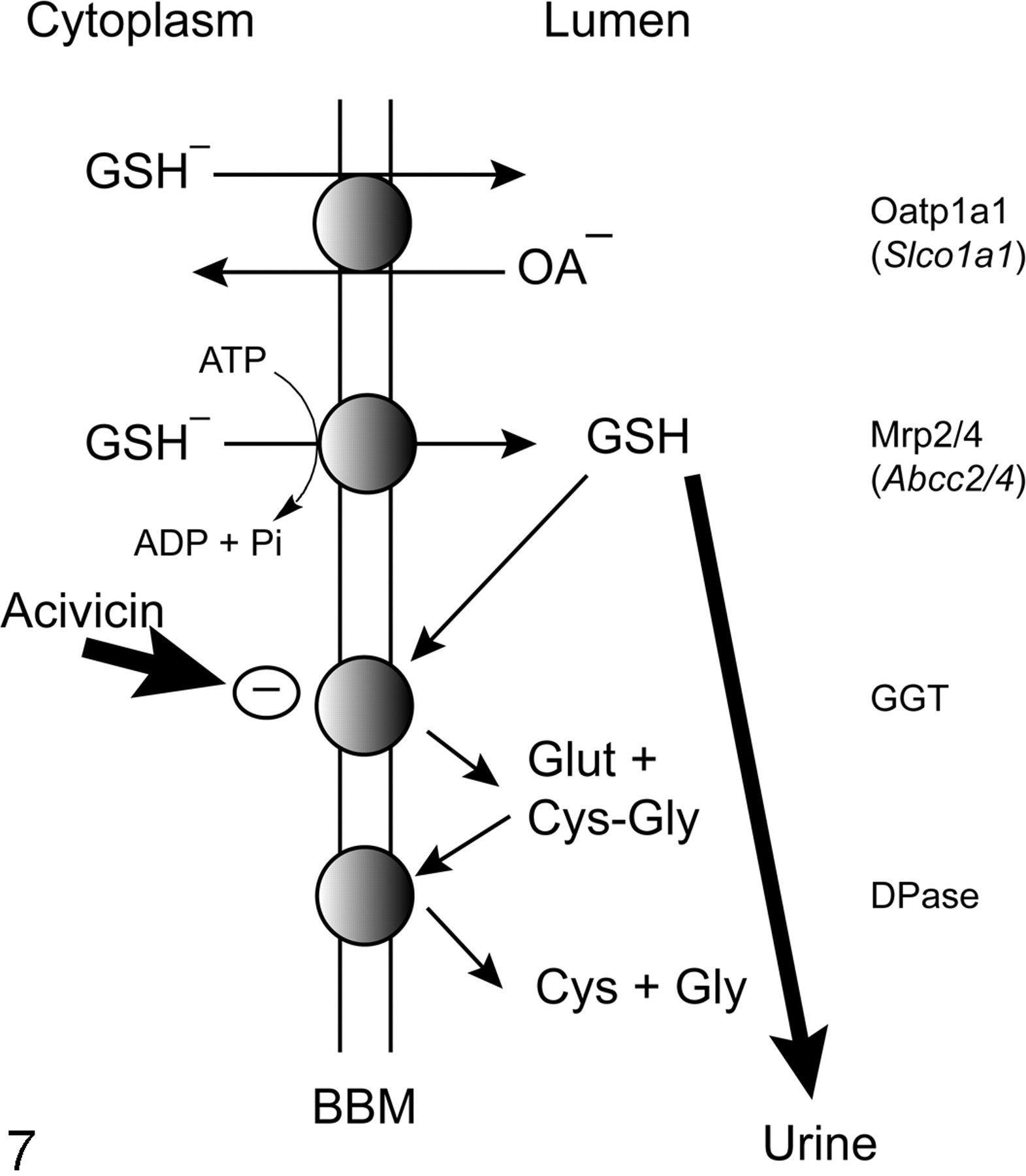
Pathways for glutathione (GSH) transport and metabolism at the renal brush-border plasma membrane (BBM). Efflux of GSH across the BBM into the tubular lumen can be mediated by either the organic anion transporting polypeptide 1a1 (Oatp1a1), which catalyzes GSH efflux in exchange for that of another organic anion (OA–), or by 1 of 2 adenosine triphosphate (ATP)–dependent multidrug resistance proteins (Mrp2/4). In the tubular lumen, GSH is avidly degraded to its constituent amino acids (see legend to Fig. 5). Inhibition of γ-glutamyltransferase (GGT) by acivicin leads to a profound glutathionuria (thick arrow). ADP, adenosine diphosphate; Pi, inorganic phosphate.
Despite the long-appreciated physiological importance of renal BBM efflux of GSH, only indirect evidence for the function of specific carrier proteins is available. Oatp1a1, which is found in other cell types besides the proximal tubules, including hepatocytes, has been shown to use the GSH electrochemical gradient to drive organic anion uptake. 61,68 Additionally, 2 isoforms of the Mrp family, Mrp2 22,23,62,74,76 and Mrp4, 43,77 have been shown to mediate efflux of GSH. As noted above, all 3 carriers are highly expressed in renal proximal tubular cells, leading one to conclude that they function similarly as they do in other cell types and are thus responsible, at least in part, for GSH efflux across the renal BBM.
Transport Across MIM Determines Mitochondrial GSH Pool and Can Modulate Susceptibility to Oxidative Injury and Disease
Whereas plasma membranes are the initial barriers that regulate the flux of material into and out of the cell and separate the cell and its extracellular milieu, intracellular organelles also possess membranes that are essential barriers that regulate their internal composition and function and may be critical for drug disposition and toxicity. This is particularly true of the mitochondria, as discussed above. It has been recognized for many years that the mitochondrial GSH pool is distinctly regulated compared with the cytoplasmic pool and that this has toxicological importance. 46 Although virtually all cells, including those of the kidney and liver, have some capacity to synthesize GSH, the enzymes that do this appear to be localized exclusively in the cytoplasm and are absent from mitochondria. 27,66 Hence, this distinctly regulated pool of GSH must derive from the cytoplasm by transport across the MIM.
Early studies on mitochondrial GSH transport from either rat liver 42,64 or kidney 81 focused on the energetics of the transport process and its relationship to mitochondrial respiratory activity. Because of the importance of GSH as the primary intracellular and intramitochondrial, low-molecular-weight thiol, knowledge of the specific carriers that can catalyze uptake of cytoplasmic GSH is critical. In particular, if a major goal is to therapeutically modulate mitochondrial GSH status to alter intramitochondrial redox status, then identification of the carrier proteins responsible for determining this GSH pool is essential. In our earliest study of GSH transport into renal cortical mitochondria, 66 we reasoned that inasmuch as GSH is a charged molecule at physiological pH, it may be a substrate for one of the members of the family of organic anion/citric acid cycle carriers in the MIM (cf Fig. 4). Accordingly, our initial strategy was to measure transport of GSH into suspensions of isolated mitochondria from rat renal cortex in the absence and presence of selective inhibitors or substrates for several putative anion carriers. Based on those studies, 19,66 we identified 2 of the 8 potential MIM carriers, the DIC and OGC, as being responsible for most if not all of the measurable transport of GSH into renal cortical mitochondria. The other carriers were excluded based on the lack of effect of selective inhibitors or competing substrates or differences in observed energetics. We confirmed this identification further by overexpressing the complementary DNAs (cDNAs) for the 2 carriers, reconstituting them into proteoliposomes, and demonstrating their function in GSH transport with properties similar to those observed in suspensions of isolated mitochondria from rat renal cortex. 20
Identification of the DIC and OGC as the MIM carriers for GSH uptake has important implications for cellular energetics, regulation of cellular and mitochondrial redox status, potential modes of regulation of activities of these carriers, and determinants of susceptibility to oxidative and other forms of chemically induced injury. The relationships between GSH transporters at both the plasma membranes and MIM and cellular energetics are illustrated in Fig. 8 . One important common link between GSH transporters at the BLM and MIM is the central role of 2-OG: 2-OG is a substrate for both the DIC and OGC at the MIM and for the NaC3, Oat1, and Oat3 at the BLM. Changes in cellular intermediary metabolism, such as might occur during starvation, as a consequence of other dietary changes, or in certain diseases such as diabetes, would be expected to alter the supplies of 2-OG, thereby potentially altering the function of the various transporters. A second common link among all these pathways is that of cellular ATP generation, which is itself altered by changes in citric acid cycle activity. Besides being required for the 2 enzymatic steps that synthesize GSH in the cell, ATP is directly required for Mrp2 and Mrp4, which likely mediate efflux of GSH across the BBM, and for the (Na++K+)-stimulated ATPase on the BLM, which helps establish the Na+ ion gradient across the BLM that is necessary for the function of the NaC3.
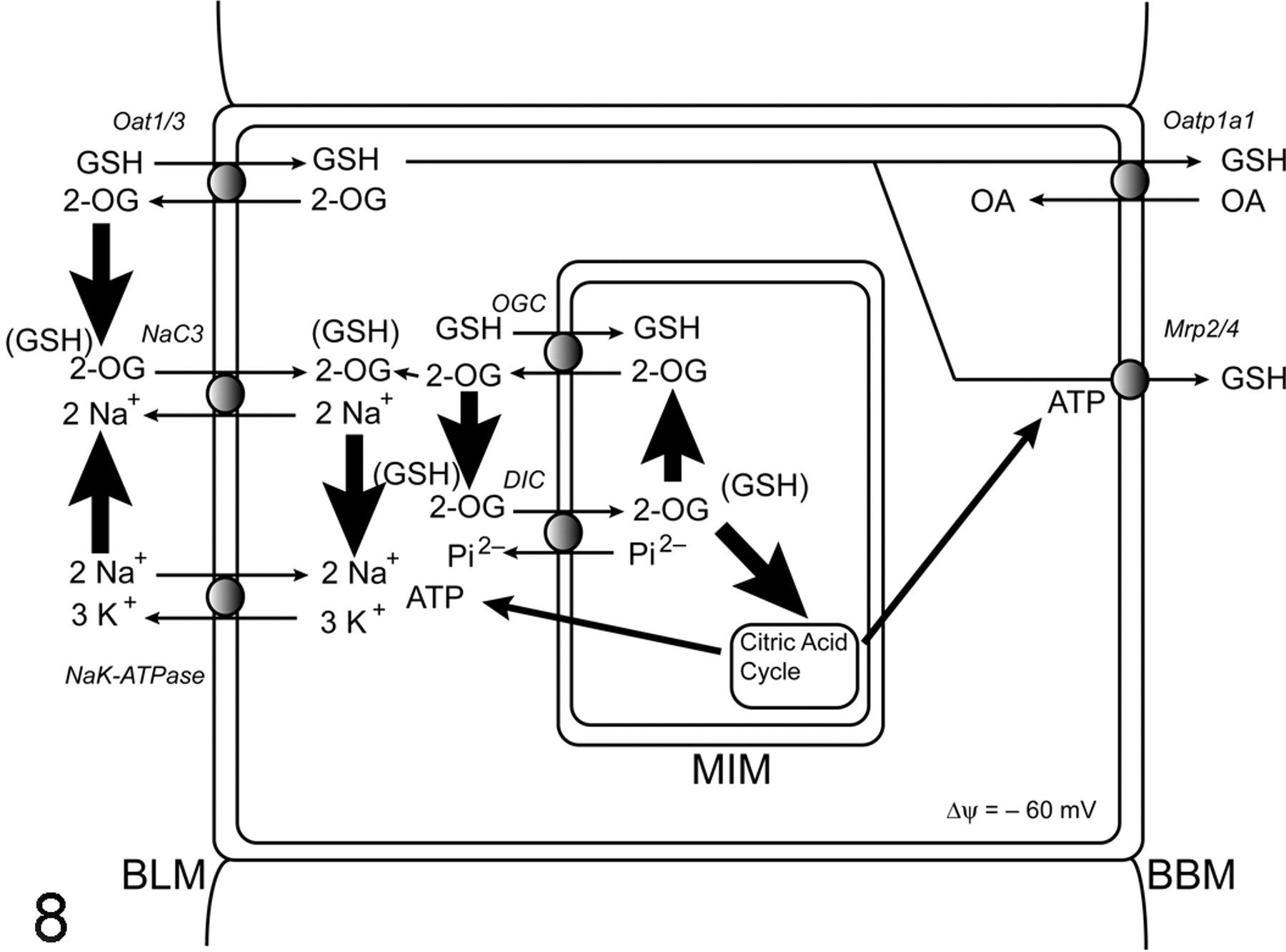
Integration of plasma membrane and mitochondrial glutathione (GSH) transport pathways with mitochondrial metabolism. The scheme highlights several common elements that play key roles in GSH transport and homeostasis in the renal proximal tubule. These include 2-oxoglutarate (2-OG), which is generated in the citric acid cycle and serves as a substrate for both mitochondrial GSH transporters, the dicarboxylate carrier (DIC) and 2-oxoglutarate carrier (OGC), and for the sodium–dicarboxylate 3 carrier (NaC3) and organic anion transporter 1 and 3 (Oat1/3) on the basolateral plasma membrane (BLM), and cellular adenosine triphosphate (ATP) supply, which is generated primarily by mitochondrial oxidative phosphorylation and is used to energize the NaK-ATPase on the BLM and the multidrug resistance proteins 2 and 4 (Mrp2/4) on the brush-border plasma membrane (BBM). GSH efflux across the BBM also can occur by function of the organic anion transporting polypeptide 1a1 (Oatp1a1).
The implications for regulation of transporter activity lie in the existence of critical cysteinyl residues on the DIC and OGC. Although both the DIC and OGC are considered to belong to a superfamily of MIM transporters with similar 3-dimensional structure, 72,73 they exhibit some differences with respect to the numbers and functions of critical cysteinyl residues. The DIC contains 286 or 287 amino acids, depending on species of origin, and a molecular mass of 31 kDa, whereas the OGC contains 314 to 322 amino acids, depending on species, and a molecular mass of 34 to 37 kDa. Both proteins are believed to exist as homodimers. The DIC from rat or bovine liver contains 5 cysteinyl residues at positions 17, 21, 22, 211, and 216. Conversion of any of these cysteinyl residues to serines or alanines by site-directed mutagenesis, however, had no effect on transport activity (J. Wang, F. Xu, D. A. Putt, L. H. Matherly, and L. H. Lash, unpublished observations). It is known, however, that the DIC readily undergoes oxidative inactivation upon purification and isolation, 12,38 suggesting that whereas oxidation of the thiol groups in the native protein affects activity, the functional significance of these cysteinyl residues is unclear. In contrast, the OGC from rat brain and heart contains only 3 cysteinyl residues at positions 184, 221, and 224, with the latter 2 forming an intramolecular disulfide bond. 13,14,17,94 Conversion of C221 and C224 to serines by site-directed mutagenesis markedly reduces transport activity, 94 indicating that formation of the intramolecular disulfide linkage is critical for function.
Another difference between the DIC and OGC is that whereas the DIC cDNA sequence appears to be invariant across tissues, with a high degree of amino acid sequence homology, the OGC exhibits much broader cDNA and amino acid sequence differences across both species and tissues. Thus, we found significant sequence differences between rat kidney and liver OGC, 97 although the functional implications of these differences are not entirely clear. One thing that is clear, however, is that although both carriers are expressed in kidney and liver mitochondria, their relative contribution to mitochondrial GSH uptake differs in the 2 tissues. Although the 2 carriers essentially account for virtually all of the detectable GSH uptake in renal cortical mitochondria, 19,66 function of at least 1 additional carrier must be invoked to account for the observable transport in rat liver mitochondria. 97
To directly demonstrate the toxicological role of mitochondrial GSH transport in renal cells, we used an immortalized cell line derived from normal rat proximal tubules (NRK-52E cells) as a convenient experimental model. 58 Overexpression of the cDNA for either the DIC 59 or the OGC 94 markedly increased mitochondrial GSH transport rates and provided significant protection from oxidant-induced cell death by apoptosis. In contrast, overexpression of the cDNA for the double-cysteine mutant of the OGC (OGC-C221,224S), which produces a protein that cannot form the intramolecular disulfide bond and exhibits a greater than 80% reduction in transport activity compared with wild-type carrier, resulted in slight decreases in mitochondrial GSH content and no effect on susceptibility to oxidant-induced cell death. 94 Thus, these results demonstrate that modulation of mitochondrial GSH transport expression and activity can directly affect the response of renal cells to chemicals that elicit cytotoxicity by a common mechanism of action.
There is increasing evidence in the literature that in addition to exposure to drugs and toxic chemicals, many pathological states are associated with significant depletion and/or oxidation of the mitochondrial GSH pool. An important advantage of identifying the specific carrier proteins that are responsible for mitochondrial GSH transport, thus providing the source of this critical pool, is that we are provided with potential therapeutic targets. One such disease state that is of considerable public health interest is diabetic nephropathy, which is a serious and frequent complication of both type 1 and type 2 diabetes. 33 Numerous studies have shown that exposure of the renal proximal tubule to chronic hyperglycemia results in both oxidative and nitrosative stress, 2,10,15,18,78 with consequent mitochondrial dysfunction. 78,79 Alterations in renal mitochondrial GSH status also appear to be associated with chronic hyperglycemia, 36,69,88,92,95 although the underlying mechanism for these changes has not been established.
One possible clue to the underlying mechanism for the mitochondrial redox and functional changes in diabetes comes from a study by Kaplan and colleagues, 39 who demonstrated that expression and activity of several mitochondrial anion transporters are altered in livers of the streptozotocin-induced diabetic rat. Accordingly, we have begun studying the effects of progressive diabetic nephropathy in the streptozotocin rat model on renal mitochondrial GSH transport. We confirmed that mitochondrial energetics and GSH status are significantly altered in kidneys from diabetic rats. 96 Surprisingly, mitochondrial GSH contents in kidneys from diabetic rats are significantly higher and expression and activity of the DIC and OGC are modestly increased compared with those from age-matched control rats. 96 We interpret this as a compensatory response to the oxidative stress that is induced by the chronic hyperglycemic state. Interestingly, however, primary cultures of renal proximal tubular cells from streptozotocin-treated diabetic rats exhibit higher levels of reactive oxygen species and are more sensitive to oxidant-induced injury than cells from age-matched control rats. 98 We interpret these findings to mean that the compensatory increases in mitochondrial GSH transport and content are insufficient to counteract the oxidative stress induced by the chronic hyperglycemic state. Hence, we hypothesize that further overexpression of the DIC and/or OGC will be beneficial to the proximal tubular cells, leading to a suppression of oxidative stress and a reversion to the nondiabetic phenotype. Studies to test this hypothesis are currently underway.
Conclusions
Membrane transport processes are a critical component of renal function and are involved in numerous aspects of drug metabolism, disposition, absorption, and excretion. Moreover, because of the remarkably high rate of renal blood flow, the process of glomerular filtration, the presence of numerous carrier proteins on renal plasma membranes (particularly those of the proximal tubule), and the presence of bioactivation enzymes in renal epithelial cells, the kidneys are frequent targets for toxic chemicals. As an example of transport processes that are significant for renal toxicology and disease, GSH transport across renal BLM, BBM, and MIM has been discussed. Some of the data discussed here demonstrate how genetic manipulation of GSH transporter expression may serve as a novel strategy for a therapeutic approach to treatment of certain pathological states. Wallace 90 has provided a convincing and comprehensive analysis and proposed that changes in mitochondrial energetics and redox status underlie a vast number of metabolic and degenerative diseases, as well as aging and cancer. In summary, this review has provided an example of how transport processes can play a central role in renal susceptibility to chemical toxicants and in pathological states.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: National Institute of Diabetes and Digestive and Kidney Diseases (grant R01-DK40725) and the Department of Defense (grant PR064340).
