Abstract
Gelatinous marrow transformation, or serous atrophy of bone marrow fat, has been noted in livestock, laboratory animals, and wildlife in association with an inadequate plane of nutrition, inanition, or intoxication. This is a report of gelatinous marrow transformation and hematopoietic marrow atrophy in a 5-year-old miniature horse stallion. The horse had oral malformations leading to poor food assimilation and emaciation. A bone marrow biopsy obtained to investigate persistent anemia and leukopenia showed hematopoietic atrophy and replacement of fat with a granular extracellular substance, which stained with alcian blue, consistent with acidic mucopolysaccharide content. Surgical correction of the dental abnormalities resulted in improved food assimilation, weight gain, and resolution of cytopenias. In humans, gelatinous bone marrow transformation and hematopoietic atrophy are commonly associated with malnutrition from anorexia nervosa and other causes. The cause of hematopoietic atrophy is unknown but may relate to a nonsupportive marrow microenvironment and inadequate hematopoietic substrate availability. Similar pathogenic mechanisms were suspected in this horse.
Gelatinous marrow transformation (GMT), or serous atrophy of bone marrow fat, has been noted as a gross lesion in ruminants, 6 male Göttingen minipigs, 4 reindeer, 12 and rabbits 25 in association with inadequate nutrition or inanition. GMT and hematopoietic tissue atrophy have also been observed in swans with lead intoxication, 21 in dairy calves born to dams with fluoride intoxication, 18 and in calves with experimentally induced sarcocystosis. 11 The cause of GMT is poorly understood. In humans it is often associated with malnutrition due to anorexia nervosa but also with cancer, infection, maldigestion, heart failure, and endocrine disorders. 3,10 GMT is frequently but not always associated with weight loss; it is therefore thought that stress and other bioregulatory derangements also contribute to its development. 3 The factors leading to hematopoietic atrophy are unknown but may relate to an inimical bone marrow environment, 9,13,25 with inadequate hematopoietic substrate availability, lack of stimulation from adipocytes or fibrocytes, and deficiency of colony-stimulating factors. 9,13 Histologically, atrophy of adipocytes and hematopoietic tissue is accompanied by an accumulation of gelatinous material that, based on histochemical staining, is hyaluronic acid-rich mucopolysaccharide without reticulin fibers. 3 Lesions are usually focal but may become diffuse with increased severity. 3 The composition of the extracellular material may differ depending on the type of associated disease 3 and whether the malnutrition involves a lack of carbohydrates and fats or a lack of protein. 10,13 Mucopolysaccharide extracellular matrix is not present in normal bone marrow. 3 In cases of starvation, the lesions are often rapidly reversible, with return to an adequate plane of nutrition. 3,25 We report here a case of GMT and hematopoietic atrophy in a horse, as diagnosed from bone marrow aspirate and core biopsy preparations.
Clinical History
A 52-kg, 5-year-old miniature horse stallion was presented to the Ontario Veterinary College Teaching Hospital with a history of nasal discharge and weight loss of 2 months' duration (day 0). The horse had had recent dental floatation, had been administered an anthelmintic, and had been treated with trimethoprim–sulfamethoxazole (TMP-SMZ, 30 mg/kg every 24 hours) for 3 weeks before presentation, with partial resolution of the nasal discharge. During that time, the horse’s daily ration was considered nutritionally adequate, as it consisted of a mixture of oats, sunflower shoots, alfalfa cubes, and a mineral supplement.
Abnormalities on physical examination included increased bronchovesicular lung sounds consistent with mild airway disease and a grade III–IV holosystolic heart murmur. There were no significant findings on thoracic radiography. Electrocardiography was within normal limits; mild mitral valve regurgitation was detected by cardiac ultrasound but was considered clinically insignificant. Oral examination revealed brachygnathia superior, bilateral lingual displacement of both maxillary fourth premolar teeth, and associated bilateral fistulae. Nasopharyngeal endoscopy was performed, and impacted feed was removed from the nasal cavity. Computed tomography confirmed misalignment of the maxillary fourth premolar teeth, impacted feed between the right maxillary third and fourth premolar teeth, an oronasal fistula between the left maxillary third and fourth premolar teeth, and left maxillary and frontal sinusitis. Additional findings included sinus asymmetry, nasal septal deviation, a left mucoid sinusitis, and inflammation of the right nasal conchae. Abnormal hematology results included a mild normocytic normochromic anemia with mild hyperproteinemia (day 0; Table 1 ).
Serial Complete Blood Cell Counts From a Horse With Bone Marrow Atrophy a
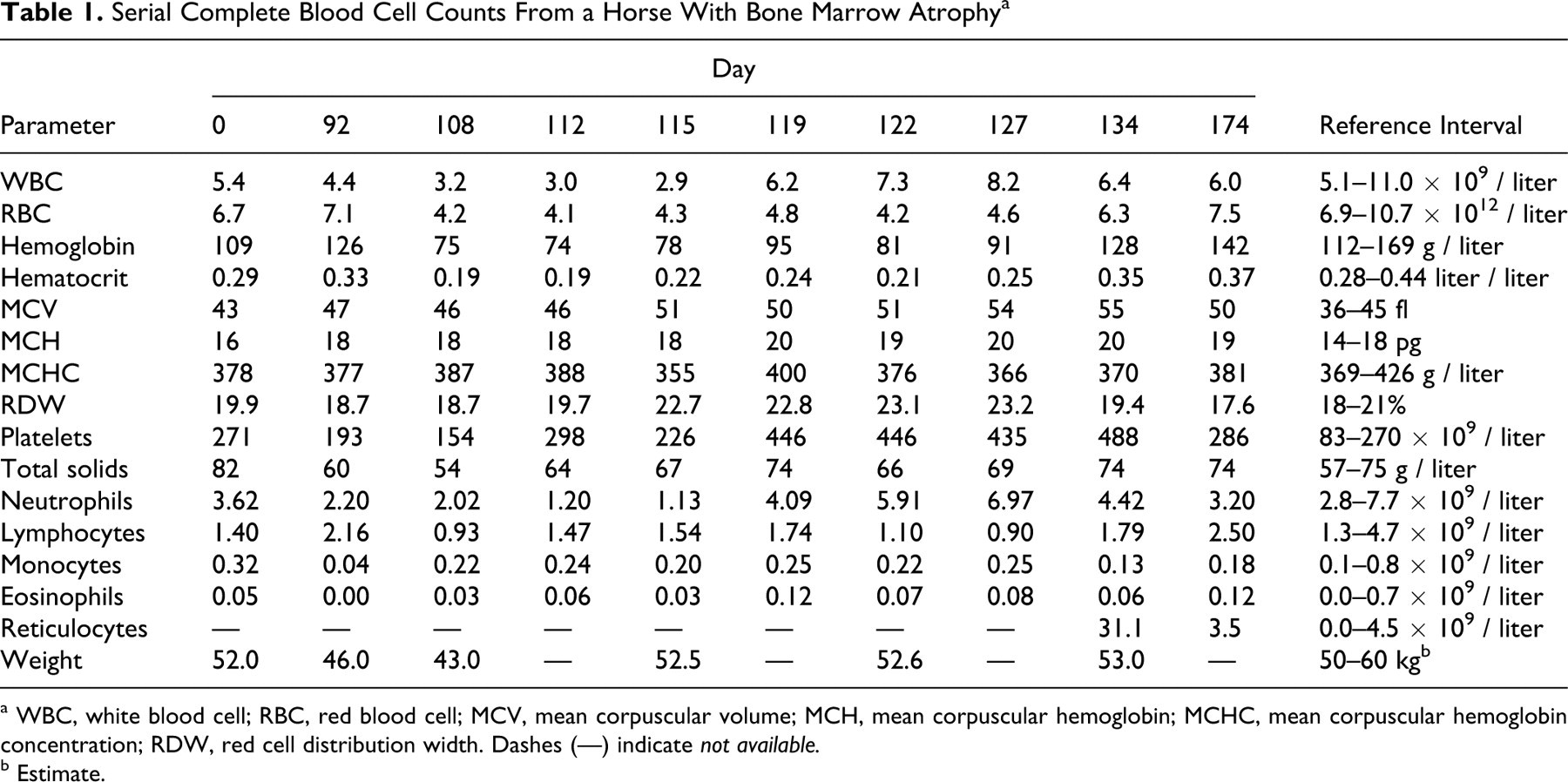
a WBC, white blood cell; RBC, red blood cell; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RDW, red cell distribution width. Dashes (—) indicate not available.
b Estimate.
On day 2, under general anesthesia, the upper-left fourth premolar tooth was removed by repulsion via a frontomaxillary sinus flap. Surgical blood loss necessitated hemostasis with cocaine-soaked gauze, a blood transfusion (with cross-matched whole blood), and administration of intravenous colloids. Perioperative drug therapy included TMP-SMZ (30 mg/kg every 12 hours over 5 days) and sodium penicillin, as well as phenylbutazone (2.2 mg/kg every 12 hours over 6 days), fluticasone propionate, and salbutamol. Recovery was uneventful. The horse returned several times over the ensuing months for ongoing care of the surgical site.
On day 92, the horse was presented with a history of recent depression and inappetence. Serum albumin (24 g/liter; reference interval, 30–37 g/liter) and creatinine (56 μmol/liter; reference interval, 80–130 μmol/liter) concentrations were low. Abnormal hematologic results included leukopenia with neutropenia (Table 1).
The horse was re-presented on day 105 with history of a regained appetite, and it was consuming a slurry diet of complete-nutrition concentrate and alfalfa cubes. Despite this, the horse had lost approximately 17% of its body weight relative to day 0 (Table 1). On physical examination, no new abnormalities were identified, but the right-sided fistula had extended into the maxillary sinus. Therefore, on day 106, the right maxillary fourth premolar was extracted under general anesthesia and the fistula repaired. Postoperatively, the horse was treated with TMP-SMZ and phenylbutazone. The horse’s nutrition was subsequently improved through gradually increased amounts of alfalfa cubes with concentrate suspended in water. The horse had a good appetite and gained 7 kg between days 106 and 112. Additional laboratory results on days 108 and 109 included negative fecal flotation results for parasites and serum iron concentration of 9 μmol/liter (reference interval, 12–47 μmol/liter), with an unsaturated iron-binding capacity of 28 μmol/liter (reference interval, 17–63 μmol/liter). Total iron-binding capacity was therefore 37 μmol/liter, with a calculated saturation of 24%; these values were interpreted to be consistent with systemic inflammation. 5
On day 112, a foul odor emanated from the right nostril of the horse, and an anaerobic infection was suspected. On thoracic auscultation, crackles and wheezes were heard. From radiographic and ultrasonic evaluation of the chest, pleuropneumonia with a consolidated ventral lung lobe was diagnosed. The horse developed tachypnea and pyrexia later that day, and intravenous antimicrobial therapy was begun with penicillin, gentamicin, and metronidazole. A moderate macrocytic normochromic anemia and leukopenia were detected by complete blood count (Table 1). The latter—in light of recent surgery, pyrexia, and pneumonia—was considered an inadequate granulopoietic response.
Bone Marrow Evaluation
To investigate the cytopenias, a bone marrow core biopsy and aspirate from the right tuber coxae were obtained on day 113. On preparation of the bone marrow aspirate smears, poorly drying gelatinous material was apparent. Microscopically, the stained preparations contained abundant pink, amorphous, granular extracellular material in addition to hematopoietic cells (Figs. 1, 2 ). Granule cellularity was estimated at 30 to 40%. Megakaryocytes were morphologically normal but slightly reduced in number. Erythroid precursors were increased relative to granulocytic precursors with an approximate myeloid:erythroid ratio of 1:4, and there were relatively few metarubricytes and polychromatophilic cells. Granulocytic maturation was unremarkable, and stained iron was present.
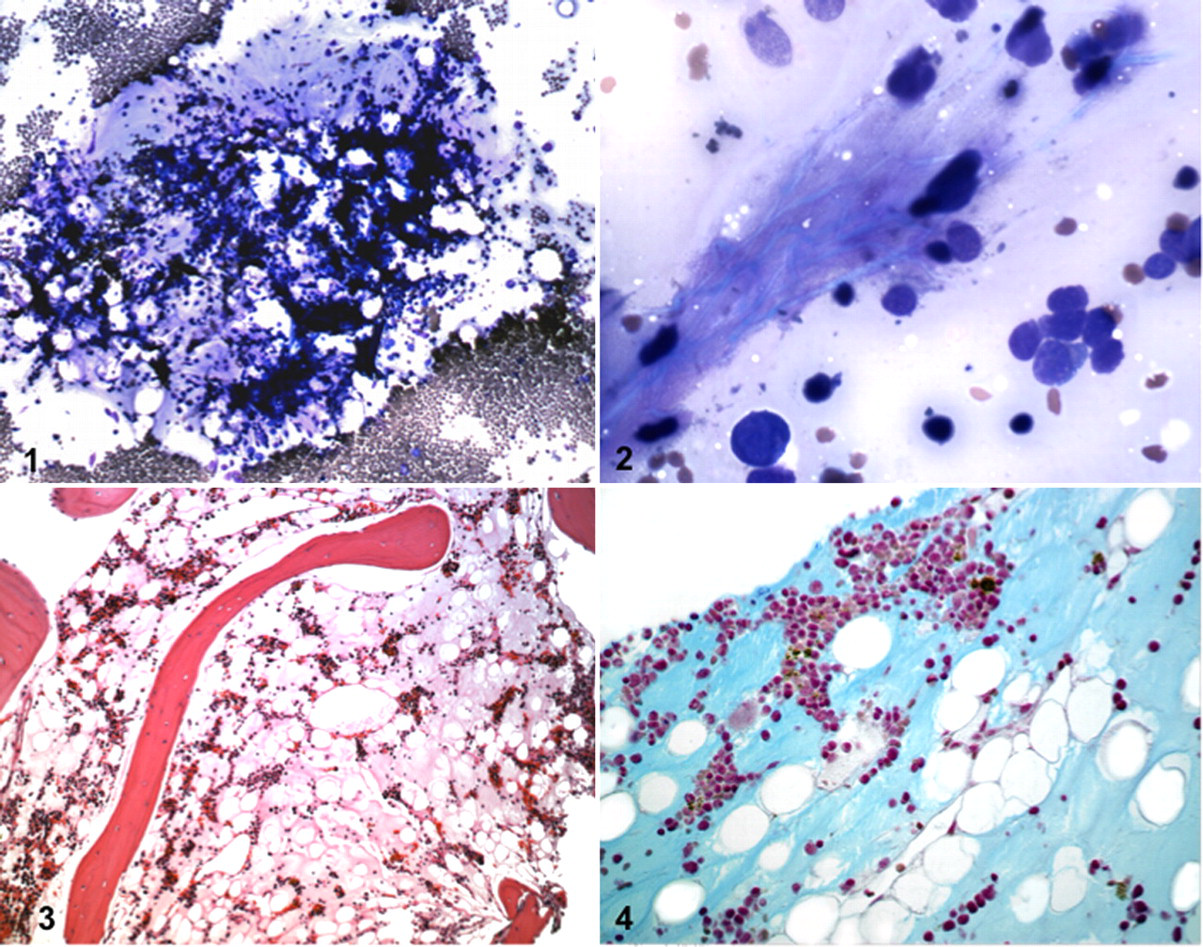
The bone marrow core biopsy was placed in rapid decalcifying solution for 20 minutes, followed by 24 hours' fixation in 10% neutral buffered formalin. The sample was routinely processed, and 4-μm sections were stained with hematoxylin and eosin. Histologically, the bone marrow was estimated to be 20% cellular; all hematopoietic cell lineages were present. Megakaryocytes were reduced in number. Adipocytes were present but generally small and separated by amorphous, eosinophilic, and finely granular extracellular material (Fig. 3 ). Blood vessels were prominent and dilated. The interpretation was atrophy of all hematopoietic cell lineages and serous atrophy of fat.
To further characterize the extracellular material in bone marrow, additional sections of the biopsy were stained with alcian blue at pH 2.5, silver impregnated to highlight reticulin fibers, and subjected to a periodic acid–Schiff reaction. The extracellular matrix was faintly and diffusely alcianophilic (Fig. 4 ). There was an absence of reticulin fibers, and the sections were periodic acid–Schiff negative except for faint staining of vascular basement membranes. These histochemical findings were consistent with an acid mucopolysaccharide extracellular matrix, as noted in cases of GMT of humans. 3,13,16
Case Outcome
Because of the bone marrow changes and in an attempt to stimulate hematopoiesis, empirical daily treatment with lithium carbonate (9 mg/kg every 12 hours over 19 days) was initiated on day 115, along with a 5-day course of intravenous multivitamins. Blood lithium concentration on day 119 was 0.23 mmol/liter; the desired therapeutic range in humans is 0.6–1.2 mmol/liter. Antimicrobial therapy was discontinued on day 122, but lithium treatment continued. Serum albumin and creatinine concentrations remained low (21–22 g/liter and 37–60 μmol/liter, respectively), whereas leukopenia and anemia gradually resolved by day 134 (Table 1). Low albumin and creatinine concentrations were attributed to a combination of low nutrient assimilation, increased protein catabolism, and inflammation, coupled with a generalized wasted state.
Discussion
The differential diagnosis for hematopoietic atrophy included an inadequate plane of nutrition caused by oral disease, anemia of inflammatory disease, suppression due to phenylbutazone or TMP-SMZ, or neoplasia. Neoplasia was ruled out by cytologic and histologic evaluation of the bone marrow.
TMP-SMZ administration has been associated with neutropenia 2 and thrombocytopenia 7 in humans. The mechanism for neutropenia was considered to be inhibition of dihydrofolate reductase under folate-deficient in vitro conditions. 2 No mechanism was suggested for induction of thrombocytopenia, which resolved within a median of 7 days after cessation of TMP-SMZ. 7 No primary references were identified that discussed the effects of TMP-SMZ on equine bone marrow. Additionally, because this horse was severely ill, no gastrointestinal absorption studies were undertaken. The possibility therefore remains that the horse was in a folate-deficient state and that TMP-SMZ could have caused the neutropenia. However, TMP-SMZ is widely used in horses and has not been associated with anemia or with GMT.
In humans, phenylbutazone administration has been associated with granulocytopenia, 1,23 which was attributed to an underlying, genetically determined sensitivity. 23 In horses, reports of hematologic effects of phenylbutazone are mixed. 14,15,17,19,24,26 Decreased albumin concentration, attributed to gastrointestinal loss, 14,17,19,24 and neutropenia, associated with a much higher dose of phenylbutazone administered over a longer time, were reported. 19 Hence, although it is possible that phenylbutazone had toxic effects on granulopoiesis in this horse, it was considered unlikely.
Anemia of inflammatory disease would explain the mild anemia coupled with an ineffective regenerative attempt, but it does not explain the decrease in granulocytes and thrombocytes. Hypoplasia was considered unlikely because the horse was mature and because it initially had normal hematologic findings, apart from a mild anemia.
Atrophy of bone marrow is well recognized in humans with anorexia nervosa, 3,13 as is serous atrophy of adipose tissue in emaciated livestock, laboratory animals, and wildlife. 4,11,12,18,21 In humans, reduced carbohydrate and fat intake is associated with gelatinous fat transformation, hematopoietic atrophy, and accumulation of acid mucopolysaccharides, whereas lack of dietary protein induces hematopoietic and fat atrophy without accumulation of mucopolysaccharide matrix. 20 In the horse presented here, it is unknown whether a lack of carbohydrates and fats, with or without a decrease in protein intake, was responsible for the hematopoietic atrophy. However, if horses respond as humans do, the accumulation of acid mucopolysaccharide matrix in the bone marrow suggests carbohydrate and fat starvation.
Bones of the appendicular skeleton are considered to be more susceptible than flat bones to fat atrophy; 27 hence, in this case, diagnosis of bone marrow atrophy in a pelvic marrow biopsy suggests that gelatinous transformation might have been quite extensive. Bone marrow atrophy secondary to reduced caloric intake in humans typically manifests with anemia, neutropenia, and lymphopenia, as noted in this horse, and less commonly with thrombocytopenia. 10 As in this case, cytopenias in anorexic humans are usually not severe, and neutrophil morphological abnormalities are absent. 10 This horse had mild macrocytosis on days 92 to 112, which became marked between days 115 to 134 (Table 1). The earlier values were considered to reflect increased erythropoiesis as a result of recurrent surgical blood loss, which did not resolve the anemia. Inflammation may have contributed to insufficient erythropoiesis. However, with resolution of emaciation and inflammation, the anemia resolved by day 134. The profound nature of the regenerative response was illustrated by marked reticulocytosis. Commonly measured hemotrophic factors such as iron, ferritin, and erythropoietin are normal in anorexic humans, 10 and marrow and serum iron were considered adequate or marginally decreased in this horse.
In summary, physical inability to assimilate feed because of the oral lesions was the likely cause of emaciation. Removal of the misaligned teeth, surgical correction of the fistulae, improvements in diet, and antimicrobial therapy were associated with resolution of anemia and leukopenia. The horse was also empirically treated with lithium carbonate, which is thought to increase production of granulocyte colony-stimulating factor and stimulate hematopoietic stem cell recruitment, leading to increased granulopoiesis and thrombopoiesis in humans. 8 Blood concentrations considered therapeutic in humans were not achieved in this horse; therefore, whether lithium contributed to hematopoietic recovery is questionable. Reversal of hematopoietic atrophy in anorexic humans occurs within 14 to 25 days after institution of proper nutrition. 3,10,22 Similarly, in this case, anemia and leukopenia resolved within approximately 3 weeks of correction of starvation (Table 1). In the diagnostic investigation of serous atrophy of fat, routine fixation may not be ideal for the identification of an extracellular matrix rich in hyaluronic acid, which is best preserved for optimal staining by fixation in 10% acid formalin with 70% ethanol rather than in 10% neutral-buffered formalin. 16
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
