Abstract
Sheep inoculated with a virulent South African strain of bluetongue (BT) virus serotype 4 developed severe clinical signs and lesions characteristic of fulminant BT, including coronitis, hemorrhage and ulceration of the mucosal lining of the oral cavity and forestomaches, hemorrhage in the wall of the pulmonary artery, and focally extensive necrosis of skeletal muscle, especially of the neck. At necropsy, up to 14 days after infection, the infected sheep exhibited striking pulmonary edema, edema of the subcutaneous tissues and fascial planes of the head and neck, and pleural and pericardial effusion of varying severity. A reliable model for experimental reproduction of fulminant BT in sheep will facilitate future studies to better characterize the pathogenesis of this disease, particularly as it regards the mechanisms responsible for the increased vascular permeability that characterizes BT and related orbiviral diseases such as African horse sickness.
Bluetongue (BT) is a noncontagious, insect-transmitted disease of sheep, certain species of wild ruminants, and rarely of cattle. 17, 18, 19, 23 The disease was first described in the Cape Colony of southern Africa after merino sheep were introduced into the region in the late 18th century, 21 and subsequently has been recognized in other parts of Africa, Europe, the Middle East, the Americas, and Asia. 23 Bluetongue virus (BTV), the causative agent of BT, has now been isolated on all continents except Antarctica, but the species of Culicoides insects that serve as principal vectors of the virus differ among regions. 1, 9, 22 The recent spread of BT throughout extensive portions of both southern and northern Europe has resulted in the deaths of some 1.5 million animals, and the inference that this epidemic is the result of climate change suggests that BT is reemerging in terms of its global significance. 7, 10, 20
Although all species of domestic ruminants are commonly infected with BTV in endemic regions, clinical BT typically occurs only in sheep breeds such as the merino, whereas cattle, goats, and sheep of most breeds usually are only subclinically infected. 13, 18, 21, 23 BT is characterized by microvascular injury that results in hemorrhage, edema, and necrosis of specific tissues of affected sheep, including the skin and hooves, oral cavity and upper gastrointestinal tract, respiratory system, and both skeletal and cardiac muscle. 17, 23 BTV replicates principally in endothelial cells (ECs) and macrophages in the tissues of infected ruminants, 13 but we recently have shown that inherent species-specific differences in BTV-induced production of inflammatory and vasoactive mediators may underlie the susceptibility of sheep and relative resistance of cattle to expression of clinical disease. 4– 6 Specifically, BTV infection of bovine lung microvascular ECs results in their activation with production of a variety of mediators, including prostacyclin, whereas ovine lung microvascular ECs more rapidly undergo necrosis and produce significantly lower amounts of the same mediators as well as lower titers of virus. 4, 6 Similarly, the ratio of plasma thromboxane to prostacyclin, indicative of enhanced coagulation as a consequence of EC injury, is significantly greater in BTV-infected sheep than in cattle at approximately 9 days after infection. 5
In addition to clearly documented differences in the susceptibility of individual species and breeds of ruminants to expression of BT, strains of BTV also vary remarkably in both their virulence and genetic constitution. 1, 8 Furthermore, experimental infections of susceptible sheep with field strains of BTV, including those strains isolated from fulminant cases of the disease, often result only in mild clinical signs. 2, 5, 8, 23 Thus, the objective of this study was to develop a reproducible experimental model of severe BT in sheep that can be used to better characterize the pathogenesis of the disease and the mechanism of microvascular injury in particular.
Nine yearling merino sheep (1 male, 8 female) that were seronegative to BTV were obtained locally. The sheep were housed in insect-secure isolation facilities at the Biomedical Research Center, Faculty of Veterinary Science, University of Pretoria. Six of the sheep (Nos. 1665, 1672, 1695, 1696, 1700, and 1706) were intravenously inoculated with 2 ml of a 10−2 dilution of BTV-infected sheep blood that contained a strain of BTV serotype 4 (5/81). The virus was originally collected in 1981 in the blood of a sheep with BT and had been previously passaged twice in sheep, thus the blood inoculum was from the second sheep passage. Three sheep (Nos. 1675, 1677, and 1689) served as uninfected, in-contact controls. The sheep were closely evaluated throughout the study in accordance with the Institutional Animal Use and Care Committee protocol, and body temperatures were recorded at least daily. Severely affected animals were humanely euthanatized. Blood was collected from all sheep in 10-ml Vacutainer tubes (Becton Dickinson, Franklin Lakes, NJ) prior to inoculation and at 3, 6, 8, 10, 12, and 14 days after inoculation (DAI). Blood collected in EDTA was evaluated using a CELL-DYN 3700 System instrument (Abbott Laboratories, Abbott Park, IL) to quantitate hemoglobin, red cell count, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin concentration, total and differential white blood cell (WBC) count, and enumeration of platelets. Citrated blood was collected to determine prothrombin (PT) and partial thromboplastin (PTT) times and fibrinogen levels, using reagents provided by DiaMed (Cressier sur Morat, Switzerland) and an Electra 750 coagulation timer (Global Medical Instrumentation Inc., Minneapolis/St. Paul, MN). Muscle injury was evaluated by measuring serum concentrations of creatine kinase (CK), aspartate aminotransferase (AST), and lactate dehydrogenase (LDH) using a VETEX analyzer (Bayer, Leverkusen, Germany) with reagents supplied by Alfa Wassermann (West Caldwell, NJ). Serum albumin concentrations also were determined. BTV was isolated from heparinized blood collected at 7, 8, and 10 DAI by intravenous inoculation of embryonating chicken eggs. 14
The body temperatures, clotting times, blood cell counts and parameters, and serum chemistry values of the BTV-infected and BTV-uninfected sheep were compared using repeated measures analysis of variance. If significant differences were found, pair-wise comparisons were made using a Mann-Whitney test. Statistical calculations were done using SPSS statistical software version 15.0 (SPSS, Inc., Chicago, IL), and a P value ≤ .05 was considered to be significant.
The BTV-inoculated sheep were confirmed to be infected by isolation of virus from their blood; BTV was isolated from the blood of 4 of 6 of the inoculated sheep at 7 DAI, 6 of 6 sheep at 8 DAI, and 5 of 5 surviving sheep at 10 DAI. The infected sheep all became febrile at 4 DAI, and by 6 to 8 DAI exhibited varying degrees of serous nasal discharge, facial edema, congestion, hemorrhage and ulceration of the oral mucous membranes, and coronitis (Table 1). These signs were especially severe in 1 sheep (No. 1706), which developed extensive subcutaneous edema of its head and neck, necrosis of much of the dental pad mucosa, and diffuse congestion of its tongue with focally extensive areas of hemorrhage and ulceration. This sheep was euthanatized for humane reasons at 8 DAI, and postmortem evaluation showed hemorrhage at the site of venipuncture; extensive edema of the head, neck, lungs, and bowel mesentery; pleural and pericardial effusion; and focally extensive hemorrhage and necrosis of the mucosa of the oral cavity and forestomachs (Figs. 1–3). The animal's tongue was uniformly purple and the mucosa extensively ulcerated, consistent with so-called “blue tongue” (Fig. 4). The clinical signs in the 5 remaining sheep were similar but milder than those exhibited by sheep No. 1706, however at 8 to 10 DAI two sheep (Nos. 1672 and 1696) developed substantial hematomas and excessive bleeding at venipuncture sites. These 5 BTV-inoculated sheep all developed increasingly severe respiratory distress that began at 10 to 12 DAI, and all exhibited profuse serous or mucoid nasal discharge, marked coronitis, and precipitous loss of body condition. One sheep (No. 1695) also developed severe torticollis. In addition to showing increasing respiratory distress, these 5 sheep became progressively inappetant, lethargic, and reluctant to stand or move; one died at 13 DAI and the others were euthanatized for humane reasons at 13 or 14 DAI (Table 1). Necropsy evaluation showed consistent but variably severe pulmonary edema, pleural and pericardial effusion, edema of the head and neck that was especially prominent in the subcutis beneath the mandibles and in the fascia of the muscles of the cervical spine including the area around the nuchal ligament, focally extensive hemorrhage in the muscles of the cervical spine, resolving ulcers and hemorrhages within the mucosa of the oral cavity and upper gastrointestinal tract, coronitis, and subintimal and adventitial hemorrhages in the pulmonary artery (Figs. 5–7).
Clinical findings and outcomes in sheep after infection with virulent BTV.

All sheep developed fever, depression, coronitis, facial edema, and variably severe hemorrhage and ulceration of the oral mucosa.
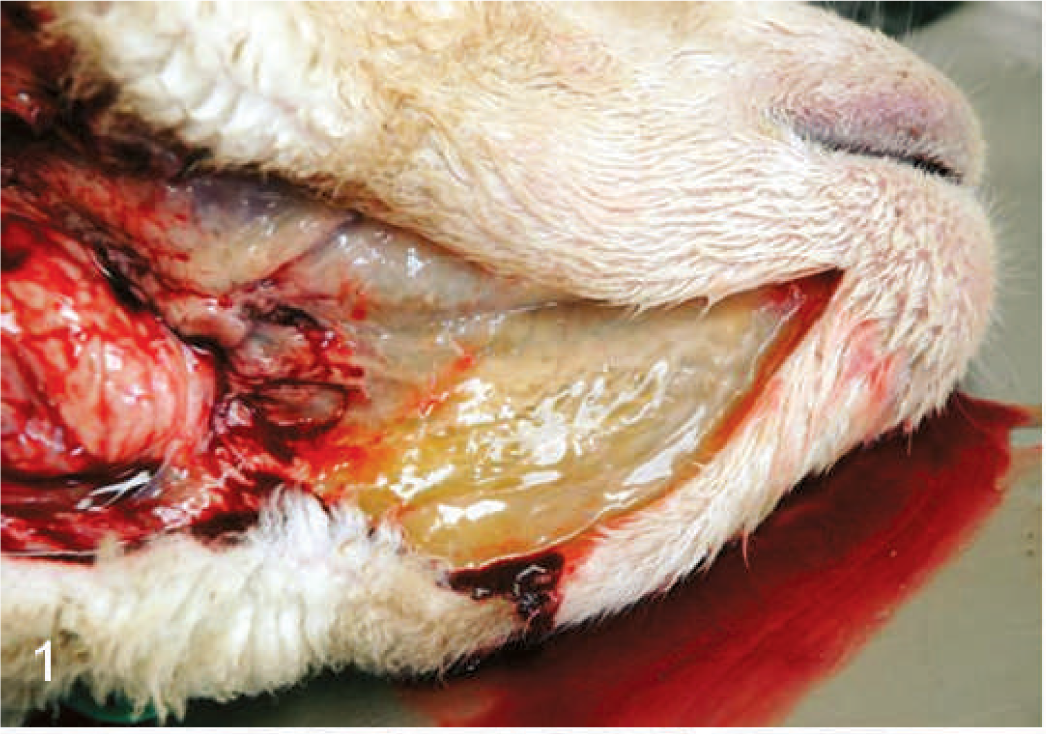
Head; sheep. Subcutaneous edema beneath the mandibles.
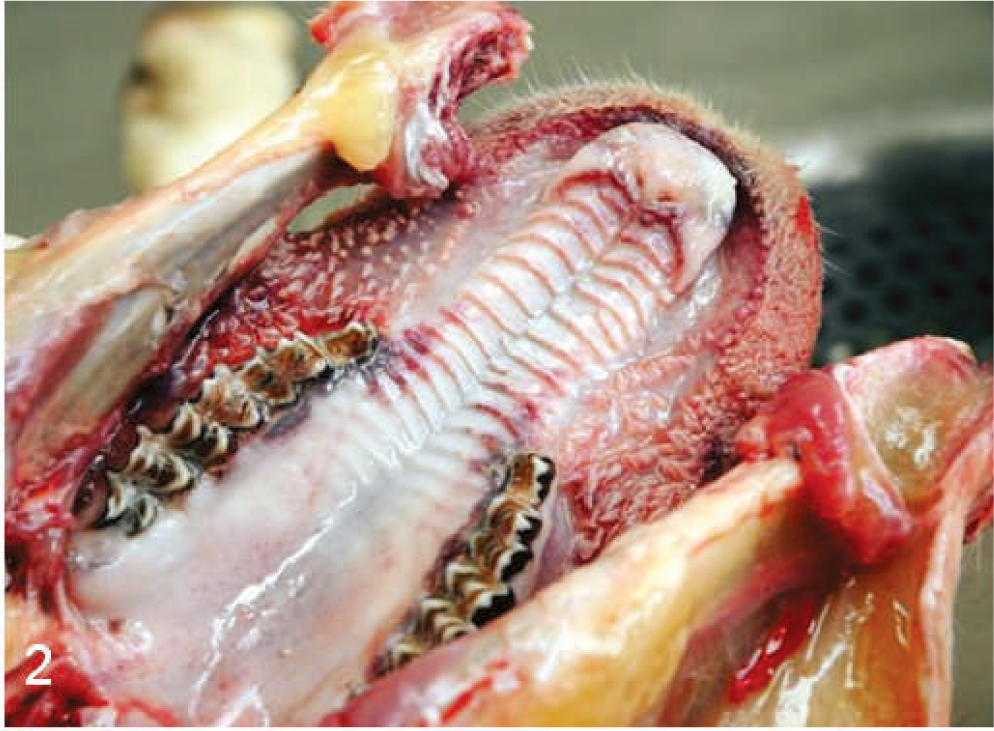
Oral cavity; sheep. Acute hemorrhage, necrosis, and ulceration.
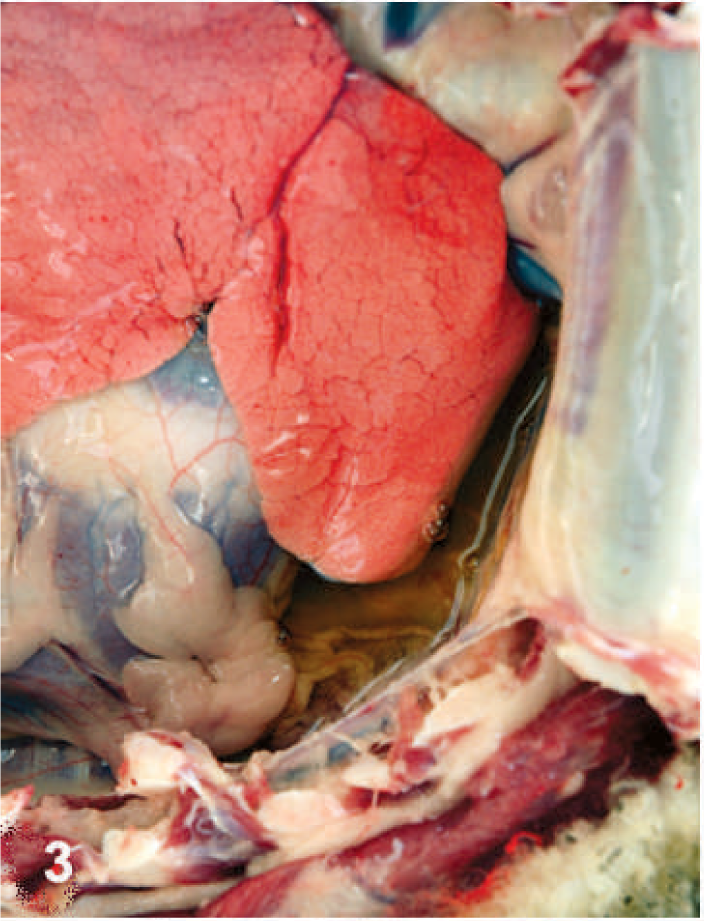
Thoracic cavity; sheep. Pulmonary edema and pleural effusion.

Tongue; sheep. Cyanosis and focally extensive mucosal necrosis and ulceration.
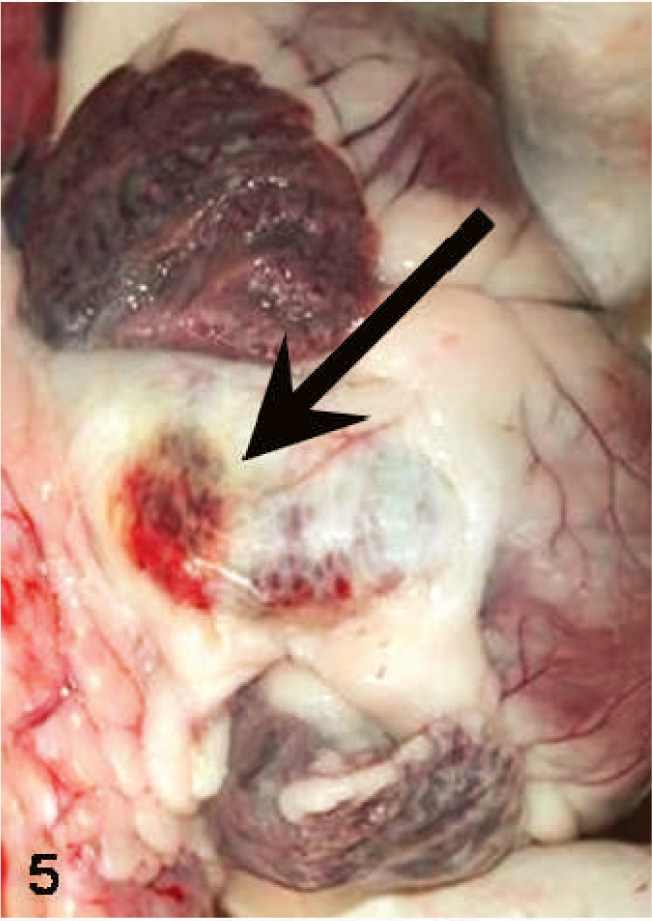
Heart; sheep. Hemorrhages on the adventitial surface of the pulmonary artery (arrow).
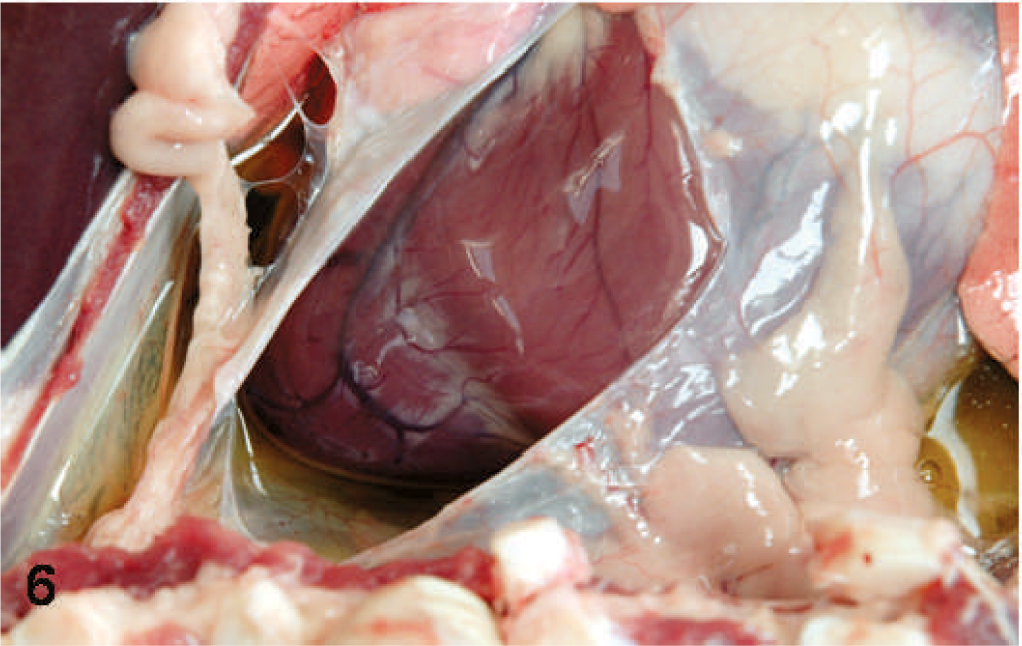
Thoracic cavity; sheep. Pericardial effusion.
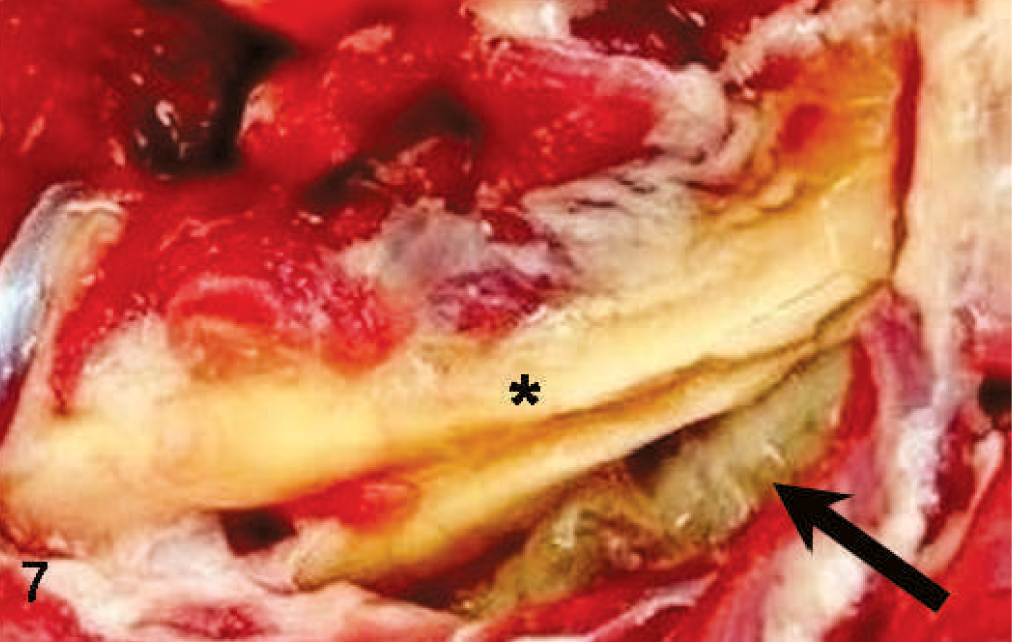
Neck; sheep. Edema (arrow) around the nuchal ligament (∗).
Histologic changes in the tissues of the infected sheep included multifocal necrosis and hemorrhage in skeletal muscles, especially those of the neck, marked pulmonary edema with distention of pleural and interstitial lymphatics, and ulceration and necrosis of the mucosal lining of the oral cavity and upper gastrointestinal tract, including the forestomachs. Other changes included acute hemorrhage in lymph nodes of the neck region, scattered hemorrhage and minimal acute myocardial necrosis in 1 sheep, and a small focus of acute suppurative bronchopneumonia in another animal. ECs lining small- to medium-caliber blood vessels adjacent to areas of muscle necrosis and mucosal ulceration were often hyperchromatic and swollen, changes that are suggestive of EC activation but without significant EC necrosis.
There was a statistically significant decrease in platelet count (Fig. 8) and increase in the PT (maximum mean of 25.4 seconds at 8 DAI; control 23.2 seconds) and PTT (maximum mean of 30.2 seconds at 8 DAI; control 23.4 seconds) of inoculated sheep at 6 to 10 DAI, although the increases were relatively modest even in the sheep (Nos. 1672 and 1696) that had pronounced coagulopathy at 8 to 10 DAI, as reflected by the excessive bleeding that occurred at venipuncture sites. The CK values in infected sheep increased significantly by 8 DAI (Fig. 8), as compared with the control sheep. The concentrations of both AST and LDH also increased at 8 to 10 DAI, although the difference in the values compared with control sheep was not statistically significant. The WBC counts of the infected sheep steadily increased after 6 DAI as the result of statistically significant mature neutrophilia, whereas both lymphocyte and monocyte counts initially decreased following infection, although the difference was not statistically significant. Serum albumin concentrations decreased in the infected sheep between samples collected before infection and prior to death (24 vs. 21 g/liter), although the difference was not statistically significant. There was no significant difference in concentration of fibrinogen or red cell parameters between the infected and control sheep.
Mean platelet numbers, body temperature, and concentration of CK in 6 sheep inoculated with virulent BT virus serotype 4. Arrow indicates onset of clinical signs in the inoculated sheep.
The inoculated sheep developed severe BT, with all but one animal having a relatively protracted course of disease that persisted up to 14 DAI. Endothelial injury is central to expression of viral hemorrhagic fevers such as BT, but the mechanisms responsible for EC dysfunction and vascular leakage are poorly defined and potentially could include direct virus-mediated damage, perturbations of anticoagulant/procoagulant activity, or the activities of cytokine and/or inflammatory mediators produced in the course of BTV infection. 16 Although direct virus-mediated EC injury is clearly critical to acute manifestations of BT, previous studies indicate that infection of ECs is both transient (up to 11 DAI) and restricted in distribution in the tissues of BTV-infected sheep. 15, 19 Similarly, viremia typically but not invariably persists less than 14 days in BTV-infected sheep. 2, 5, 14, 17, 21, 23 Thus, most sheep in this study developed fulminant disease in the late, putatively convalescent stages of BTV infection, which is consistent with the original description of BT and those in modern reviews. 21, 23
Consumptive coagulopathy and disseminated intravascular coagulation (DIC) subsequent to BTV-induced EC injury may contribute to the manifestations of severe BT. Howerth et al. described microvascular injury and DIC after experimental BTV infection of white-tailed deer (WTD), which are very prone to expression of BT. 11 However, BTV-infected WTD rapidly succumbed to BT and had considerably more pronounced perturbation of their clotting parameters than did the sheep in this study, and these WTD had severe and disseminated hemorrhages at postmortem. Furthermore, the platelet counts, fibrinogen concentrations, and PT and PTT times in the inoculated sheep were not consistent with the development of acute DIC, and platelet counts had returned to preinoculation levels by 14 DAI, when surviving BTV-infected sheep exhibited severe clinical signs (Fig. 8).
The BTV-infected sheep all developed severe pulmonary edema, pleural and pericardial effusion, and edema of the tissues of the head and neck. The extensive edema that occurred in these sheep was not associated with widespread histologic evidence of destruction of small blood vessels, suggesting that the edema resulted from increased vascular permeability without obvious EC necrosis. The absence of significant myocardial necrosis in most sheep further suggests that the extensive edema they exhibited was not simply a consequence of cardiac failure, as previously proposed. 15 The clinical signs and postmortem lesions in the inoculated sheep are strikingly similar to those of acute, fulminant African horse sickness (AHS), which like BT is an endotheliotropic viral disease characterized by fatal pulmonary edema without consistent, widespread histologic evidence of small blood vessel destruction. 3, 12 Together, these findings suggest that indirect effects on ECs mediated by soluble mediators produced in the course of BTV infection likely contribute to the increased vascular permeability and edema that characterizes severe BT caused by virulent virus strains. 4– 6 Mediators such as tissue necrosis factor (TNF-α), interleukin-1 (IL-1), interleukin-6 (IL-6), and interferon (IFN-β) can all potentially mediate the tissue injury, procoagulant activity, shock, increased vascular permeability, and death that characterizes viral hemorrhagic fevers, as can other mediators such as tissue factor, complement derivatives, and individual viral proteins. 16
In summary, the availability of an experimental infection model that reproduces severe BT will facilitate future studies to better characterize the pathogenesis of the disease and, in particular, the role of specific host and viral factors in mediating the increased vascular permeability that characterizes BT and related diseases such as AHS.
Footnotes
Acknowledgements
We gratefully acknowledge the indispensable assistance provided by Roelf Greyling, Elsbe Myburgh, Meagan Kay, Clifton Drew, and Patricia Wong. These studies were supported by funds provided by the Center for Food Animal Health, University of California, and the United States Department of Agriculture under the Animal Health Act, 1977, Public Law 95-113.
