Abstract
The variation in histologic pattern of meningiomas can make their diagnosis challenging. The immunohistochemical profile of 28 canine and 8 feline meningiomas was examined. Tumor types included anaplastic (6 dogs), angiomatoid (1 cat), fibroblastic (3 dogs, 1 cat), meningothelial (1 dog), microcystic (2 dogs), myxoid (3 dogs), psammomatous (4 cats), and transitional (13 dogs, 2 cats). The authors compared the expression of novel markers (CD34, E-cadherin, claudin-1, glucose transporter 1 [GLUT-1], laminin, and protein gene product [PGP] 9.5) with published markers (cytokeratins, glial fibrillary acidic protein [GFAP], progesterone receptor, S100, and vimentin). Neoplastic cells were immunohistochemically positive for vimentin in 100% of the meningiomas; CD34, 94%; GLUT-1, 86%; E-cadherin, 81%; S100, 75%; laminin, 72%; claudin-1, 60%; PGP 9.5, 55%; progesterone receptor, 44%; pancytokeratins, 39%; cytokeratins 8/18, 17%, and GFAP in 9%. Ki67 index did not correlate well with mitotic index. Based on these results and those in the human literature, immunohistochemistry for vimentin, CD34, and E-cadherin is proposed to support a diagnosis of meningioma. Immunohistochemistry for claudin-1, albeit of only moderate to low sensitivity in canine and feline meningiomas, may help to distinguish meningioma from some mesenchymal neoplasms involving the brain and associated structures, such as schwannomas, which in humans express claudin-1 poorly or not at all. Further studies with CD34, E-cadherin, and claudin-1 in canine and feline tumors that may mimic meningiomas are needed to determine the adequacy of this approach.
Keywords
Meningiomas are tumors arising from cells of the arachnoid membrane and pia mater of the nervous system. 22 They are one of the most common primary neoplasms of the central nervous system in dogs and cats but are rare in other species. 30,40,41,54,57,61 Meningiomas in domestic species are mostly intracranial and less commonly spinal, paranasal, or cutaneous. 22,40 In dogs, meningiomas are typically solitary, whereas in cats, meningiomas can be single or multiple masses. 22,25
The histologic diversity of meningiomas, with mesenchymal and epithelial patterns, reflects mesodermal and neural crest contributions to their formation. 37 The current World Health Organization (WHO) classification of nervous system tumors of domestic animals includes 9 types of meningioma: meningotheliomatous, fibrous (fibroblastic), transitional (mixed), psammomatous, angiomatous (angioblastic), papillary, granular cell, myxoid, and anaplastic (malignant). 21 An additional type, microcystic, used in the WHO classification of human meningiomas, has been reported in the dog. 22,33,57 Because of this diversity in pattern, meningiomas may resemble other tumors, such as astrocytomas, oligondendrogliomas, metastatic carcinomas, germ cell tumors, and peripheral nerve sheath tumors. 11,22,31
The number of immunohistochemical markers used in human meningiomas is large, ranging from generic markers (such as vimentin and cytokeratins) to more specialized markers (such as CD34, epithelial membrane antigen [EMA], E-cadherin, and claudin-1). 6,15,31,43,52,53 To our knowledge, antibodies to EMA that cross-react with formalin-fixed, paraffin-embedded (FFPE) canine or feline tissues are not available. An antibody to CD34 that reacts with canine endothelial cells on FFPE tissues has been recently reported. 55 The main focus of immunohistochemistry in human meningioma is to distinguish this tumor from its mimics (eg, hemangiopericytoma, solitary fibrous tumor, schwannoma, metastatic carcinoma) with a secondary interest in prognostic markers, such as proliferation markers. 11 Markers used in meningiomas of domestic species include cytokeratins, estrogen receptor, glial fibrillary acidic protein (GFAP), Ki67, neuron-specific enolase, progesterone receptor, proliferating cell nuclear antigen, S100, synaptophysin, h-telomerase, and vimentin. 1,3,27,28,42,59 Combining the results of 2 large series of canine meningiomas, vimentin was expressed in all (37 of 37), followed by neuron-specific enolase (29 of 37), S100 (28 of 37), GFAP (6 of 37), and cytokeratins (11 of 37). Synaptophysin was not reactive in any case (0 of 15). 3,33 Although the number of cases and markers studied in cats is fewer than that in dogs, results are similar (eg, consistent labeling for vimentin and S100 and no labeling for cytokeratins). 25
There are few reported immunohistochemical studies of large series of canine or feline meningiomas. The purpose of this study was to evaluate the immunohistochemical reactivity of canine and feline meningiomas with novel markers, such as CD34, E-cadherins, claudin-1, glucose transporter 1 (GLUT-1), laminin, and protein gene product (PGP) 9.5, in addition to classic markers (cytokeratins 8 and 18, GFAP, Ki67, pancytokeratins, progesterone receptor, S100, and vimentin) on FFPE tissues.
Methods
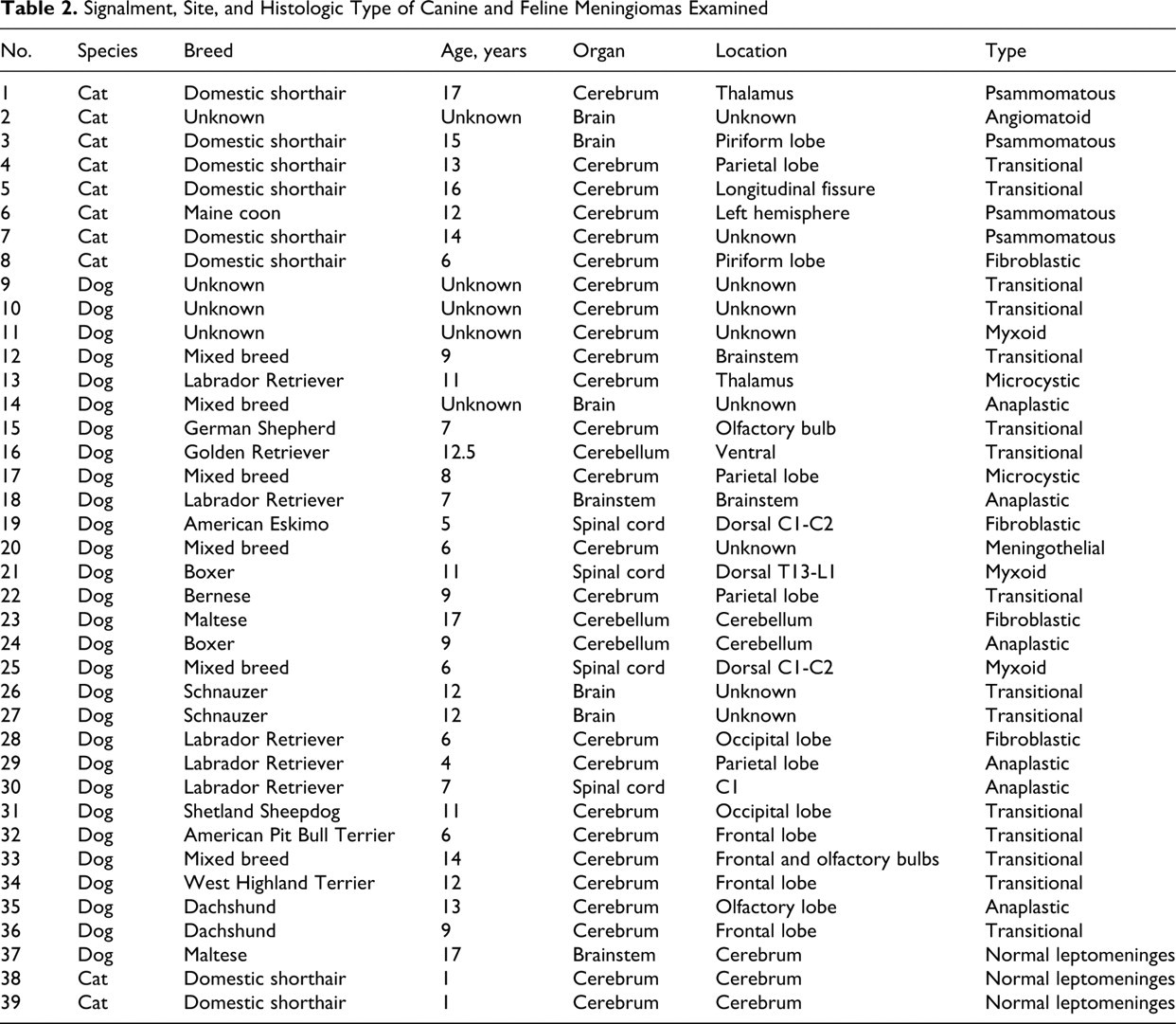
The databases of the Animal Disease Diagnostic Laboratory at Purdue University and the Diagnostic Center for Population and Animal Health at Michigan State University were reviewed for canine and feline meningioma cases from 1998 to May 2009. Of a total of 54 cases with a diagnosis of meningioma, 36 were selected for this study on the basis of the following case definition: A superficial mass in the central nervous system adhered to the leptomeninges or dura mater; it did not involve a nerve; and it comprised histologic features of one of the described subtypes of meningioma. 21 In addition, selected cases needed to have enough relevant tissue for immunohistochemistry. In some cases, the exact location of the tumor was not included in the pathology report or the clinical history; however, histologic evaluation determined that the mass was superficial and adhered to meninges. Leptomeninges from the cerebrum of 2 cats and 1 dog with no neurologic disease were included in this study. All tissues were fixed in 10% neutral buffered formalin for an undetermined period, estimated at fewer than 2 weeks for all the samples, and stored in paraffin blocks at room temperature for 1 week to 11 years. In our experience, immunoreactivity for all the markers examined is not significantly affected by formalin fixation up to 1 month. 62 For CD34, immunoreactivity is not reduced in tissues fixed for at least 2 weeks (personal observations). When multiple slides of the same tumor were available, the most representative sample was selected by 1 author (J.A.R.). Cases were reviewed and classified by 2 pathologists in each laboratory without knowledge of the classification in the pathology report. Any discrepancies between the authors in the same laboratory were resolved by consensus among the pathologists reviewing the cases. Classification in some cases was challenging because of the rather common presence of several patterns within the same tumor. 3 A final classification was produced by consensus between 2 pathologists (J.A.R, M.A.M) following the current WHO classification for domestic animal meningiomas. 21 Key descriptions for each tumor type follow: anaplastic (lack of distinct growth pattern, increased mitotic index, and/or extensive necrosis and parenchymal invasion), angiomatous (numerous and variably sized vessels among aggregates of meningeal cells), fibroblastic (spindle cells forming long interlacing fascicles), meningothelial (solid whorls of polygonal cells), microcystic (intercellular pale vacuoles progressing to cysts, admixed with spindloid cells), myxoid (neoplastic cells separated by myxomatous matrix), psammomatous (whorls of meningothelial cells around hyaline or mineralized concretions), transitional (including features of meningothelial and fibroblastic types).
Table 1 summarizes the 13 primary antibodies used in this study and the immunohistochemical protocol. Immunoreactivity for CD34, claudin-1, E-cadherin, GLUT-1, laminin, S100, and vimentin was examined in most tumors. In addition, antibodies to pancytokeratins, cytokeratins 8 and 18, GFAP, Ki67, PGP 9.5, and progesterone receptor were evaluated in a subset of cases. Immunohistochemistry was performed with a Dako autostainer; all the incubations were at room temperature. Immunoperoxidase procedures with diaminobenzidine as chromogen have been published. 38,45,46 Positive tissue controls from each species were used simultaneously. The negative reagent control was rabbit immunoglobulins (for polyclonal antibodies) or mouse immunoglobulins (for monoclonal antibodies). The expected staining pattern for each antibody was as follows: CD34 (cytoplasmic), claudin-1 (cytoplasmic and/or membrane), cytokeratins 8 and 18 and pancytokeratins (cytoplasmic), E-cadherins (cytoplasmic and/or membrane), GFAP (cytoplasmic), GLUT-1 (cytoplasmic), Ki67 (nuclear), laminin (cytoplasmic, interstitial), PGP 9.5 (nuclear and/or cytoplasmic), progesterone receptor (nuclear), S100 (nuclear and/or cytoplasmic), vimentin (cytoplasmic). After the immunohistochemical procedure, slides were counterstained with Mayer’s hematoxylin. Immunoreactivity was scored as follows: 3 (high), > 60% positive cells; 2 (moderate), 30 to 59% positive cells; 1 (low), 10 to 29% positive cells; or 0 (negative), < 10% positive cells. 46 Immunoreactivity in normal leptomeninges was graded as positive or negative. The sensitivity of a given marker was the percentage of positive cases relative to all meningiomas examined for the given marker. We did not attempt to determine the specificity of the examined markers (ie, the reactivity of meningioma-like tumors or meningioma mimics). Mitotic index (average number of mitotic figures in 10 fields [400×]) and Ki67 index (average number of positive nuclei in 10 fields [400×]) were also recorded.
Antibody Reagents, Antigen Retrieval, and Detection Systems Used in Immunohistochemistry
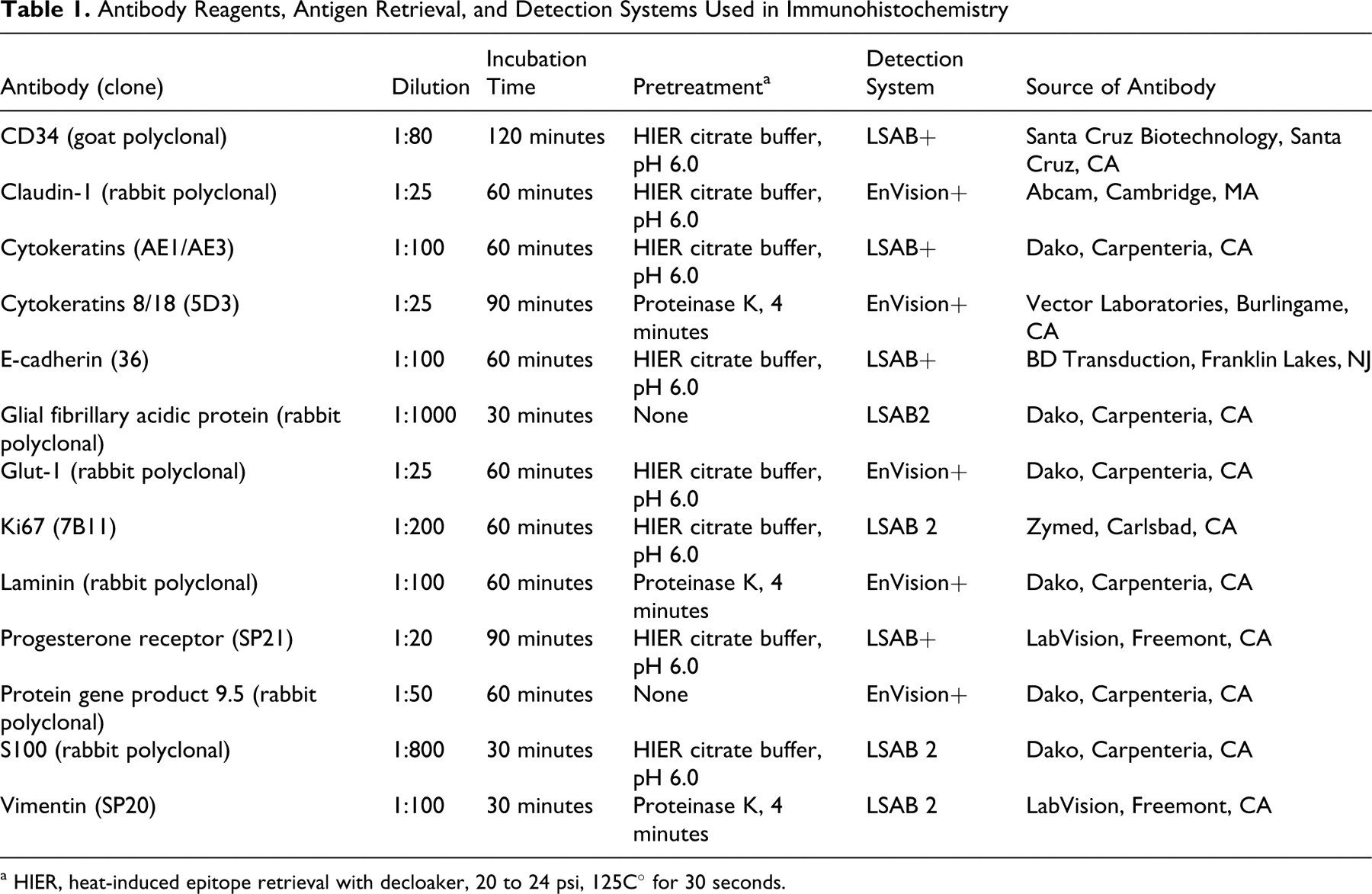
a HIER, heat-induced epitope retrieval with decloaker, 20 to 24 psi, 125C° for 30 seconds.
Results
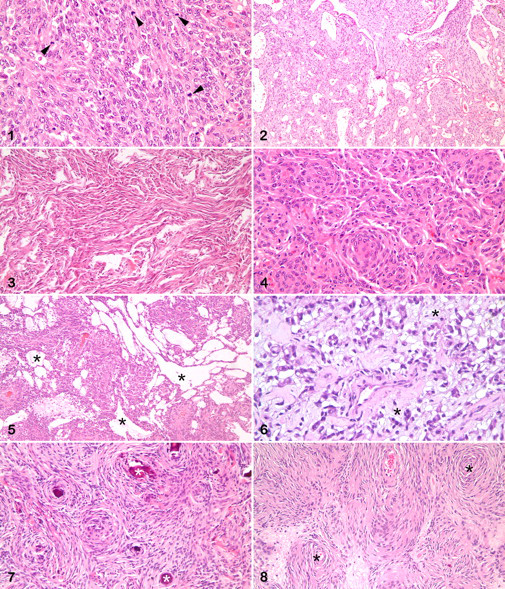
In sum, 8 feline and 28 canine meningiomas were included in this study (Table 2 ). There were 12 male dogs, 13 female dogs, and 3 of unknown sex. Cats were equally distributed in terms of sex, with 2 unknown. The average age at diagnosis for meningiomas was 8.1 years for dogs (range, 4 to 14 years) and 13.3 years for cats (range, 6 to 17 years). A variety of canine breeds was represented, mostly medium size to large. The breeds of 3 dogs were unknown. All cats with recorded breed were domestic shorthair, except for 1 Maine coon cat. Thirty-two meningiomas were intracranial (24 dogs, 8 cats); 4 were spinal (all dogs). The meningiomas were classified as follows: anaplastic (6 dogs), angiomatoid (1 cat), fibroblastic (3 dogs, 1 cat), meningothelial (1 dog), microcystic (2 dogs), myxoid (3 dogs), psammomatous (4 cats), transitional (13 dogs, 2 cats) (Figs. 1–8). Except for 3 anaplastic meningiomas with 32, 19, and 9 mitotic figures per field (400×), mitotic index was 3 per field or fewer, with 15 tumors having no identified mitotic figures. Table 3 summarizes the immunohistochemical results.
Signalment, Site, and Histologic Type of Canine and Feline Meningiomas Examined
Canine and Feline Meningiomas Examined: Immunohistochemical Results a
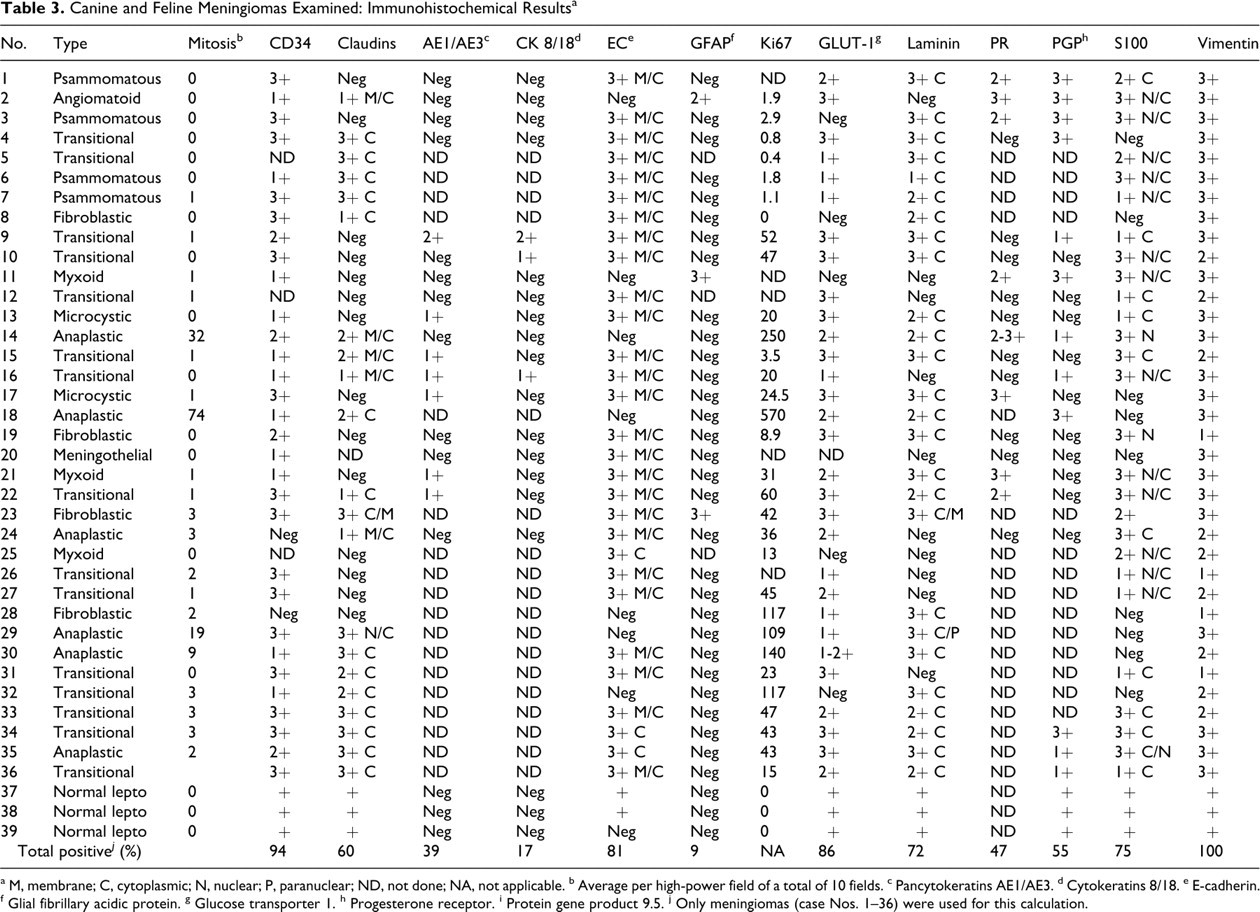
a M, membrane; C, cytoplasmic; N, nuclear; P, paranuclear; ND, not done; NA, not applicable.
b Average per high-power field of a total of 10 fields.
c Pancytokeratins AE1/AE3.
d Cytokeratins 8/18.
e E-cadherin.
f Glial fibrillary acidic protein.
g Glucose transporter 1.
h Progesterone receptor.
i Protein gene product 9.5.
j Only meningiomas (case Nos. 1–36) were used for this calculation.
Normal Meninges
Immunoreactivity of normal canine meninges resembled that of normal feline meninges. CD34 antibody strongly labeled the external limiting layer of the leptomeninges and scattered cells in the deeper layers. Endothelial cells were also labeled. For E-cadherin, the reactivity was intense in the outer layer and patchy and less intense in cells in the inner aspect of the leptomeninges. Claudin-1 was not detected in the outer leptomeninges and only in some cells in the inner aspect. There was mild GLUT-1 granular cytoplasmic labeling of some cells in the external limiting layer and fewer labeled cells in the deeper aspect of the leptomeninges. Expression of laminin in normal leptomeninges was mostly located in cells of the outer layer with less reactivity in cells in deeper aspect. The leptomeninges were highly and diffusely labeled for PGP 9.5 and S100. Vimentin expression was moderate throughout. There was no labeling of normal leptomeninges with antibodies to pancytokeratins, cytokeratins 8 and 18, and GFAP.
Meningiomas
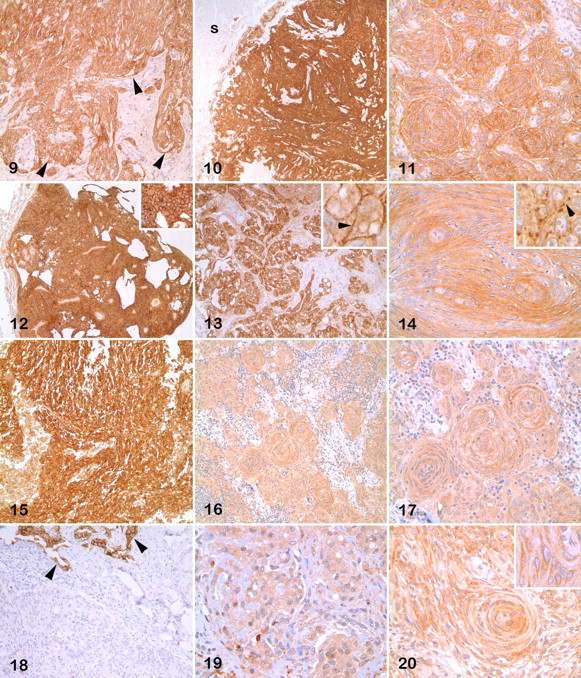
Antibody to E-cadherin decorated > 60% of neoplastic cells in 29 of 36 (81%) meningiomas (Figs. 9–14). The labeling was strong and present in the cytoplasmic membrane and the cytoplasm, with the cytoplasmic membrane more intensely labeled and with a granular pattern (see insets of Figs. 13, 14). Seven meningiomas did not react with the antibody to E-cadherin, including 3 anaplastic, 1 myxoid, 1 angiomatoid, 1 transitional, and 1 fibroblastic. Of 35 meningiomas, 21 (60%) expressed claudin-1. The reactivity for claudin-1 was cytoplasmic in all cases, with cytoplasmic membrane reactivity in 5 cases (Figs. 15–20). Membrane staining was diffuse or granular and more intense than cytoplasmic labeling. For claudin-1, 4 anaplastic meningiomas had a moderate or high reactivity, and 1 had low reactivity. The staining was high in 11 cases and moderate or low and patchy in the rest.
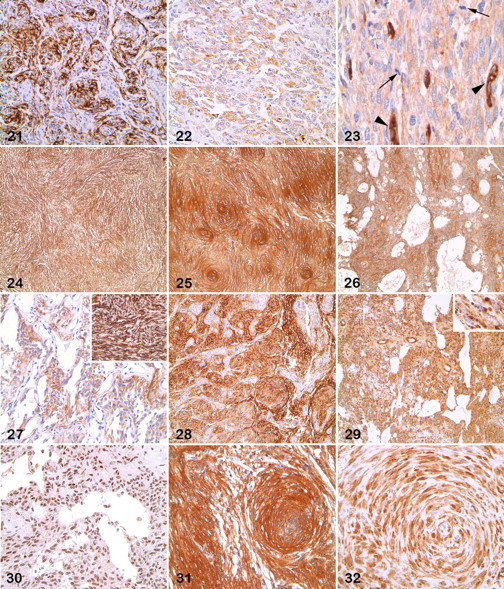
Pancytokeratins and cytokeratins 8 and 18 were detected in 7 of 18 meningiomas (39%; 4 transitional, 2 microcystic, 1 myxoid) and 3 of 18 meningiomas (17%; 3 transitional), respectively (Figs. 21, 22). Neither of the 2 anaplastic meningiomas examined for cytokeratins was positive. None of the tumors that expressed cytokeratins had strong immunoreactivity.
Of 33 meningiomas, 31 (94%) expressed CD34 with a diffuse cytoplasmic or granular pattern in most neoplastic cells or multifocally (Figs. 23–26). All feline meningiomas and all but 2 canine meningiomas (1 fibroblastic, 1 anaplastic) were positive. One anaplastic meningioma had a punctate pattern. Twenty meningiomas had moderate to high reactivity. Transitional meningiomas usually had a strong and diffuse reactivity, although in some instances the meningothelial cells were more strongly labeled than spindloid cells.
Of 33 meningiomas, 3 (9%) were reactive to antibody to GFAP: 1 feline angiomatoid, 1 canine myxoid, and 1 canine fibroblastic meningioma had moderate to strong cytoplasmic labeling (Fig. 27). Two GFAP-positive meningiomas were negative for E-cadherin and weakly positive for CD34, whereas the third tumor was strongly positive for these 2 markers.
Of 35 meningiomas, 22 (63%) had moderate to high reactivity to GLUT-1; 8 (23%) had weak reactivity; and 5 had no detectable expression. GLUT-1 expression did not correlate with type of meningioma (Fig. 28 ). The cytoplasm of GLUT-1 positive cells was diffusely labeled. GLUT-1 internal positive control was perivascular mesenchymal structures.
Although tumors with the highest mitotic index had high Ki67 index, in most cases there was little correlation between these 2 indices. Of 5 anaplastic meningiomas, 4 had a Ki67 index > 100. Other types with a Ki67 index > 100 were transitional (1 case) and fibroblastic (1 case). Tissue from 5 meningiomas was not available for Ki67 evaluation. One fibroblastic meningioma had a Ki67 index of 0. Interestingly, of 5 meningiomas with a Ki67 index > 100, 4 did not express E-cadherin, whereas 3 of 4 meningiomas with a Ki67 index > 100 expressed claudin-1.
Laminin labeled the cytoplasm and, much less commonly, the cytoplasmic membrane of neoplastic cells with moderate to high scores (25 cases, 69%) or a low score (1 case, 3%) (Fig. 29 ). Ten meningiomas were negative for laminin, including 5 of 13 canine transitional types. The laminin internal control was vascular basement membrane.
Progesterone receptor antibody decorated the nucleus of neoplastic cells in some tumors. Of 18 cases, 8 (44%) had moderate to high labeling; 10 cases (56%) were negative (Fig. 30 ).
PGP 9.5 usually labeled both the nucleus and the cytoplasm of neoplastic cells: Of 22 tumors, 7 (32%) had moderate to high labeling and 5 (23%) had low labeling (Fig. 31 ).
Of 36 meningiomas, 19 (53%) had moderate to high labeling with antibody to S100 protein; 8 (22%) had low scores; and 9 (25%) were negative. Labeling was nuclear and cytoplasmic in 13 cases, cytoplasmic in 9, and nuclear in 2 (Fig. 32). Two fibroblastic meningiomas were positive and 2 were negative. Other S-100-negative cases were evenly distributed among other meningioma types.
There was diffuse cytoplasmic vimentin labeling of all meningiomas in this study. The immunoreactivity was moderate to high in 29 cases and low in 4.
Discussion
In this study, we examined the immunoreactivity of 28 canine and 8 feline meningiomas using novel markers (CD34, E-cadherin, claudin-1, GLUT-1, laminin, and PGP 9.5) and previously reported markers (cytokeratins, GFAP, progesterone receptor, S100, and vimentin). Vimentin, CD34, GLUT-1, E-cadherin, and S100 were expressed in at least 75% of meningiomas examined.
With some exceptions, transitional meningioma is the most commonly reported meningioma type in dogs and cats. 1,3,22,27,30,33,41 In our series, transitional meningioma was the most common type in dogs, but psammomatous meningioma was the most common type in cats. Meningiomas in cats tend to occur at a later age than do those in dogs. 22 Similar results were observed in our series.
Two epithelial markers, EMA and cytokeratins, have been used in the immunohistochemical characterization of human meningiomas. EMA is expressed in 75% to 100% of meningiomas. 24,31,58 Although EMA is more consistently expressed in carcinomas than meningiomas, it is used to rule out some meningioma mimics, such as schwannomas, hemangiopericytomas, and solitary fibrous tumors in which this marker is not expressed. 11,24,31 Unfortunately, to our knowledge, no antibodies to EMA cross-react with canine and feline tissues. 22 However, we examined the cytokeratin immunoreactivity in this group of meningiomas using pancytokeratins (clones AE1 and AE3) and a monoclonal antibody to cytokeratins 8 and 18. In human pathology, cytokeratins are detected in 6% to 80% of meningiomas. 2,24,31,32,58 Interestingly, several studies comparing benign and malignant human meningiomas demonstrated high cytokeratin expression in malignant tumors, whereas cytokeratin expression in benign meningiomas was low or undetectable. 20,24 The expression of cytokeratins seemed to be inversely correlated with that of EMA in meningiomas, with increased cytokeratin expression and decreased EMA expression as tumor grade increased. 20,31 In a comprehensive study of cytokeratin expression in human meningiomas, cytokeratin 18 was the most commonly expressed cytokeratin in most meningioma types, followed by clone AE1. 32 The 2 cytokeratin antibodies used in our series labeled few meningiomas and, in positive cases, usually a low or (rarely) moderate number of cells. One explanation could be that the use of antibodies that recognize multiple cytokeratins (eg, AE1 and AE3, cytokeratins 8 and 18) reduced the chances of detecting the cytokeratin more commonly expressed in meningiomas (eg, clone AE1, cytokeratin 18). Our results with cytokeratin immunohistochemistry of meningiomas are somewhat contradictory to those in human studies; none of the anaplastic meningiomas in our series were positive for cytokeratins, whereas human malignant or anaplastic meningiomas tend to have strong cytokeratin expression. 20,24 Studies in canine meningiomas have reported higher immunoreactivity for cytokeratins than that in this series, ranging from 18% to 73%, although the number of positive cells is usually low. 3,33 Based on our and others' results, cytokeratins are not a reliable marker for meningiomas, although the patchy reactivity typically observed in meningiomas can be used to rule out carcinoma, which usually expresses these proteins strongly and in most neoplastic cells.
To our knowledge, CD34 expression in meningiomas of domestic species has not been reported. CD34 was the second most common marker expressed in our series, after vimentin. The expression of CD34 in human meningiomas varies from 0% to 40%. 11,15,53 Contrary to CD34 expression in human meningiomas, which is typically observed in the fibroblastic type only, all types of canine and feline meningiomas strongly expressed CD34. This result is not surprising considering that, in our study, normal canine and feline leptomeninges strongly expressed CD34. CD34 is a protein in hematopoietic cells of the bone marrow and in endothelial cells. 13,23,44 This protein was recently reported in vascular endothelial cells of canine FFPE. 55 Using the same antibody, we detected CD34 expression in endothelial cells in a various canine and feline tissues. Interestingly, this marker is not expressed in human peripheral nerve sheath tumors (eg, neurofibromas, schwannomas). 11,15 Additional studies are needed to characterize CD34 reactivity in canine and feline peripheral nerve sheath tumors and other meningioma mimics to determine its specificity for meningiomas.
E-cadherin, a glycoprotein of the adherens junction, is expressed in most epithelial cells. 14,51 E-cadherin is consistently expressed in most human meningiomas 12,18,51,52 and has been used to distinguish human meningiomas from their mimics. E-cadherin is not expressed in human hemangiopericytoma, although it is consistently detected in schwannomas. 17,52 In addition, E-cadherin has not been detected in other human brain tumors except choroid plexus papilloma. 51 Though not reported in meningiomas of domestic species, E-cadherin was detected in most meningomas in our series. Some studies of human meningiomas indicate a tendency to stronger expression of this protein in meningotheliomatous versus fibroblastic areas 12,51 or even lack of expression in the fibroblastic meningioma. 60 E-cadherin expression did not correlate with meningioma types in our series, although 2 of 4 anaplastic meningiomas were negative. The significance of expression (or lack thereof) of E-cadherin in human malignant meningiomas is controversial. Some reports indicated that many malignant meningiomas do not express this protein whereas all benign types do; tumors undergoing malignant transformation lack E-cadherin expression. 51 However, differences in E-cadherin expression among different meningioma tumor grades have not been found in other studies. 6,12,52 In our series, all but 1 E-cadherin-positive tumor had both cytoplasmic membrane and cytoplasmic expression. The cytoplasmic expression was distinguished from the cytoplasmic membrane expression by its typical granular pattern. In a recent study, 34% of human meningiomas had cytoplasmic expression of E-cadherin without cytoplasmic membrane expression. The lack of cytoplasmic membrane expression did not correlate with tumor grade. 6 Based on the consistent expression of E-cadherin in epithelial cells and their tumors, this marker is not useful as a single marker to distinguish meningiomas from metastatic carcinomas.
To our knowledge, the expression of claudin-1 in meningiomas of domestic species has not been reported. Claudins are tight junction–associated proteins. There are more than 15 types of claudins, and each is associated with certain cells and tissues. 14 Claudin-1 is variably expressed in 50% to 100% of human meningiomas. 4,15,43,48 Of 33 meningiomas, 19 (58%) in our series reacted with antibody to claudin-1. Claudin-1 seems to be an excellent marker to distinguish human meningiomas from schwannomas, given that the latter do not express this protein. 4,15 Zero to 13% of human hemangiopericytomas, another possible mimic of meningiomas, have low expression of claudin-1 15,43 ; however, it is expressed in most perineuriomas. 26 Based on our results with meningiomas and on results in the human literature with meningiomas and their mimics, claudin-1 immunohistochemistry has high specificity (if epithelial neoplasms are excluded) and low to moderate sensitivity for meningiomas. Additional studies with canine and feline mesenchymal neoplasms are necessary to evaluate the specificity of claudin-1 for meningioma.
GFAP expression was moderate to high in 3 meningiomas: 1 feline angiomatoid, 1 canine myxoid, and 1 canine fibroblastic. GFAP immunoreactivity in meningiomas has been examined in 3 studies totaling 56 canine meningiomas, with only 3 cases being reactive: 2 fibroblastic and 1 anaplastic. 3,33,42 GFAP immunoreactivity in human meningiomas, albeit rare, has been reported. 7,56 We used the same antibody and a similar immunohistochemical protocol as in previous studies. 3,7,33,56 GFAP is considered to be a specific marker for glial cells and their tumors; however, based on our results and published studies, some meningiomas can be strongly immunoreactive to GFAP, when a differential diagnosis includes glioma or peripheral nerve sheath tumor.
GLUT-1 is expressed in erythrocytes, blood–brain barrier, liver, placenta, perineurial cells, germinal centers of lymphoid follicles, and renal tubules. 50,53 Human perineurial tumors commonly express GLUT-1 and may mimic meningiomas, particularly, meningothelial type. 16,50,53 This marker is expressed in 100% of human meningiomas, albeit with a wide range in the number of positive cells in a given tumor. 53 In our series, of 32 meningiomas, 27 (85%) were positive for GLUT-1. All anaplastic meningiomas expressed this protein to various degrees. Although GLUT-1 is a moderately to highly sensitive marker for meningiomas, it is expressed in some human and bovine meningioma mimics and should therefore be used in conjunction with other markers. 34
Ki67 immunohistochemistry is used to determine the growth fraction of a tumor. 5 It has been used in human meningiomas as a prognostic marker, with conflicting results. 11 However, it appears that Ki67 index is associated with tumor behavior and recurrences. 11,20 Ki67 and proliferating cell nuclear antigen, a cell cycle–specific marker, were inversely correlated with progesterone receptor expression in canine meningiomas. 27,59 The apparent lack of correlation between the mitotic and Ki67 indices in this series of meningiomas might be explained by differences in the duration of the cycling phases of tumor cells or by difficulties in distinguishing mitotic cells from pyknotic nuclei. 5
Laminin is a protein that is commonly present in basement membranes and the extracellular matrix. It is produced by a variety of mesenchymal and epithelial cell types and their tumors. The expression of laminin and other extracellular matrix proteins has been studied in human meningiomas 8,35,36,49 but, to our knowledge, not in meningiomas of domestic animals. This marker was included in our series to determine whether it could be used to distinguish meningiomas from some mimics, such as schwannomas, which typically express this marker. 29,39 In our series, 67% of meningiomas expressed laminin with no significant differences among histologic subtypes, although 50% of the transitional meningiomas did not express this marker. In an ultrastructural study of human meningiomas, the meningothelial type did not express laminin except in vascular structures, whereas in the fibroblastic type, laminin was present among the tumor cells but not within their cytoplasm. 36 In our series, all fibroblastic meningiomas had strong or moderate cytoplasmic expression and, less common, cytoplasmic membrane expression. Based on the presence of laminin in human schwannomas (and its expected expression in animal schwannomas), the expression of laminin in many canine and feline meningiomas makes it unsuitable to distinguish between meningioma and schwannoma.
Progesterone receptor is expressed in 70% to 100% of canine and feline meningiomas. 27,59 We examined progesterone receptor expression in only 19 meningiomas, and less than 50% were positive (3 of 4 feline and 5 of 19 canine meningiomas). All meningioma types in our series had at least one positive case except for the sole meningothelial type. In human meningiomas, lack of progesterone receptor expression is linked to an aggressive behavior. 19 The significance regarding the loss of progesterone receptor expression in domestic animals is unknown.
PGP 9.5 is a ubiquitin carboxyl-terminal hydrolase-1 originally reported to be expressed only in neurons and neuroendocrine cells. 9 It is currently accepted that PGP 9.5 is expressed in a variety of tissue and cell types, including nerve sheath cells and their tumors. 9,46 We have observed PGP 9.5 immunoreactivity in canine peripheral nerve sheath tumors (unpublished observations). PGP 9.5 has not been used in the characterization of meningiomas. Our interest in using PGP 9.5 was to determine if it could be used to distinguish meningiomas from nerve sheath tumors. Of 22 meningiomas, 12 (55%) were positive for PGP 9.5 in our series, including most histologic types. Based on these results, PGP 9.5 is not a good marker to distinguish between nerve sheath tumors and meningiomas.
S100 protein is one of the most commonly used markers in human and animal meningiomas. It is expressed in human meningiomas but less commonly than other markers, such as vimentin, EMA, E-cadherin, and claudin-1; however, up to 80% of fibrous meningiomas express S100 protein. 2,11,31,47 In our series, S100 protein was detected in 24 of 32 meningiomas (75%), with more frequent nuclear and cytoplasmic labeling, rather than just cytoplasmic. In 2 series of canine meningiomas, most tumors expressed S100 protein 3,33 ; however, the number of positive cells varied, and the pattern was typically multifocal with labeling being nuclear, cytoplasmic, or both. 3 Montoliu et al 33 reported a stronger labeling of fibroblastic than meningothelial cells. In many meningiomas in our series, there were no differences among cellular phenotypes or patterns, although in a limited number of cases, bundles of fibroblastic cells had stronger reactivity than that of adjacent whorls of meningothelial cells. S100 was a moderately sensitive marker in our series. However, this marker is expressed in multiple mesenchymal tumors of domestic animals, including some meningioma mimics (eg, schwannoma). 10
Vimentin, a generic marker of mesenchymal neoplasms, is consistently expressed in human and animal meningiomas. In dogs and cats, vimentin is expressed in all histologic subtypes of meningiomas, and its reactivity is usually higher than with S100. 3,25,30,33 Our results are consistent with those in the literature. All meningiomas in this series were positive for vimentin, and most had a strong and diffuse reactivity. The use of vimentin in the characterization of meningiomas is still valuable when a differential diagnosis includes metastatic carcinoma, but it should be used in conjunction with other markers.
In summary, the markers used in this study labeled meningiomas in the following descending order based on the percentage of tumors that were positive: vimentin (100%), CD34 (94%), GLUT-1 (86%), E-cadherin (81%), S100 (75%), laminin (72%), claudin-1 (60%), PGP 9.5 (55%), progesterone receptor (44%), pancytokeratins (39%), cytokeratins 8 and 18 (17%), and GFAP (9%). Given these results and observations on human meningiomas and their mimics, we propose a basic immunohistochemical panel of vimentin, CD34, and E-cadherin for the characterization of canine and feline meningiomas. The additional use of claudin-1, albeit of moderate to low sensitivity in meningiomas of small animals, can help to rule out some mesenchymal neoplasms involving the brain and associated structures, such as schwannomas. CD34 was consistently expressed in canine and feline meningiomas; as such, it may be used to distinguish them from peripheral nerve sheath tumors. However, validation of these and other markers for meningiomas will require their evaluation in canine and feline tumors that could resemble meningiomas.
Footnotes
Acknowledgements
We very much appreciate the technical expertise of Dee DuSold in performing histologic preparations and immunohistochemical testing.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
