Abstract
Sialyl Lewis x–modified core 2 branched O-glycans (C2-O-sLex) on human leukocytes mediate much higher-affinity adhesion to selectins on activated vascular endothelium than does sialyl Lewis x on other structures. In some canine and human carcinomas, high expression of sLex-decorated carbohydrates has been associated with metastasis and, in humans, a poor prognosis, but detection in canine gastric carcinomas is unreported. The authors hypothesized that these carbohydrates are highly expressed in more malignant types of canine gastric carcinomas, they promote metastasis, and they are associated with a poorer prognosis for dogs. The objectives were to determine the presence and importance of C2-O-sLex expression in canine gastric carcinomas. Routine histological sections of 16 canine gastric carcinomas were categorized on the basis of 3 classification schemes: World Health Organization, Lauren, and Goseki. Serial sections were stained with antibodies directed against C2-O-sLex (CHO-131 monoclonal antibody), cytokeratin (Lu-5 monoclonal antibody), and stains to detect neutral and acid mucins (periodic acid–Schiff and alcian blue). Whereas normal gastric mucosal epithelial cells were negative for C2-O-sLex, 56% of the tumors examined were positive for C2-O-sLex. Importantly, the majority of more poorly differentiated tumor types had more numerous and larger intensely stained areas of C2-O-sLex expression compared with moderate to well-differentiated tumor types. Signet ring–type carcinomas had markedly higher distribution and intensity of periodic acid–Schiff and alcian blue staining than did tubular carcinomas. These findings suggest that C2-O-sLex is a tumor-associated antigen that may play a role in the invasiveness and metastatic potential of certain types of canine gastric carcinomas.
In cancers of dogs and humans, a greatly increased metastatic potential and, in some instances, malignant behavior and a poor prognosis are associated with the high expression of carbohydrates modified with sialylated and fucosylated structures, including sialyl Lewis x (sLex). 21,23,27,28 In humans, the sLex antigen on leukocytes functions as a ligand for the selectin family of cell adhesion molecules and mediates the binding of leukocytes to vascular endothelium at sites of inflammation. These selectin-binding interactions are greatly enhanced when leukocytes display sLex on a core 2 branched O-glycan carbohydrate structure (C2-O-sLex). 16 The role of C2-O-sLex in the growth and metastasis of canine gastric carcinomas has not been examined, but we expect that these carbohydrates are tumor-associated antigens that bind to selectins and influence the malignant properties of canine gastric cancer cells.
Although the incidence of gastric cancer is much lower in dogs than in humans, this uncommon canine disease has increasingly been diagnosed in recent years with the more routine use of endoscopic techniques. 2,7,25 The majority (70 to 90%) of canine gastric tumors are carcinomas. 26,32 In both dogs and humans, prognosis is generally poor owing to the advanced state of the disease at the time of diagnosis. Treatment involves surgical resection but is often unsuccessful because the cancer metastasizes to regional lymph nodes and liver. Recurrence of disease is common after resection of tumors; in dogs, survival ranges from 3 months to 5 years depending on variables such as histologic tumor type, occurrence of metastasis, and location and size of the tumor. 1,4,5,20
Human and canine gastric carcinomas have been categorized according to 3 schemes: The World Health Organization (WHO) scheme is useful but offers little prognostic significance. 11,36 The Goseki and Lauren human classifications may more accurately predict prognosis based solely on histopathologic features, 9,15,29 and the latter scheme has been successfully adapted to the dog. 7,26 The Goseki and Lauren classifications have also been used to investigate the pathogenesis, metastasis, and prognosis of human gastric carcinomas. 10,19,29 Thus far, the TNM classification system has been the most accurate predictor of prognosis, wherein T indicates the size or direct extent of the tumor; N, the degree of spread to regional lymph nodes; and M, the presence of metastasis. 29,39 It is difficult to apply this latter system to dogs because the status of nodal and distant metastasis in dogs is often unclear at the time of clinical diagnosis.
As a first step to address whether a relationship exists between C2-O-sLex expression and tumor differentiation status, we examined the expression patterns of C2-O-sLex in 16 canine gastric carcinomas categorized by histologic type according to the WHO, 11 Lauren, 15 and Goseki 9 classifications, and we compared C2-O-sLex expression with that observed in 5 normal gastric tissues. We hypothesized that carbohydrates thought to play a role in tumor invasion and metastasis in humans are highly expressed in more malignant types of canine gastric carcinomas and promote metastasis.
Materials and Methods
Tissue Specimens
We examined tissue samples from 16 canine gastric carcinomas submitted to the University of Minnesota’s Veterinary Diagnostic Laboratory between 2001 and 2007, which represented all cases submitted during this time for which paraffin tissue blocks were available; we also examined 5 normal gastric tissues from archived samples. All tissues were used in accordance with the University of Minnesota’s Institutional Animal Care and Use Committee regulations.
Antibodies
CHO-131 monoclonal antibody (mAb) is a mouse immunoglobulin M (IgM) that binds the glycan structure C2-O-sLe and was kindly provided by Dr Bruce Walcheck, University of Minnesota. Mouse mAb Ab-3, clone Lu-5, is a mouse immunoglobulin G1 (IgG1; Thermo Fisher Scientific, Fremont, California) and is a broad-spectrum anti-pan-keratin antibody that stains epithelial cells and differentiates epithelial and nonepithelial tumors. The mu chain–specific alkaline phosphatase–conjugated goat anti-mouse IgM and gamma chain–specific horseradish peroxidase–conjugated goat anti-mouse immunoglobulin G (IgG) antibodies (Chemicon International Inc, Billerica, Massachusetts) were used as secondary antibodies in the CHO-131 and Lu-5 mAb staining procedures, respectively. Mouse IgM and mouse IgG1 (Invitrogen, Carlsbad, California) were used as negative-control antibodies for CHO-131 and Lu-5 mAb staining at the same concentrations as the corresponding primary antibodies.
Immunohistochemistry
Serial sections of gastric tissues from each dog were stained using CHO-131 mAb at a concentration of 15 µg/ml and Lu-5 mAb (mouse IgG1, Thermo Fisher Scientific) at a concentration of 0.5 µg/ml. CHO-131 mAb was used to detect C2-O-sLex in tumors, and Lu-5 mAb was used to detect cytokeratin to confirm the diagnosis of carcinoma and to identify tumor cells that were obscured by an intense scirrhous reaction. Tissue sections were deparaffinized, hydrated through graded concentrations of ethanol, incubated with protein block (ScyTek Laboratories, Logan, Utah) for 10 minutes, and treated with the primary antibodies for 60 minutes. Sections were incubated for 30 minutes with the secondary antibody: mu chain–specific alkaline phosphatase–conjugated goat anti-mouse IgM (16 µg/ml; Chemicon International Inc) for CHO-131 mAb preparations and gamma chain–specific horseradish peroxidase–conjugated goat anti-mouse IgG (Chemicon International Inc) for Lu-5 mAb staining. Vector Red (Vector Labs, Burlingame, California) and diaminobenzidine substrates (Invitrogen) were used as chromogens. Tissues were counterstained with Mayer’s hematoxylin. Incubations with protein block and primary and secondary antibodies were carried out in a humidified chamber at 27°C to 30°C. The remaining staining procedures were performed at room temperature. For comparison with the tumor tissues, samples of stomach from 5 normal dogs were processed and stained similarly using the CHO-131 and Lu-5 antibodies. Negative control sections were prepared by replacing the primary antibody with the corresponding isotype-matched, negative-control antibody.
Histopathology and Histochemistry
Tissues were processed routinely. Representative 4-µm-thick formalin-fixed, paraffin-embedded serial sections were stained according to standard procedures with hematoxylin and eosin (HE) to assess tissue morphology and with alcian blue (AB) and periodic acid–Schiff (PAS) to evaluate the presence of acid and neutral mucins, respectively.
Tumor Classification Systems
Based on HE-stained sections, the same 16 tumors were histologically classified according to the WHO, Lauren, and Goseki classifications. Classification per WHO criteria and assessment of tumor differentiation were determined by the most prominent histological pattern and principal cell type of the tumor: tubular, papillary, tubulopapillary, mucinous, signet ring cell, or undifferentiated carcinoma. 11 The tumors examined in our study were primarily signet ring or tubular types. Canine gastric carcinomas were classified as signet ring type when greater than half the tumor was composed of epithelial cells containing intracellular acid or neutral mucin displacing the nucleus to one side of the cell. 11,14 Carcinomas were categorized as tubular when they contained prominent neoplastic tubules. 11 HE-stained slides were examined and classified according to the Lauren classification, which has the following categories: intestinal type, diffuse type, and other (eg, tumors with equal proportions of intestinal and diffuse characteristics and others with a solid growth pattern). Intestinal carcinomas contained rudimentary glands that superficially resembled intestinal glands. Diffuse carcinomas contained cells that failed to form distinct structures. 15,26
The Goseki classification included evaluation of PAS- and AB-stained slides in addition to HE and contained the following categories: group I (good tubular differentiation, poor mucus amount in cytoplasm), group II (good tubular differentiation, rich mucus amount in cytoplasm), group III (poor tubular differentiation, poor mucus amount in cytoplasm), group IV (poor tubular differentiation, rich mucus amount in cytoplasm), and a category for others. 9
Differentiation Status
By definition, all diffuse or signet ring gastric carcinomas are poorly differentiated. 6 In this study, this feature was verified by histopathological examination of the canine signet ring gastric carcinomas. Tubular carcinomas were categorized as being well, moderately, or poorly differentiated: Well-differentiated tumors were carcinomas with well-formed glands. Moderately differentiated tumors were intermediate between well and poorly differentiated tumors. Poorly differentiated tumors were carcinomas composed of highly irregular, poorly formed glands that were difficult to recognize or contained infiltrates of many single cells that were isolated or arranged in clusters.
Immunohistochemical Staining Scores
The presence of CHO-131 mAb staining in gastric tissues was evaluated in a semiquantitative manner. For each tissue, 5 noncoincident microscopic fields at 400× magnification were consecutively analyzed and assessed for staining with CHO-131 mAb. Positive staining was defined as tumor cells in which cytoplasmic and/or cell membrane labeling with CHO-131 mAb was detected. Sections in which less than 25% of all tumor cells were positive were given a distribution score of 1+; tumors with 25 to 50% positive cells, 2+; tumors with 50 to 75% positive cells, 3+; and tumors with > 75% positive, 4+.
The intensity of CHO-131 mAb staining for C2-O-sLex and PAS and AB staining in reactive tissues was scored using a semiquantitative system from 0 to 3: intensity score 0, no visible staining; 1, mild staining; 2, moderate staining; and 3, strong staining. An intensity score of 0 to 1 indicated low reactivity, and 2 to 3 indicated high reactivity with CHO-131 mAb.
Statistical Analysis
For each tumor type within a classification system, the distribution scores for CHO-131 mAb staining were compared with the nonparametric Wilcoxon signed rank test and Mann–Whitney rank sum test. The numbers of tumors with high (2 to 3) or low (0 to 1) intensity scores for each tumor type within a classification system were compared with a chi-square test. A P value of ≤ .05 was considered significant.
Results
Overall, 9 of 16 canine gastric carcinomas (56%) stained positively with CHO-131 mAb, and the staining intensity was most prominent in the cytoplasm and cell membranes of individual neoplastic cells within each tumor. In contrast, none of 5 normal gastric tissues stained with CHO-131 mAb.
The 16 carcinomas were typed according to the WHO, Lauren, and Goseki classifications, and Table 1 presents the results. The same tumors were compared with each classification, and statistical analysis was performed to determine whether differences in CHO-131 mAb staining existed among tumor types within each classification. Individual tumors that were tubular carcinomas in the WHO classification correlated with the intestinal type in the Lauren classification and with group I in the Goseki system. The signet ring carcinomas in the WHO classification were classified as diffuse type within the Lauren classification and as group III or IV within the Goseki classification, depending on whether the cells contained a low or high amount of intracellular mucin, respectively. For each classification, we did not find statistically significant differences in CHO-131 mAb distribution or intensity scores among tumor types. Nevertheless, we did observe a trend in CHO-131 mAb staining patterns among groups within each classification, as described below.
CHO-131 Monoclonal Antibody Staining Scores for Canine Gastric Carcinomas: Grouped by Pathological Classifications
a Tissues were stained with CHO-131 monoclonal antibody. The percentage of tumor area stained and the intensity of staining for each tumor were determined as described in Materials and Methods.
As shown in Table 1, within the WHO classification, 13 of 16 gastric carcinomas were categorized as either signet ring or tubular; therefore, statistical analysis with CHO-131 mAb distribution scores was performed on these 2 types only. The other 3 tumors included 2 undifferentiated carcinomas (one of which was intensely stained with CHO-131 mAb; intensity score 3) and 1 moderately differentiated mucinous carcinoma that did not stain with CHO-131 mAb. We classified 9 tumors as poorly differentiated signet ring carcinomas; 6 of these 9 tumors (67%) demonstrated high-intensity staining with CHO-131 mAb (intensity score 3). The remaining 3 signet ring carcinomas did not stain with CHO-131 mAb (intensity score 0). Of the 4 tubular carcinomas, 1 well-differentiated tumor displayed highly positive, intense staining with CHO-131 mAb (tumor 10, intensity score 3, Table 1). Of the 3 tubular carcinomas that did not stain with CHO-131 mAb, 1 was well differentiated and 2 were moderately differentiated.
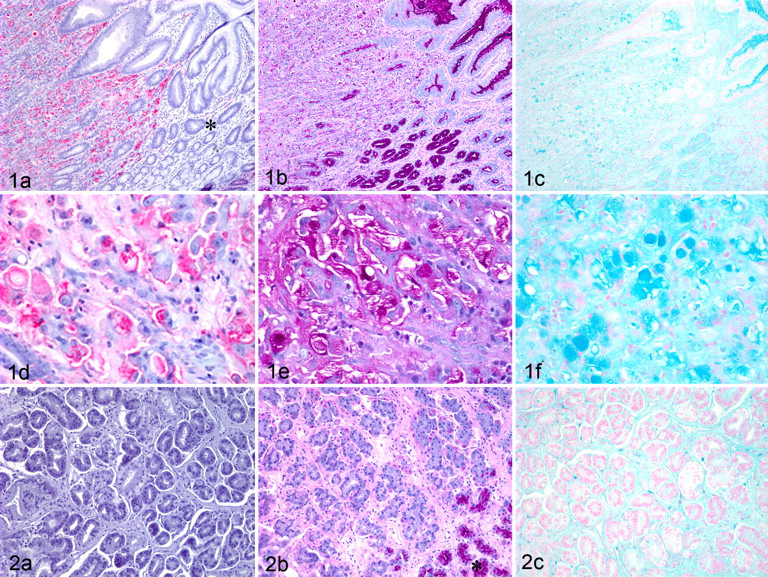
Figure 1 shows a signet ring–type gastric carcinoma (tumor No. 6, Table 1). The pattern of CHO-131 mAb staining corresponded to the tumor area. Normal gastric epithelium adjacent to malignant cells within the same tissue did not stain with CHO-131 mAb (Fig. 1A, asterisk). Importantly, the 6 signet ring gastric carcinomas that exhibited the strongest staining intensity with CHO-131 mAb also had the strongest staining with PAS and AB. Indeed, the intensity of PAS and AB staining, the number of cells stained, and the tissue area of staining in the signet ring gastric carcinomas were markedly increased compared to those of tubular carcinomas (Figs. 1 and 2). Normal gastric mucosal epithelial cells stained strongly with PAS and AB but, as mentioned earlier, did not stain with CHO-131 mAb. This pattern is evident in Fig. 1, which includes an adjacent area of normal gastric epithelium for comparison. Figure 2 shows a representative tubular carcinoma (tumor No. 10, Table 1) that demonstrates the diffusely negative immunostaining for CHO-131 mAb (Fig. 2A) and very faint staining with PAS and AB (Fig. 2B, 2C).
When the tumors were assessed with the Lauren classification, 11 were classified as diffuse type, 4 as intestinal type, and 1 as unclassified (Table 1). The unclassified tumor was omitted from the statistical analysis. Of the 11 diffuse type, 7 (64%) demonstrated areas of positive staining with CHO-131 mAb (distribution score 1+ to 4+), 6 of which were stained at high intensity (intensity score 3). Only 1 of 4 intestinal-type carcinomas (tubular type, WHO; type I, Lauren) was strongly stained positive with CHO-131 mAb (tumor No. 10, intensity score 3).
In the Goseki classification system, 4 tumors were categorized in group I, with good tubular differentiation that corresponded to the intestinal type (Lauren) and tubular type (WHO). None of the tumors was group II. Twelve tumors were categorized in groups III and IV, with poor tubular differentiation. Of 12 carcinomas, 8 (67%) were positively stained with CHO-131 mAb; of these, 7 were intensely stained (Table 1, intensity score 3).
As expected, epithelial cells in normal gastric mucosa and within the tumors were positive for cytokeratin, as identified with the Lu-5 mAb.
Discussion
The study reported here was designed to determine whether C2-O-sLex is consistently expressed in canine gastric carcinomas and whether relationships exist between the extent of C2-O-sLex expression and tumor differentiation status. This information addresses the need to understand the molecules involved in the pathogenesis of these tumors, and it could lead to the development of strategies to interrupt disease progression.
We postulate that C2-O-sLex as displayed on canine gastric carcinoma cells allows strong adhesion to selectins and promotes adherence to vascular endothelium, extravasation, and metastasis. Evidence to support our hypothesis is based on the ability of C2-O-sLex on human leukocytes to allow high-affinity selectin-mediated binding to endothelium. 16,17 Evidence is also based on our earlier studies wherein we found that C2-O-sLex is highly expressed on human colon carcinomas and liver metastases; expression increases with advancement of tumors to a less-differentiated status; C2-O-sLex is predominantly localized to the advancing, invasive front of colon carcinomas; and colorectal carcinoma cells expressing C2-O-sLex but not those cells lacking C2-O-sLex expression bind avidly to E-selectin. 30,31 The expression of C2-O-sLex on canine carcinoma cells, including gastric carcinomas, may similarly promote adhesion to selectins, attachment of cancer cells to vascular endothelium, and metastasis.
In human gastric carcinomas, the presence of C2-O-sLex has not been examined, but both sLex and sialyl Lewis a have been implicated in the high-metastatic potential and poor prognosis of these carcinomas on the basis of biochemical and molecular methods. 8,22,33 Furthermore, high levels of sLex in human gastric carcinomas have been correlated with advancing depth of invasion. 34 In dogs, recent reports have confirmed that sialyl Lewis carbohydrates are detectable in canine malignant mammary tumors and that elevated levels of sLex expression in these cancers are associated with lymph node metastases. 21,27 To our knowledge, this study is the first report of the detection of C2-O-sLex carbohydrates in canine gastric carcinomas.
We demonstrated that 50% of the canine gastric carcinomas examined here highly expressed C2-O-sLex, whereas all normal gastric tissues were negative, indicating that C2-O-sLex is a tumor-associated antigen. We did not observe significant statistical differences in distribution or intensity scores among tumor types within the WHO, Lauren, and Goseki classification systems, namely, because the power of the statistical evaluation to detect true differences in distribution or intensity scores was limited by the small sample size.
Of interest is that in the more poorly differentiated types of tumor (signet ring carcinoma in the WHO system; diffuse type in the Lauren system; groups III and IV in the Goseki system), 60 to 70% of these tumors had more numerous and larger areas of intense expression of C2-O-sLex, as evidenced by higher distribution and intensity scores, than did the moderate to well-differentiated types, in which only 25% of tumors highly expressed C2-O-sLex (tubular type, WHO; intestinal type, Lauren; group I, Goseki). Thus, increasing amounts of C2-O-sLex in canine gastric carcinomas may indicate their greater metastatic potential.
The CHO-131 mAb that recognizes C2-O-sLex is well characterized; in addition to our observation of positively staining canine gastric carcinomas, it has reacted positively with other cancer cells and immune cells. 3,24,30,31,35 The detection of C2-O-sLex in tissues was specific, as evidenced by the relationship between CHO-131 mAb staining and histochemical staining for neutral and acid mucins (PAS and AB, respectively) and between staining of neoplastic and normal cells. Normal gastric mucosal epithelial cells stained strongly with PAS and AB but not with CHO-131 mAb, indicating that the mucins present in normal gastric epithelium did not express C2-O-sLex. In addition to strong staining with CHO-131 mAb, the neoplastic poorly differentiated signet ring–type carcinomas stained strongly with PAS and AB, indicating that CHO-131 mAb selectively labeled cells uniquely expressing C2-O-sLex rather than nonspecifically binding to mucins. Furthermore, C2-O-sLex may decorate a mucin glycoprotein in canine gastric carcinomas. Aberrant glycosylation of mucins has been implicated in cell signaling and adhesion, 37,38 and together, C2-O-sLex and mucins may regulate malignant behavior of cancer cells.
Canine gastric carcinomas frequently metastasize to gastric lymph nodes, liver, spleen, peritoneum, and lung, leading to a poor prognosis for patients. 7 The most commonly occurring canine gastric tumor is a poorly differentiated signet ring gastric carcinoma (WHO) with excessive fibrosis and invasive growth; these generally have a worse prognosis than that of the less frequently occurring well-differentiated tubular carcinomas (WHO). 11,12 In humans, more extensive invasion of tumor cells and more frequent metastasis are associated with advanced signet ring (WHO) and diffuse (Lauren) gastric carcinomas. 13,15,18
For this report, information was not available on the clinical history of the dogs such that we could evaluate the relationship between expression of C2-O-sLex and prognosis. However, given our findings of the role of C2-O-sLex in human colorectal carcinomas and other reports on the relationships between sialyl Lewis structures and malignancy, 30,31 we believe that the more poorly differentiated canine gastric carcinomas strongly expressing C2-O-sLex are likely more invasive and have a higher metastatic potential, which could result in a poorer prognosis for affected dogs. The impact of C2-O-sLex expression on the prognosis of dogs with gastric carcinomas and the presence of C2-O-sLex in other types of canine carcinomas remains to be investigated. In future studies, we will investigate the potential ligands in the tumor microenvironment that bind C2-O-sLex and the role of these molecular interactions in tumor growth and metastasis.
Footnotes
Acknowledgements
We thank Dr Mariya Farooqui, Gregory Mitcheltree, and Kathleen Bolender-Neumann for assistance with the immunohistochemistry; Laurie Reichel, Anne Undersander, and Josh Parker for technical assistance with the histological tissue sections; Dr Karin Matchett for review of the article; and Dr Qing Cao for assistance with the statistical analysis. This work was supported in part by funds from the National Institutes of Health, National Cancer Institute, grant No. K08 CA111829-03.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
