Abstract
Solanum bonariense intoxication is characterized by cerebellar neuronal vacuolation, degeneration, and necrosis. Cerebellar Purkinje cells seem especially susceptible, but more research is needed to determine the pathogenesis of neuronal necrosis and the mechanism of Purkinje cell susceptibility. Calbindin D28k (CbD28k) is highly expressed in Purkinje cells and has been used as a marker for normal and degenerative Purkinje cells. The goal of this study was to describe S bonariense–induced disease by ascertaining Purkinje cell–specific degenerative changes using CbD28k expression and to correlate this with apoptosis in Purkinje cells, as determined using TUNEL (transferase-mediated dUTP-biotin nick end-labeling) and ultrastructural changes. In all cases, an increase in both dose and duration of S bonariense intoxication resulted in a decrease in the number of Purkinje cells. CbD28k immunohistochemistry was an excellent marker for Purkinje cells because immunoreactivity did not change in normal or degenerative tissues. This finding suggests that excessive calcium excitatory stimulation does not induce rapid neuronal degeneration and death. As found in previous studies, TUNEL tests and electron microscopy suggest that Purkinje cell degeneration and death are not occurring via an apoptotic process. These findings suggest that S bonariense poisoning induces progressive Purkinje cell death that is not mediated by excitotoxicity or apoptotic activation.
Keywords
Cattle eating Solanum bonariense (S bonariense) in western Uruguay developed periodic episodes of ataxia, hypermetria, hyperesthesia, head and thoracic limb extension, opisthotonos, nystagmus, and fall to the side or backward. 14 Reported histological lesions include Purkinje cell vacuolation, degeneration, and necrosis. Similar cerebellar cortical degeneration has been described in other countries in which cattle were poisoned with different solanaceae species. 7,12,13 Little is known of the degenerative mechanism, although the lack of apoptotic markers in degenerative neurons in Solanum kwebense–intoxicated cattle suggests that apoptosis is not involved. 13
Neuronal calcium may play a role in neuronal survival as well as in apoptosis, pathological neuronal, degeneration and necrosis. 6 Sustained increases in intracellular calcium levels, such as those observed after excitatory amino acid treatment or attributed to reduced transport activity, have been shown to correlate with increased protein and lipid degradation, which progresses to cell death. 1
In the cerebellum, where rapid firing neurons experience high levels of calcium influx, controlled calcium regulation is essential to avoid rapid neuronal death owing to excessive excitatory stimulation. 1 Intracellular calcium is mainly regulated by calcium binding proteins (CaBPs). Interaction of calcium with these proteins represents one of the mechanisms by which a second messenger controls many biological processes. 1
Calbindin D28k (CbD28k) is a CaBP highly expressed in Purkinje cells. Recent work suggests that CbD28k is a marker of Purkinje cells in normal and degenerative cerebellar tissue. 4,5 Researchers have speculated that in Purkinje cells, CbD28k expression is neuroprotective against degeneration in a variety of acute and chronic disorders. 1,2
The goals of the present work were to describe the effects of S bonariense in cattle by monitoring Purkinje cell populations using CbD28k expression. Labeled cells were also evaluated for evidence of apoptosis using the TUNEL (transferase-mediated dUTP-biotin nick end-labeling) test and ultrastructural evaluation.
Study details regarding plant description and experimental methodology have been reported. 14 Two control steers, 2 heifers naturally poisoned with S bonariense, and 4 steers experimentally intoxicated with S bonariense were prepared and examined. In the experimental portion of the study, each of the 4 experimentally intoxicated steers received S bonariense at varying doses and durations to produce different degrees of cerebellar degeneration. Steer No. 1 received 0.512 kg of fresh leaves of S bonariense per kilogram body mass (kg BM) during 61 days (0.50 times the cerebellar sign-inducing threshold [CSIT] dose); steer No. 2, 1.024 kg plant per kg BM for 128 days (1.00 times CSIT dose); steer No. 3, 1.28 kg plant per kg BM for 160 days (1.25 times CSIT dose); and steer No. 4, 2.048 kg plant per kg BM for 198 days (2.00 times CSIT dose).
Slices from 6 selected areas of the cerebellum of all animals in the study (3 from the cranial, medial, and caudal areas of the vermis; 1 from each lateral hemisphere; and 1 from the flocculus) were formalin fixed, paraffin embedded, and sectioned at 5 μm. One slice from each location was stained with hematoxylin and eosin for histological observation. Immunohistochemistry of the remaining slices was performed using a mouse monoclonal primary antibody, anti-CbD28k (clone NLC, Novocastra, Newcastle Upon Tyne, UK). Briefly, slices were incubated with primary antibody overnight. Following 3 washings of the slides with phosphate buffered saline, samples were incubated with a peroxidase-labeled polymer system (EnVision Universal Peroxidase, DakoCytomation, Glostrup, Denmark) for 45 minutes. Diaminobenzidine was used as a chromogen. Immunohistochemically stained slides were counterstained with Mayer’s hematoxylin.
Evaluation for possible apoptosis in degenerative Purkinje cells was performed using the in situ TUNEL apoptosis test (kit ApoTag, Chemicon, Temecula, CA) following the manufacturer’s suggested protocol on additional serial cerebellar sections from these animals. All TUNEL-labeled samples were counterstained with methylene green. The TUNEL protocol included positive controls obtained from samples of bovine small intestine and a negative control obtained by excluding the terminal deoxynucleotidyl transferase step of the original method.
For transmission electron microscopy (TEM) evaluation, 1-mm3 duplicate tissue block samples from each cerebellum were fixed in cacodylate-buffered 4% glutaraldehyde, postfixed in 1.5% osmium tetroxide, dehydrated via alcohols and propylene oxide, and embedded in epon. Semithin and thin sections were stained with 1% toluidine blue and uranyl acetate/lead citrate, respectively. 10
Each cerebellar sample was subjectively compared, and the histological studies confirmed the loss of Purkinje cells in natural and experimentally intoxicated animals (Figs. 1, 2). With a subjective evaluation, the loss of Purkinje cells in this study was similar to that of previous studies that reported degeneration or loss of Purkinje cells. 8,9,14
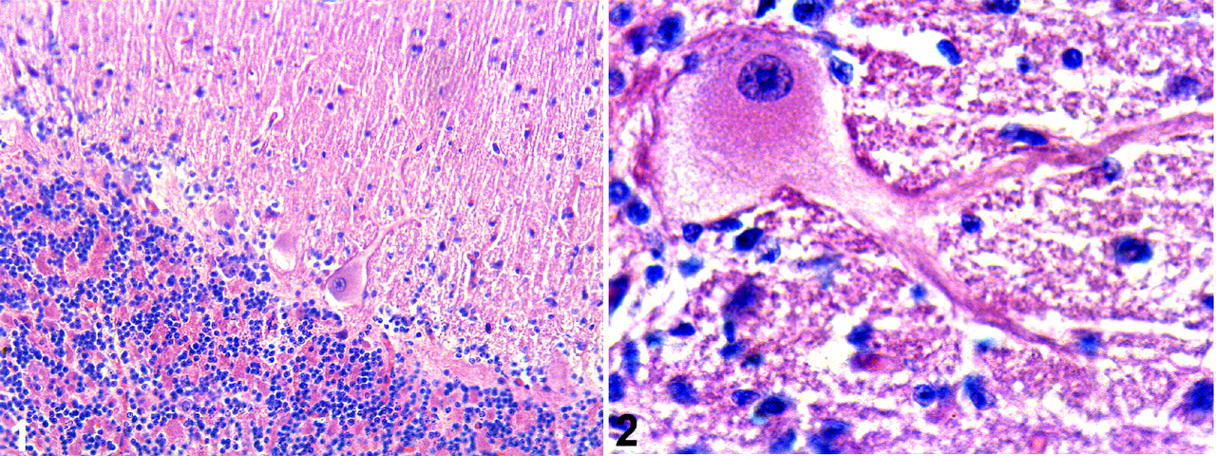
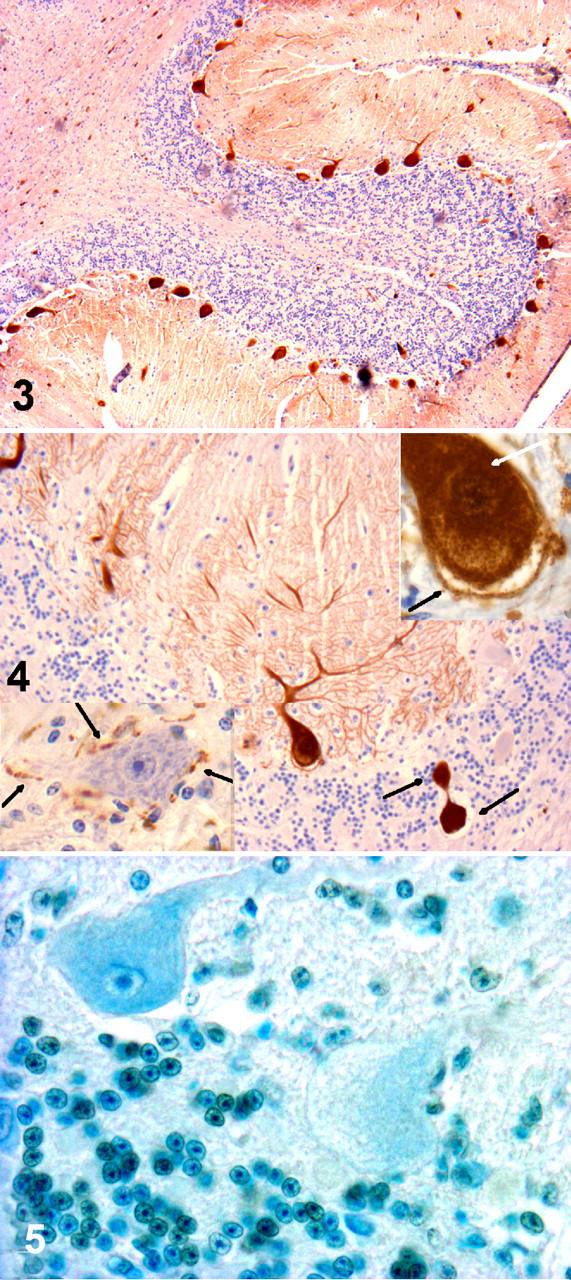
CbD28k immunoreactivity was always intense and regular in Purkinje cells from all sections of the cerebellum in control animals as well as animals experimentally and naturally poisoned. CbD28k immunoreactive intensity was observed in Purkinje cell bodies, axons, and dendrites and was similar in controls as well as experimentally poisoned and naturally poisoned animals (Figs. 3, 4). Degenerative axons or spheroids in the cerebellar white matter were also CbD28k positive. In all analyzed animals, neurons of the deep cerebellar nuclei were negative for CbD28k, whereas the surrounding synaptic terminals were positive (Fig. 4, lower inset).
Populations of Purkinje cells from equivalent cerebellar areas in intoxicated and control bovines showed no positive reaction to TUNEL apoptotic reagents despite obvious progressive Purkinje cell loss (Fig. 5). This finding was supported by TEM, which showed an absence of apoptotic figures in spite of signs of Purkinje cellular damage (data not shown).
The present study confirms a loss of Purkinje cells in S bonariense intoxication. Similar Purkinje cell loss has been observed after intoxication by other solanaceae species. 7,12 –14 In advanced cases, the progressive disappearance of dendritic processes from degenerative Purkinje cells results in a corresponding thinning of the molecular layer. 9 This study demonstrated that longer intoxication times resulted in correspondingly fewer Purkinje cells in the cerebellar laminae of all experimentally intoxicated steers, confirming the progressive disappearance of Purkinje neurons and their process.
As with other species, we found that CbD28k expression is high in bovine Purkinje cells. Purkinje cells have intense CbD28k immunoreactivity, which appears to be independent of the degree of cerebellar degeneration. Although similar findings have been reported, 8 Haworth et al 4 and Ishikawa et al 5 reported a significant decay in CbD28k immunoreactivity in other neurodegenerative models. Differences between these studies may be due to the dependence of CbD28k immunoreactivity on the degenerative process. For example, some excitotoxic degenerative Purkinje cells may have disruption of the calcium-buffering system. CbD28k plays a protective role against calcium-mediated excitatory amino acid neurotoxicity by reducing the levels of intracellular free calcium. 5 Disruption of the calcium-buffering system may result in CbD28k depletion and loss of immunoreactivity. This does not seem to be the case in S bonariense poisoning.
The negative reaction observed in samples where the TUNEL test was applied indicates that Purkinje cells do not die by apoptosis. Van der Lugt et al 13 reached a similar conclusion for bovines intoxicated with Solanum kwebense. We confirmed these findings using TEM, which showed the absence of typical apoptotic signs in the nucleus of affected Purkinje cells. Results suggest that the changes observed in degenerative neurons are more indicative of necrosis, although this hypothesis must be confirmed by other methods.
Neurons in cerebellar nuclei were negative for CbD28k antibody, but the surrounding Purkinje cell axonal projections were strongly positive. The same pattern of immunoreactivity was described in primates and rats 1,3,11,15 and is probably due to specific expression of other CBPs that buffer calcium in these neurons (ie, calretinin or parvalbumin). 1,3
In summary, CbD28k antibodies appear to specifically react with bovine antigens, thereby suggesting an accurate marker for bovine Purkinje cells. The combination of CbD28k expression, lack of TUNEL staining, and reported TEM findings suggests that S bonariense poisoning induces progressive neuronal death that is not mediated by excitotoxicity or activation of apoptosis. Additional studies using neurodegenerative markers may be useful in characterizing the cerebellar lesions.
Footnotes
Acknowledgements
This work was supported by project No. FPR/F/BI/49/28 (SECTIP, Argentina; PDT-MEC, Uruguay) and CSIC-UdelaR (Uruguay). C.G.B., C.N.Z., E.L.P., and E.J.G. are members of CONICET, Argentina. We thank Dr B. Stegelmeier (Poisonous Plants Research Laboratory, US Department of Agriculture, Logan, Utah) for his constructive criticism and proofreading. Our gratitude to Alba Román and Rosa Villegas for their technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
