Abstract
Scleromyxedema—the generalized form of lichen myxedematosus, a primary mucinosis—is a rare disease in human patients. It is characterized by dermal mucin deposits, increased numbers of fibroblasts, and variable fibrosis in the absence of thyroid disease. It is accompanied in 80% of cases by a monoclonal gammopathy. To date, scleromyxedema with systemic involvement has not been documented in domestic animals. This is the first report of a scleromyxedema-like syndrome in a cat, which had a substantial deposition of mucin in the dermis of the head and paws with a mild gammaglobulinemia of 2.25 g/dl (reference range, 1.39–2.22 g/dl). At necropsy, multiple nodules of connective tissue intermingled with mucin deposits were conspicuous on the surface of thoracic and abdominal organs. Such severe systemic accumulations of mucin have not been reported in human or veterinary medicine.
Scleromyxedema is the generalized form of lichen myxedematosus, a primary dermal mucinosis. Lichen myxedematosus is characterized by lichenoid papules, nodules, and/or plaques of dermal deposition of mucin and variable fibrosis in the absence of thyroid dysfunction. 17 It is divided into two clinicopathologic subsets: a localized, papular form and a generalized form called scleromyxedema. Scleromyxedema is a rare disease of middle-aged humans. Patients have a widespread, symmetric eruption of small, waxy, firm, closely spaced papules most commonly located on hands, forearms, face, neck, upper trunk, and thighs. 17 With progression, skin stiffness may occur and become generalized. 1,10,17
Most cases of scleromyxedema are associated with a monoclonal gammopathy. Of 26 cases in one study, 23 patients had a monoclonal gammopathy that, in most cases, was characterized by immunglobulin G lambda light chains. 7 In the bone marrow, a mild plasmacytosis may be noted, but progression to multiple myeloma occurs in less than 10% of patients. 16 Systemic disorders include disturbances of the central nervous system, peripheral neuropathy, arthropathies, hematological disorders, a scleroderma-like renal disease, gastrointestinal problems (mostly dysphagia), restrictive or obstructive pulmonary disease, endocrine abnormalities, proximal muscle weakness, inflammatory myopathy, and vascular abnormalities. 7,10,17 It is believed that some systemic manifestations of scleromyxedema might be due to aggressive therapy. 7
In humans, the typical histologic findings in the dermis consist of a diffuse deposit of mucin in the upper and midreticular dermis, an increase in collagen deposition, and the presence of irregularly arranged fibroblasts, often associated with a mild perivascular, superficial, lymphoplasmacytic dermal infiltrate. 16 The mucin deposits are characterized by an amorphous mixture of acidic glycosaminoglycans that consists mainly of hyaluronic acid. 16
Autopsy findings of scleromyxedema patients have rarely been reported and are often nonspecific. Mucin deposition in organs other than the skin is not a consistent finding. However, mucin has been reported in all layers of blood vessels of several organs, such as the heart, lungs, spleen, and kidney; in vessels associated with nerves; in the connective tissue beneath the pleura, peritoneum, and Glisson’s capsule; in the interstitium of kidney, pancreas, adrenal gland, and nerves; and even in the cornea. 3,10,13,16 Seldom does the mucin deposition seem to account for the systemic symptoms. The diagnosis of scleromyxedema is based on the clinical and histological features, the presence of monoclonal gammopathy, and the absence of thyroid disease. 16
Here, we present the clinical and pathologic findings in a domestic shorthair cat with a scleromyxedema-like disorder. The 13-month-old ovariectomized cat was presented with slowly progressive skin lesions on all four legs and the head. Anamnestically, the cat had variable signs of illness during its first few months of life; therefore, at the age of 5 months, when it was ovariectomized, a blood cell count was performed. It revealed a leukocytosis (23,150/µl). A differential leukocyte count was not done. Serum biochemistry values were within normal range. The total T4 was 28.4 nmol/L (reference range, 10.0–46.3 nmol/L). Tests for feline leukemia virus and feline immunodeficiency virus were negative.
The cat was presented at 11 months of age for clinical examination, which revealed mild interdigital erythema, hypotrichia, hyperkeratosis, and severe lichenification on the palmar and plantar aspects of the paws. The head of the cat, with the exception of the pinnae, was diffusely enlarged owing to thickening of the skin (Fig. 1 ). The cat was never pruritic. In addition to the cutaneous changes, the tongue was diffusely thickened and broader than normal. Skin scrapings were negative for mites. Impression cytology was unremarkable. Skin biopsy specimens were taken interdigitally and from the chin and the lips. Serum electrophoresis revealed a mild gammaglobulinemia of 2.25 g/dl (reference range, 1.39–2.22 g/dl), which was not further differentiated.
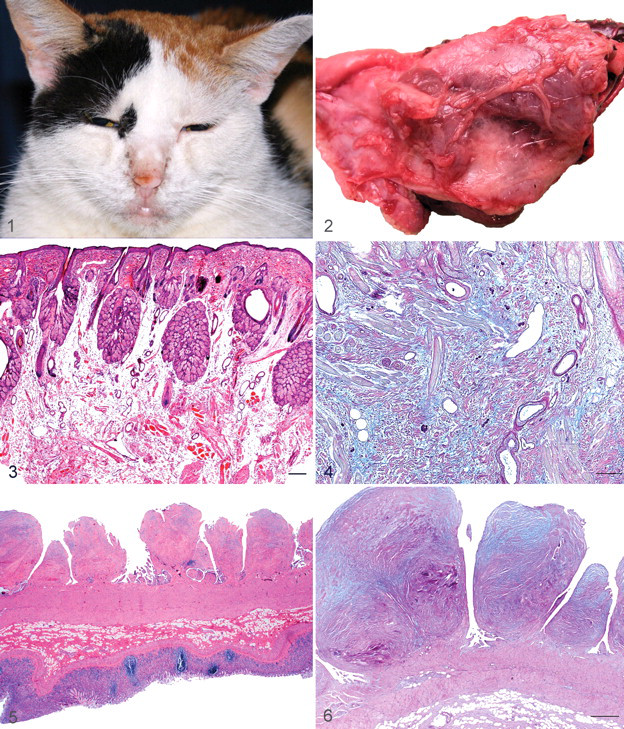
Two months later, the cat developed progressive dyspnea without fever. Between 50 and 120 ml modified transudate was aspirated from the left thoracic cavity daily on 5 consecutive days (specific gravity, 1.025; protein content, 3.1 g/dL). Multiple cystic lesions on the left lung, mild ascites, and distension of the uterine horns by liquid was detected by ultrasound. Owing to the progressive nature of the disease and the severe systemic signs, the cat was euthanized 5 days after the onset of dyspnea.
Necropsy revealed a diffuse moderate swelling of the skin of the head and all four feet. The thoracic cavity contained approximately 20 ml of transparent yellowish transudate. The left lung lobes were diffusely atelectatic; the overlying pleura was thickened by dense connective tissue with fluid-filled cysts up to 2 cm in diameter (Fig. 2). On the surface of the right lung, multiple, randomly distributed white firm nodules up to 0.2 cm in diameter were seen. The thoracic surface of the diaphragm was affected by thickening and nodules like those on the left lung. White nodules (diameter, 0.1–2.0 cm) were also in the peritoneum as well as the serosa of the stomach, liver, spleen, omentum, uterus, urinary bladder, and small and large intestines. Bilaterally, the fascia of muscles around the humeri were thickened. The thyroid gland and lymph nodes were macroscopically normal.
For histologic examination, tissue specimens were taken from skin of the head, chin, and paw upon presentation at the clinic and from all gross lesions and major organs at necropsy. Samples were processed routinely, and sections were stained with hematoxylin and eosin; selected sections were evaluated with periodic acid–Schiff (PAS), alcian blue, or toluidine blue.
In affected skin, amorphous ground substance separated the collagen fibers in the mid to deep dermis (Fig. 3). The section from the chin had a moderate, subepidermal fibrosis. Intermingled with the ground substance was an increased number of irregularly arranged spindloid to stellate well-differentiated fibroblasts. The superficial dermis comprised a moderate perivascular to interstitial infiltrate of mast cells and very few neutrophils, eosinophils, lymphocytes and plasma cells. The epidermis and the outer root sheath of some hair follicle infundibula were moderately and irregularly hyperplastic. The ground substance was PAS negative, alcian blue positive at pH 2.5 (Fig. 4), and toluidine blue negative at pH < 2.0. These staining characteristics are consistent with hyaluronic acid, the main constituent of dermal mucin. 16 Immunohistochemistry for CD3 and S100 was performed using a rabbit antibody to bovine S100 (Dako Z0311, 1:800, Dako, Glostrup, Denmark) and a polyclonal rabbit antibody to CD3 epsilon (RB-9039-P9, 1:200, LabVision Neomarkers, Fremont, CA). Immunhistochemistry for CD3 and S100 was negative.
A skin section from the back revealed myositis of the subcutaneous musculature. The affected musculature had severe vacuolar degeneration of myocytes with replacement by granulation tissue. In the sections of stomach, small and large intestine, omentum, and lung, the serosal nodules were composed of abundant mucin, intermingled with polygonal to fusiform fibroblasts (Figs. 5, 6). The large intestine serosal nodules had hyperplastic mesothelium. The myocytes of heart and skeletal muscle (Mm. biceps and triceps) were multifocally atrophic with signs of degeneration (hypereosinophilia, loss of cross striation, fragmentation). The uterine wall was diffusely thickened by fibrous tissue intermingled with abundant ground substance. There were also multiple uterine serosal nodules composed of connective tissue. Sections of diaphragm and lung contained a diffuse proliferation of pleural connective tissue with abundant mucin. Findings of the spleen, thymus, thyroid gland, parathyroid gland, liver, and kidney were within normal limits.
Pathologic findings were consistent with a scleromyxedema-like syndrome. The cause of scleromyxedema is unknown. Increased fibroblast activity probably contributes to the mucin accumulation. 1 However, the stimulating factors have not yet been defined, and the role of the monoclonal gammopathy is still debated because its severity does not correlate with extent or progression of the disease. 16 It was shown that serum of scleromyxedema patients with a monoclonal gammopathy stimulated the production of hyaluronic acid and prostaglandin E in fibroblast cultures but that isolated serum IgG failed to do so. 11,19 This suggests a pathogenetic role of other, yet-unknown circulating factors, such as interleukin 1, tumor necrosis factors, and transforming growth factor β, which are known to stimulate glycosaminoglycan synthesis. 16 Clinical remission of scleromyxedema after transplantation of autologous stem cells points to the bone marrow as a source of these circulating factors. 16
Several findings in our cat support the diagnosis of a scleromyxedema-like syndrome. These include the distribution of the skin lesions (which resembles that described in humans) and, especially, the histologic findings and the staining characteristics of the ground substance. In addition, our cat presented with subcutaneous myositis of the back and degenerative changes in cardiomyocytes and skeletal musculature. Similar muscular lesions are reported in human patients. 1,7 Furthermore, thyroid and renal disorders were excluded. Unfortunately, the mild gammaglobulinemia in our case was not further analyzed; thus, another characteristic of human scleromyxedema—monoclonal gammopathy—was not verified. However, about 20% of reported human cases are not associated with a monoclonal gammopathy. 7,13,14,18 In these cases, the diagnosis is based on clinical presentation, histologic characteristics, and, if present, the systemic involvement. The course of the disease in our cat was chronic and progressive, and the systemic involvement severe. A prominent finding in our cat was the widespread distribution of nodular, mucin-containing lesions on almost all abdominal organs, lung, and diaphragm. Such tumorlike nodules have not been reported in human scleromyxedema, although mucin deposits have been reported in the connective tissue beneath the pleura and peritoneum. 13 Because the putative fibroblast-stimulating factors of scleromyxedema are in the serum, such a widespread, systemic activation of fibroblasts, as seen in our cat, might well be a conceivable presentation of the disease.
Apart from cutaneous mucinosis in the Shar-Pei and myxedema in hypothyroid animals, cutaneous mucinoses rarely occur in domestic animals, and few such cases have been described. 4,8 These include the cases of papular and plaquelike mucinotic lesions in a puppy, focal nodular mucinoses in 7 dogs, and generalized papular mucinosis in 1 dog. 2,4,6 In all but the last case, the clinical and histological features of the skin lesions differ from those in our cat. The dog with the generalized papular mucinosis had diffuse cutaneous edema and mucinous papules throughout the dermis with multifocal hyperplasia of fibroblasts in the superficial dermis. In addition, this dog had an abnormal serum gamma-globulin profile. 4 These findings appear to fulfill the qualifications for a scleromyxedema-like syndrome. In cats, a case of a generalized scleredema has been reported in which the dermis was severely thickened by collagen fibers. However, mucin deposits were scarce, and no increase in fibroblast number was noted. 4
The differential diagnosis for dermal mucin accumulations includes mucin-producing mesenchymal tumors such as myxomas, mucinous hamartomas, and tumors of neural origin. Furthermore, mucin accumulation may be associated with epitheliotropic lymphoma, mucopolysaccharidoses, systemic scleroderma, and scleredema. 5,16 The neoplastic entities were ruled out by the clinical, histological, and immunohistochemical findings. Mucopolysaccharidoses are characterized by accumulation of glycosaminoglycans, such as dermatan and heparin sulfate, which have different staining characteristics from hyaluronic acid. 16 Human scleroderma may clinically resemble scleromyxedema. It is a collagen vascular disease leading to induration of the skin caused by excessive collagen deposition in the dermis and subcutis, with only small amounts of mucin, if present at all. 9 The typical histologic finding in scleredema is a thickening of the reticular dermis with large collagen bundles that extend into the subcutis. 16
An important entity in the differential diagnosis of scleromyxedema in human medicine is nephrogenic fibrosing dermatopathy, which may develop with chronic renal disease and lead to skin lesions histologically identical to those of scleromyxedema. 12 In contrast to scleromyxedema, nephrogenic fibrosing dermatopathy typically does not include severe systemic involvement, monoclonal gammopathy, or facial involvement. 12 Furthermore, in our case, the cat did not have chronic renal disease.
The young age at onset in this cat merits consideration of cutaneous mucinosis of infancy, a localized form of lichen myxedematosus. However, the focal well-circumscribed dermal mucin deposits of lichen myxedematosus are unassociated with fibroblast proliferation or systemic disease. 16,17 Some authors argue that cutaneous mucinosis of infancy might not be a separate entity but, rather, an infantile presentation of lichen myxedematosus. 15
Thus, in conclusion, the clinicopathologic findings in our case support the diagnosis of a scleromyxedema-like disorder and so eliminate other entities in the differential diagnosis.
Footnotes
Acknowledgements
We deeply appreciate the discussion of this case with Dr Helga Nievergelt from the Universitätsklinik für Dermatologie at the Inselspital Bern and the technical staff in the clinic, the necropsy hall, and the histology laboratory.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
