Abstract
The present study describes pathologic and virologic findings in 15 sheep and 6 goats that died of natural peste des petits ruminants virus infection in Turkey. Pathologic findings included erosiveulcerative stomatitis, fibrino-necrotic tracheitis, bronchointerstitial pneumonia, multifocal coagulation necroses in the liver, and severe lymphocytolysis in lymphoid tissues. Syncytial cells were conspicuous, especially in the oral mucosa, pulmonary alveoli, liver, and lymphoid tissues. In addition to the typical tissue distribution, eosinophilic intracytoplasmic and/or intranuclear inclusions were observed in epithelial cells lining the renal pelvis and abomasal mucosa. Immunolabeling of the viral antigen was observed in the kidney, brain, rumen, abomasum, heart, and myocytes of the tongue besides its more typical locations. In this study, we report and describe in detail the first peste des petits ruminants endemic in Kirikkale Province, Central Anatolia of Turkey. In conclusion, these previously unreported pathologic findings in natural peste des petits ruminants virus infection establish a basis for resemblance to other morbillivirus infections, such as canine distemper and distemper of sea mammals. Reverse transcriptase-polymerase chain reaction analyses indicated that the 448-bp genome fragment was amplified in 18 cases (18/21, 85.7%). Phylogenetic analysis showed that viruses belong to lineage 4 in the peste des petits ruminants virus common phylogenetic tree.
Introduction
Peste des petits ruminants (PPR) is a highly contagious viral disease of goats and sheep that has been widely reported throughout the Middle East, India, and sub-Saharan North Africa during the last 2 decades. 1– 4, 9, 22, 24, 26, 31, 32, 34 The PPR virus (PPRV) is the fourth member of Morbillivirus, which belongs to the family Paramyxoviridae, and has a close relationship with rinderpest, measles, canine distemper, and phocine distemper viruses. 5, 17 Histopathologic findings resulting from PPRV infection include pseudomembraneous, erosive, and ulcerative stomatitis; necrotic tonsillitis; fibrinohaemorrhagic enteritis; and bronchointerstitial pneumonia. 6, 7, 9, 12, 27, 28 Among the characteristic histopathologic findings of PPRV infection are syncytial cells in affected oral mucosa and lungs, as well as eosinophilic nuclear and cytoplasmic inclusion bodies, especially in the respiratory and/or alimentary tract epithelia. 6, 8, 12, 21, 28 Noncharacteristic pathologic findings, such as multifocal necrosis in the liver and inclusion bodies in hepatocytes and abomasal epithelial cells, are rarely reported. 2, 32 Little is known about the resemblance of the pathologic findings of PPRV infection to those resulting from the other morbillivirus infections in terms of lesions in the central nervous system, kidney, and abomasum. 11, 21, 28, 32, 33
The first PPRV infection in Turkey was reported in 4 sheep 2 , and since then many outbreaks have been reported. 23, 24, 32, 34 Özkul et al. 24 isolated PPR viruses from 2 separate field cases in Turkey. These viruses belonged to lineage 4, to which goats seem to be more susceptible than sheep. The disease results in high mortality, especially among young goats, although the frequency of the disease is higher in older goats. 18, 32
In this study, we report and describe in detail the first PPR endemic in Kirikkale Province, Central Anatolia of Turkey. The objectives of the study were to describe the histopathologic, immunohistochemical, and virologic findings of the natural PPRV infections in sheep and goats and to reconsider the tissue localization of the PPRV.
Materials and Methods
Animals
The materials for the study were collected during necropsies of 15 sheep and 6 goats submitted from 15 different flocks raised in Kirikkale Province and suspected to have PPR (Table 1). In December 2004, the initial PPR cases came from a flock (Flock A) containing 250 hair goats and 15 Angora goats. Four goats (case Nos. 1–3, 6) died within a month after exhibiting clinical signs, such as coughing and diarrhea. Between December 2004 and September 2005, 9 dead animals (case Nos. 4, 5, 8, 9, 12, 18–21) and 8 organ samples (case Nos. 7, 10, 11, 13–17) from 14 different flocks (Flocks B−N) suspected to have PPR were submitted to the Kirikkale University Veterinary Faculty Pathology Laboratory for necropsy and histopathologic examination. According to the history, the animals had not been vaccinated against PPRV infection. There were no reports of abortion, but there had been a case of dead-born twin lambs (case Nos. 4, 5) as a result of artrogriposis and scoliosis. Necropsy was performed within 12 hours after death, and tissue samples were fixed in 10% neutral formalin for 48 hours, routinely processed, embedded in paraffin wax, and sectioned at 5 μm. For virologic analyses; fresh samples of the lung, spleen, and lymph nodes were stored at −20°C until assayed with reverse transcriptase−polymerase chain reaction (RT-PCR).
Severity and localization of the immunohistochemical findings in natural PPRV infection.
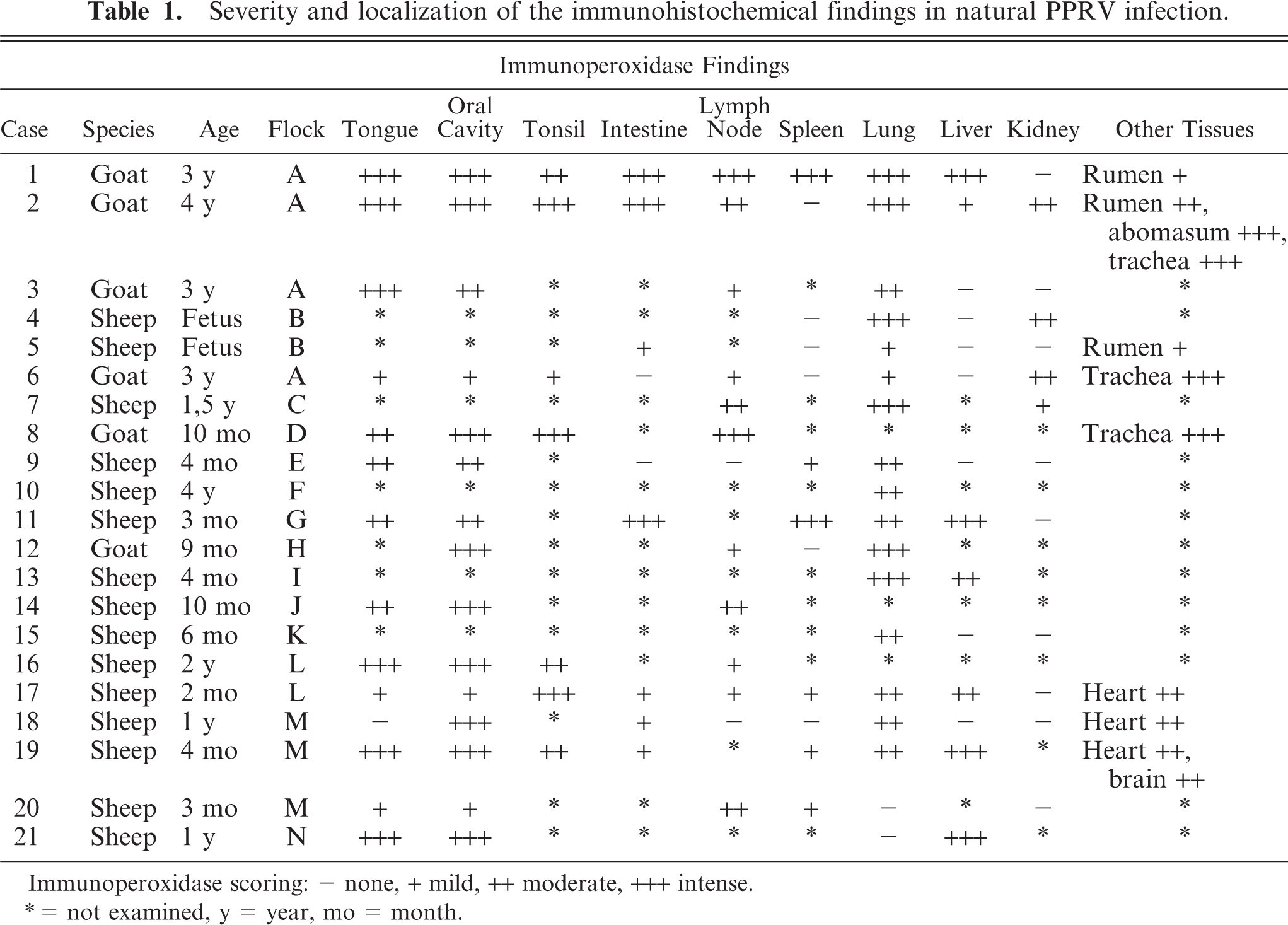
Immunoperoxidase scoring: − none, + mild, ++ moderate, +++ intense.
∗ = not examined, y = year, mo = month.
Immunohistochemistry
A commercial streptavidin/biotin immunoperoxidase kit (LSAB 2 system, HRP, DacoCytomation, Denmark) was used, and all procedures were performed according to the instruction manual. Tissue sections were digested with Proteinase K (0.1%) and incubated with a rabbit antirinderpest antibody (Institute for Animal Health, Pirbright, UK) at a dilution of 1/500. An aminoethyl carbazole chromogen substrate system (Labvision Corp., Fremont, CA) was applied for color reaction.
Peste des petits ruminants virus−positive lung tissues, previously confirmed with RT-PCR, were used as positive controls. Lung sections from 2 lambs (3 and 9 months old) with no clinicopathologic evidence of PPRV infection were used as negative controls. Immunoperoxidase scoring was made on the basis of positively stained cells observed in 3 different areas at 40 × microscope objective. The scores were as follows; − (none): absent, 1+ (mild): a few immunopositive cells, 2+ (moderate): focal prominent immunopositivity, and 3+ (intense): strong immunopositivity in more than 50% of the cells.
Reverse Transcriptase−Polymerase Chain Reaction
Detection of PPRV RNA was performed as described elsewhere. 14 Briefly, organ suspensions were used as a source of RNA for RT-PCR detection of viral genomes. Virus RNA was extracted using acid guanidium-phenol-chloroform-isoamyl alcohol mixture as described by Chomczyňski and Sacchi. 10 The RNA pellet was dissolved in 20 μl of sterile distilled water and used for cDNA synthesis. Complementary DNA synthesis was initiated by incubation of tubes at 70°C for 5 minutes to denature probable secondary structures in the RNA. The synthesis of cDNA was carried out in a mixture of 25 mM Tris-HCl, 25 mM KCl, 4 mM MgCl2, 10 mM DTT, 50 ng random hexamere primers, 200 U Moloney murine leukemia virus reverse transcriptase (MMLV-RT) (MBI, Fermentas, Lithuania), and 10 U of RNase inhibitor. The reaction mixture was incubated first at 25°C for 10 minutes, which was followed by second step incubation at 37°C for 1 hour. Moloney murine leukemia virus reverse transcriptase was then inactivated by holding at 70°C for 10 minutes.
Polymerase chain reaction amplification was carried out by adding 3 μl of cDNA in the master mix containing 75 mM Tris-HCl (pH8.8), 20 mM NH4(SO4)2, 1.5 mM MgCl2, 15 pmole of each primer, 0.2 mM dNTP, and 0.5 U of Taq DNA polymerase (MBI, Fermentas, Lithuania). The amplification was completed in 30 μl of total reaction mixture in the thermal cycler (Techne, Oxford, UK). The steps of amplification on thermal cycler were set up as follows: initial denaturation for 6 minutes at 94°C was followed consecutively by 1 minute at 65°C, 1 minute at 72°C, and 45 seconds at 94°C, which were repeated 40 times. Amplification was terminated by final extension at 70°C for 10 minutes. A PPR-specific primer set used in the study was selected from the F protein-coding gene sequence. Designed upper (sense) and lower (antisense) primers were as follows; PPRF1b - 5′-ATCACAGTGTTAAAGCCTGTAGAG-3′ (positions 760–784) and PPRF2d - 5′-GAGACTGAGTTTGTGACCTACAAG-3′ (positions 1183–1207). Using this pair of primers, it was expected to amplify a 448-bp DNA product. The resulting DNA products (amplicon) were analyzed on agarose gel (1.5%) after electrophoresis at 80 V for 30 minutes. The DNA bands were observed under ultraviolet light and photographed.
Sequencing and Phylogenetic Analysis
The DNA products obtained from RT-PCR were cleaned using a commercial kit (Sigma, Germany) and sequenced using Sanger's method in CEQ 8000 Genetic Analyser (Bechman-Coulter, USA). 30 Representative viruses for each endemic year (2004/2005) were used for sequencing. The nucleotide sequences were aligned with those obtained from cases of previous years, and phylogenetic analysis were performed using DNADIST and NEIGHBOR programs of PHYLIP v3.6 software. 13
Results
Macroscopic Findings
Fibrinopurulent nasal discharge and crusts were present around the nostrils and on the muzzle (case Nos. 1–3, 8, 16, 18). When the crusts were removed, the surface appeared ulcerative and hemorrhagic. Lesions of the oral cavity were localized to the dorsal and ventral surfaces of the tongue, lips, and soft and hard palates and consisted of gray to yellow pseudomembraneous foci, which were 1 to 5 mm in diameter (Figs. 1, 2). Prominent erosions were detected on the inner surfaces of the lips and mechanical papillae. Nasal conchae was diffusely hyperemic, and multifocal erosions were present on the mucosa. The oropharyngeal mucosa and the neighboring tonsillar surfaces were covered by fibrinous exudates and plaques (Fig. 3), which were caseous in severe cases (case Nos. 1, 2, 8, 17, 19). Fibrino-necrotic tracheitis was detected in a case (case No. 2) (Fig. 4). Cranial lung lobes were consolidated and hemorrhagic. The intestinal mucosa was congested and covered with mucous. The Peyer's patches were hemorrhagic and necrotic. Multifocal pale areas were evident at the cut surfaces of the liver in 6 cases (case Nos. 1, 11, 13, 17, 19, 21).
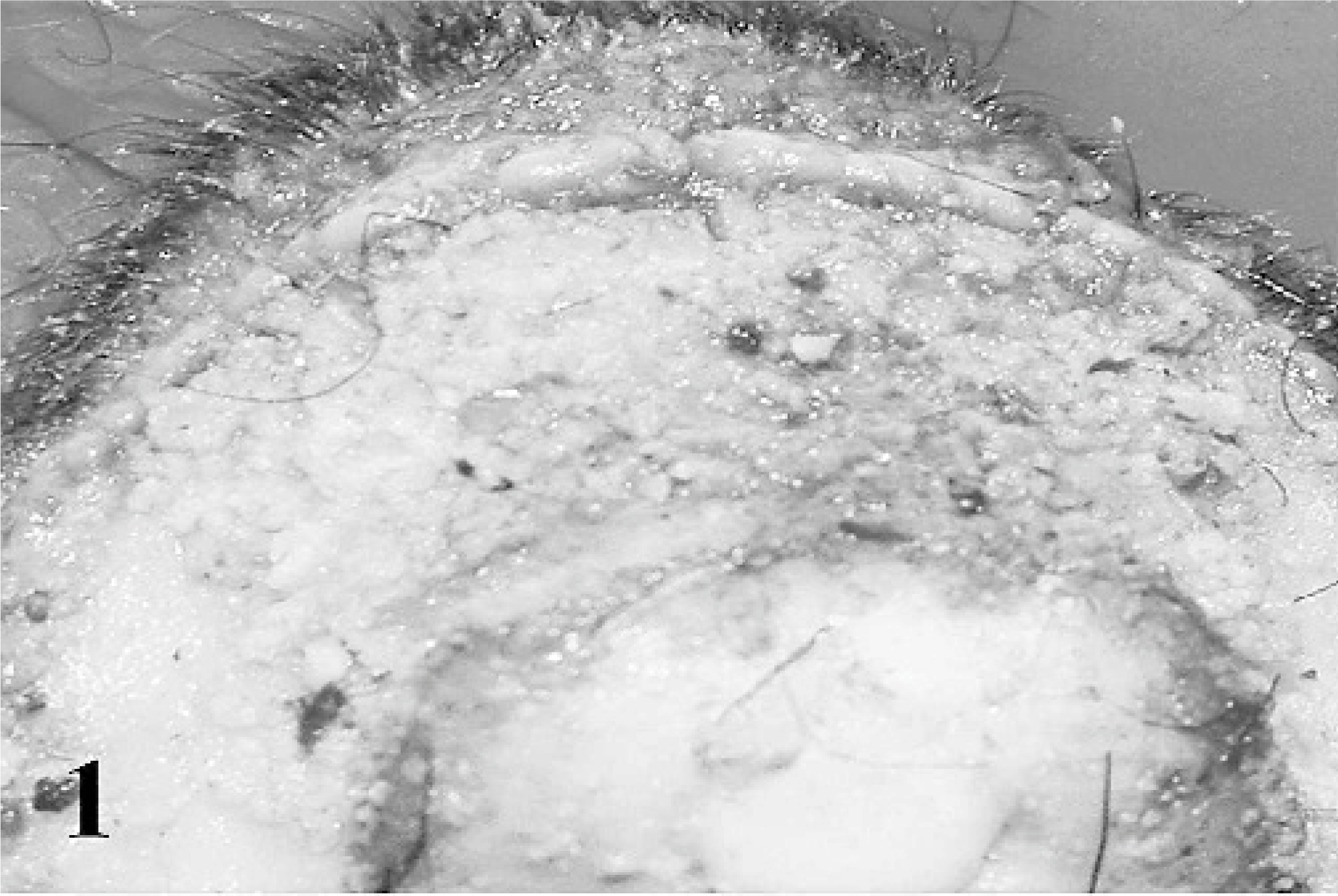
Oral mucosa, goat. Necrotic and pseudomembraneous foci are on the upper lip mucosa.

Tongue, goat. Pseudomembraneous foci, 1 to 5 mm in diameter and creamy in color are noted on the ventrolateral surfaces of the tongue.
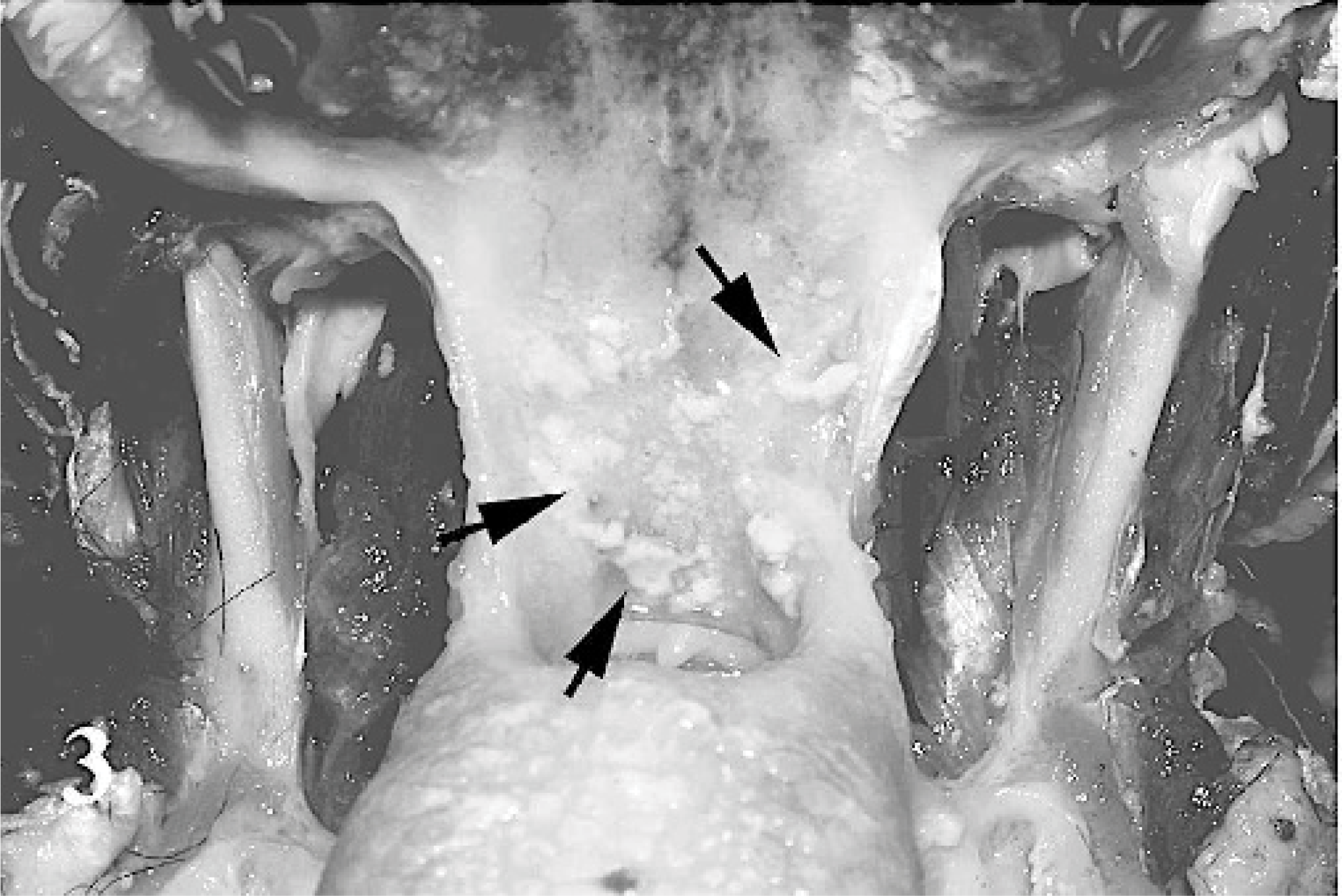
Oral mucosa, goat. White-yellowish necrotic material (caseous) is deposited on the oropharynx mucosa (arrows).
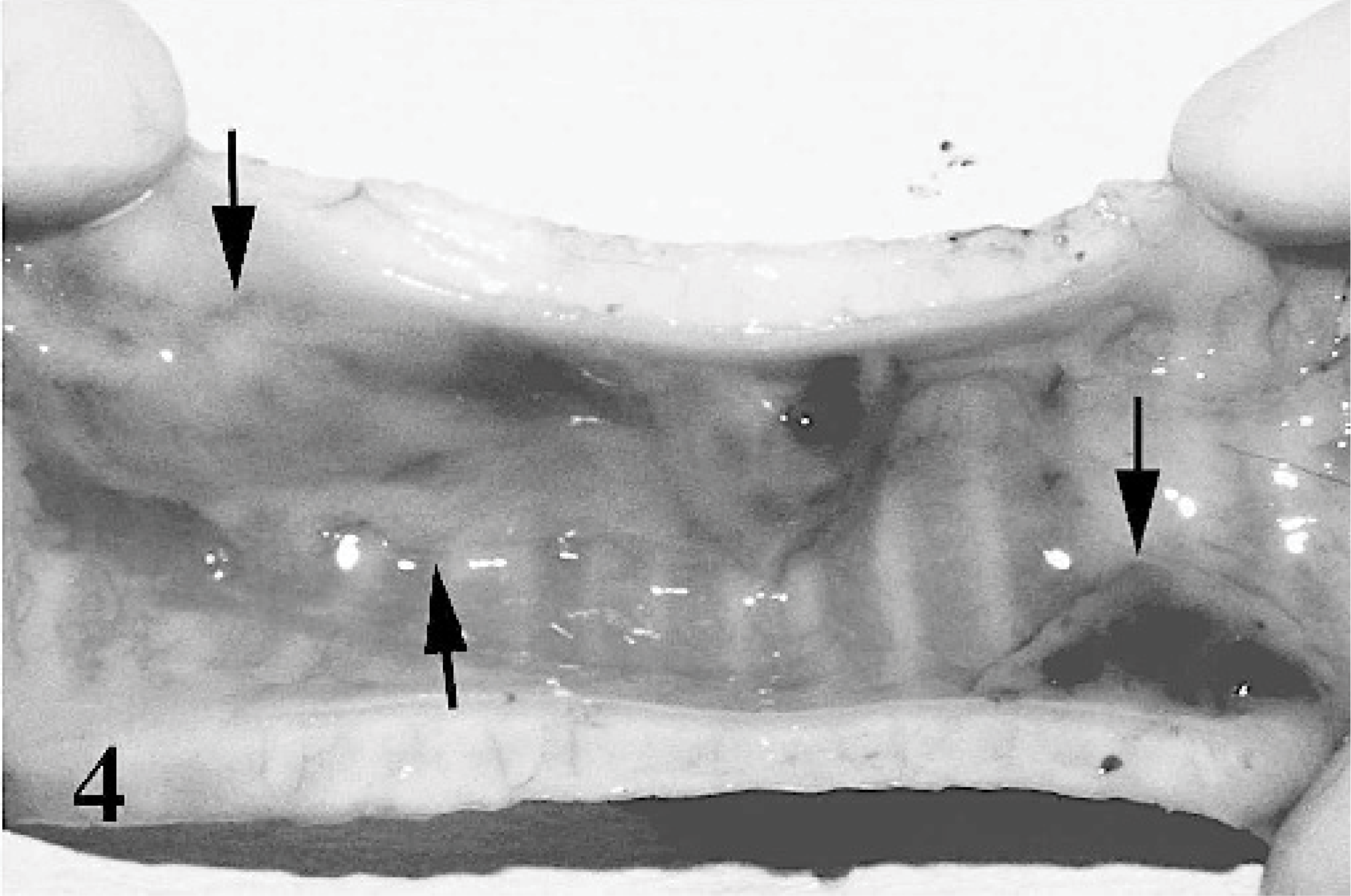
Trachea, goat. Extensive fibrinopurulent, necrotic foci (arrows) on the trachea mucosa. Mucosa is diffusely hyperaemic and oedematous.
Histopathology
Erosive-ulcerative lesions were observed on the oral mucosa (Fig. 5) (case Nos. 1–3, 8, 9, 16, 18, 19), nasal conchae, and trachea (case Nos. 2, 6, 8). These ulcerated areas contained numerous syncytial cells with 2 to 10 nuclei and eosinophilic cytoplasmic inclusion bodies. In the rumen of 3 cases (case Nos. 1, 2, 5) and abomasum in a case (case No. 2), there was vacuolar and hydropic degeneration, as well as intracytoplasmic inclusion bodies in the mucosal epithelial cells. The most conspicuous pathologic findings in lymphoid tissues, including Peyer's patches (case Nos. 1, 2, 6, 11), lymph nodes (case Nos. 1–3, 7, 8, 16, 17), and spleen (case Nos. 6, 9, 11, 19, 20), was lymphoid depletion. Syncytial cells, eosinophilic debris, and intranuclear inclusion bodies were also noted in the affected lymphoid tissues (Fig. 6). Tonsils (case Nos. 1, 2, 8, 16, 17, 19) had numerous intracytoplasmic inclusions in crypt epithelia. Bronchointerstitial pneumonia was characterized by syncytial cells in alveoli (Fig. 7) and inclusions in syncytial cells, alveolar macrophages, bronchial/bronchiolar epithelia, and peribronchial glands.
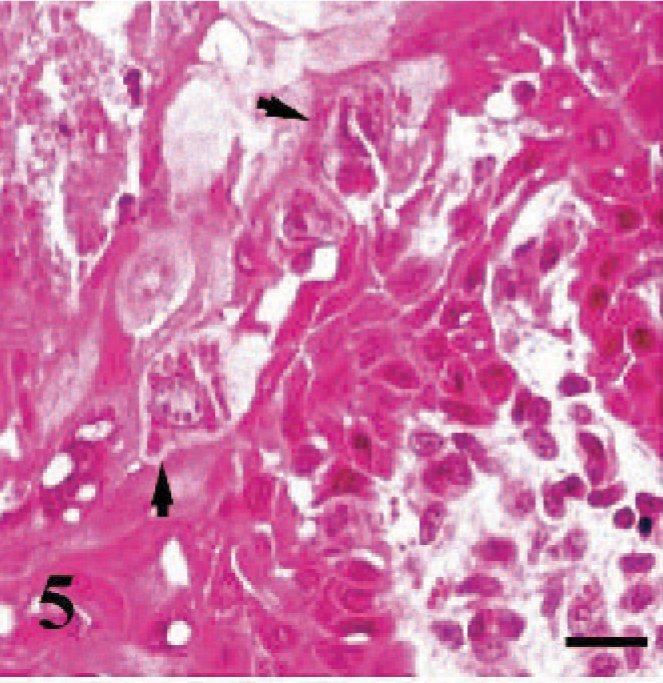
Buccal mucosa, goat. The photomicrograph demonstrates the pseudomembraneous changes, hydropic degeneration, and necrosis in some epithelial cells. In the stratum spinosum, intracytoplasmic inclusions are present (arrows). HE, bar = 20 μm.
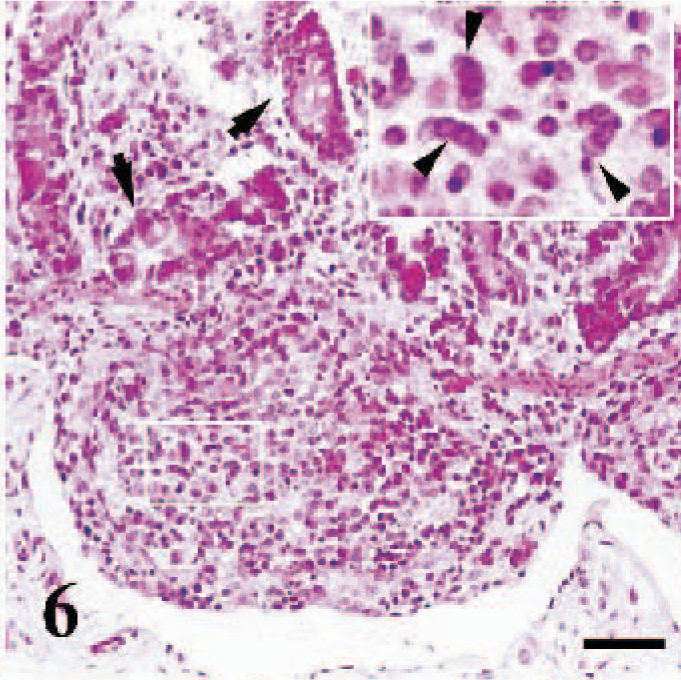
Ileum, sheep. Lymphocytolysis in Peyer's patch. Also notice numerous reticular syncytial cells (arrowheads), and necrotic intestinal glands (arrows). HE, bar = 180 μm.
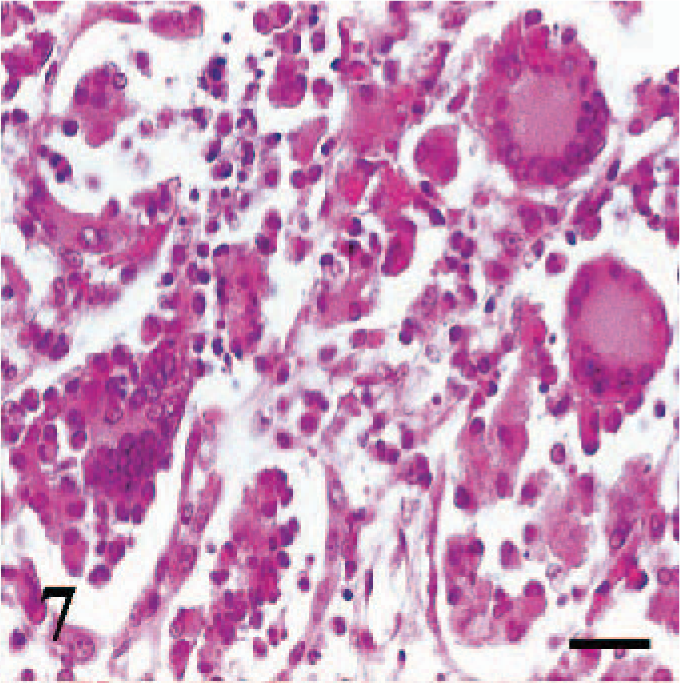
Lung, goat. Syncytial cells are prominent. HE, bar = 60 μm.
Multifocal midzonal-periportal hepatocyte coagulation necroses and rare calcification (case Nos. 17, 19, 20) were detected in affected livers (case Nos. 1–3, 11, 13, 17, 19, 21). Syncytial cells were formed by fusion of 2 to 6 hepatocytes around the necrotic areas. Eosinophilic intranuclear inclusions were also noted in hepatocytes and syncytial cells.
Vacuolar degeneration (case Nos. 17, 18, 20) and eosinophilic inclusion bodies (case No. 2) were observed in transtitional epithelial cells of the renal pelvis. There was fragmentation and hyalinization of cardiac myocytes with infiltrates of lymphocytes and macrophages in 3 cases (case Nos. 17–19).
Immunoperoxidase Findings
Tissue localization of immunolabeling indicating viral presence was complementary to histopathologic findings. In oral mucosa, immunolabeling was detected in nuclei and cytoplasm of the stratum spinosum and syncytial cells. Additionally, necrotic cell debris on the lip and tongue mucosa were immunopositive (Fig. 8). Submucosal seromucous glands also showed immunoreactivity. In the tongue (case No. 19), macrophages in the propria and myocytes in the muscularis mucosa were immunopositive. Intense immunolabeling was observed in epithelial cell debris, inflammatory cells in the epidermis, hair follicles, and sweat glands of the muzzle near the mucocutaneous junction of the lip (case Nos. 1–3, 8, 9, 16, 18, 19).
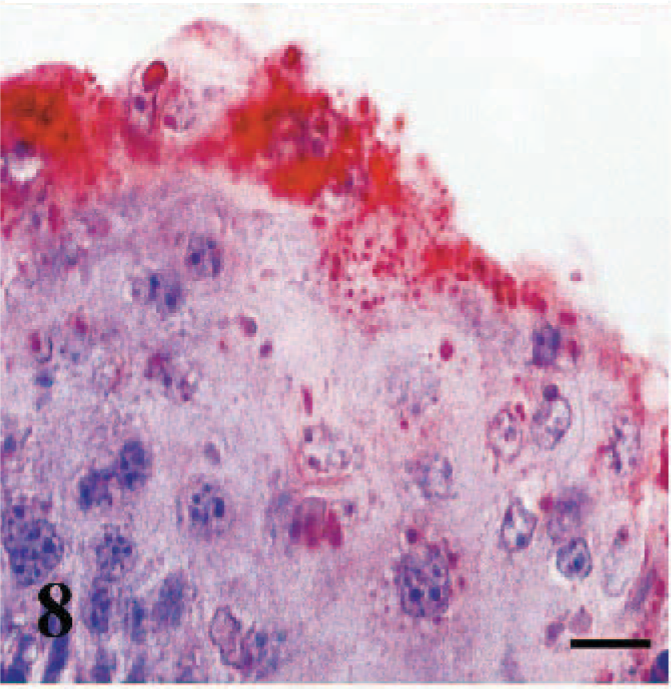
Oral mucosa, sheep. Cytoplasmic granular immunostaining. The density of the reaction is much higher in the surface epithelium. ABC technique, Mayer's hematoxylin counterstain. Bar = 20 μm.
Immunoreactivity was present in pseudostratified ciliated columnar epithelial cells lining the conchae and tracheal mucosa (Fig. 9), epithelia of the proprial mucous glands, and secretory ducts. In the lung, widespread immunostaining was evident in alveolar macrophages and bronchial/bronchiolar epithelial cells, but immunostaining in alveolar syncytial cells was less intense.
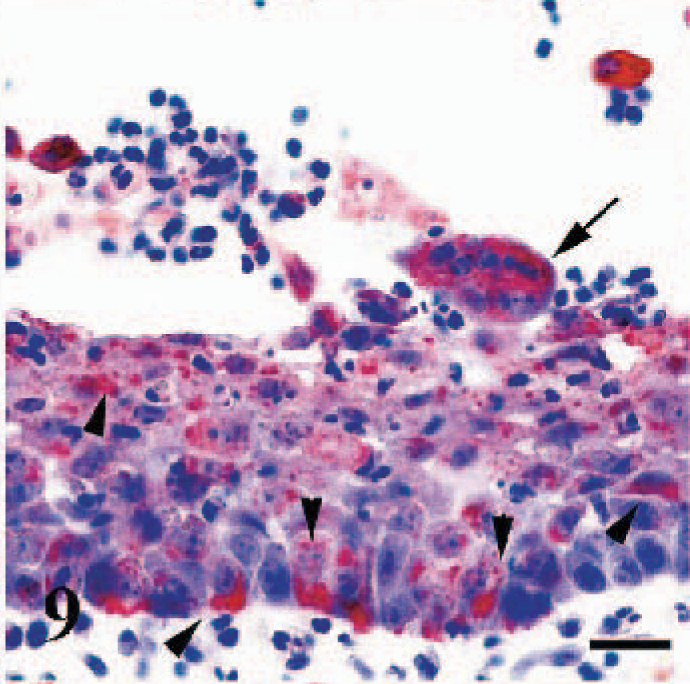
Trachea, goat. Cytoplasmic immunoreactivity in the mucosal epithelium (arrowheads) and in a syncytial cell (arrow). Anti-RPV antibody, ABC technique, Mayer's hematoxylin counterstain. Bar = 30 μm.
In the intestines, immunolabeling was detected in Lieberkuhn crypt cells, necrotic material deposited in the lumen of cystic glands, and remnants of necrotic lymphocytes in Peyer's patches and in some in syncytial cells. Diffuse epithelial staining of the ruminal papillae was seen in 3 cases (case Nos. 1, 2, 5) and in the abomasal mucosa of 1 case (case No. 2).
Spleenic and lymph node immunoreactivity was detected in sinusoidal macrophages, necrotic lymphoid follicles, and syncytial cells. Tonsillar crypt epithelium and necrotic material in the crypt lumens were intensely immunopositive; however, the lymphoid tissue stained weakly. Areas of coagulation necrosis in the liver exhibited a granular pattern of immunostaining. The immunoreactivity in the adjacent hepatocytes and syncytial cells was homogenously localized in nuclei (Fig. 10) and cytoplasm (case Nos. 1, 2, 11, 13, 17, 19, 21). Biliary duct epithelium was also immunopositive (case No. 11).
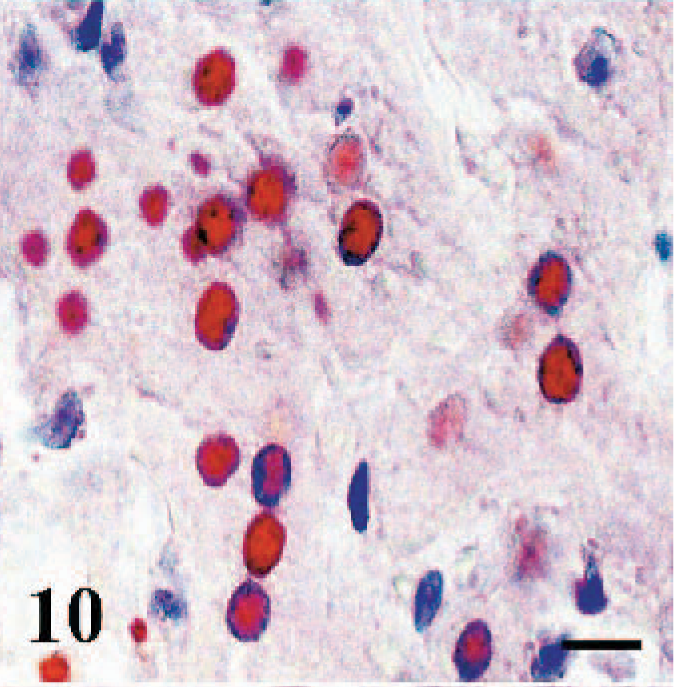
Liver, sheep. Immunopositivity in the nuclei of hepatocytes. Anti-RPV antibody, ABC technique, Mayer's hematoxylin counterstain. Bar = 25 μm.
Immunopositive staining was detected in the epithelium of the renal pelvis (Fig. 11) (case Nos. 2, 4), parietal layer of Bowman's capsule, and proximal tubules (Fig. 12) (case Nos. 4, 7). In addition, endothelial cells of cortical vessels (case Nos. 4, 6) and numerous perivascular macrophages (case No. 4) showed immunoreactivity.
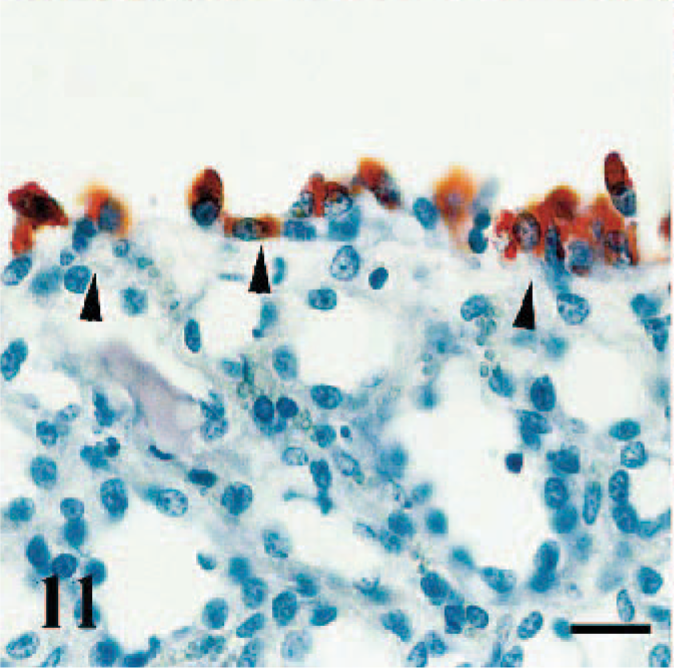
Kidney, goat. Renal pelvis transitional epithelium exhibit immunoperoxidase positive staining in the cytoplasms (arrowheads). Anti-RPV antibody, ABC technique, Mayer's hematoxylin counterstain. Bar = 40 μm.
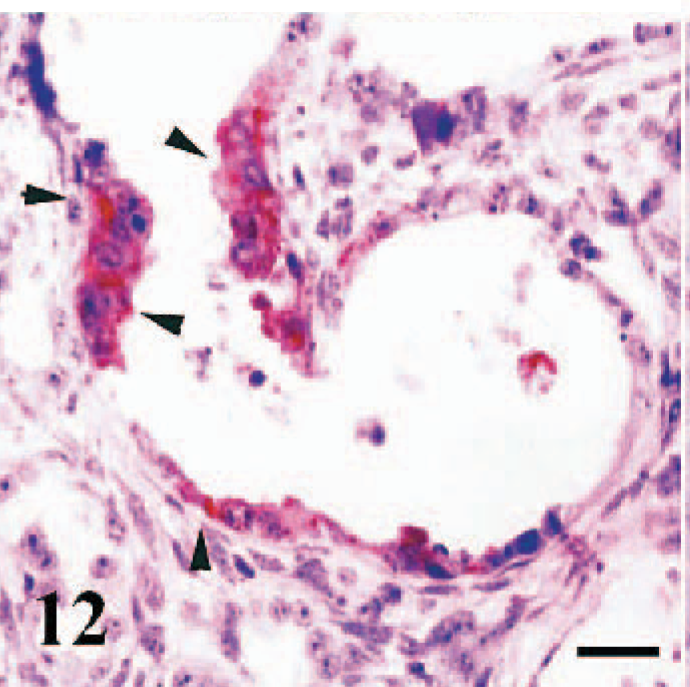
Kidney, sheep fetus. Strong immunopositivity in hyperplasic epithelial cells lining a cystic proximal tubule (arrowheads). Anti-RPV antibody, ABC technique, Mayer's hematoxylin counterstain. Bar = 40 μm.
Ependymal cells of the rostral colliculi and meningeal macrophages were immunopositive in a case (case No. 19). Three animals exhibited interstitial myocarditis (case Nos. 17–19), and myocytes were immunopositive in a granular and linear patterns (Fig. 13). There were no immunostaining in the negative control lung sections obtained from 2 healthy lambs.
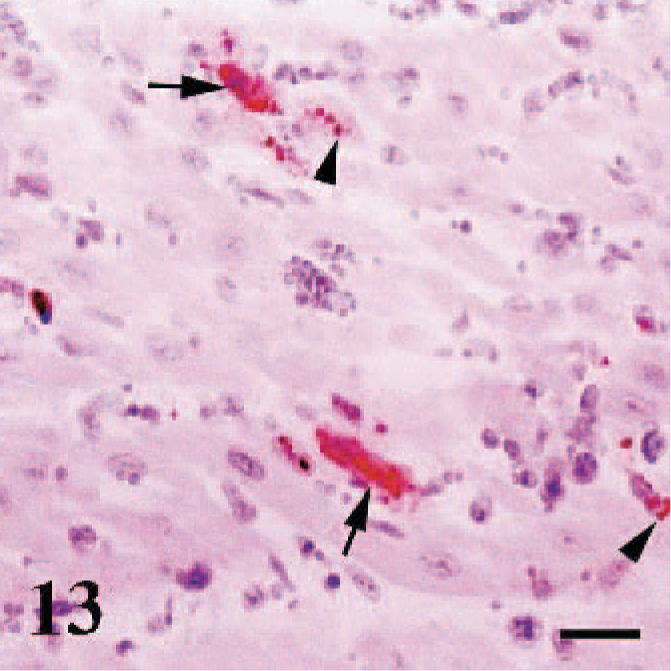
Heart, sheep. Granular (arrowheads) and linear staining pattern in some myocytes (arrows). Anti-RPV antibody, ABC technique, Mayer's hematoxylin counterstain. Bar = 40 μm.
Virology
Positive RT-PCR results, indicated by amplification of a 448-bp genome fragment, were obtained in 18 cases (case Nos. 1–4, 6–9, 11–14, 16–21) (18/21, 85.7%). Nucleotide analysis of the isolates revealed a high homology between the viruses. On the other hand, 2 stable mutations at positions of 5999 (G→A) and 6083 (C→T) of respecting local reference virus of PPR (Gen Bank Acc. No: AJ849363) were found in all viruses detected in cases (data not shown). However, putative aminoacid sequence showed no change in its composition, indicating antigenic stability for at least mentioned F protein. Phylogenetic analysis showed that the viruses are closely related not only to each other but also to the local reference virus (Fig. 14), and they all belong to the lineage 4 in PPRV common phylogenetic tree.
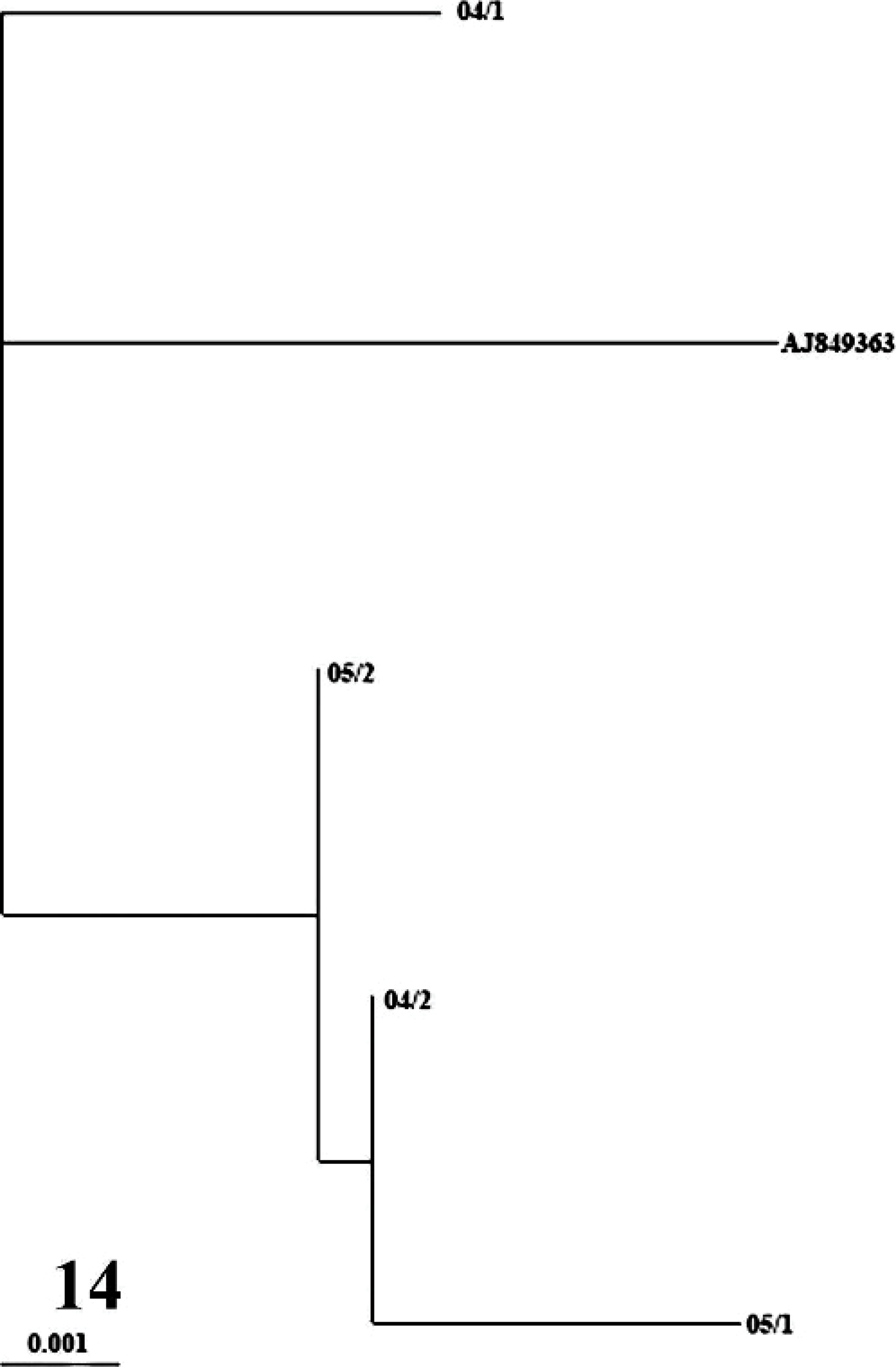
Phylogenetic relationship between local PPR virus isolates and ancestor PPR virus (GenBank Accession Number AJ849363) isolated in Turkey in 2000. The tree drawn with Treeview (Page, 1996) is based on partial sequence data from the fusion (F) protein gene and was derived by using the PHYLIP DNADIST and FITCH programs (Felsenstein, 1989). Branch lengths are proportional to the genetic distances between viruses and the hypothetical common ancestor at the nodes in the tree. The bar represents nucleotide substitutions per position.
Discussion
Peste des petits ruminants virus infection has been one of the most prevalent infectious diseases among small ruminants in Turkey for 10 years and has been acknowledged as an emerging disease with an increasing number of outbreaks from the different regions. 23, 24, 32, 34 In this study, the first PPR outbreak in Kirikkale Province was detailed. First, we found that PPRV infection was more prevalent in sheep (15/21, 71.4%) than goats. This finding seems contrary to phylogenetic information obtained from a previous field study. 24 The PPRV strains in this study were identified as lineage 4, which is more pathogenic in goats. 24
It has been almost 6 years since the first detailed genetic analysis of the local PPRV Turkey 2000 (Tu00) that indicated the virus is closely related to those in lineage 4. 24 The full genome sequence of Tu00 has recently been documented in detail by Bailey et al. 5 The results of the present study showed a high genetic homology to the local reference virus (Tu00) and an antigenic stability for at least the corresponding region of F protein.
Peste des petits ruminants virus infection resembles rinderpest in terms of clinicopathologic findings and pathogenesis. However, lung lesions resulting from PPRV infection differ from those resulting from rinderpest. 6 According to reports on the mechanisms of the disease, the virus enters through nasopharyngeal mucosa and localizes to and replicates in the tonsils and regional lymph nodes. 6, 12, 27 After a viremic stage, virus can be identified in most lymphoid tissues, alimentary and respiratory mucosa, and it subsequently causes pneumoenteritis. 6, 12, 32 In the present report, characteristic erosive ulcerative lesions that include sincytial cells and inclusions in the alimentary and respiratory mucosa and bronchointerstitial pneumonia reveal affinity of the PPRV to the epithelial cells. Peste des petits ruminants virus can cause severe lymphocytolysis in lymphoid tissues, such as tonsils, spleen, Peyer's patches, and mediastinal and mesenterial lymph nodes, and a subsequent immunodeficiency, to lymphoid depletion. 6, 12, 21, 28 We observed conspicuous pathology in Peyer's patches in 4 cases, lymph nodes in 7 cases, and spleen in 5 cases, which was characterized by lymphoid depletion and apparent reticular cells.
This study revealed a novel localization of the PPRV in the kidney, brain, rumen, abomasum, heart, and tongue muscle. In the kidney, the presence of immunopositive staining in the cortical vessels (case Nos. 4, 6), adjacent proximal tubules (case Nos. 4, 7), and transtitional epithelia of renal pelvis (case Nos. 2, 4) suggested that the virus might have reached to the kidney via the bloodstream, passed through glomerular filtrate, and been excreted in the urine. Immunoperoxidase positive staining for PPR in the renal pelvis in 2 cases is comparable to urinary system localization of the other morbillivirus infections (e.g., canine distemper, distemper in seals). 19, 20, 29, 33 Such affinity is important because the virus may pass into the urine at a higher concentration and subsequently can be excreted at a significant amount via urine and consequently contaminate the environment. 29
Neurovirulence of the morbilliviruses, including canine distemper virus and phocine distemper virus, is described in detail. 11, 19, 20, 33 All morbilliviruses can infect the central nervous system, but some morbilliviruses are easily eliminated by the developing host immunity, and temporary central nervous system infection occurs. 11, 16 The entrance of the morbilliviruses into any cell is dependent on the immune status of the host and virus-specific receptors, such as membrane cofactor protein CD46. 15, 16 However, rinderpest virus and PPRV neurovirulence still remains an obscure entity because of insufficient data available. Galbraith et al. 16 reported that rinderpest virus (Saudi/81) and PPRV (Nigeria 75/1) showed neurovirulence when the viruses were experimentally inoculated into the mouse brain. In our study, we detected the viral antigens exclusively in meningeal macrophages and ependymal cells. This is an interesting finding because the presence of the virus in the ependymal cells may indicate a potential of the viruses to pass into the cerebrospinal fluid. We could not draw conclusions on the presence of the PPRV neurovirulence in natural cases because of limited information obtained from only 1 animal and the absence of the neuronal localization of the virus.
Intersititial myocarditis, hyalinization of the myocytes, and immunopositivity in the heart in 3 cases in the present study are considered novel pathologic features of PPRV infection. Similar nonspecific heart lesions, such as hemorrhages and mild myocardial degenerations, are also reported for rinderpest. 6 Perhaps the unusual virus localization in myocytes resulted from the prolonged period of the infection, and it might have been due to high cellular tropism of PPRV.
We did observe dense immunopositivity in the abomasum in 1 case (case No. 2) and rumen mucosa in 3 cases (case Nos. 1, 2, 5) that were similar to well-known gastric mucosal lesions of canine distemper. 33
In conclusion, uncommon and novel pathologic findings in the kidney, brain, rumen, and abomasum are similar to those resulting from other morbillivirus infections. Phylogenetic analysis showed that the virus belongs to lineage 4 in the PPRV common phylogenetic tree.
Footnotes
Acknowledgement
This work was supported by grants from the Kirikkale University, Scientific Research Council (Project no: 2005/15). We thank Associate Professor Nihat Toplu from Adnan Menderes University, Faculty of Veterinary Medicine, for supplying rinderpest virus antibody and Dr. Siyami KARAHAN from Kirikkale University, Faculty of Veterinary Medicine, for linguistic help.
