Abstract
Peliosis hepatis is a vasculoproliferative disorder of the liver with infectious and noninfectious causes. In humans and dogs, Bartonella henselae has been linked to peliosis hepatis. Although domestic cats are the natural reservoir of B. henselae and although peliosis hepatis is common in this species, an association between this condition and infection with B. henselae has never been investigated in cats. In this study, 26 cases of peliosis hepatis in cats were tested for B. henselae infection by nested polymerase chain reaction and immunohistochemistry. The authors failed to detect B. henselae nucleic acid or antigen in any of the affected liver specimens. These findings suggest that, unlike in humans and dogs, peliosis hepatis in cats may not be significantly associated with a B. henselae infection.
Peliosis hepatis is a vasculoproliferative disorder characterized by blood-filled cystic structures in the liver parenchyma, often lined by prominent endothelium. In humans, the condition is associated with both noninfectious causes (such as malignant tumors and certain pharmaceuticals) and infectious causes (primarily Bartonella henselae infection). 10 There has been one report linking B. henselae to peliosis hepatis in a dog. 4 The pathogenesis of peliosis hepatis is still under investigation, but there is evidence of an association with high levels of vascular endothelial growth factor. 12 Vascular endothelial growth factor and other angiogenic growth factors, such as IL-8, are triggered by the proangiogenic hypoxia-inducible factor 1, which has been shown to be activated by Bartonella adhesin A–expressing B. henselae. 3 Among domestic cats (the natural reservoir for B. henselae), peliosis hepatis is a relatively common condition. 5 However, an association of B. henselae infection with peliosis hepatis has not been addressed before in this species. In this study, we hypothesized that B. henselae infection may also cause peliosis hepatis in cats, similar to the situation in humans and dogs.
Material and Methods
Twenty-six cats with peliosis hepatis, ranging from 2 to 20 years of age, were examined for the presence of B. henselae by immunohistochemistry and polymerase chain reaction (PCR). Cats were necropsied, and histopathology was performed on all relevant organ systems using formalin-fixed, paraffin-embedded tissue specimens of 4-µm sections and hematoxylin and eosin stains. For immunohistochemistry, a labeled streptavidin–biotin–peroxidase system (Vectastain Elite ABC-Kit, Vector Laboratories Inc., Burlingame, CA) was used with 3′3′-diaminobenzidine tetrahydrochloride as chromogen (DAB buffer tablets, Merck, Darmstadt, Germany). Endogenous peroxidase was blocked with methanol containing 3% hydrogen peroxide. Antigen retrieval was performed in 0.1% pronase E (AppliChem GmbH, Darmstadt, Germany) in phosphate buffered saline for 12 minutes at 37°C. Unspecific binding was blocked by 20% normal goat serum and 0.2% bovine serum albumin for 15 min at room temperature. B. henselae–specific affinity-purified rabbit immunoglobulin was used as primary antibody, diluted 1:5,000. 8 Biotinylated goat anti-rabbit immunoglobulin G (IgG) was used as secondary antibody, diluted 1:200 (Vector Laboratories Inc.). Vero cells infected with B. henselae were used as positive controls, whereas uninfected Vero cells served as negative controls. Cells were embedded in agar, formalin fixed, and embedded in paraffin. Tissue sections from 2 formalin-fixed, paraffin-embedded lymph nodes obtained from human patients with cat scratch disease were used as additional control (kindly provided by Christoph Thorns, Luebeck, Germany).
Both lymph node specimens tested positive for B. henselae DNA by nested PCR targeting the htrA gene, as described below. Negative controls included affinity-purified rabbit IgG directed against ec10r, an irrelevant antigen (Eurogentec, Seraing, Belgium). For PCR detection of B. henselae, 2 nested and 1 standard assay targeting 3 genes were used. All assays are employed routinely in our laboratories to successfully amplify B. henselae DNA from fresh, frozen, or formalin-fixed, paraffin-embedded tissues. Table 1 summarizes primers and annealing temperature. DNA was extracted from three 8-µm paraffin sections per sample using a commercial kit (QiaAmp DNA Mini Kit, Qiagen, Hilden, Germany) after deparaffinization with xylene. First a genus-specific PCR for pap31 of Bartonella spp. was employed. For each sample, 10 µl of template DNA were amplified in a 50-µl reaction mixture containing 1X GoTaq Flexi Buffer (Promega, Madison, Wisconsin), 10 mM of each dNTP (dNTP-Mix, Fermentas, St.Leon-Roth, Germany), 1 mM MgCl2 (Promega), 1 U GoTaq Flexi Polymerase (Promega), and 12.5 pmol of each primer. PCR was carried out with 5 minutes at 94°C, followed by 35 cycles of 30 seconds at 94°C, 30 seconds at 58°C, 90 seconds at 72°C, and 10 minutes at 72°C. The limit of detection was approximately 10 colony-forming units. The integrity of DNA was verified for each sample by amplification of a 68–base pair (68-bp) EF-1 alpha fragment localized between nucleotides 1,007 and 1,074 of the feline EF-1 alpha gene (GenBank No. AY712790), using primers 5′-caa aaa tga ccc acc aat gg-3′ and 5′-ggc ctg gat ggt tca gga ta-3′. Cycling conditions were the same as described, except for an annealing temperature of 61°C. Results were confirmed by a species-specific nested PCR targeting the heat-shock protein (htrA) gene of B. henselae (GenBank No. L20127). Primers for this assay were designed using the Primer3 software (http://biotools.umassmed.edu/bioapps/primer3_www.cgi). Primer htrA3+ was adapted from a primer previously described. 1 Amplification was performed as described above, except for 100 pmol for each primer and 2 mM MgCl2. Two separate regions on the htrA gene were targeted by this assay. The first region, amplified by htrA3+ and htrA4– as outer primers and htrA1+ and htrA2– as inner primers, was located between nucleotides 1,209 and 1,372 with amplicon lengths of 163 bp and 82 bp, respectively. The second was located between nucleotides 511 and 693 and targeted by htrA6+ and htrA5– as the outer pair and htrA7+ and htrA8– as the inner pair, with amplicon lengths of 182 bp and 93 bp, respectively. In the first amplification step (outer primer pairs), 10 µl of sample DNA was used as template. In the second amplification step (inner primer pairs), 1 µl of the undiluted PCR product from the first step was used as template. Cycling conditions were the same as described, with an annealing temperature of 58°C and with 25 cycles for the first amplification step and 35 cycles for the second. A further species-specific nested PCR targeting the 16S rDNA of B. henselae was performed as described by Dauga et al, 2 using the following conditions: 94°C for 2 minutes, followed by 35 cycles of 1 minute at 91°C (first amplification step) and 1 minute at 94°C (second amplification step), 1 minute at 62°C (first amplification step) and 1 minute at 65 °C (second amplification step), 1 minute at 72°C (first amplification step) and 5 minutes at 72°C (second amplification step). The detection limit of both nested PCR assays was determined to be approximately 1 colony-forming unit by serial dilution of culture-grown, heat-deactivated B. henselae. As positive controls, bacterial DNA from culture-grown hydrolyzed B. henselae strain Berlin-1 (kindly provided by Mardjan Arvand, Rostock, Germany) was used in all PCR assays. Negative controls in all amplifications were performed without template DNA.
Oligonucleotide Primers and Annealing Temperatures of Polymerase Chain Reaction Assays Used for Detection of Bartonella henselae
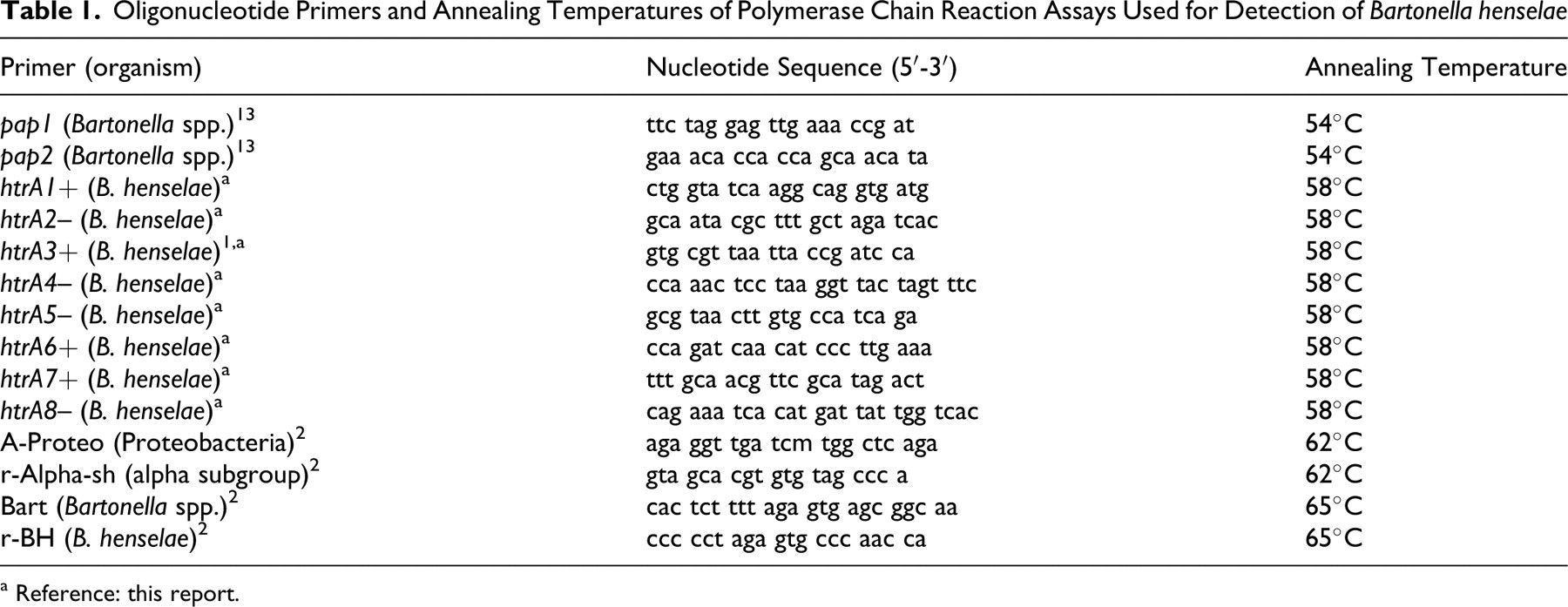
a Reference: this report.
Results
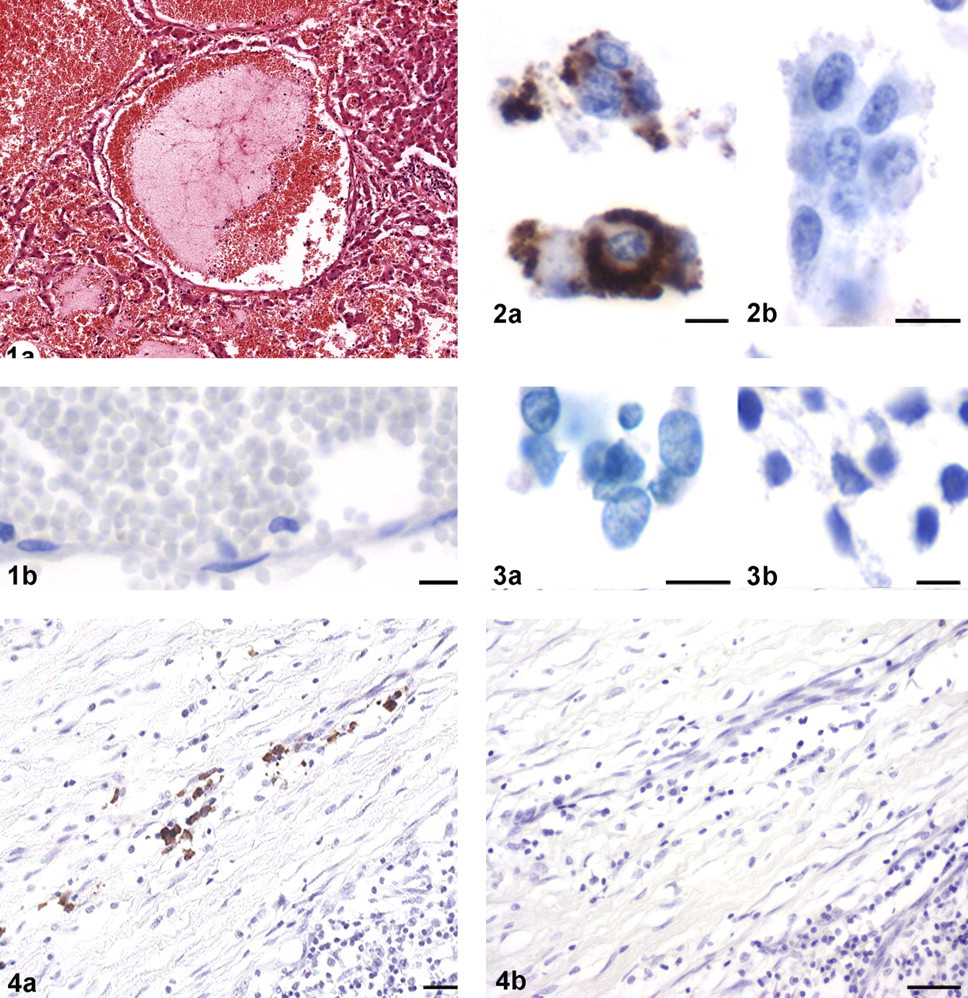
The median age of the 26 cats in this study was 15 years. Hypertrophic cardiomyopathy and neoplastic diseases were each identified in 6 cats; 2 additional cats had both. Three cats had massive intraabdominal hemorrhage with hypovolemic shock. Two of these cats had focal rupture of peliosis hepatis. One cat had pyogranulomatous polyserositis consistent with feline infectious peritonitis. Four cats had focal or extended inflammatory processes; 2 had miscellaneous conditions. In one case, no data were available on necropsy or clinical findings. All lesions of peliosis hepatis consisted of multifocal to coalescing, predominately subcapsularly located blood-filled cysts of up to 2 mm in diameter lined by endothelial cells (Figure 1a). In all specimens, immunohistochemistry failed to detect B. henselae antigen (Figure 1b). In contrast, all positive controls had strong specific signals using anti–Bartonella adhesin A rabbit IgG. B. henselae–infected Vero cells had strong, finely granular to coalescing cytoplasmic signals (Figure 2a), whereas no signals were observed using the irrelevant antibody (Figure 2b) or uninfected Vero cells (Figure 3). Sections from lymph nodes of 2 human patients with cat scratch disease had strong cytoplasmic signals within histiocytic cells (Figure 4). Correspondingly, B. henselae DNA was not amplified from any of the 26 specimens, neither in a genus-specific PCR targeting the pap31 gene or in 2 species-specific nested PCR assays targeting the htrA gene and the 16S rDNA gene, respectively. In contrast, EF-1 alpha was successfully amplified from all samples.
Discussion
Given that B. henselae has been associated with peliosis hepatis in human beings and dogs, we hypothesized that a similar association may exist in cats. However, both immunohistochemistry and PCR failed to detect B. henselae in any of the 26 specimens of feline peliosis hepatis. The reason for this unexpected result is unclear. Possibly, peliosis hepatis in cats may not be associated with B. henselae infection at all. Alternatively, different roles of B. henselae in this disease have to be considered in different species. For example, the absence of B. henselae in all feline specimens tested here may be linked to early clearance of the agent from the lesion by the host. This would clearly differ from the condition in humans where B. henselae is present in peliosis hepatis over prolonged periods of time. 6 These lesions resolve upon antimicrobial treatment effective against B. henselae, but several months of treatment are recommended for therapeutic success owing to frequently relapsing infections. 9 Based on the controls included here, false-negative results attributed to technical problems can largely be excluded. The anti–Bartonella adhesin A rabbit IgG, as directed against an adhesin of B. henselae present on the surface of early-passage B. henselae isolates, has been used to detect B. henselae by immunofluorescence, immunoblotting of patient sera, and immunoelectron microscopy. 7,11 Although it had never been used in an immunohistochemistry assay, it proved effective using slides from lymph nodes with B. henselae detection by PCR and formalin-fixed, paraffin-embedded agar pellets of B. henselae–infected Vero cells as positive controls. In conclusion, our studies did not identify an association between B. henselae infection and peliosis hepatis in cats, suggesting that cats have limited value as models to study the role of B. henselae in angioproliferative orders.
Footnotes
Acknowledgements
This study was supported by the H. Wilhelm Schaumann Foundation, Germany (A. U. Buchmann), and the DFG (V. A. J. Kempf). Selected tissue specimens were kindly provided by Paula Grest, Department of Veterinary Pathology, Zurich, Switzerland.
