Abstract
A 7-year-old spayed female schnauzer dog with chronic hematuria had a soft tissue mass within the wall of the bladder. The mass was excised and, when examined histologically, was determined to be a discrete well-organized mural mass consisting of spindloid cells arranged in swirling sheets and palisades punctuated by aggregates of principally eosinophils. The overlying transitional mucosa was extensively ulcerated and focally hyperplastic and nodular with subjacent solid down growths, superficial cysts, and the extension of tubular structures deep into the submucosa. The histologic appearance of this mass is consistent with canine polypoid eosinophilic cystitis, also known as benign inflammatory fibrous polyp.
A solitary ulcerated red polypoid intramural mass (2.3 × 1.5 cm) was visualized by ultrasound and surgically removed from the cranioventral fundus of the urinary bladder of a 7-year-old spayed female schnauzer dog, which had a 2-month history of bacterial cystitis and hematuria that was unresponsive to antibiotics. All clinical chemistries and blood values were within reference ranges. Sections of the mass (5µm thick) were prepared using standard methodology (formalin fixation and hematoxylin and eosin, van Gieson and trichrome staining).
Differential Diagnoses
Differential diagnoses for the urinary bladder mass included fibroma, leiomyoma, fibroleiomyoma, and inflammatory polyp.
Microscopic Findings
The examined sections of urinary bladder were extensively ulcerated. In nonulcerated areas, there was hyperplasia of transitional cells in various configurations (Fig. 1). These included solid down growths of epithelium, some of which were contiguous with the surface epithelium (Brunn nests), cystic structures closely apposed to the surface epithelium (cystitis cystica), and tubular and acinar structures composed of transitional epithelium extending deep into the submucosa (cystitis glandularis). The transitional cells forming these structures were columnar and stratified. The acinar structures were centrally lined by cells with proteinaceous content and cystic degeneration similar to areas of the hyperplastic bladder lining. Cell margins were generally distinct, and cytoplasm was fibrillar, amphophilic, and finely vacuolated. Desmosomal bridges were present. Nuclei were ovoid to polygonal and euchromatic with finely stippled chromatin and multiple, dense, and variably sized nucleoli. There were moderate numbers of necrotic/apoptotic cells. Anisokaryosis was moderate; giant syncytial cells were present; and mitotic activity was low.
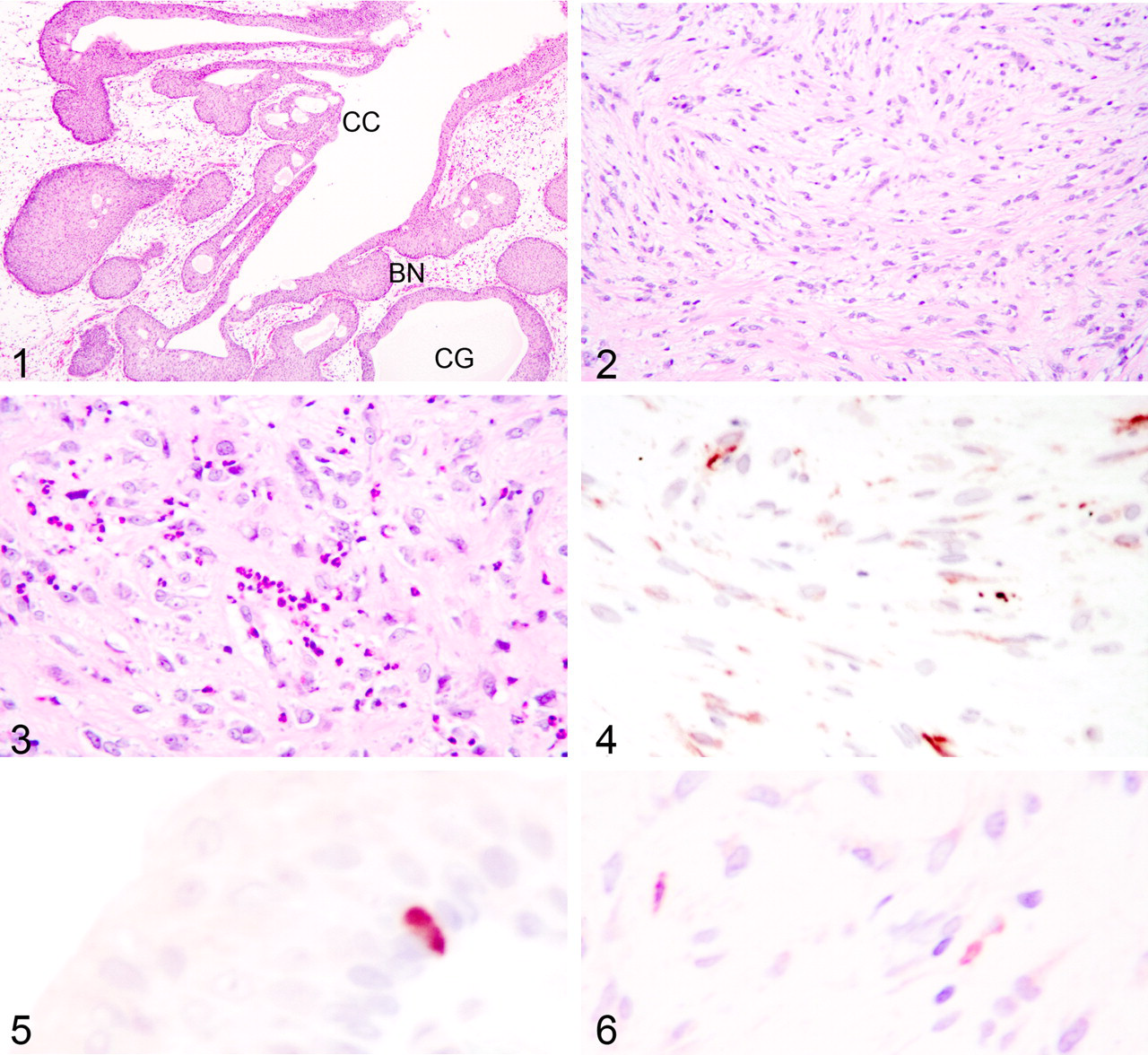
Extending from the ulcerated and hyperplastic surfaces (but not associated with all areas of transitional cell hyperplasia) and occupying the submucosa was a discrete unencapsulated mass that did not invade the tunica muscularis. The mass consisted of broad, interweaving, somewhat-areolar sheets and fascicles of spindloid cells (Fig. 2). Most cells were indistinctly bordered with fibrillar eosinophilic cytoplasm. Nuclei were irregularly ovoid with ruffled nuclear membranes and dispersed, dust-like to granular, chromatin, and they generally contained a single, prominent magenta nucleolus. Punctuating the mass were multiple loose aggregates of inflammatory cells, most of which were eosinophils (Fig. 3). Nodular aggregates of lymphocytes were present at the periphery of the mass. Van Gieson and trichrome staining of the spindloid cells delineated a mixture mainly composed of fibroblasts with fewer smooth muscle cells in addition to the van Gieson–positive neovasculature.
Immunohistochemistry
Unstained, paraffin-embedded bladder was sectioned at 3 to 5 µm and placed on positively charged glass slides. Immunohistochemical staining was performed using an automated staining device (Dako Autostainer, Dako, Carpinteria, CA) with Nova Red (Vector Labs, Burlingame, CA) as the chromogen and a hematoxylin counterstain. Antibodies tested were monoclonal mouse antihuman muscle actin (1:50), polyclonal rabbit antihuman chromogranin A (1:1000), mouse monoclonal antihuman cytokeratin (AE1–AE3; 1:200), rabbit polyclonal antibovine cytokeratin (wide spectrum screening [WSS]; 1:500), monoclonal mouse antiswine desmin (1:50), monoclonal mouse antirabbit sarcomeric actin (1:20), monoclonal mouse antihuman smooth muscle actin (1:300), polyclonal rabbit antihuman synaptophysin (1:200), and mouse monoclonal antibovine vimentin (1:100; all from Dako). High-temperature antigen retrieval 2 was employed for chromogranin A, synaptophysin, vimentin, desmin, and sarcomeric actin, whereas proteinase K retrieval (5 minutes) was an added step for muscle actin and both cytokeratins. Positive control tissue for chromogranin A and synaptophysin was normal canine adrenal medulla. Positive control tissues for the muscle antibodies were internal structures within the examined tissue as well as canine heart, canine intestine, and canine rhabdomyosarcoma. The positive control for vimentin and the cytokeratins was normal canine skin. The negative control for all tissues and antibodies was the universal negative control provided by Dako. With the exception of sarcomeric actin, which stains striated muscle, all immunohistochemical smooth muscle markers (muscle actin, desmin, 3 smooth muscle actin) and vimentin were positive to varying degrees in the cytoplasm of the spindloid cells composing the mass (Fig. 4), and the cytokeratin (AE1–AE3, WSS) and endocrine (chromogranin A, synaptophysin) markers (Fig. 5) were positive in the cytoplasm of the transitional epithelium. Additionally, nests of cells in the spindloid mass were positive for cytokeratin (WSS) (Fig. 6).
Diagnosis
The diagnosis was as follows: polypoid eosinophilic cystitis with Brunn nests, cystitis cystica and cystitis glandularis.
Discussion
In the current case, the presence of eosinophils—within an organized, noninvasive mass of spindloid cells that are immunohistochemically characterized as myofibrocytic or mixed fibrocytic and leiomyocytic—is highly suggestive of an inflammatory, rather than neoplastic, process. Four months postsurgical removal of the mass, the dog is free of clinical signs suggesting functional removal without regrowth of the mass. There are a variety of appellations applied to nonneoplastic nodular growths in the submucosa of the urinary bladder.
These include polypoid cystitis, eosinophilic cystitis, inflammatory pseudotumor, and pseudosarcomatous stromal reaction. 6,7,11,12 All appear to be a result of chronic irritation, potentially from primary cystitis, uroliths, drug reactions, or (rarely) catheterization, parasites, and autoimmune disease. 5,13 Polypoid cystitis in dogs appears to have a predilection for females and presents as macroscopic hematuria. 10 Most lesions are present in the cranioventral bladder wall.
The masses are single to multiple and may be polypoid, pedunculated, or nodular. In humans and animals, eosinophilic cystitis may occur without a proliferative connective tissue component. 4,13 In humans, 43% of these cases have peripheral eosinophilia. 14 Therefore, it appears that the term eosinophilic cystitis should be reserved for those cases without stromal proliferation. The presence of eosinophils in polypoid stromal lesions is often critical in differentiating inflammatory disease (polypoid cystitis) from neoplastic disease. There is speculation that urinary inflammatory polyps will regress if the inciting cause is removed. 1
In humans, the proliferating stromal cells may be immunohistochemically positive for antibodies to fibroblasts, smooth muscle markers, and occasionally cytokeratins. In many cases, mixed-cell populations of spindloid cells are present. 6 In the current case, a battery of immunohistochemical stains indicated cytoplasmic staining for smooth muscle, and vimentin were present with occasional cytokeratin-positive staining. Metaplastic changes in transitional epithelium, including Brunn nests, are common in humans and dogs.
Although they occur in association with inflammatory disease, up to 80% of normal canine bladders 9 and 93% of normal human bladders contain such lesions. 15 They may therefore be considered a variant of normal. There has been speculation over the years that Brunn nests, cystitis cystica, and cystitis glandularis are preneoplastic changes. 16 Evidence to support this hypothesis is weak in the human literature and nonexistent in the veterinary literature. Transitional cells in these lesions in humans are sometimes immunologically positive for endocrine/paracrine markers. 8 In this case, they were positive for both chromogranin A and synaptophysin, indicating that these cells in the dog are similar to those of humans and have an endocrine/paracrine function.
Footnotes
Acknowledgements
I thank Kay Fischer and the personnel of the histopathology section of the Veterinary Diagnostic Laboratory and Jill Bartlett for their excellent technical assistance. Dr Brett Hamilton is thanked for the clinical information.
The author declared no conflicts of interests with respect to the authorship or publication of this article.
The author declared no financial support for the research and/or authorship of this article.
