Abstract
Objective:
Investigate preferences of childhood cancer survivors (CCS) or of their caregivers (CG) about functionalities of a to be developed mobile app intended to improve compliance with a personalized survivorship care plan (Survivorship Passport) delivered according to the PanCareFollowUp model.
Study setting and design:
A 19-item Likert-type questionnaire was distributed by Childhood Cancer International among its affiliated associations. App functionalities were grouped into four modules (My Passport; My Hospital; Communication/Telehealth; My Health) and assigned to one of the Quality of cancer Survivorship Care Framework (QCSCF) domains.
Data source and analytic sample:
A decreasing score was given to each answer with “very useful” having the score of 4 and “not useful” the score of 1. Response data were reported with their mean (μ) score and values >3.5, between 3.2 - 3.5, 2.6 – 3.1 and <3.1 were defined as “very appreciated” “appreciated”, “fairly appreciated” and “not appreciated”, respectively.
Principal findings:
One hundred and fifty-four subjects (55% CG) completed the survey. Six functionalities were very appreciated and belonged either to the My Passport (n=3), My Hospital (n=2) or Communication/Telehealth (n=1) modules. Nine functionalities were appreciated (Communication/Telehealth n=6, My Passport n=3). Functionalities of the My Health module were the least appreciated. When functionalities were pooled according to the QCSCF, those pertinent to the health care delivery contextual domain were very appreciated, while those related to cancer and its treatment were not appreciated.
Conclusions:
This survey provided useful information which contributed to the development of the Cancer Survio Smart Card prototype by the SmartCARE consortium.
Introduction
The population of childhood and adolescent cancer survivors (CCS) has been increasing over time both in terms of absolute number as well as attained age. 1 Although epidemiological data on the total number of CCS living in Europe are not available yet, it is reasonable to believe that this population accounts for almost 500,000 individuals 1 and that their median age is between 25-29 years. 2
Compared to their peers in the general population, CCS are at higher risk of developing long-term health and psychosocial conditions resulting in excess morbidity and mortality.3-5 In this framework, personalized care and surveillance for early detection of treatable diseases is pivotal in preserving health and improving quality of life (QoL) of this fragile population.
The EU funded PanCareFollowUp project (PCFU - www.pancarefollowup.eu, grant number: 824982) was designed to improve the quality of life of CCS by bringing evidence-based, person-centered care into clinical practice.6,7 As part of the PCFU’s personalized approach, each survivor when entering the late effects clinic receives the PanCareFollowUp Survivorship Care Plan (PCFU-SCP), also known as Survivorship Passport (SurPass). This includes a Treatment Summary (TS), providing details on the cancer diagnosis and treatment history, and an individualized Survivorship Care Plan (SCP) 7 providing recommendations for long-term follow-up care based on internationally approved guidelines.8,9 These recommendations have been further converted into information summaries (educational brochures) meant to be Person-centered, written in Lay language, Accessible, Internationally relevant, and Navigable (PLAIN) 10 to be made available to CCS through the PanCare website (https://www.pancare.eu/plain-language-summaries/). Within the PCFU project, the individualized TS and SCP are being delivered in Italy using the digital Survivorship Passport (SurPass – v1.2) implemented in a secure platform accessible to survivors (www.survivorshippassport.org). 11
Nowadays the use of mobile apps has become more and more popular by providing quicker and more convenient access to services provided by web-based platforms. There are several apps dedicated to cancer patients available either on the Google Play or App Store, often developed by commercial organizations, but only few seem to have been downloaded in a significant number by intended users. 12 In a systematic review of manuscripts reporting on the impact and utility of such smartphone applications, Changrani et al. 13 concluded that they are particularly useful in supporting the transition between active treatment and follow-up care; however, the evaluation of their utility is limited because of the low number of randomized clinical trials (RCT).
Within the PCFU project we decided to launch an online questionnaire among European CCS and their caregivers (CG) to investigate their preferences regarding possible functionalities of a to-be-developed SurPass-specific mobile app.
Study setting and design
A 19-item Likert-type questionnaire, with one additional free text section (Online Supplementary Table 1, first column) was developed by a group of physicians, nurses, psychologists, and patient advocates participating in the PCFU project. The questionnaire aimed to investigate the preferences of CCS or their CG about the functionalities (F) of a to-be-developed SurPass mobile app, to help them in the management of their survivorship care. Respondents had to report their evaluation about usefulness (very useful, useful, fairly useful, not useful) of each of the 19 proposed functionalities (18 in the case of male survivors since F15 applied only to females). The last item (F20) allowed respondents to indicate in a free text format any other functionality they would suggest.
To facilitate the organization of the mobile app in modules, the functionalities were subsequently grouped in four categories: I) My Passport (six F); II) My Hospital (two F); III) Communication/Telehealth (seven F) and IV) My Health (four F) (Online Supplementary Table 1, second column). The free text answers (F20), if completed, were reviewed by one psychologist (S.O.) and one physician (R.T.) and grouped based on similarity to one of the four modules.
Each functionality was further categorized as pertinent to one of the Quality of Cancer Survivorship Care Framework (QCSCF) domains published by Nekhlyudov et al. 14 Among the individual factors included in the QCSCF, the authors identified five domains pertaining to Cancer and its Treatment: i) prevention and surveillance of recurrence and new cancers; ii) surveillance and management of physical effects; iii) surveillance and management of psychosocial effects; iv) surveillance and management of chronic medical conditions; v) health promotion and disease prevention, and four contextual domains of the Health-Care Delivery system that influence cancer survivorship care quality: i) clinical structure; ii) communication/decision making; iii) care coordination; and iv) patient/caregiver experience. As reported in Online Supplementary Table 1, third column, of the 19 functionalities included in our questionnaire, only four were considered pertinent to the Cancer and its Treatment domain while the remaining fifteen were considered pertinent to the Health Care Delivery contextual domain.
The SurPass App questionnaire was distributed by Childhood Cancer International Europe (CCI-E) among its affiliated survivors and/or parents associations along with an introductory letter explaining the objectives of the survey and the framework in which it was developed. The letter was shared with each association requesting the circulation of the questionnaire through the mailing list of its members who could contribute to the survey by clicking on a hyperlink to a dedicated Google platform. Respondents had to qualify themselves either as a CCS or a CG and were informed that the participation was on a voluntary and anonymous basis and that result will be used only in aggregate format. Those who participated in the survey were not directly involved in the PCFU trial. The consent to participate in the survey was considered as given by completing the questionnaire. No ethical approval was required because no personal data (e.g. identification code, original cancer type, country of residence, dates of birth and diagnosis) were requested.
The survey started on February 2022 and closed on August 2022 after two reminders.
Data source and analytic sample
A decreasing score was given to each possible answer with “very useful” having the higher score of 4 and “not useful” the score of 1. Response data were reported as absolute frequencies and percentages, with their mean (μ) and median (Me) score overall and by respondent type (CCS or CG). Since the obtained scores were generally high, for reporting purposes, four levels of appreciation were arbitrarily defined for functionalities as well for modules and domains, based on their mean score. For modules and domains this was a weighted mean score (
The Pearson’s chi-square test or Fisher’s exact test, were applied when appropriate to compare differences between respondent types (CCS vs. CG) in their appreciation of functionalities. A Student’s t-test was applied to compare difference between CCS and CG in the mean scores of each answer. All statistical tests were two-sided and a p-value <0.05 was considered significant. All analysis were performed using the statistical package Stata (version 13.0, Stata Corporation, College Station, TX).
Principal findings
The survey was completed by 154 subjects, of which 84 (55%) were CG and 70 (45%) CCS. The results on the usefulness evaluation of each app functionality are reported in Online Supplementary Table 2 stratified by respondent type. There were no significant differences (p value always >0.05, data not shown) between CCS and CG in the frequency and score values for all the proposed app functionalities; for this reason, results will be reported here all together independently of respondent type.
In Table 1 and Figure 1 Panel A, functionalities are listed ordered by their mean score (from very useful to not useful) and its standard deviation; color codes in Figure 1 report the frequencies (%) of levels of appreciation evaluation. The highest mean score was 3.7 (F2) and the lowest 2.9 (F15). Six functionalities were very appreciated. The first three functionalities: downloading the Portable Document Format (PDF) of the SurPass (F2), making queries about the personal treatment history (F18) and reading the last follow-up report (F3) belonged to the My Passport module and all had a μ score of 3.7. They were followed by two functionalities belonging to the My Hospital module: contact details of the late effects clinic (F5; μ 3.6) and of the hospital where treatment was given (F4; μ 3.6) and by one: alert the follow-up clinic in case of emergencies (F11; μ 3.6) belonging to the Communication/Telehealth module.
SurPass app functionalities evaluation by adult childhood cancer survivors or their caregivers.
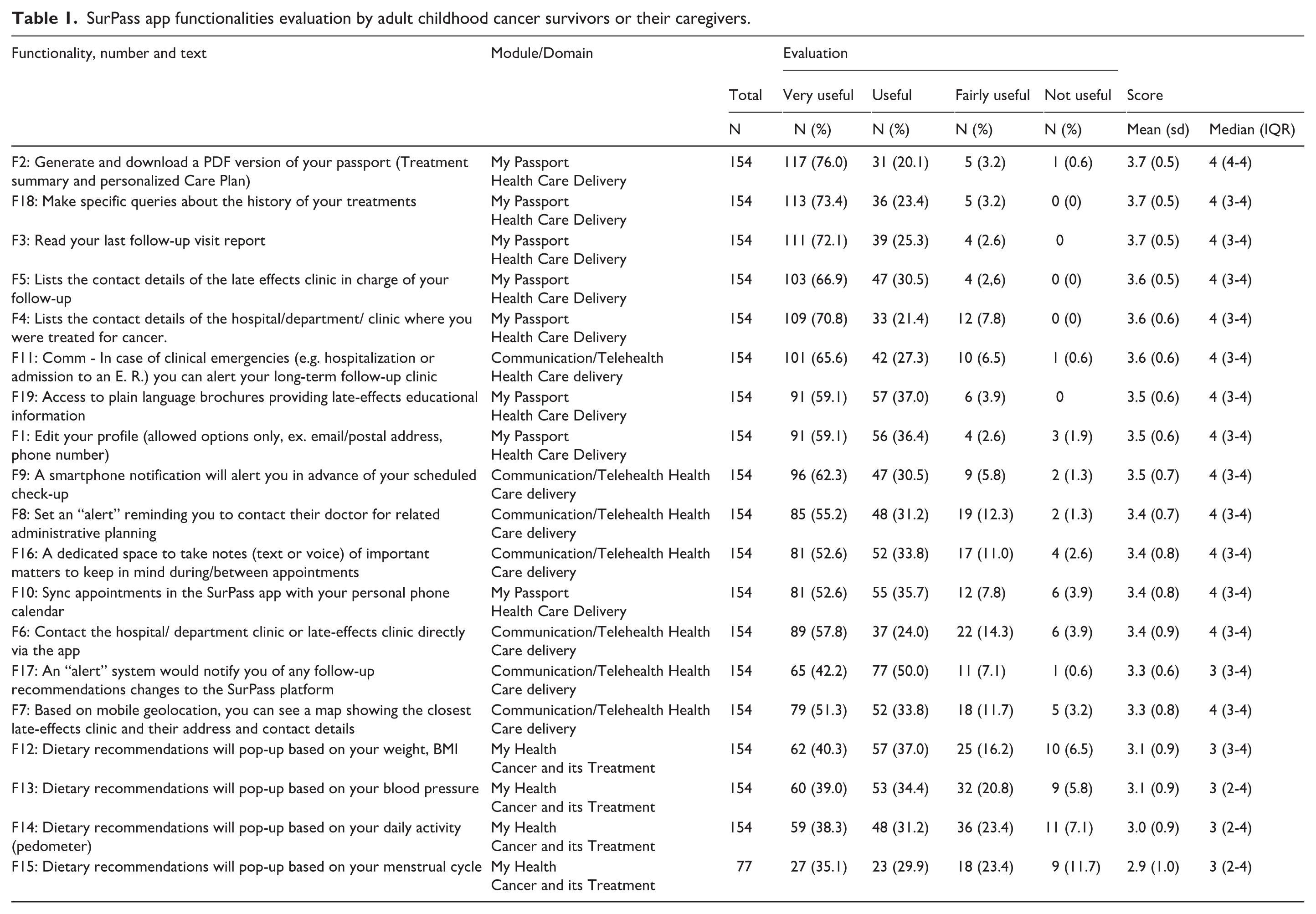
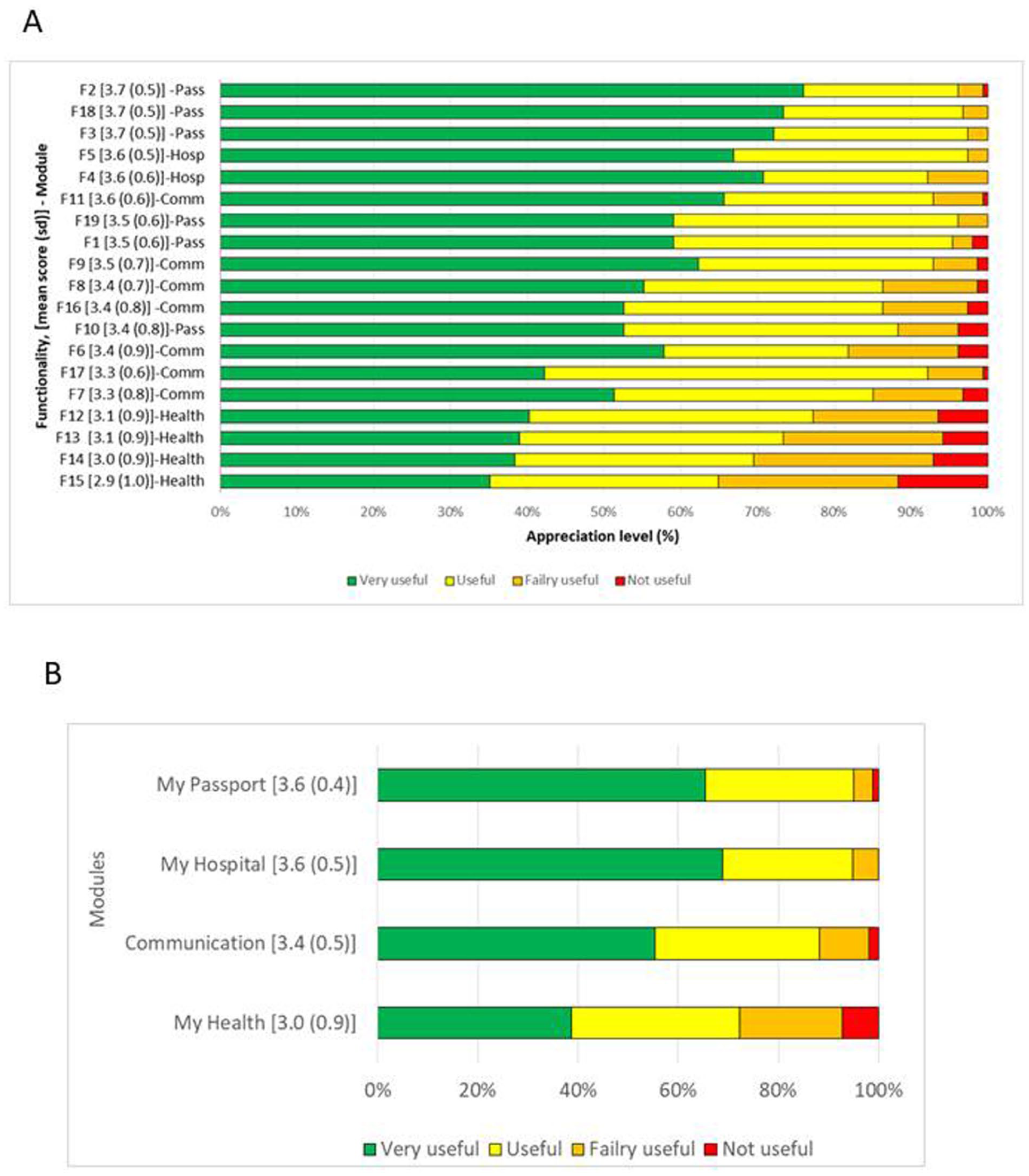
Appreciation scores of the SurPass App functionalities (A) and modules (B) as reported by Childhood Cancer Survivors and/or their Caregivers.
Nine functionalities were appreciated, having a μ score between 3.5 and 3.2. Three of them belonged to the My Passport module and six to the Communication/Telehealth one. In detail, access to plain language educational brochures (F19; μ 3.5), editing the profile (F1; μ 3.5) and synchronize the medical visit with the phone agenda (F10; μ 3.4) were pertinent to the My Passport module, while smartphone notification to remember scheduled check-up (F9; μ 3.5) and smartphone notification for administrative planning (F8; μ 3.4), dedicate space for taking notes on clinical issues (F16; μ 3.4), contact details of the late effects clinic (F6; μ 3.4), receive an alert in case of modification of any follow-up recommendation (F17; μ 3.3); geolocation of the closest late effects clinic when traveling (F7; μ 3.3) belonged to the Communication/Telehealth module.
The remaining four functionalities (dietary recommendations based on BMI (F12), on blood pressure (F13), on daily activity (F14) and menstrual cycle (F15)) were all part of the My Health module and were fairly appreciated or not appreciated (F15).
Figure 1 Panel B, reports on the mean appreciation scores of the App modules, which in Table 2 are also cross matched with those of the QCSCF domains after pooling each functionality to the pertinent ones. The My Hospital (
Cross tabulation reporting the mean scores of the mobile app modules and the Quality of Cancer Survivorship Care Framework domains.
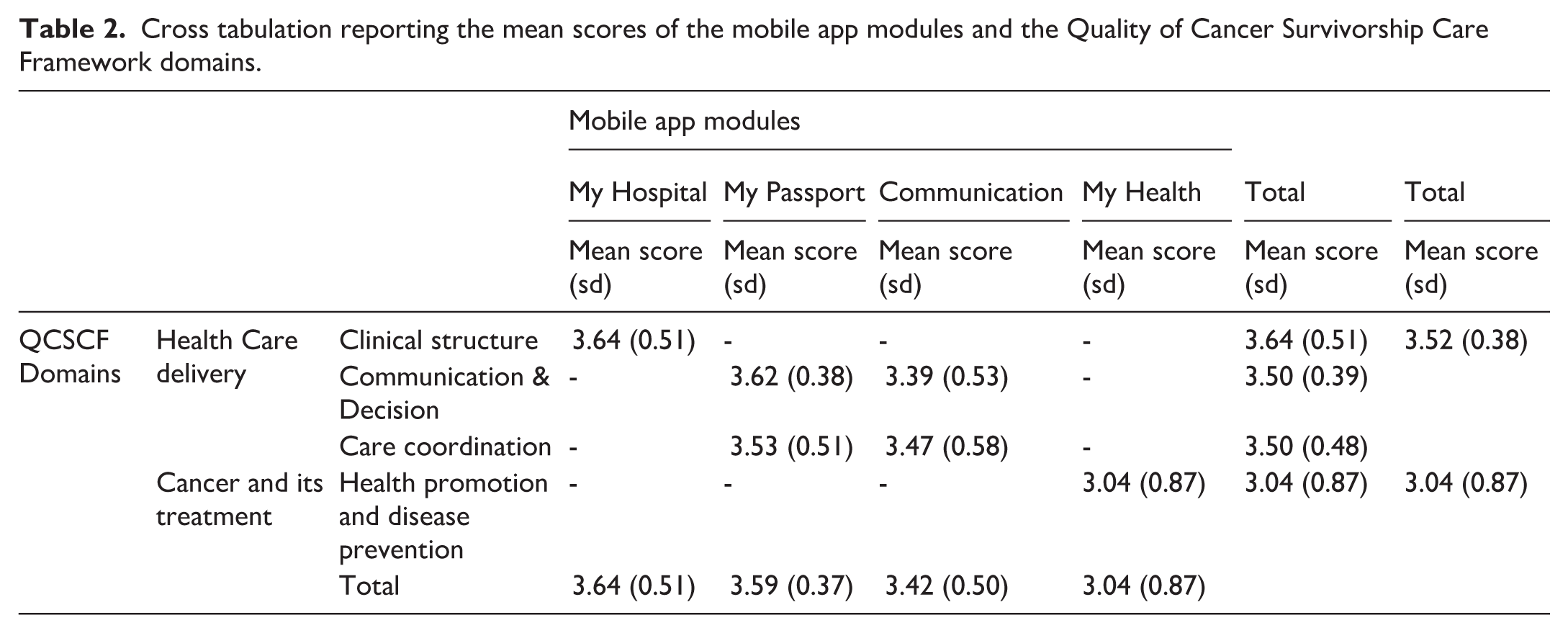
When Functionalities were pooled according to the QCSCF domains, those pertinent to the Health Care delivery contextual domain were very appreciated (
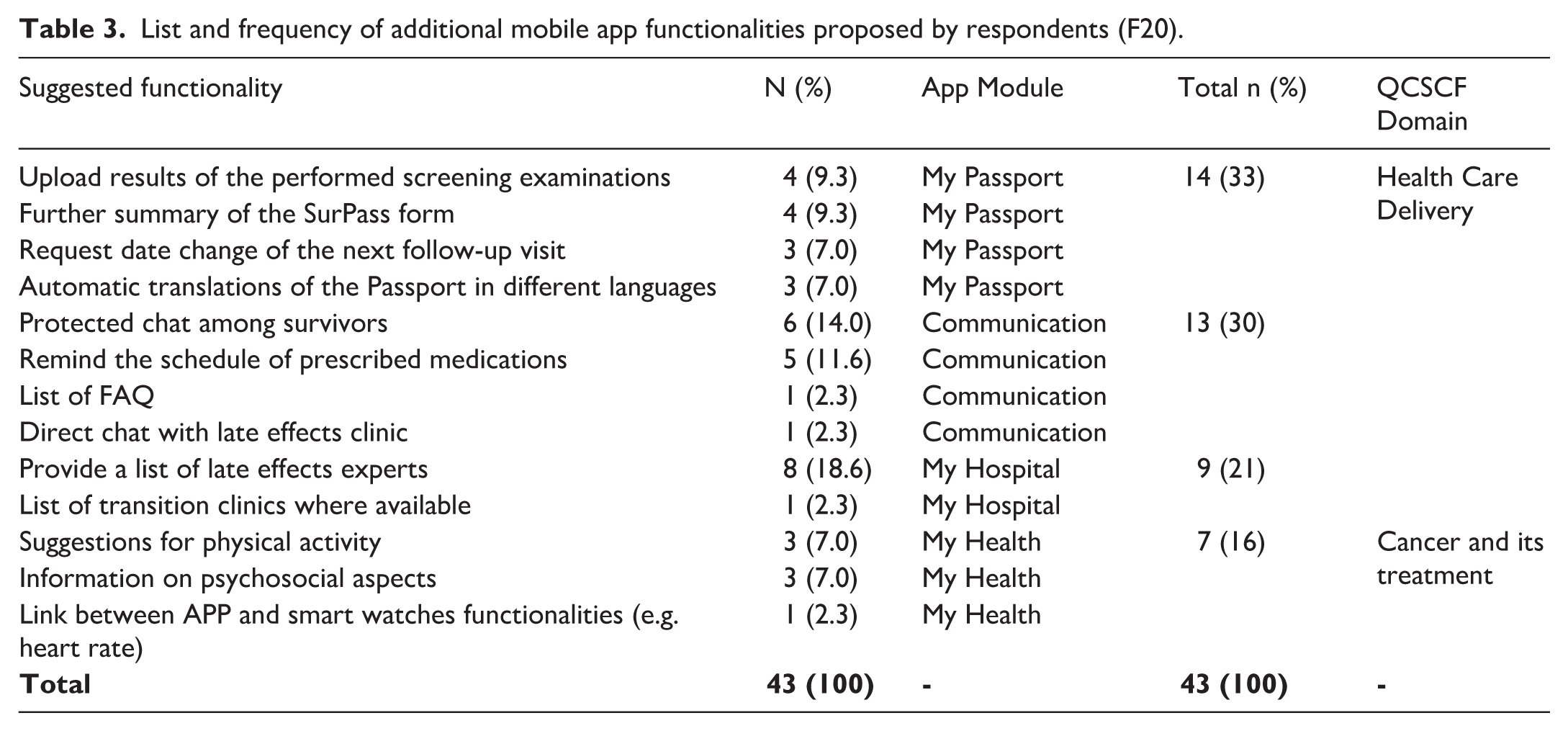
The free text question (F20) asking for any other suggested functionality was completed by 58 (38%) subjects. Of these, 15 (26%) expressed only positive comments about the survey and the possibility of accessing the SurPass tool. Table 3 reports on the functionalities suggested by the remaining 43 respondents with their corresponding SurPass app module and QCSCF domain. Overall, 14 (33%) suggestions were attributable to the My Passport module, 13 (30%) to the Communication, nine (21%) to the My Hospital and seven (16%) to the My Health one. If QCSCF domains were considered, 36 (84%) belonged to Health Care Delivery and seven (16%) to Cancer and its Treatment.
List and frequency of additional mobile app functionalities proposed by respondents (F20).
Conclusion
The PCFU project, in its commitment to promoting an evidence-based and person-centered approach, aimed to explore the interest of CCS and their CG in the potential development of a SurPass mobile app. By actively involving CCS and their CG in this process, this project tried to ensure that the resulting app will not only be relevant but also aligned with the lived experiences of its future users. Patient involvement is essential for creating tools that will likely be used and enhance both quality of life and long-term health outcomes of CCS and their CG. The survey was designed in the context of the PCFU care approach. 6 In this context, each survivor reaching the five year since diagnosis time point, while still in complete remission, receives the SurPass. 7 It includes the treatment summary and the long-term screening recommendations as agreed, after a shared decision with the late effects specialist at the moment of the SurPass delivery. Survivors are also informed about the possibility of accessing PLAIN language informational material on the PanCare website. 10 In our opinion, long-term screening according to SurPass recommendations should continue even after the critical transition period from pediatric- to adult- care, guaranteed by a late effects clinic in collaboration with the general practitioner and/or the local hospital. This approach is pivotal for successful long-term survivorship care. We believe that such approach addresses the five domains of cancer survivorship care pertaining to Cancer and its Treatment identified by Nekhlyudov et al. 14
The most appreciated modules and functionalities in our survey were those pertaining to the contextual domains of Health-Care Delivery that influence cancer survivorship care. This observation is further confirmed when the functionalities proposed by respondents (free text) are considered, which in most cases (36/43; 84%) pertained to the Health-Care Delivery contextual domain (Table 3).
In more detail, the My Hospital and the My Passport modules, which were the two most appreciated modules, offer the possibility of accessing the contact details of the late effects clinic as well of the treating hospital, together with the possibility of accessing the CCS-specific SurPass data (e.g. edit the personal profile, see the last follow-up visit summary; access to the educational brochures). The high scores of the functionalities included in these modules confirm the survivors’ request for a quick access to personalized information about their reference clinic, and their treatment history, as well as of their desire for guidance on self-care practices as reported by Knijnenburg et al. in a survey on CCS expectations of a website on late effects. 15 In the PCFU model, besides the shared decision process made together with the late effects specialist, the guidance on self-care is also provided by the condition-specific educational brochures which can be either downloaded from the PanCare website or be accessed by the SurPass app. 10
The third most appreciated module was Communication/telehealth. In this module functionalities are included which help facilitate a bilateral “conversation” between survivors and the late effects clinic allowing efficient communication regarding both changes in international guidelines and their corresponding algorithms, as well on survivor’s health status. For example, this module can help long-term follow-up clinics in efficiently informing survivors of the publication of any important new guideline or modification of old ones. This information is usually always available on the IGHG and PanCare websites, however any update directly available from the app might be more convenient to the survivors.
Another observation regarding the Communication/telehealth module is that two functionalities (F6 and 11) intended for survivors’ messages to the LTFU clinic either for urgent information (F11) or for consultation (F6) were “very appreciated” by CCS. However, their implementation in real life might be controversial as it would require clinics to be organized in a way that guarantees the prompt management of any alerts. This fact might include legal as well as ethical issues which should be carefully addressed before their real implementation.
Another appreciated functionality was the “mapping” of the closest late-effects clinic when traveling. In this context, the Survivorship Work Package 7 of the recently approved Joint Action on Network of Expertise (Jane-2 – grant N. 101183265) will provide an exhaustive map of the survivorship programs across the EU countries covering specificities related to each country, patient age class (pediatric vs. adult) and type of tumor. There should be no impediments to include such a map in the App.
Functionalities included in the My Health module were the least appreciated (μ 3.04). This is probably due to the fact that they referred to some basic tools which are often already implemented in almost all the mobile phones that can be purchased in the shops (e.g. pop-up notification remembering the schedule of prescribed drugs, of appointments and of daily physical activity). Instead, in our experience matured during several PanCare meetings and projects as the recently concluded smartCARE (grant N 101080048) focusing on the development of the prototype of the so called Cancer Survivor Smart Card, it is clear that many survivors would very much like an app in which everything is managed together, including functionalities focused on managing their health. Indeed survivors urgently need opportunities to take care of their health, obtain information and thus be at the center of their own care as facilitated with the person-centered approach of PCFU.
Similar to the SurPass, the Passport for Care (PFC) developed by Poplack et al. 16 is in use in several clinics affiliated with the Children's Oncology Group (COG) across the USA. The PFC can be generated, either by the CCS itself or by the HCP, through a dedicated website. Once created, it possible to access COG recommended follow-up screenings, access to Health Links (educational information for survivors), and a list of relevant guidelines. King et al, in a recent study investigating the opinion of clinicians using PFC, 17 reported their recommendation of developing a mobile health app to enable survivors to access and review their SCP on their personal device to augment survivor engagement. We believe that the functionalities of the SurPass App we investigated, in addition to aligning with the HCP recommendations reported by King et al., further provide services similar to those included in the Health Care Delivery contextual domain, which might further strength the “alliance for care” between survivors and their HCPs.
To our knowledge, there are two relevant studies addressing nature and scope, 12 or utility in improving quality of life, 13 of survivor-specific mobile apps. In more detail, in a scoping review on publicly available apps for cancer survivors downloadable either from Google Play or App Store, Adam et al. 12 identified 151 apps, most of which address individuals living with and beyond cancer in general (59%), or breast cancer (15%) or prostate cancer (6%). The review also reported that none of the Apps were focusing specifically on CCS except one addressing only retinoblastoma survivors. Most of the Apps were available from commercial or private organizations while only 27% were developed either by healthcare or academic organizations or clinical research societies. Features advertised by these Apps refer in most cases (63%) to imparting information about cancer, planning and organization of cancer care (56%) and to enacting management strategies (46%) or interacting with others (including others affected by cancer and healthcare professionals) (29%). Most statistics on their actual use or downloads are not always available and 43% of Apps had <100 downloads while only <9% had >5000 downloads. 12 Adam et al. concluded that most of the Apps that promise empowerment in the ‘fight’ against cancer are “tempered by the potential for exaggerated claims and exploitation”. 12
In another systematic review of manuscripts published in the period 2015-2021 on smartphone applications to support, educate and empower cancer survivors, Changrani et al. identified only 24 studies. 13 Among these 46% (n=11) focused on breast cancer survivors, 25% (n=6) on other specific tumors or treatments (head and neck, chronic myeloid leukemia, colon cancer, HSCT) and the remaining 29% (n=7) on combination of cancer types. For each of the 24 Apps, these authors identified which of the QCSCF domains of cancer survivorship care pertaining to cancer and its treatment 14 were addressed by each app. Most of them focused on health promotion and disease prevention and/or surveillance and management of physical or psychosocial effects. The domains of prevention and surveillance for cancer recurrence and new cancers were addressed in 5 Apps, and management of chronic medical conditions (after HSCT) was addressed in only one App. These authors concluded that mobile apps are both feasible and acceptable, in particular supporting the transition between active treatment and follow-up care. However, the evaluation of their utility is limited by the low number of randomized clinical trials and concluded that practical guidance to help survivors choose the appropriate App is needed.
We recognize some weaknesses of our study. Indeed, although our survey was meant to be distributed across Europe, due to privacy issues, it has not been possible to have exhaustive information about country of residence, gender of the CCS, type of the original tumor, age at diagnosis, and at interview, and type of survivorship care received by respondents. In any case, we believe that our survey was useful since its findings fed into the Cancer Survivor Smart Card (CSSC) prototype developed in the form of a Mobile App by the SmartCARE consortium (https://www.europeancancer.org/hpv/impact/smartcare.html) represented by health care professionals and patients, survivor and caregiver representatives. Details of the findings will be reported in the final Blueprint report, available on the website (https://www.europeancancer.org/eu-projects/impact/resource/lessons-learnt-next-steps.html). Functionalities most appreciated in our survey such as uploading and tracking medical information, telehealth and communication and useful informational resources were among the SmartCARE conclusions and recommendations for the CSSC.
Supplemental Material
sj-docx-1-tmj-10.1177_03008916261425443 – Supplemental material for Survey among childhood cancer survivors about their preferences on functionalities of a to-be-developed Survivorship Passport Mobile App
Supplemental material, sj-docx-1-tmj-10.1177_03008916261425443 for Survey among childhood cancer survivors about their preferences on functionalities of a to-be-developed Survivorship Passport Mobile App by Sara Oberti, Giorgia Manuzi, Martina Fragola, Hannah Gsell, Monica Muraca, Andrea Beccaria, Anita Kienesberger, Carina Schneider, Zuzana Tomasikova, Jack Latteur, Leontien C.M. Kremer, Riccardo Haupt and Ramona Tallone in Tumori Journal
Footnotes
Author contributions
All authors contributed to the conception and design of this study. Ethical approvals were not obtained because all the data were completely anonymous. Data were analyzed by Martina Fragola, Sara Oberti, Riccardo Haupt, Ramona Tallone and Hannah Gessel, and results were interpreted by all authors. The manuscript was drafted by Sara Oberti, Ricardo Haupt, Ramona Tallone Hannah Gessel, Jack Latteur and critically revised by all authors. All authors approved the final version of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Union’s Horizon 2020 research and innovation programme (grant number: 824982). The funding source was not involved in study design, data collection, data analysis and interpretation, writing of the manuscript, and the decision to submit this article for publication.
A.B., R.H. and M.M. were partially supported by the Associazione Oncologia Pediatrica e Neuroblastoma - OPEN OdV within the SurPass-DOPO project.
Members PanCare FollowUp Consortium
• Leontien C.M. Kremer, Helena J.H. van der Pal, Renee L. Mulder, Saskia M.F. Pluijm, Rebecca J. van Kalsbeek, Selina R. van den Oever, Jaap den Hartogh, Tessa Fuchs
Princess Maxima Center (PMC) for Paediatric Oncology, Utrecht, the Netherlands; Department of Paediatrics, Emma Children's Hospital, Amsterdam
• Lars Hjorth, Cecilia Follin, Lill Eriksson, Thomas Relander, Jacob Engellau, Karin Fjorden, Karolina Bogefors, Anna S. Holmqvist
Lund University, Skane University Hospital, Department of Clinical Sciences Lund, Paediatrics, Lund, Sweden
• Riccardo Haupt, Monica Muraca, Sara Oberti, Andrea Beccaria, Ramona Tallone, Brigitte Nicolas, Marina Benvenuto
DOPO Clinic, Department of Hematology/Oncology, IRCCS Istituto Giannina Gaslini, Genoa, Italy.
• Vera Araujo-Soares
Center for Preventive Medicine and Digital Health, Department for Prevention, Medical Faculty Mannheim, Heidelberg University, Mannheim, Germany
• Tomas Kepak, Katerina Kepakova, Hana Hrstkova, Viera Bajciova, Marta Holikova, Lucie Strublova
International Clinical Research Center, St. Anne's University Hospital Brno, Czech Republic.
• Anne Uyttebroeck, Sandra Jacobs, Heidi Segers, Marleen Renard, Monique van Helvoirt
Department of Paediatric Haematology and Oncology, University Hospitals Leuven, Leuven, Belgium
• Jeanette F. Winther. Line E. Frederiksen, Luzius Mader, Elisabeth A. W. Andersen
Childhood Cancer Research Group, Danish Cancer Society Research Center, Copenhagen, Denmark; Department of Clinical Medicine, Faculty of Health, Aarhus
• Gisela Michel, Stefan Boes, Katharina Roser
University of Lucerne, Faculty of Health Sciences and Medicine, Lucerne, Switzerland
• Jacqueline Loonen, Rosella Hermens, Irene Gottgens, Eline Bouwman, Iridi Stollman, Adriaan Penson, Dionne Breij
Radboud University Medical Center, Radboud Institute for Health Sciences, Department of Hematology, Nijmegen, the Netherlands
• Roderick Skinner
Newcastle University Centre for Cancer, Wolfson Childhood Cancer Research Centre, Newcastle upon Tyne, United Kingdom; Great North Children's Hospital, Royal Victoria Infrmary, Newcastle upon Tyne, United Kingdom; Translational and Clinical Research Institute, Wolfson Childhood Cancer Research Centre, Newcastle upon Tyne, NE1 7RU, United Kingdom
• Morven C. Brown
Population Health Sciences Institute, Newcastle University, Sir James Spence Institute, Royal Victoria Infrmary, Newcastle upon Tyne, United Kingdom; Newcastle University Centre for Cancer, Wolfson Childhood Cancer Research Centre, Herschel Building, Brewery Lane, Newcastle upon Tyne, NE1 7RU, United Kingdom
• Samira Essiaf, Anne Blondeel, William Sciberras, Giorgia Manuzi
European Society for Paediatric Oncology, Brussels, Belgium
• Joke Korevaar, Mieke Rijken
Netherlands Institute for Health Services Research (Nivel), Utrecht, the Netherlands
• Anita Kienesberger. Hannah Gsell Carina Schneider
Childhood Cancer International Europe, Vienna, Austria
• Edit Bardi, Jeroen te Dorsthorst
PanCare, Jacobus Bellamylaan 16, 1401 AZ Bussum, the Netherlands
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
