Abstract
Background
Multimorbidity is a concern for people living with cancer, as over 90% have at least one other condition. Multimorbidity complicates care coming from multiple providers who work within separate, siloed systems. Information describing high-risk and high-cost disease combinations has potential to improve the experience, outcome, and overall cost of care by informing comprehensive care management frameworks. This study aimed to identify disease combinations among people with cancer and other conditions, and to assess the health burden associated with those combinations to help healthcare providers more effectively prioritize and coordinate care.
Methods
We used a population-based retrospective cohort design including adults with a cancer diagnosis between March-2003 and April-2013, followed-up until March 2018. We used observed disease combinations defined by level of multimorbidity and partitive (k-means) clusters, ie groupings of similar diseases based on the prevalence of each condition. We assessed disease combination-associated health burden through health service utilization, including emergency department visits, primary care visits and hospital admissions during the follow-up period.
Results
549,248 adults were included in the study. Anxiety, diabetes mellitus, hypertension, and osteoarthritis co-occurred with cancer 1.1 to 5.3 times more often than expected by chance. Disease combinations varied by cancer type and age but were similar between sexes. The largest partitive cluster included cancer and anxiety, with at least 25% of individuals also having osteoarthritis. Cancer also tended to co-occur with hypertension (8.0%) or osteoarthritis (6.2%). There were differences between clusters in healthcare utilization, regardless of the number of disease combinations or clustering approach used.
Conclusion
Researchers, clinicians, policymakers, and other stakeholders can use the clustering information presented here to improve the healthcare system for people with cancer multimorbidity by developing cluster-specific care management and clinical guidelines for common disease combinations.
Introduction
Multimorbidity is the co-existence of two or more conditions, and rates of multimorbidity are high and increasing. In Ontario, Canada, for example, one in four people had multimorbidity in 2009, and since then, the prevalence of multimorbidity has increased in all population groups, including children and youth, as well as older men and women.1,2 Complexity within individuals is also increasing, with multimorbid patients increasingly presenting with three, four, or even five or more conditions. 2 Multimorbidity is particularly a concern for people with cancer, since more than 90% people with cancer have at least one other condition, and 37% have four or more conditions beside cancer. 3
Multimorbidity adds to the complexity of patient care 4 that creates challenges for individuals and the healthcare system, especially among cancer patients for whom prescriptions and recommendations come from multiple healthcare providers who are systemically disconnected from one another. In Ontario’s healthcare system, primary care and cancer specialty clinics are physically and administratively separate, and they lack resources to effectively coordinate or integrate cancer and primary care. 5 Indeed, most clinical treatment guidelines and management protocols are designed to address a single disease, making them a poor fit with cancer patients’ multiple, co-occurring conditions and wider range of needs.6-8 This single disease focus can negatively impact patients’ compliance with treatment (eg, if protocols are redundant, incompatible, or overly burdensome), and also responses to treatment.7,9 Research shows that patients with cancer and comorbidities, including mental illness, have higher use of services3,10,11 but also reduced survival.9,12,13
One way to improve the care provided to patients with cancer and multimorbid chronic conditions is for healthcare providers to attend to the impacts of specific co-occurring conditions. Specific combinations of multimorbid conditions differentially affect survival,12,14-16 suggesting that information about specific disease combinations may be useful for organizing and prioritizing aspects of care. Strategies to help healthcare providers prioritize multimorbidity among cancer patients is particularly important because cause of death among cancer patients is more likely to be other, non-cancer conditions than cancer itself.9,17 In order to do this effectively, healthcare providers and health systems may benefit from information describing common clusters of diseases1,18-20 that they can use to identify common risk factors and patient preferences for groups of individuals with the same multiple conditions, and then take steps to optimize and streamline multimorbidity-focused, patient-centred care.20,21 Such information could also be used to design comprehensive frameworks for care management of commonly occurring combinations of multiple chronic conditions.22,23 Healthcare providers who organize care according to individual patients’ specific combination of co-occurring conditions are also orienting towards providing patient-centered care, because the needs and desires of the patient — not healthcare system siloes 24 — guide the provision of services.
Thus, in order to inform improvements to care and care management for increasing populations of complex cancer patients, the aims of this study are to: 1-Identify the most common disease combinations as observed in individuals and grouping of diseases statistically most similar (statistical clustering), among people with cancer and one or more other condition (ie, complex cancer), overall and for specific cancer types; and 2-Assess the related health burden (ie, morbidity as indicated by health service utilization 25 ) of identified disease clusters within cancer patients. The findings from this study have potential to inform recommendations (eg risk prediction or stratification, medication conciliation, provider and patient awareness), for more effectively treating complex cancer patients in a person-centred way.
Materials and Methods
This population-based retrospective cohort study consisted of eligible OHIP (Ontario Health Insurance Plan, Ontario, Canada) patients over the age of 18 years, with a clinical diagnosis of cancer between March 2003 and April 2013. All eligible OHIP adults in the province were included in the study if they were diagnosed with cancer and at least one of 17 selected chronic conditions occurring before or up to 15 years after the original cancer diagnosis (ie follow-up until March 2018). Information on health services utilization was also obtained during the follow-up from administrative data. Those with missing age were not included.
Data were obtained through provincial health administrative databases and linked as described in our previous studies.2,3 The use of these provincial databases allows for the collection of medical information for the entire eligible provincial population.
In addition to cancer, 17 chronic conditions were identified (Supplementary Appendix 1) using hospital discharge (DAD), physician billings (OHIP) and prescription dispensing (ODB) data and included: acute myocardial infarction (AMI), asthma, cardiac arrhythmia, chronic obstructive pulmonary disease (COPD), congestive heart failure (CHF), chronic coronary syndrome, dementia, diabetes, hypertension, non-psychotic mood disorders, anxiety, other mental illnesses (including schizophrenia, delusions and other psychoses; personality disorders; and substance abuse), osteoarthritis, osteoporosis, renal failure, rheumatoid arthritis, stroke (excluding transient ischemic attack). 2
The study was approved by Lakehead University’s (Thunder Bay, Ontario, Canada) Research Ethics Board (#1466523). The study was first approved May 31, 2018; then renewed every year with current validity until March 2023. The need for consent to participate was waived by the REB for the use of secondary health administrative data, authorized by the Institute for Clinical and Evaluative Sciences (IC/ES) Privacy and Legal Office for external researchers, under section 45 of Ontario’s Personal Health Information Protection Act. IC/ES is a prescribed entity under section 45, authorized to collect personal health information, without consent, for the purpose of analysis or compiling statistical information with respect to the management of, evaluation or monitoring of the allocation of resources to or planning for all or part of the health system. All individual-level data were anonymized and only available to authorized researchers through a secure platform. We are reporting aggregated data that precludes identification of any individual person. The study is reported according to STROBE guidelines. 26
Measures of Complexity
As in our earlier papers, we described multimorbidity by level (ie, counts of 1, 2, 3, 4, or 5 (+) conditions besides cancer) as an indicator of patients’ complexity. We further examined complexity in two ways, using (1) observed disease combinations, within each level of multimorbidity, and (2) statistically-informed clustering of conditions, using a Partitive K-means approach. This approach maximizes homogeneity within clusters and heterogeneity between clusters thereby creating prototypes for succinctly describing large data sets. 27
Observed combinations were disease combinations with the highest prevalence. Combinations more prevalent than expected (ie with a high observed to expected prevalence ratio) were also assessed. We calculated the expected number of disease combinations and count of people in each combination, within each level of multimorbidity, based on the random chance of a given condition to co-occur with cancer and any of the 16 remaining conditions (Supplementary Appendix 2). Expected prevalence of each condition was then estimated accordingly ([study population in each multimorbidity level/expected number of combinations in that level]*number of possible combinations where the condition appears). Assuming that within multimorbidity level k, each condition has the same chance to occur with cancer, regardless of its real prevalence, the possible number of disease combinations would be n = C (k, 17). We assessed that in multimorbidity level 1, the number of combinations would be n = C (1,17) = 17 (ie If someone has 1 condition beside cancer, there would be 17 disease combinations by chance); in multimorbidity level 2, n = C (2, 17) = 136 combinations by chance; in multimorbidity level 3, n = C (3, 17) = 680; and in multimorbidity level 4, n = C (4, 17) = 2380.
Similarly, the number of combinations where a specific condition would be included by chance is: in multimorbidity level 1, n = [C (1, 17) – C (1, 16)] = 1 (ie Removing all combinations where the given condition is excluded); in multimorbidity level 2, n = [C (2, 17) – C (2, 16)] = 16; in multimorbidity level 3, n = [C (3, 17) – C (3, 16)] = 120; in multimorbidity level 4, n = [C (4, 17) – C (4, 16)] = 560.
Statistically-informed clusters were assessed using a Partitive (k-means) clustering algorithm accounting for the observed prevalence of each condition. Appropriate numbers of clusters were informed by preliminary hierarchical clustering with annual data (Supplementary Appendix 3).
Patient Outcomes
Health services utilization variables, including emergency department (ED) visits, primary care (PC) visits, and hospital admissions (HA) during the follow-up period after cancer diagnosis (up to 15 years), were used as proxies of patients’ outcomes or health burden; we did not focus on specific diagnosis or clinical manifestation for these visits. These variables were calculated per person-year to account for the different length of follow-up and/or death.
Analyses
Statistical analyses included a description of patients’ clinical and sociodemographic characteristics, multimorbidity level, and crude prevalence of each comorbid condition (overall, and by cancer type). For the complexity analysis, we reported the top 10 observed combinations by multimorbidity level, along with the ratio of observed-to-expected count of patients in each combination, and the number of combinations with high ratios (ie much more prevalent than expected). Then, we analyzed partitive clusters by age group or sex, using clustering algorithm based on the prevalence of each condition. Lastly, we analyzed bivariate associations between disease combinations or clusters and health services utilization (HSU) to support potential risk stratification. These statistics are appropriate for the design and we are sufficiently powered to enable reliable statistical results.
Our methodology, including study population, measures and analyses, may be reproduced by other researchers.
Results
Population Characteristics and Co-occurring Conditions
Characteristics of the Study Population of Patients with Cancer and Another Condition in Ontario, 2003-2013 (N = 549,248).
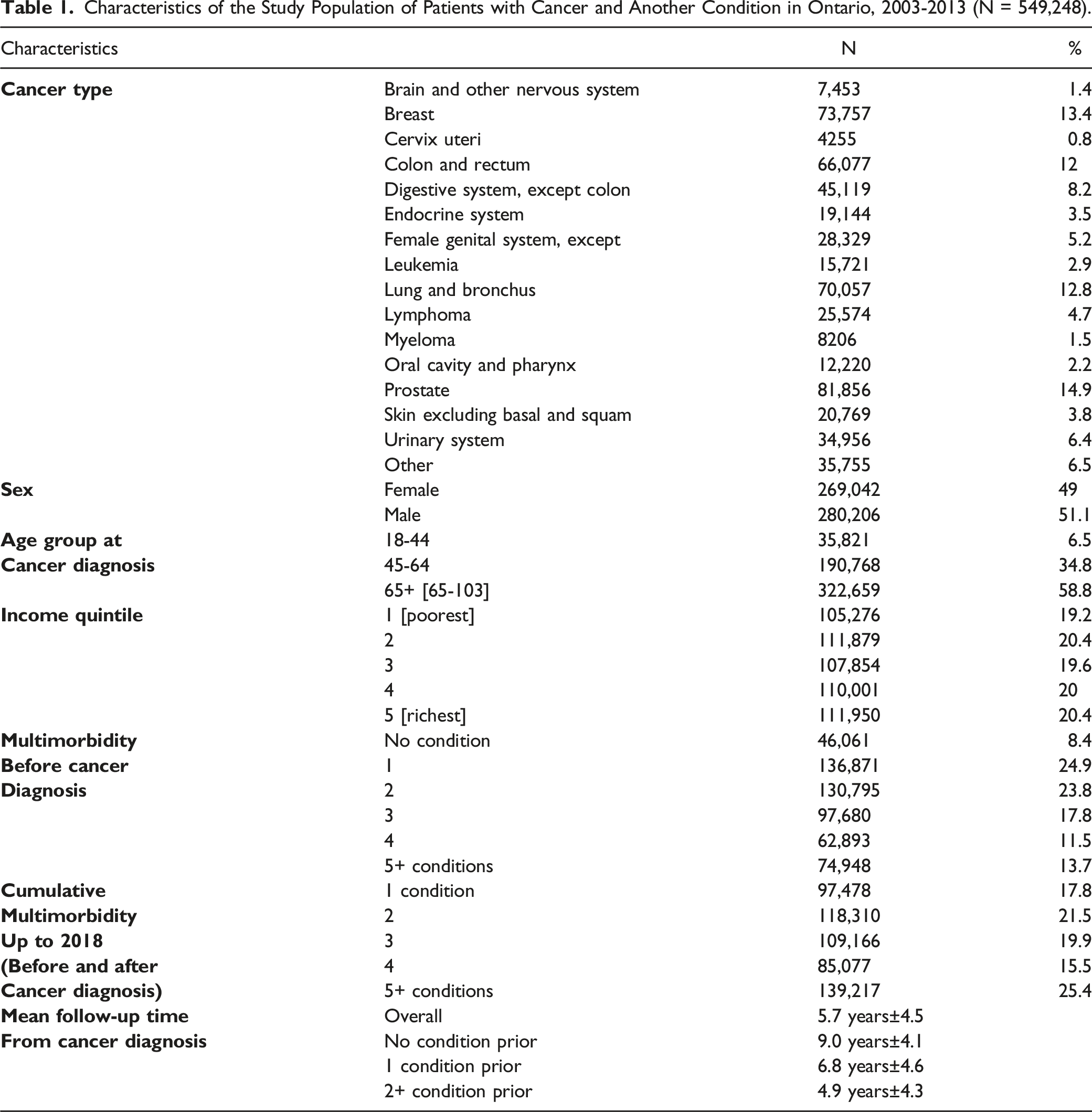
The temporal overlap between cancer and other chronic diseases is important for understanding the presence and burden of multimorbidity. In this study, data showed that the vast majority (91.6%) of the study population had a chronic condition prior to cancer, while 48.8% developed another condition after cancer diagnosis. On average, those with a chronic condition prior to cancer diagnosis received their last diagnosis 3.6 years before cancer (−1327 days; IQR: −1955 to −256). Of those who developed at least one new condition after cancer, the first diagnosis occurred on average 2.4 years (892 days; IQR: 222 to 1309) following cancer. To illustrate the degree of overlap in timing between cancer and other chronic conditions, data showed that 56% and 68% of non-cancer chronic condition diagnoses were made within 3 years, prior to or following cancer diagnosis, and that broadening the window to five years showed that 73% and 86% of chronic disease diagnoses occurred before and after cancer diagnosis, respectively.
Ratio of Observed to Expected Prevalence of Each Co-Occurring Condition Within Level of Cumulative Multimorbidity, i.e Both Pre or Post Cancer Diagnosis (N = 549,248).

Specific pre-existing (prior to cancer) conditions were more prevalent among patients later diagnosed with certain cancers, while other conditions were most frequently diagnosed following the diagnosis of certain types of cancer. For example, women with breast cancer were more likely to have anxiety or osteoporosis prior to their cancer diagnosis, and more likely to develop osteo-arthritis following breast cancer. Among people with lung cancer, COPD was the condition most commonly diagnosed prior to cancer. Patients with prostate cancer developed any chronic conditions more frequently than average, likely due to their longer survival (Supplementary Appendix 4).
Observed Disease Combinations
Top 10 Observed Disease Combinations Among Patients with Cancer, and Ratio of Observed to Expected Prevalence Based on Random Combinations.

aOccurred more often than expected by chance (ratio >3).
Abbreviations: Hypertension (hyper); Cardiac Arrhythmia (arryth); Coronary syndrome (coron); Diabetes Mellitus (dm); Osteo-arthritis (ostarth); Osteoporosis (osteopor); (Other) Mental health condition (othermen); renal disease (renal).
Disease clustering varied by individual characteristics. For example, among young adults (18-44 years) with cancer, combinations that occurred more frequently than expected primarily included asthma, anxiety, mood disorders and/or other mental illness. In older adults (>65 years), however, disproportionally prevalent combinations often include dementia and/or hypertension.
Top 10 Observed Disease Combinations by Sex and Multimorbidity Level, Among Patients with Cancer.

aOccurred more often than expected by chance (ratio >3).
Abbreviations: Hypertension (hyper); Cardiac Arrhythmia (arryth); Coronary syndrome (coron); Diabetes Mellitus (dm); Osteo-arthritis (ostarth); Osteoporosis (osteopor); (Other) Mental health condition (othermen); renal disease (renal).
Partitive Disease Clustering
Given the large sample size, we performed hierarchical clustering using annual data to inform the choice of appropriate number of clusters for the partitive (k-means) clustering approach.
Considering the number of clusters deemed appropriate for multiple years of data, and with the highest “Expected Overall R-Squared” and “CCC value”, 61 clusters seemed adequate for the k-means clustering (Supplementary Appendix 3). Even though 15 clusters appeared acceptable for almost all the years of analysis, they were not optimal based on various criteria and lack of specificity in the clusters’ composition.
Description of Disease Clusters Based on Partitive (K-Means) Method with 61 Clusters (n = 549,248).

Abbreviations: Hypertension (hyper); Cardiac Arrhythmia (arryth); Coronary syndrome (coron); Diabetes Mellitus (dm); Osteo-arthritis (ostarth); Osteoporosis (osteopor); (Other) Mental health condition (othermen); renal disease (renal) ** Cluster # automatically assigned by software, based on their emergence through the statistical steps. However, we have presented the clusters in the table according to their size (most populous first), column N (highest to lowest).
Health Outcomes Associated With Disease Clustering
It is important to consider disease clusters in care management because of the impact that such clusters may have on health services utilization and outcomes. As shown in Figures 1 and 2, there were differences in healthcare utilization (HA, ED and PC visits) between clusters, regardless of the number of combinations or clustering approach (top five observed combinations vs 61 partitive clusters). Variations were observed both within and between age groups. For example, the highest numbers of PC visits or HA per person-year in any age group were observed for clusters including all of asthma, CHF, COPD, diabetes, hypertension, osteoarthritis, and anxiety. On the other hand, the lowest healthcare utilization occurred among clusters including asthma, osteoarthritis or hypertension. For the same disease cluster, ED visits were generally lower among older adults (>65) than younger people. Health services utilization among the top 5 observed combinations, by multimorbidity level and age group. (a) Primary care visits, Age group 18-44. (b) Hospitalization and emergency department visits, Age group 18-44. (c) Primary care visits, Age group 45-64. (d) Hospitalization and emergency department visits, Age group 45-64 (e) Primary care visits, Age group 65 +. (f) Hospitalization and emergency department visits, Age group 65+. Note: Hypertension (hyper); Cardiac Arrhythmia (arryth); Coronary syndrome (coron); Diabetes Mellitus (dm); Osteo-arthritis (ostarth); Osteoporosis (osteopor); renal disease (renal) Patients’ health service use by 61 partitive (k-means) clusters, and age groups.

Discussion
The purpose of this study was to use a large, retrospective cohort design to identify the most common disease combinations and statistical clustering, among complex adult cancer patients (ie people who have cancer and one or more other conditions), overall and for specific cancer types, and to assess the related health burden of identified disease clusters within cancer patients. Our findings have implications for clinical care of people with multimorbidity.
Consistent with previous research (eg,1,2), data from this study showed that multimorbidity was highly pervasive, with more than 90% of the sample having cancer and at least one other chronic health condition, and with 25% having five or more co-occurring chronic health conditions. These data reinforce the idea that multimorbidity among people with cancer is the norm and not the exception. Within our study design, we also demonstrated that chronic health conditions were frequently present prior to cancer diagnosis, making multimorbidity care important from the outset of cancer care. However, almost half of participants were diagnosed with additional chronic health conditions after cancer diagnosis, suggesting that cancer patient complexity is dynamic, and that healthcare providers and future treatment protocols must be flexible and adjustable as patients’ health and complexity is likely to increase with survival and age.
As predicted, data also showed that some chronic health conditions are more likely to co-occur with cancer than others, and that the most prevalent conditions present prior to cancer diagnosis were somewhat different than those most likely to emerge after cancer was diagnosed. Using both observational and statistical clustering methods, diseases such as anxiety, diabetes, hypertension and osteoarthritis occurred at rates that far exceed those expected by chance, providing strong evidence that care protocols that take disease clusters into account are needed to advance clinical care. Further validating the existence and clinical importance of cancer multimorbidity clusters were the identification of clusters that are highly intuitive and consistent with large bodies of epidemiological research. For example, we found women were more likely than men to have diagnosed anxiety and osteoporosis before cancer, and osteoarthritis following breast cancer. Anxiety28,29 and bone density diseases 30 are well known to occur more often in women than men. Similarly, the risk of developing COPD was much higher among people who had lung cancer, both of which share exposure risks (eg smoking, mining). Also, not surprisingly, rates of dementia and hypertension were higher among older persons with cancer than youth, as the risk of both conditions is well known to increase with age.31,32
Similar to previous research,33-35 and further reinforcing the importance of considering disease clusters in health-system, and personalized care, we found that patients within different disease clusters used healthcare services differently and had differing healthcare outcomes including emergency and hospital admissions. Our observations were robust against the number of clusters or clustering approaches that we employed. For example, we showed that people with cancer and asthma, CHF, COPD, diabetes, hypertension, osteoarthritis, or anxiety were likely to have more PC visits and hospital admissions across age groups. Perhaps surprisingly, ED visits were generally lower among older adults (>65) than younger people. One possibility is that older adults have more advanced care planning including strategies to avoid rehospitalization. A related possibility, however, is that young people have less primary care support, including less access overall and/or fewer established relationships with primary care providers. Future research is needed to explore the range of factors contributing to lower ED use in older adults. Information about trends in health services utilization and outcomes are extremely important for health system factors such as estimating care needs, cost of care, and the types of supports and services to provide patients with the most appropriate (ie, lowest restrictive level of) care possible. For instance, our data suggests that younger patients may benefit from additional community supports (eg, nurse visits) to reduce hospital admissions. More research is needed to empirically investigate the specific kinds of supports and services that would best meet the wants and needs of patients within specific cancer multimorbidity groups.
While our study does not suggest the specific ways in which care protocols and pathways should be enhanced for individual disease clusters or groups, the general existence of robust disease clusters has numerous implications for how future care might be conceived, managed, coordinated, and delivered. For example, cancer clinics may begin to routinely screen for highly comorbid conditions (anxiety, diabetes mellitus, hypertension, and osteoarthritis) at the start of care and include case management, scheduled communications between primary care and other healthcare providers (including mental health), to ensure that care for non-cancer conditions can be integrated (or at least continue in parallel) with cancer care, in a way that is acceptable to and sustainable for patients and their families. Similarly, if CHF and renal failure are common emergent conditions following cancer diagnosis and care, care protocols may also include patient education about signs and symptoms and/or systematic communications to follow-up (ie, primary care) providers to monitor problems that are objectively and statistically likely to occur. Other chronic health conditions that were likely to co-occur might also be flagged for screening or monitoring among certain populations where the general risk for certain chronic conditions remains high (eg, COPD among people with lung cancer). Also, existing Healthcare Information Technology (HIT) functionalities such as dashboards, patient relationship managers, event alerts, referral tracking, and care plans (eg, 36 ) could be used to facilitate such changes in cluster-informed care. While these kinds of prompts and reminders are not at all novel, they remain important since cancer diagnosis and care often supersedes care for other conditions 37 - even when the risk of morbidity and mortality from non-cancer conditions often remains high.9,17,38 An important next step in improving services for people with cancer multimorbidity is to bring together experts in the specific cancer and chronic disease types represented in clusters, plus experts in integrated and coordinated care, to create effective and efficient protocols for improving complex cancer care, including medication reconciliation, symptoms monitoring, self-advocacy and self-care. Since cancer treatments including surgery, radiation and chemotherapy can lead to chronic illnesses such as lymphedema, osteoporosis, 39 cardiac problems, as well as secondary cancers,40,41 future research should better assess treatment sequelae and resulting chronic conditions, to inform better care management and improve health for cancer survivors.
Strengths and Limitations
Overall, this study has several notable strengths including the size, representativeness of the population, and 10+ years follow-up period. Additional strengths of the study include our use of two different and complementary approaches to identifying disease clusters, both of which produced similar findings and validated the importance of considering cancer disease clusters in future work. Compared to other studies, we included all documented cancer types and multiple chronic conditions, enabling broader and deeper exploration of the concept of disease clusters in a population with cancer. Our focus on disease clusters also is an important step towards normalizing and integrating patient-centered care practices into healthcare systems that have traditionally been single-disease focused and siloed.
While this study is the first to systematically identify cancer multimorbidity clusters in a large population-level data set spanning more than 15 years, we recognize several limitations of our design. First, our results identified many more clusters than might be addressed with specific disease protocols. However, strategies to address some of the largest and more common than expected clusters, with the highest healthcare utilization and worst outcomes, could be targets for interventions with the possibility of greatly improving care and related outcomes. In any case, though top observed combinations can be informative, they remain numerous and future studies are needed to identify the highest priority clusters as well as the most feasible, highest yield interventions for trial, cluster-based health system reform. A related issue was operationalizing cancer complexity since cancer remission is possible and may have occurred for some individuals within the study timeframe. Although the majority (76%) of the entire cohort had a chronic condition diagnosed within 5 years either before or after cancer occurrence, our analyses have potential to overestimate of the degree of overlap between cancer and other chronic diseases and hence the presence and extent of cancer complexity. At the same time, since cancer is a chronic, relapsing condition, although someone may be in remission, they still carry the designation of being a cancer survivor and such experience is likely taken into consideration in clinical decision making and personal experience. As such, we thought it prudent to overestimate (ie, include all instances of cancer and not remove individuals from analyses due to potential remission) than to underestimate its role in this research. Indeed, multimorbidity complicates and has the potential to worsen patients’ health status and quality of care. Examples of this include neglecting care for pre-existing conditions and/or failure to recognize and adequately treat new ones.
We also recognize that other methodological approaches to identifying clusters in population data may be useful. While partitive clustering helped to create key groupings with essential conditions co-occurring with cancer and while this approach is most appropriate given the sample size and number of conditions, it is limited by the need to choose a predefined number of clusters k 42 . While not reported here, we tried different k’s suggested by a hierarchal clustering analysis with annual data and selected the most performant k to perform the partitive clustering.
Finally, the study presents a limited potential for selection bias since it is a population-based cohort. However, possible misclassification regarding multimorbidity should be considered because we are using a limited (but large) number of conditions. Nevertheless, these conditions have been largely used and proven to provide a reasonable picture of multimorbidity in the study population.1,2 The use of administrative data is also susceptible to information bias; however, the universal coverage in the province and very long follow-up period contribute to minimizing this bias. Despite using validated algorithms to operationalize the 17 chronic conditions, other relevant conditions may be missing and some of the conditions may not be adequately represented in the data, including risks of under- or over-diagnosis of co-occurring conditions in people with cancer.43,44 Nonetheless, the findings from this study may be applicable to similar settings with universal coverage and comparable access to care. However, given the selected conditions and differential needs and access to health services for certain population groups, further, more specific explorations are necessary.
Implications and Conclusions
People with cancer are overwhelmingly likely to have other chronic health conditions, including those that are present before cancer diagnosis, and those that develop after cancer. Data from this study showed that certain types of cancer are likely to systematically co-occur with other chronic health conditions and that overall conditions such as hypertension, osteoarthritis and anxiety are likely to co-occur with cancer overall. Data also showed that people with different clusters of diseases are at differential risk for increased health burden, with the highest numbers of PC visits or HA per person-year in any age group occurring among people with all of asthma, CHF, COPD, diabetes, hypertension, osteoarthritis, and anxiety. Overall, our findings reinforce the fact that multimorbidity complicates and has the potential to worsen patients’ health status and quality of care, including neglecting care for pre-existing conditions and/or failure to recognize and adequately treat new ones and iatrogenic effects of treatment on chronic conditions or secondary cancers.39-41 Researchers, clinicians, policymakers and other stakeholders can use the clustering information presented here to begin to improve the healthcare system for people with cancer multimorbidity. Cluster-informed actions and strategies can be privileged to resolve conflicting or redundant recommendations in clinical treatment guidelines, create consulting and referral relationships with specialists skilled in treating clustered conditions, define roles and responsibilities for managing shared care tasks, create connections to complementary services, and define other elements of a comprehensive care plan that patients and caregivers can review, approve, or modify to meet their individual needs towards patient-centered care.
Abbreviations
Acute Myocardial Infarction
Cubic Clustering Criterion
Congestive Heart Failure
Chronic Obstructive Pulmonary Disease
Discharge Abstract Database
Emergency Department
Hospital Admissions
Healthcare Information Technology
Health Services Utilization
Ontario Health Insurance Plan
Primary Care
Supplemental Material
Supplemental Material - Multimorbidity and Complexity Among Patients with Cancer in Ontario: A Retrospective Cohort Study Exploring the Clustering of 17 Chronic Conditions with Cancer
Supplemental Material for Multimorbidity and Complexity Among Patients with Cancer in Ontario: A Retrospective Cohort Study Exploring the Clustering of 17 Chronic Conditions with Cancer by Anna P. Kone, Deborah Scharf, and Amy Tan in Cancer Control
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This work was supported by the Ontario Ministry of Health (MOH) through grant No. 06034 to the Health System Performance Network (HSPN). The opinions and statements expressed herein are solely those of the authors and do not reflect those of the MOH; no endorsement is intended or should be inferred.
Data Availability
The study data were made available through ICES Data & Analytic Virtual Environment (IDAVE), however restrictions apply to the availability of these data, which were used by the authorized investigators under the service agreement ICES DAS # 2020–727. Access may only be granted under specific conditions and ethics approval; please visit ![]() for more information.
for more information.
Disclaimer
Parts of this material are based on data and information compiled and provided by the Canadian Institute for Health Information (CIHI). However, the analyses, conclusions, opinions, and statements expressed herein are those of the authors, and not necessarily those of CIHI. No endorsement by Institute for Clinical and Evaluative Sciences (IC/ES) or the Ontario Ministry of Health is intended or should be inferred.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
