Abstract
Background:
Recent advancements in identifying druggable molecular drivers in lung adenocarcinoma (LUAD), have transformed treatment paradigms. In recent years, Next Generation Sequencing (NGS) has gained momentum as an essential tool for in-depth simultaneous analysis of multiple genes, thereby streamlining the diagnostic process in LUAD. Despite this, the implementation of NGS testing in both the US and Europe remains suboptimal.
Aims:
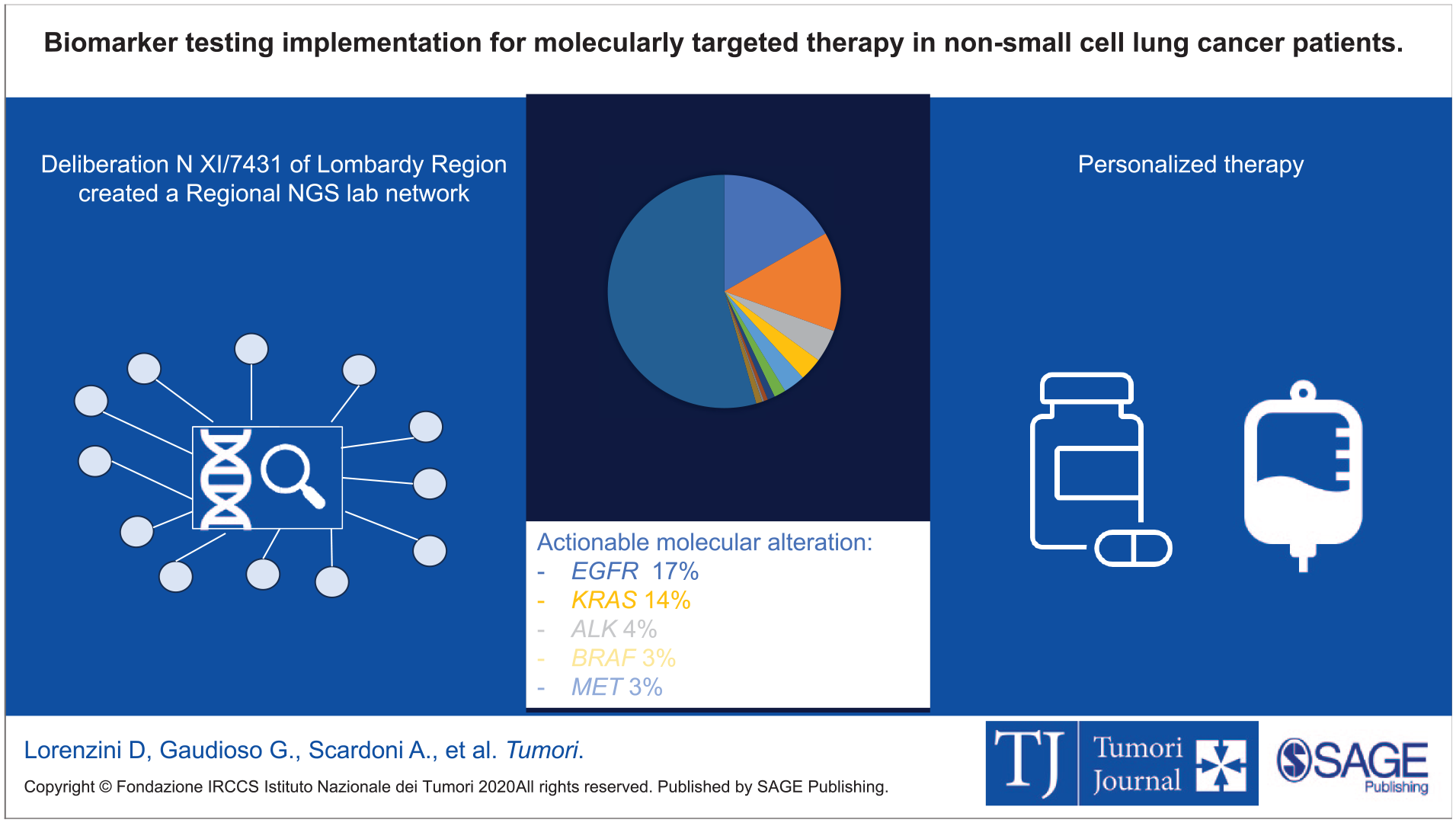
In compliance with a decree issued by the Italian Ministry of Health, Lombardy Region recently launched an initiative to implement NGS testing in patients with advanced LUAD. In this context, a real-world prospective observational study was planned to assess the efficacy of the regional network of molecular laboratories in testing nine biomarkers (
Results:
In 2023, out of the 2784 advanced/metastatic LUAD patients expected in Lombardy, 2343 (84.2%) were successfully evaluated with an NGS panel including all the nine biomarkers for on-label drugs. Actionable aberrations were identified in 45.5% of the patients (1068/2343), predominantly involving
Conclusion:
Our data provide evidence that establishing a structured network of NGS hubs is mandatory to ensure access of advanced LUAD patients to molecularly targeted treatments.
Visual abstract
Introduction
Lung cancer is one of the major causes of cancer mortality worldwide, with most patients diagnosed in a locally advanced or metastatic stage. 1 However, the identification of druggable molecular drivers has dramatically improved the survival of lung cancer patients. 2 Next Generation Sequencing (NGS) allows to detect multiple gene aberrations, reducing turnaround time (TAT) and improving patient outcome. 3 Accordingly, the European Society of Molecular Oncology (ESMO) guidelines recommend the use of NGS in the diagnostic workflow of patients with advanced stage lung adenocarcinoma (LUAD). 4 Unfortunately, the percentage of cases tested with NGS worldwide is still unsatisfying. 5 Sadik et al reported that in the US, almost half (49.7%) of advanced lung cancer patients are excluded from personalized therapy, mainly because of hurdles in biomarker testing. 6 Likewise, many studies reported that NGS is underutilized in Europe, prompting the launch of several initiatives for its implementation. In the UK, the 100,000 Genomes Cancer project profiled by whole genome sequencing (WGS) 13,880 solid tumors, including 1383 patients with lung cancer. 7 Similarly, among 2500 patients profiled by WGS in the context of the Drug Rediscovery Protocol in The Netherlands, over 1500 started treatment with targeted therapy or immunotherapy. 8
In Italy, the implementation of NGS is governed by the National Health System (NHS), aiming to ensure equitable, standardized, and appropriate access to molecular profiling. In compliance with a Decree of the Ministry of Health, in November 2022 the Lombardy Region issued a Deliberation (XI/7431) to promote NGS testing in patients with advanced LUAD.
9
The initiative was designed to guarantee the analysis of nine biomarkers (
Here, we describe the procedures carried out to put the Regional Deliberation into action, to create a structured network of molecular labs accredited by the NHS, and to report the results of NGS sequencing in a prospective real-world cohort of 2343 LUAD patients.
Methods
Regional Deliberation XI/7431
Based on the recommendations of a working group including pathologists and oncologists, the Regional Deliberation 9 established that laboratories accredited to perform NGS testing in histologically confirmed LUAD patients had to fulfill the following requirements: be a pathology laboratory with a somatic molecular diagnostic unit; be integrated with an oncology unit to ensure an interdisciplinary approach; have at least two years of documented experience with NGS testing; guarantee a turnaround time (TAT) NGS workflow equal to or less than 10 working days (15 days for comprehensive panels).
NGS testing had to be performed with NGS panels able to identify variants in the following genes:
Each laboratory was asked to provide the following items in a dedicated database for each LUAD patient subjected to NGS testing: clinical data (sex, age); contact of the referral oncologist and center; date of the NGS prescription; clinical setting (diagnosis or progression after targeted therapy); type of specimen (tissue or liquid biopsy); date of sample acceptance; date of the molecular report; type of NGS panel used; failure rate for DNA and RNA panels; molecular alterations identified; and actionability of the molecular alterations classified according to ESMO Scale of Clinical Actionability (ESCAT). 10 Each center appointed one or more person in charge of data collection and transmission to the database. Data were collected, aggregated and analyzed with descriptive statistics.
LUAD incidence in Lombardy Region
Lombardy Region collects health records in its comprehensive healthcare system data WareHouse (DWH), according to a star scheme characterized by a central fact table (i.e., the Population Registry dataset) surrounded by a set of dimensional tables (i.e. all administrative informational flows), thus enabling detailed longitudinal analyses at the individual level. To identify the number of patients with a first diagnosis of lung cancer in the Lombardy Region, we interrogated the Hospital Discharges Registry (SDO), using the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM). To further refine the analysis, we retrieved historical epidemiological data from all eight territorial cancer registries within the Regional Healthcare System, which collect cancer diagnoses from pathology laboratories. The 2017–2019 timeframe was selected to avoid the impact of the SARS-Coronavirus-2 pandemia.
Results
Laboratory network
Based on the aforementioned criteria, Lombardy Region identified 12 molecular pathology laboratories: ASST Spedali Civili di Brescia, Fondazione IRCCS San Gerardo dei Tintori in Monza, ASST Grande Ospedale Metropolitano Niguarda, ASST Lariana, ASST Santi Paolo e Carlo, ASST dei Sette Laghi, Fondazione IRCCS Ca’ Granda Ospedale Maggiore Policlinico, Fondazione IRCCS Istituto Nazionale per lo Studio e la Cura dei Tumori di Milano, Fondazione IRCCS Policlinico San Matteo, Humanitas Mirasole S.P.A., Istituto Europeo di Oncologia S.R.L. and Ospedale San Raffaele SRL.
NGS testing in Lombardy Region
According to DWH records, 10,563 patients received a diagnosis of lung cancer, any histology, any stage, in 2023. This figure also included alive patients who received the diagnosis in previous years. In the 2017-2019 period, the Lombardy Region cancer registry reported 15,235 cases with a cytological or histological diagnosis of lung cancer (mean 5078 cases per year). Of them, 8353 had a diagnosis of advanced LUAD (mean 2784 per year), that was considered a proxy for the yearly incidence of LUAD in the Lombardy Region.
According to our prospectively annotated database, the network of 12 laboratories carried out 2343 NGS tests in advanced LUAD patients (84.2% of estimated cases; Figure 1). In 1760 (75.1%) of these patients, NGS was prescribed by a multidisciplinary team. In most cases (2207; 94.2%) NGS testing was performed at diagnosis, or before first-line systemic therapy, while in the remaining 136 cases (5.8%) NGS profiling was carried out after progression to molecularly driven targeted therapy. NGS was performed in tissue samples from primary tumor or metastatic sites in most of the cases (2313; 98.7%) and in liquid biopsy in the remaining cases (20; 1.3%). The percentage of cases analyzed by liquid biopsy was slightly higher (4.4%) in patients progressing upon target therapy.
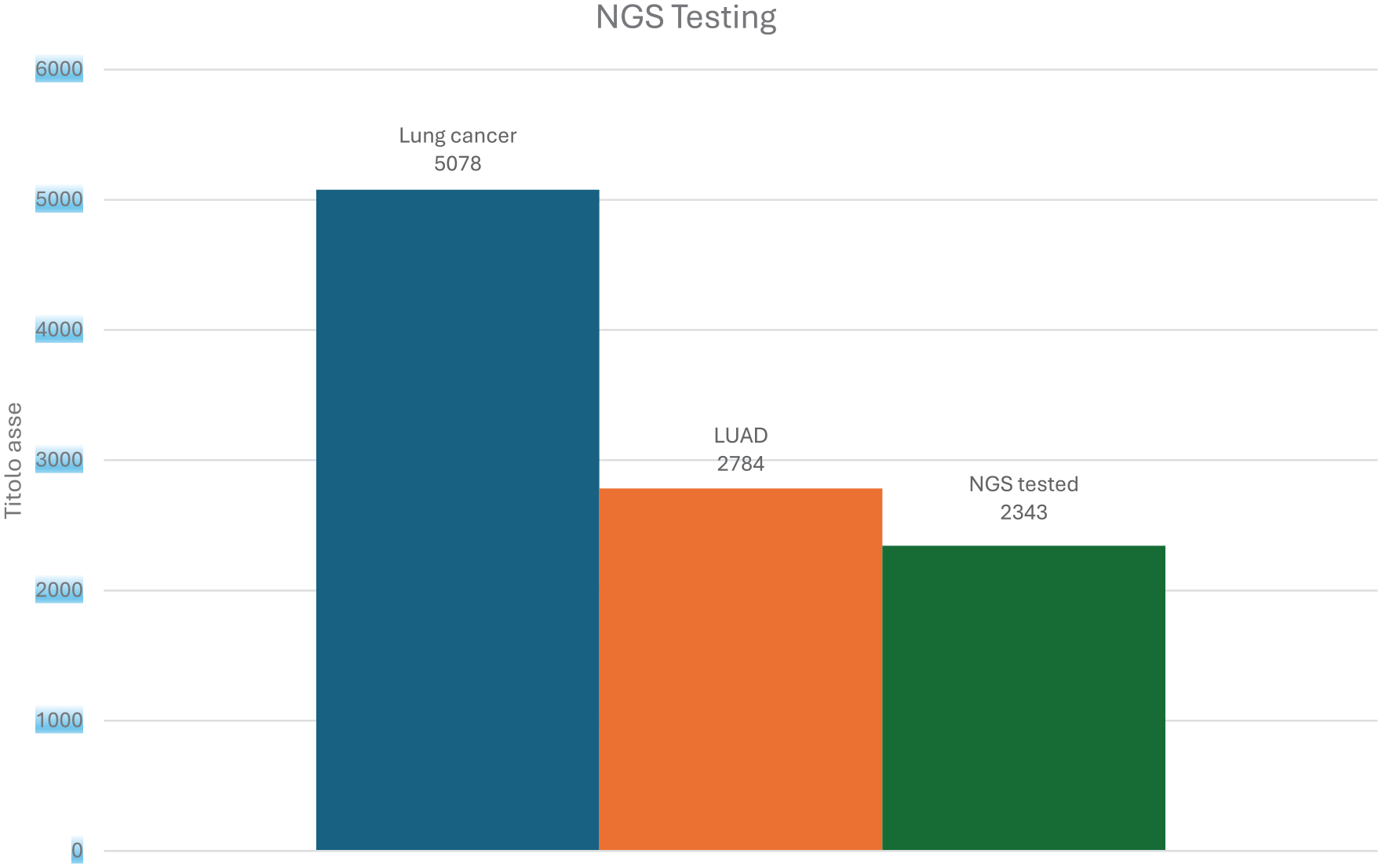
Estimated total cases of lung cancer per year in Lombardy Region (blue bar) and LUAD (orange bar), compared to NGS test performed in 2023 (green bar). LUAD: Lung adenocarcinoma. NGS: Next-generation Sequencing.
NGS panels
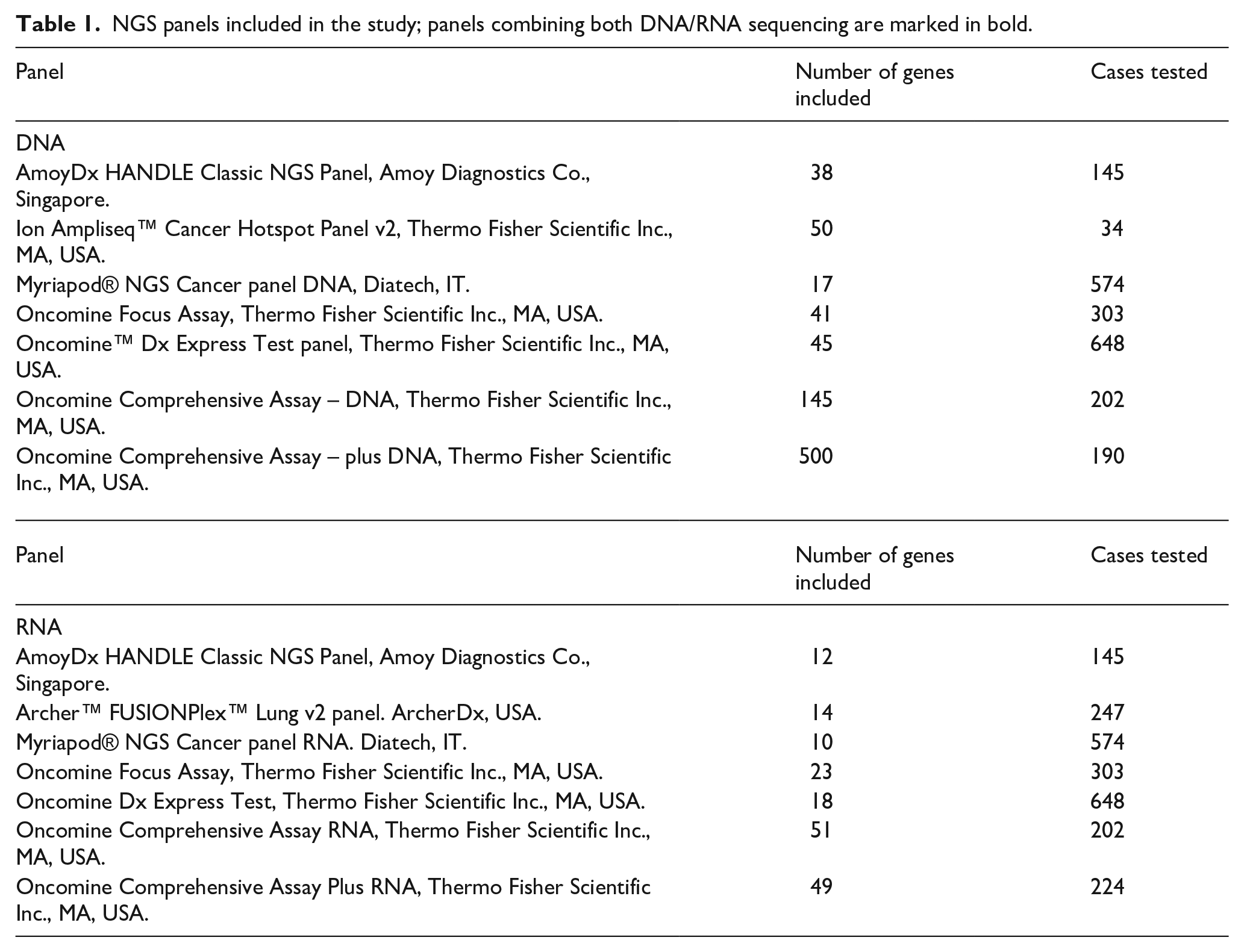
Most cases (1951, 83.3%) were profiled by a targeted DNA/RNA approach by using six different panels including up to 100 genes (Table 1). The remaining cases (392, 16.7%) were analyzed by comprehensive DNA/RNA genomic profiling (CGP) panels including 196 (in 202 cases) or 549 (in 190 cases) genes (Table 1). One center performed an RNA-based panel only (
NGS panels included in the study; panels combining both DNA/RNA sequencing are marked in bold.
Results of NGS testing
ESCAT tier I or II aberrations actionable by targeted on-label drugs identified in our cohort are summarized in Table 2 and in Figure 2. Out of the 2343 patients analyzed, 1068 (45.6%) bore an ESCAT tier I or II alteration, mostly
Actionable (ESCAT I and II) molecular alteration identified.
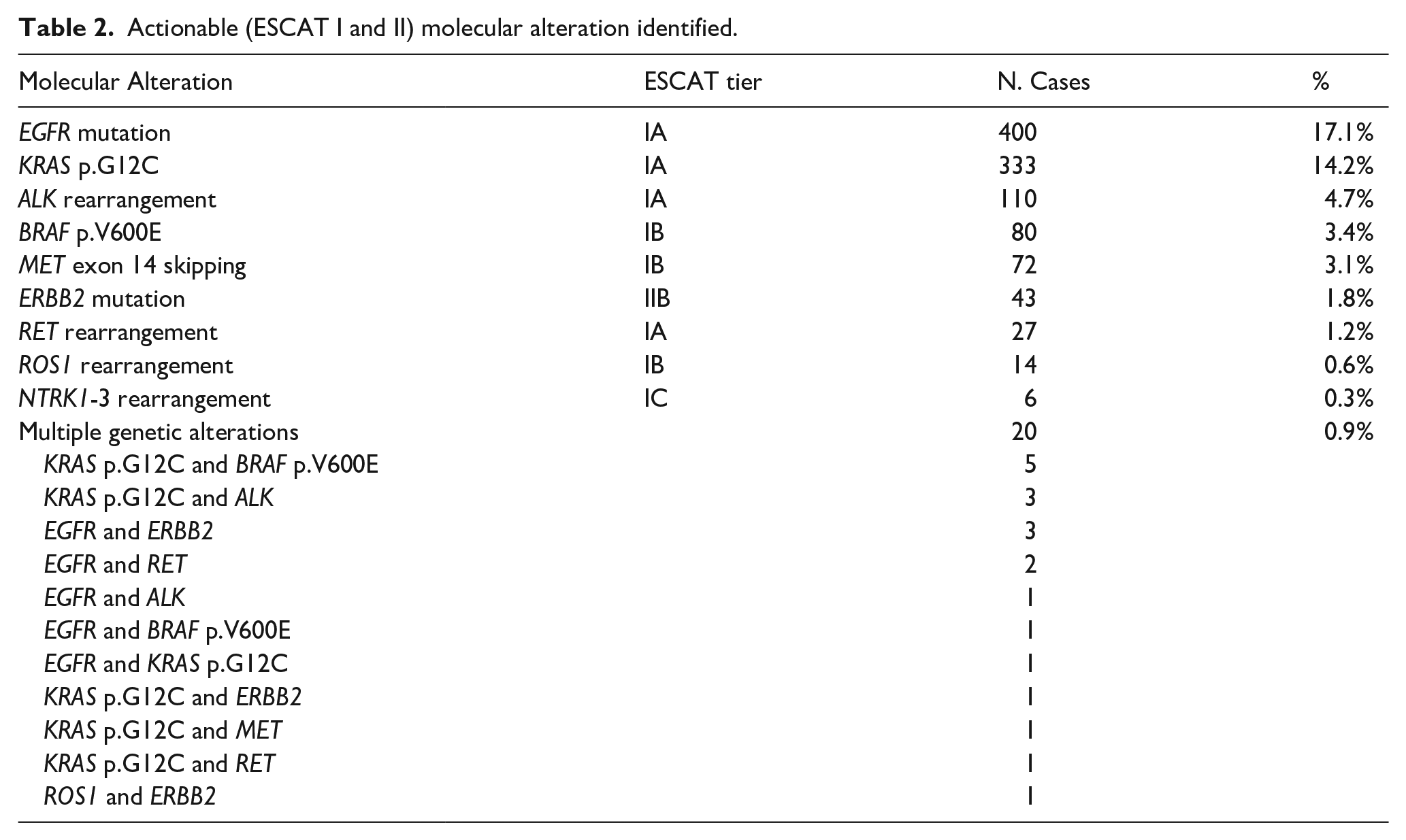
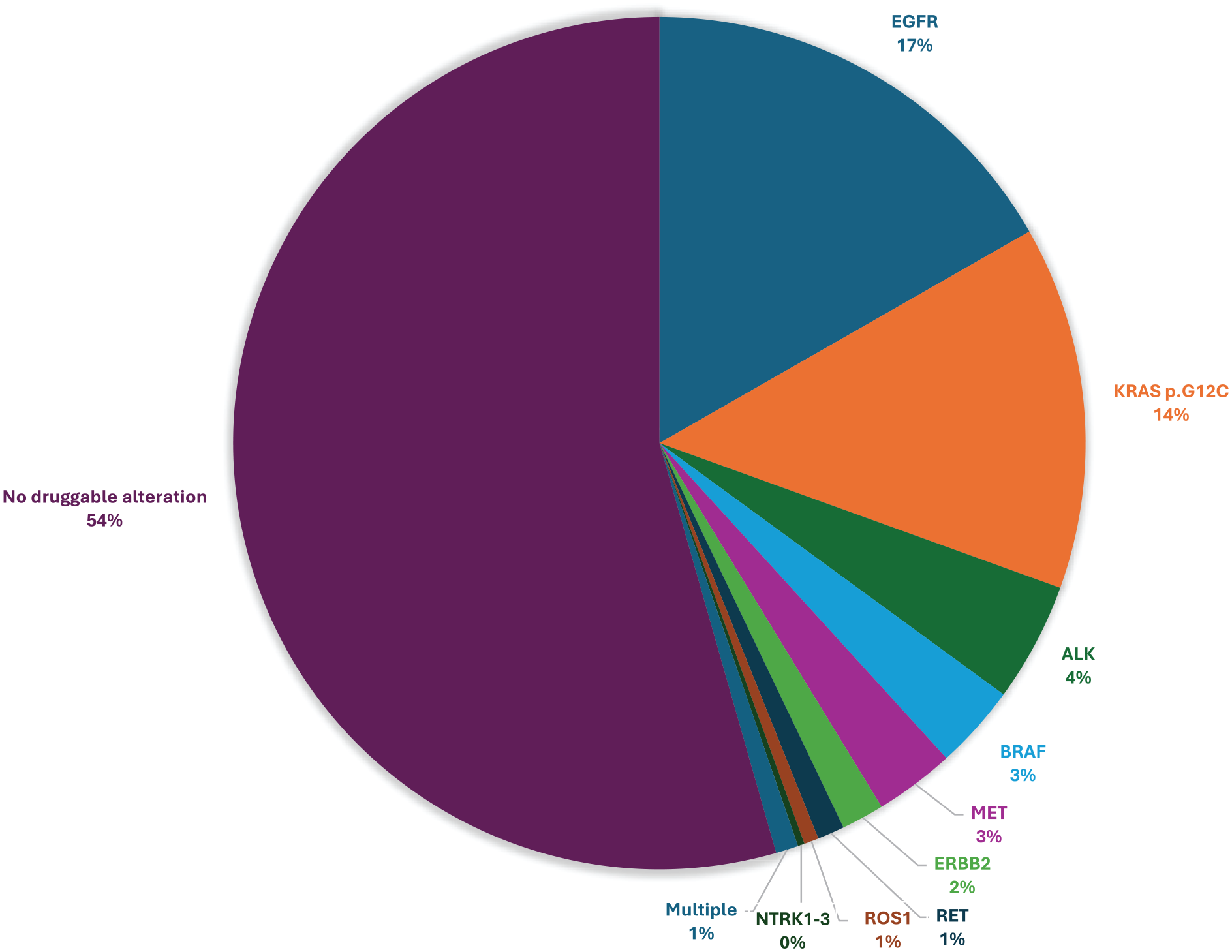
Actionable molecular alterations.
Comparison of actionable molecular alteration identified by targeted and comprehensive panels.
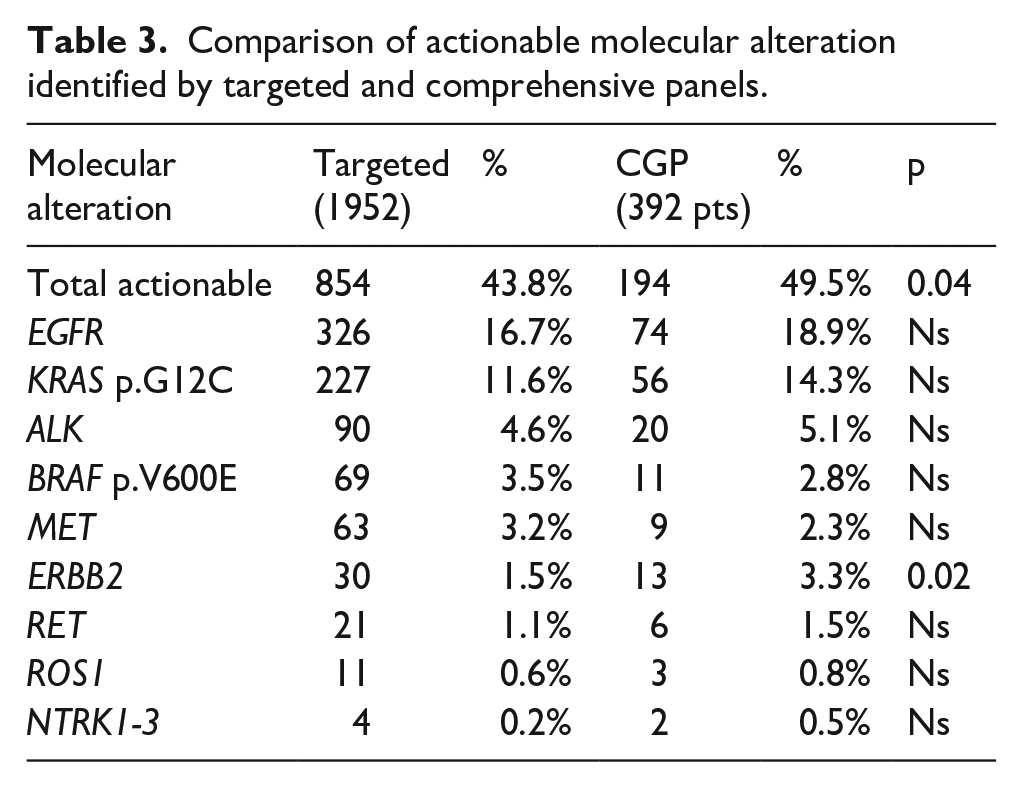
Moreover, in 151/391 patients (38.7%), DNA CGP testing found 222 additional molecular alterations potentially actionable with off-label drugs (in nine patients, 2.3%, ESCAT II; in 147 patients, 67.7% ESCAT IIIa/b; and in 66 patients, 30.4%, ESCAT IV), that were not included in most of the targeted NGS panels used in this study. The most common alterations were:
Discussion
The proper treatment of advanced or metastatic lung cancer patients relies on tumor molecular characterization. In Europe, there is a heterogeneous approach in biomarker testing, with most of the laboratories still performing single gene testing by a combination of immunohistochemistry, ISH and RT-PCR. The percentage of patients with advanced lung cancer receiving any molecular test ranged from 65% to 85% in Germany, Italy, and Spain (2011-2016).11,12 Likewise, a Swiss observational study showed that 79% of the patients were analyzed by single-gene
A recent study in the US reported that a 10 percent increase in the use of NGS panels (including
In our study, we report the impact of NGS testing in Lombardy Region, the most populated area in Italy with roughly 10 million inhabitants. To implement molecular profiling in advanced LUAD patients, a Regional Deliberation established a diagnostic workflow, formalized a dedicated network of pathology laboratories and introduced a specific reimbursement code.
9
This organizational framework allowed to obtain a complete molecular characterization in the majority of LUAD patients. In fact, out of the 2784 advanced/metastatic LUAD patients expected in Lombardy in 2023, 2343 (84.2%) were tested with an NGS panel including all the biomarkers for on-label drugs, achieving the main goal of the Regional project. Most (83.3%) of the cases were tested with smaller targeted DNA NGS panels including up to 50 genes, while a lower percentage of patients (16.7%) were assessed upfront with CGP including up to 549 genes. As expected, CGP led to the identification of a higher number of biomarkers potentially actionable with off label drugs or experimental treatments in clinical trials at progression to standard therapies.
21
Moreover, CGP panels were also able to identify ESCAT tier I or II variants in a slightly higher percentage of patients, in particular involving
Integrating DNA NGS panels with RNA panels provides a more detailed picture of the mutational landscape in cancer, because RNA sequencing is able to directly assess oncogenic fusion transcripts at high sensitivity: accordingly, we observed that the combined DNA/RNA approach identified actionable fusion transcripts in 7% of the cases. In particular,
NGS in liquid biopsy is a non-invasive approach for tumor profiling at diagnosis and for non-invasive monitoring of relapsing patients to uncover resistance mechanisms and is recommended for tumor profiling in advanced LUAD at diagnosis when fine needle biopsy/aspirate is not feasible or unsafe, or when NGS in tissue biopsy failed because of DNA/RNA poor quality or low quantity.26,27 Our real-world dataset demonstrates that liquid biopsy is still underused in clinical practice, with only a small fraction (1.3%) of patients tested with targeted panels, even in the setting of patients progressing upon targeted therapy.
The main limitation of our study lies in the absence of data relative to the clinical outcome of the advanced LUAD patients molecularly profiled by NGS. Nevertheless, we are in the process of completing our dedicated database with data related to patient treatment and outcome, including the prevalence of patients treated with molecularly targeted on-label and off-label drugs, the number of patients recruited in clinical trials, as well as objective response rate and patient progression-free survival.
In conclusion, in this observational prospective study we describe a real-world cohort of patients with LUAD who underwent NGS testing in a structured framework of regional labs. Our data provide evidence that a network of specialized NGS labs in the context of the NHS allows cost-effective NGS molecular profiling of the overwhelming majority of LUAD patients, overcoming traditional limitations and ensuring access to molecularly targeted therapy with on-label drugs to a wider number of patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
