Abstract
Introduction:
This cross-sectional study was aimed at estimating the number of Italian incident cancer patients in 2020 eligible for, and respondent to, immune checkpoint inhibitors (ICI).
Methods:
The study is based on publicly available data: the ICI approved until August 2022 by the Italian Medicines Agency (AIFA) with their specific indications and overall observed responses, rther details can be found in the Online Supplementary Materi cancer incidence estimates at 2020 and observed cancer deaths, and published papers with estimates on the frequency of different cancer stage/histology/markers etc. corresponding to AIFA authorizations.
Results:
In the analyzed period, a total of seven ICI were authorized in Italy for 20 cancer types. The estimated number of ICI-eligible patients in 2020 was 48,400, 14.3% of those tumors (including skin epitheliomas) that may fit AIFA-indications, and 10.5% of all the incident malignant tumors, including skin epitheliomas. The number of patients who may benefit from ICI therapy was 24,052, 49.7% of the ICI-eligible ones, or 5.2% of the overall estimated incident cancers in 2020.
Conclusions:
In conclusion, although the number of ICI-eligible patients is a relatively small proportion of the yearly burden of cancers, about half of them may respond to ICI-treatment.
Introduction
The immune checkpoint inhibitors (ICI) are immunotherapy drugs acting on the Programmed Cell Death Protein 1 (PD-1) and Cytotoxic T-Lymphocyte Antigen (CTLA) pathways repressing negative immune regulation and reactivating T-cell functions. Therefore, they allow more effective cancer cell elimination. 1 The use of these drugs has improved outcomes for patients affected by several advanced solid tumors. 2 In Italy, seven ICI have been approved by the Italian Medicines Agency (AIFA) until August 2022: ipilimumab (YERVOY), 3 nivolumab (OPDIVO), 4 pembrolizumab (KEYTRUDA), 5 atezolizumab (TECENTRIQ), 6 avelumab (BAVENCIO), 7 durvalumab (IMFINZI), 8 and cemiplimab (LIBTAYO). 9 Overall, these drugs were approved for the following indications: head and neck squamous cell carcinoma (SCCHN), squamous cell esophageal cancer, adenocarcinoma esophagus, junction and stomach HER2- PD-L1 cps ⩾5, stomach microsatellite instability–high (MSI-H)/mismatch repair deficient (dMMr) gastric cancer, MSI-H/dMMr colorectal cancer, MSI-H/dMMr small bowel cancer, hepatocellular carcinoma, MSI-H/dMMr biliary tract cancer, non-small cell lung cancer (NSCLC), small cell lung cancer (SCLC), skin squamous cell carcinoma, skin basal cell carcinoma, melanoma, Merkel cell carcinoma, mesothelioma, triple negative breast cancer (TNBC), MSI-H/dMMr endometrial cancer, cervical cancer, urothelial carcinoma, renal cell carcinoma, and Hodgkin lymphoma.3 -9
The use of ICI has rapidly entered clinical practice. 10 In Italy, in 2021, antineoplastic and immunomodulatory drugs represented the therapeutic category with the highest public expenditure (6,663,000 euro). Among them, ICI ranked first with a per capita cost of 12.33 euro and a compound annual growth rate of +50.5% from 2020 to 2021. 11
Although the evaluation of the theorical target population for ICI and the possible responders has been performed in the US and China,12 -14 to our knowledge no information is available for Italy.
The aim of this study is to estimate the proportion of Italian cancer patients diagnosed in 2020 who may be eligible for and responsive to ICI therapy according to the AIFA indications.
Materials and methods
We performed a cross-sectional analysis to estimate the number of Italian cancer patients who are eligible (according to the type of tumor and other specified criteria, e.g., stage, histological type, PD-L1 level, Hormonal Receptors and HER2 status, or MSI-H/dMMr) for AIFA-approved ICI.
This study was performed from July 2022 through April 2023 based on the AIFA drugs approval data by August 2022 and it follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.
As the denominator, we considered the cancer-specific estimates of incident cases (including skin epitheliomas) provided for 2020 by the Italian Network of Cancer Registries (Airtum). 15 For estimating specific histologic/biologic sub-types within tumor types (adenocarcinoma, squamous, NSCLC, SCLC, TNBC, Merkel cell) we relied on the assumptions (and the supporting references) listed in the Online Supplementary Materials. For example, NSCLC and SCLC were considered as the 85% and 15% of all lung cancers, according to the American Cancer Society. 16
As regards patients eligible for ICI, considering that the majority of ICI indications refer to advanced or metastatic cancers we used deaths for specific cancers as a proxy for metastatic cases at incidence. This is an approximation considering that deaths may be due to metastasis at diagnosis but also to the progression to metastasis of cases diagnosed in less advanced stages. In Italy, in fact, there are no national data on either condition, except for an anecdotal estimate. 17 The known difficulties with the registration of tumor stage at diagnosis apply especially to the M status, because initial staging of cancer patients takes place in a variety of healthcare facilities with unconnected information systems. Post-treatment follow-up data are even more dispersed, which makes the registration of progression and recurrences virtually impossible. Therefore, we applied the official observed number of citizens who died due to distinct cancers in Italy in the last year available (2020), 18 when available (ISTAT), or on assumption based on Airtum19,20 or other population-based data. 21
Concerning specific indications, we did not perform a formal revision of the literature for each combination cancer-marker, but searched in PubMed for the terms: type of cancer + metasta*+ type of marker and preferred information coming from Italy,19,22 when available, rather than foreign countries,16, 23 -37 and from population-based19,22,33,34,38 rather than clinical datasets,16,23 -32,35 -38 as well. Further details can be found in the Online Supplementary Materials. For example, regarding cervical cancer, pembrolizumab may be used for recurrent or metastatic cancer with PD-L1 CPS ⩾ 1. Therefore, we considered the number of deaths due to cervical cancer (as a proxy for metastatic cases) to which we applied the proportion of PD-L1 CPS ⩾ 1 reported by Steiniche et al. 38 Or, for skin melanoma pembrolizumab is authorized not only for metastatic tumors but also for those staged IIB, IIC, and III. Therefore, we applied the stage specific (IIB-IV) proportions observed in an Italian population-based series (18.5%) 22 to the estimated number of melanoma incident in 2020 (n. 14,863).
To estimate the potential benefit of ICI therapy, we defined a patient respondent based on the best available observed response rate (including complete or partial response) (ORR) reported by AIFA for each specific ICI indication3 -9 (further details in Online Supplementary Materials). This provided a cancer-specific number of patients who may derive advantage, in terms of complete or partial response, from ICI therapy.
These methods follow those applied in similar previous studies carried out in the US and China.12 -14,39
We reported the number and proportion (with 95% confidence intervals) of cases eligible for and responsive to ICI therapy, within each specific cancer.
The study is a secondary analysis based on publicly available data; therefore, it was not submitted for the evaluation to an Ethical Committee.
Results
Table 1 shows the estimated numbers of patients with a cancer diagnosis in 2020 in Italy who are hypothetically eligible for checkpoint inhibitor treatments.
Cancer sites and type for which some checkpoint inhibitor (ICI) drug is authorized, estimated number of incident cases (in 2020) and estimated number and percentage of patients whose cancer fit, or not, the eligibility criteria.
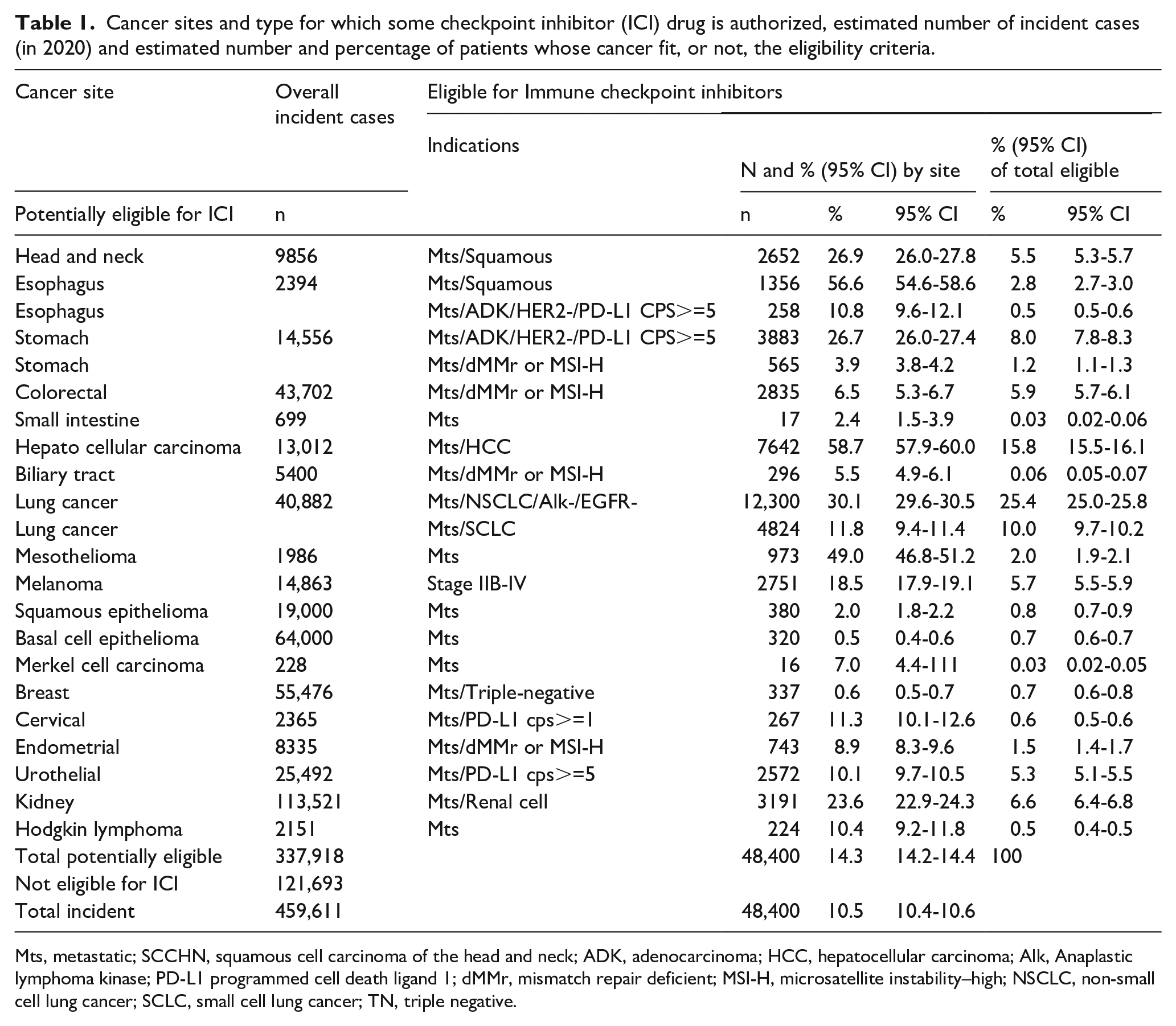
Mts, metastatic; SCCHN, squamous cell carcinoma of the head and neck; ADK, adenocarcinoma; HCC, hepatocellular carcinoma; Alk, Anaplastic lymphoma kinase; PD-L1 programmed cell death ligand 1; dMMr, mismatch repair deficient; MSI-H, microsatellite instability–high; NSCLC, non-small cell lung cancer; SCLC, small cell lung cancer; TN, triple negative.
Overall, 337,918 cancer patients (73.5% of the 459,611 newly diagnosed) suffered from a type of tumor for which ICI may be used in case stage, histology and/or specific markers fit the AIFA indications. Among them the estimated number of patients with such characteristics and theoretically eligible for ICI was 48,400 (14.3%; 14.2-14.4), that is 10.5% (10.4-10.6) of all the malignant tumors in 2020, or 46.6% (46.3-46.9) of the deaths due to theoretically ICI-specific cancer types (103,766).
According to the eligible patients the indications that most contributed to the eligibility estimate were NSCLC (25.4%), hepatocellular carcinoma (15.8%) and SCLC (10.0%).
Within a single cancer the highest proportions of patients eligible for ICI therapy were: esophagus (56.6% squamous cell and 10.8% adenocarcinoma), hepatocellular carcinoma (58.7%), mesothelioma (49,0%), and lung (30.1% NSCLC and 11.8% SCLC), and 26.7% for stomach (adenocarcinoma); for the other cancers the proportions varied from 26.9% for head and neck cancer to 0.5% for basal cell skin carcinoma.
In Table 2, for each cancer for which ICI use is authorized, the best overall observed response (ORR) in AIFA evaluated trials is presented.
Cancer sites and type for which some checkpoint inhibitor (ICI) drug is authorized, estimated number of eligible cases in 2020, best overall response rate (ORR) in AIFA-authorization trials and estimated number and percentage of patients who may benefit from ICI therapy.
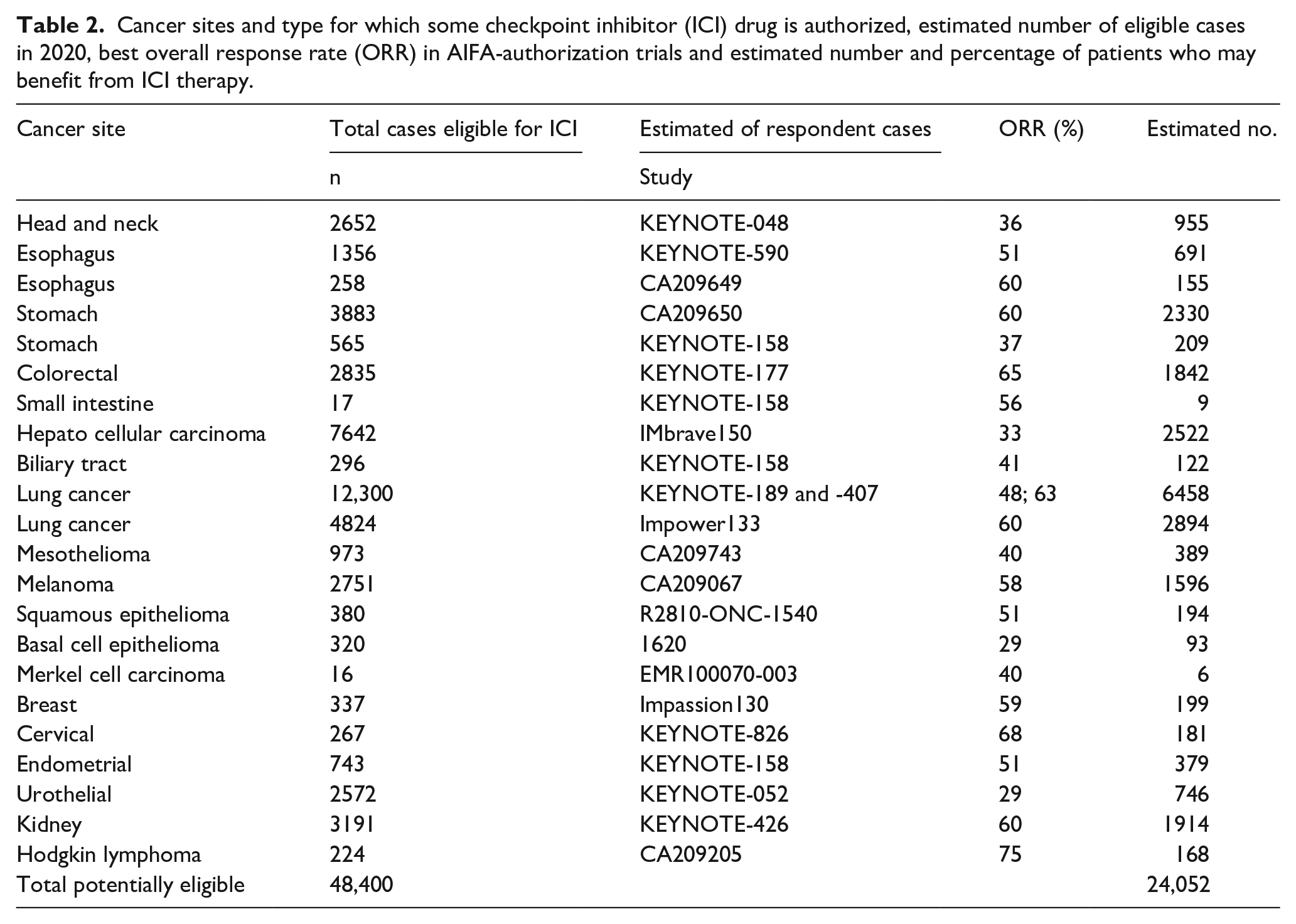
The number of cancer patients who may benefit for ICI therapy is 24,052, 49.7% (49.3-50.1) of all patients eligible for ICI, or 5.2% (5.2-5.3) of the estimated overall cancers in 2020 in Italy. The cancers for which there was the highest number of respondents were NSCLC, (n. 6458 either squamous and non-squamous, 26.9% of the total), SCLC (2894, 12.0%), hepatocellular carcinoma (2522, 10.5%), stomach cancer (2539, 10.6%), and renal cell carcinoma (1914, 8.0%), see Table 2.
Discussion
The use of checkpoint inhibitors is rapidly diffusing due to their potential benefit, even in metastatic disease. 40 Also, in Italy ICI use is speedily growing; for example, the number of ICI defined daily doses (DDD) has increased by 33.8% from 2020 to 2021 (in 2021 DDD/day = 0.3/1000 inhabitants). 11
Our study showed that the ICI approved in Italy could – in theory – be applied to almost 50,000 patients affected by the cancers included in the AIFA indications, that is 10.5% of around 460,000 estimated overall newly diagnosed cancers, including skin epitheliomas, in 2020. 15
In Italy AIFA produces an annual drug consumption report full of information, that employs standardized measures, e.g., DDD/1000 inhabitants or cost per inhabitants, for allowing comparisons among single and groups of drugs. 11 Unfortunately, the number of drug users, including ICI, is not provided. Moreover, ICI are among the drugs which have a more personalized dosing based on individual patient and cancer factors, including patient’s response and tolerability to the medication. Therefore, we were unable to compare estimates with published data on their DDD consumption and cost.
Referring to ICI-eligible patients, the proportion is 46.6%, close to those reported in China, 55.2% 14 and in the US 43.6%. 12
The type of cancer mattered, following their incidence and prevalence of regulatory indications (e.g., stage, histology, bio-markers). According to a similar evaluation carried out in the US and in China,12,14 the three cancers which contributed the most were NSCLC, hepatocellular carcinoma, and SCLC.
In Italy we estimated that about 24,052 patients may benefit from ICI-therapy, 5.2% of all the incidents of cancer in 2020. This proportion, if reported as deaths due to ICI-eligible cancers, represented 23.2%, comparable to the estimates from China, 19.2%, 14 and the USA, around 12.5% in 2018. 12 However, such comparison should be considered just for the order of magnitude due the different number of ICI authorized in US (6) 12 and China (11) 14 at the time the studies were carried out, respectively. The unavailability of complete tumor stage data suggests further caution when comparing Italy with these countries.
Our estimates are prone to a certain amount of inexactness; therefore, they have to be considered just for the order of magnitude they provide. In particular, miscalculations may come from both over and under estimations in relation to related assumptions.
In fact, we were not able to measure the actual individual uptake of ICI. Therefore, to estimate patients eligible for ICI treatment we relied on a number of assumptions aimed at translating into the real-world the drug-specific indications of AIFA. When available we counted on reliable information grounded on the Italian network of cancer registries (Airtum). Italian population-based cancer registries belonging to Airtum passed a formal data quality check, and they are also included in International Agency for Research on Cancer publications (https://ci5.iarc.fr/Default.aspx) and European Cancer information system (ECIS) of the European Commission (https://ecis.jrc.ec.europa.eu). The cancer type/sites involved in ICI therapy were generally estimated on Airtum data19-20 or on other population-based cancer registries data both Italian 17 or from US (SEER Surveillance and end results program).
Italian cancer registries, like many others, do not extensively collect information on predictive bio-markers as PD-1/PD-L1, EGFR, ALK, or dMMr/MSI-H. Therefore, we had to use data from the literature 22 -38 (see Online Supplementary Materials) taking a chance of possible individual, tumor and technical variabilities. 41
ICI are usually authorized for advanced or metastatic cancers, therefore we used, according to US and China authors,12 -14 the number of cancer deaths provided by the Italian Institute of Statistics as a proxy of incident metastatic cases. This assumption may have caused a not negligible underestimation of the eligible cases. In fact, we did not consider cases diagnosed in a less advanced stage and then progressed to diffusion in the year under evaluation (2020) and, especially, the prevalent metastatic cases surviving from previous years. For breast cancer the difference between deaths and incident metastatic cases is about -18%, but it is 3-times greater for prevalent metastatic cases. 17 Moreover, advanced, but not yet metastatic cases (indication for many ICIs) may be not considered using deaths as a proxy.
On the other hand, some of the assumptions may have overestimated our numbers. For example, we did not exclude young patients (who are not included in several ICI indications), or we did not consider that some ICI are not authorized as first-line therapy, but only after previous treatments (surgery, chemotherapy, autologous stem cell transplantation, etc.). Besides, we did not make allowance for exclusions of subjects not or barely represented in clinical trials, like patients with autoimmune diseases, hematopoietic/solid organ transplant, long-term immunosuppression, chronic viral infections, organ dysfunction, old age, or brain metastases. 42 Finally, the use of ICI is associated with a high proportion of toxicity that may cause discontinuation of the therapy. 40 Not having included this assumption may have contributed to the inflation of our estimates. Furthermore, the number of patients who could benefit from ICI may be overestimated by the ORR from clinical trials, presumably higher than that in the real-world setting.
In the meantime, new drugs targeting additional immune checkpoints are under study, 43 the diffusion of ICI in Italy is growing incredibly, for example the Defined Daily Dose of ICI almost doubled from 2020 to 2021, from 3.4 to 6.5 million. 11 Considering the growing trend in the use of ICI it is reasonable to expect a corresponding increase in spending. In the meantime, the necessary evidence is being collected on the cost-effectiveness of the various ICI treatments also in real world Italy.44 -48 However, it still only covers some indications. Therefore, even if some specific indications appear economically advantageous,44 -48 these therapies must be guaranteed to all patients who can benefit from them. In this challenge, the estimates provided in the present research can contribute to the evaluation of the sustainability of these therapies for all cancer patients.
Conclusions
The results of the present study may be useful for oncologists, policy makers, and any other stakeholder, in the debate about ICI, that includes the enthusiasms for the expected results but also doubts about their toxicity,2,49 their effective real-world benefit 12 and the sustainability of their costs. 50
Supplemental Material
sj-pdf-1-tmj-10.1177_03008916241229649 – Supplemental material for How many Italian cancer patients are eligible for, and may respond to, Italian Medicines Agency-approved checkpoint immune inhibitors?
Supplemental material, sj-pdf-1-tmj-10.1177_03008916241229649 for How many Italian cancer patients are eligible for, and may respond to, Italian Medicines Agency-approved checkpoint immune inhibitors? by Emanuele Crocetti, Alessandra Ravaioli, Fabio Falcini, Rosa Vattiato, Silvia Mancini, Flavia Baldacchini, Federica Zamagni, Benedetta Vitali, Chiara Balducci, Lauro Bucchi and Orietta Giuliani in Tumori Journal
Footnotes
Author contributions
Conceptualization, E.C.; Methodology, E.C., A.R., O.G., F.Z.; Formal Analysis, A.R., O.G., F.Z., S.M.; Data curation: R.V., S.M., C.B., F.B.; Writing – Original Draft Preparation, E.C.; Writing – Review & Editing, L.B., A.R., O.G., R.V., S.M., F.Z., B.V., C.B. F.B., F.F. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported thanks to the contribution of Ricerca Corrente by the Italian Ministry of Health within the research line “Appropriateness, outcomes, drug value and organizational models for the continuity of diagnostic-therapeutic pathways in oncology” (ID: Wfn.75L1).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
