Abstract
This study quantified the incidental dose to the first axillary level (L1) in locoregional treatment plan for breast cancer. Eighteen radiotherapy centres contoured L1-L4 on three different patients (P1,2,3), created the L2-L4 planning target volume (single centre planning target volume, SC-PTV) and elaborated a locoregional treatment plan. The L2-L4 gold standard clinical target volume (CTV) along with the gold standard L1 contour (GS-L1) were created by an expert consensus. The SC-PTV was then replaced by the GS-PTV and the incidental dose to GS-L1 was measured. Dosimetric data were analysed with Kruskal-Wallis test. Plans were intensity modulated radiotherapy (IMRT)-based. P3 with 90° arm setup had statistically significant higher L1 dose across the board than P1 and P2, with the mean dose (Dmean) reaching clinical significance. Dmean of P1 and P2 was consistent with the literature (77.4% and 74.7%, respectively). The incidental dose depended mostly on L1 proportion included in the breast fields, underlining the importance of the setup, even in case of IMRT.
Introduction
The relationship between the unintentional dose to the axillary levels and the outcome, in terms of toxicity and regional control, in the radiation treatment (RT) of breast cancer (BC) has been a subject of investigation of growing interest, 1 following the publication of the Z0011 trial. 2 The low incidence of axillary nodal failure in the above mentioned and other similar trials3,4 suggested that tangential fields (TFs) for the whole breast RT (WBRT), alongside systemic therapy, contributed to the handling of positive non-sentinel lymph nodes (SLNs), if any, in the undissected axilla. Although the quantification of incidental dose to the axilla is hindered by the paucity of RT technical parameters for dosimetric correlations, it has been investigated in small, dedicated reports or in studies reproducing the condition of treatment of the landmark BC trials.5-10 The SENOMAC trial, which has recently closed the accrual, challenged the need to irradiate L1 for slight sentinel node involvement and recommended limiting the regional RT field to L2, L3 and L4, regardless of the arm of randomisation. 11 In the locoregional RT, L1 is expected to receive a certain amount of the dose from the contribution of the breast/chest wall and the infra/supraclavicular fields, especially when this latter includes the whole or part of the second axillary level (L2). 12 This study aimed to quantify the unintended dose to L1 in the setting of increased anatomical complexity in intensity modulated radiotherapy (IMRT)-based locoregional treatments, using a multicentric platform, which served as basis for three previous investigations,13 -15 with the endorsement of the Italian Association of Radiotherapy and Clinical Oncology (AIRO).
Patients and methods
The dataset of three representative BC patients (P) with increasing anatomical complexity (P1 with clear anatomy, P2 with obesity and P3 with impaired arm mobility) treated at the coordinating centre (IEO, European Institute of Oncology IRCCS, Milan, Italy), was used to investigate the unintentional dose to L1 in the context of locoregional RT. Here follows a brief description of the selected patients:
- P1 low level of complexity (right BC); underwent breast conserving surgery and sentinel node biopsy. lymphatic drainage was basically intact. Arm abduction set-up was >90°. Body mass index (MBI) was 27.
- P2 medium level of complexity (right BC) underwent skin sparing mastectomy and axillary dissection with less than 10 nodes removed. She had immediate breast reconstruction with an expander and suffered from obesity (BMI 48). Arm abduction set-up was >90°.
- P3 high level of complexity (left BC) underwent total mastectomy and axillary node dissection, with microscopic disease left behind in axilla. She had difficulty in lifting the arm above her head because of neuropathy. Set-up was the same as the two above patients, but the arm was kept at <90°. BMI was 30.
The methodological procedure was the following. The expert radiation oncologists (ROs) of the single centres (SC) delineated the L1 contour and the clinical target volume (CTV) of L2 to L4 t (namely, SC-CTVs), 13 which was expanded according to their clinical practice to create the planning target volume (PTV, single centre: SC-PTV) and a loco-regional treatment plan was performed.
The corresponding gold standard (GS) contours for each axillary level of the three patients were created as shown in a previous paper. 14 The SC-CTV including L2-L4 was replaced by the corresponding GS-CTV and then expanded to form the GS-PTV using the same margins as those used by each RT centre. 15 Likewise, to evaluate the incidental dose to the GS-L1, the SC-L1 contour was replaced with the GS-L1. To take into account the different fractionation schemes used by the participating centres, the equivalent total dose in 2-Gy fraction (EQD2) using the alfa/beta ratio of 4 calculated for all the analyzed plans. All plans were imported in DICOM format into MIM software (version 6.1.7) and dose-volume histograms (DVHs) were extracted for L1. The study was conducted within the research project on intensity-modulated RT (IMRT) and hypofractionation on BC notified to the Ethical Committee of the IEO-IRCCS (26 May 2016, Milan, Italy), the patients gave the consent for use of their CT scans for research purposes.
Statistical analysis
For the dosimetric evaluation, dose-volume-histograms were analysed. The following parameters relative to L1 volume were extracted: mean dose delivered (Dmean, %, Gy), dose delivered to 95% of the volume (D95, %, Gy), near maximum absorbed dose (Dnear-max = D2, %, Gy), and the volume receiving 95% of the prescribed dose (V95%). Median value, min-max range and standard deviation are reported for continuous variables.
The non-parametric Kruskal-Wallis test was applied in order to investigate whether the dosimetric parameters differ significantly among the considered patients. Significance level was set to 0.05.
Results
All plans but one were performed with IMRT techniques using either forward- or inverse-planning or arc volumetric RT. Fifteen out of 18 centers used a conventional fractionation (45-50 Gy in 25 fractions) while the remaining ones adopted a moderate hypofractionation (40.05 Gy – 47.25 in 15-21 fractions).
Out of 54 plans, four were excluded for technical reasons. The first axillary level (L1) percentage dose for the selected planning objectives for the three patients is shown in Figure 1.
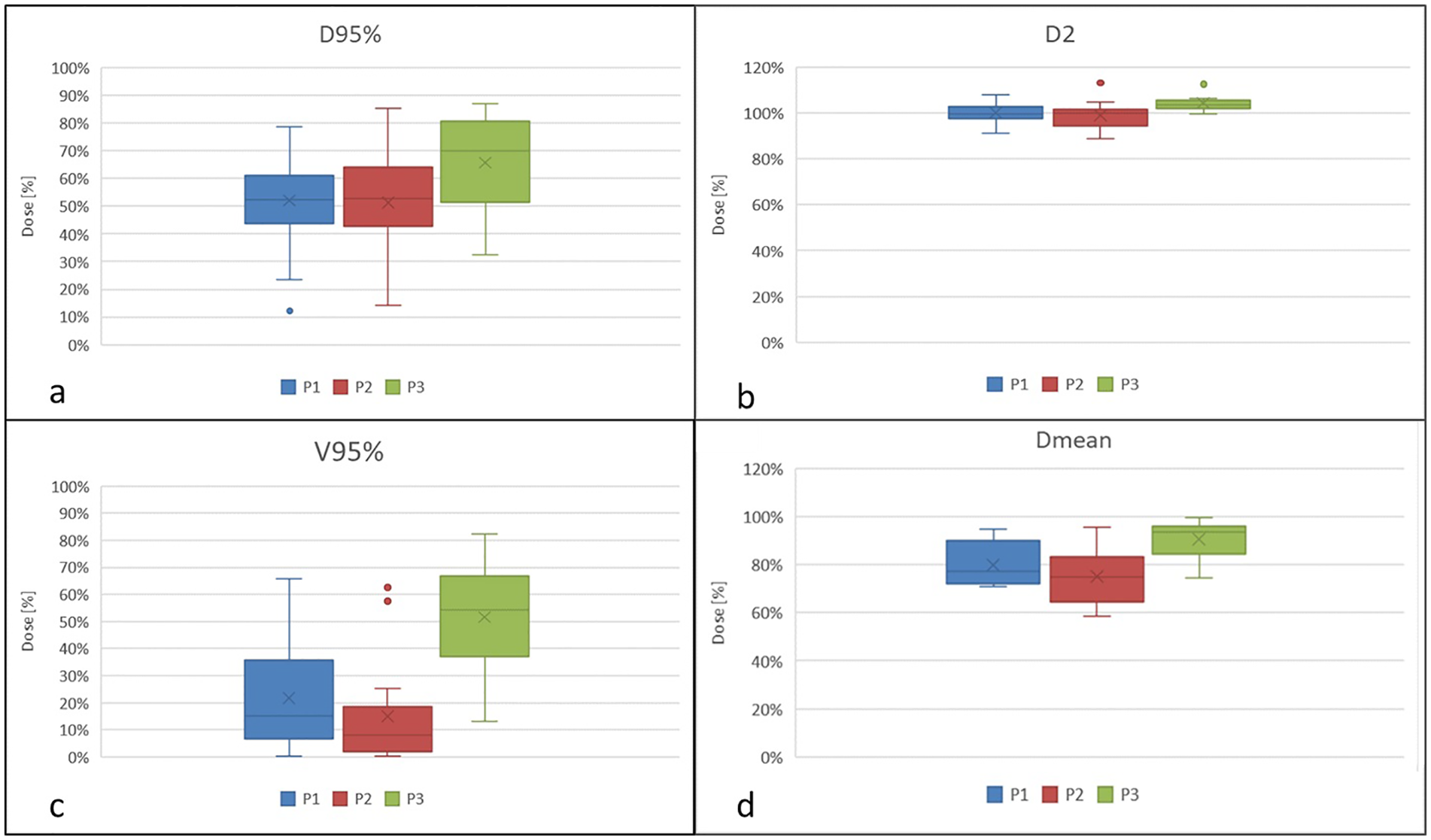
First axillary level (L1) percentage dose in terms of near-min dose (D95, a), near-max dose (D2, b), V95% (c), and mean dose (Dmean, d) for the three case patients.
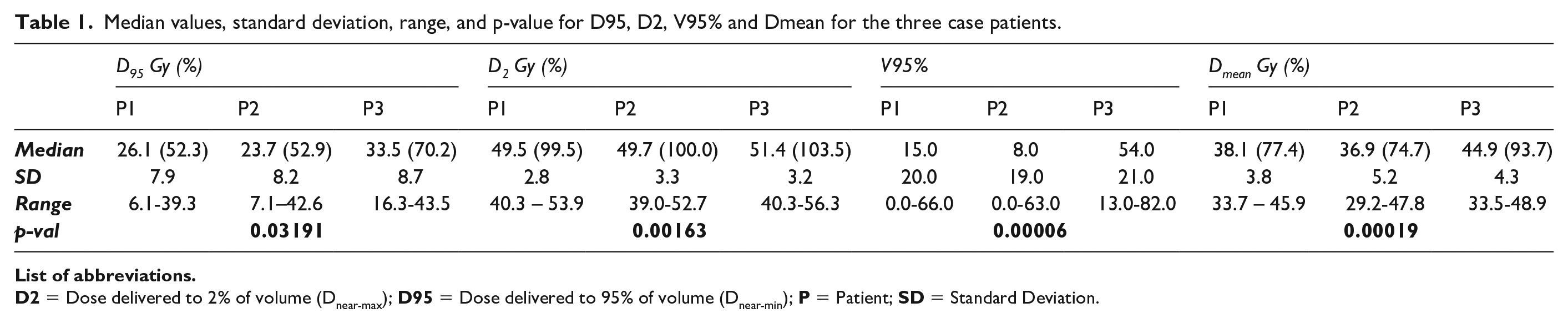
Dose to L1 resulted statistically significantly higher for the patient with impaired arm mobility (P3) considering D95 (p=.032), D2 (p=.002), V95% (p=.00006) and Dmean (p=.0002). All the results are reported in Table 1. D95, Dmean and V95% did not significantly statistically differ between the non-obese-P1 (BMI 27) and the obese P2 (BMI 48).
Median values, standard deviation, range, and p-value for D95, D2, V95% and Dmean for the three case patients.
Discussion
This study aimed at reporting real-world data on the incidental dose to L1 in locoregional IMRT treatments using a multicentric platform and patients with different anatomical complexity. Results showed that the unintended dose to L1 was mostly influenced by the unconventional arm set up of the patient with limited arm mobility while the BMI played a negligible role in dose variations.
In literature there is paucity of data reporting the contribution of the third field to the incidental dose to the lower axilla in the locoregional RT.9,10,16 The third field typically includes L3 and L4, while L2, as a whole or in its half medial part, is left to the ROs’ discretion, according to the national or institutional guidelines. 12 In the current study, the whole L2 was considered part of the nodal CTV, as required by the SENOMAC trial protocol. 11 The SENOMAC trial randomised patients with 1-2 macrometastatic SLNs to either completion axillary lymph node dissection (ALND) (addressing L1 and L2) or no further axillary surgery: the participating centres were explicitly required not to change RT regardless of the treatment arm the patient was randomised to, because only SLN biopsy (SLNB) should be regarded as a substitute for ALND. Therefore, L1 was omitted from the nodal CTV. In the present study, the median value of the mean lateral margin given to create the L2-L4 PTV was 5 mm, resulting in a certain proportion of L1 to be encroached upon by the CTV-to-PTV expansion. 17 Therefore, it might be expected that the unintentional dose to L1 is higher than that reported from the TFs of breast/chest wall RT with implications on both local control and arm toxicity.17-19 In a simulation of the axillary dose distribution according to different treatment plans, the authors 10 found that the V90% and the Dmean delivered to L1 with the high TFs were as high as those delivered in the AMAROS study, which specifically targeted L1, with V90% being 79.5% and Dmean ⩾45 Gy. Conversely, for the infra-supraclavicular field (L2-L4), the L1-V90% was about 39.4% and Dmean was about 35 Gy, which was similar to the Dmean values of 36.8-38.6 Gy found in the present study for P1 and P2. The Dmean of 44.9 Gy for P3, with the arm set up at 90°, was comparable to the Dmean observed using high TFs in the study by Wang et al. 10 In both cases, a larger proportion of L1 remained included in the breast/chest wall target volume when the upper border of the relative RT fields drew near the humeral head. In another comparative study, 9 the authors found that the amount of incidental dose depends on the TFs’ height and the body shape (lower values for small breast and slender patient). Other authors showed that BMI influenced the amount of unintended dose to L1, which was significantly higher in obese women. 8
In the current study, the unintended dose to L1 was similar between P1 with BMI of 27 and the obese P2 with BMI of 48. Axillary maximum dose D2 resulted statistically significant different across the three patients, but it was not associated with obesity, as also reported by Hildebrandt et al. 8
While it is expected that a dose near to that of prescription can exert adequate control alongside systemic therapy, it can be argued that even lower doses to the inferior part of the axilla are sufficient to handle small tumor burden. However, it is difficult to identify a threshold since the incidental dose to L1 is subject to a great variability, depending on several anatomic and technical factors.1,19,20 In addition, in the light of an increasingly effective and targeted systemic therapy, it cannot be excluded that doses considered non tumoricidal can actually have a killing effect on microscopic disease.
Among the strengths of the study, the multicentric participation provided a glimpse of real-world clinical practice, using IMRT- based techniques. However, it must be stressed that the study findings cannot cover the great variability of the incidental dose to L1.
As L1 was never intended to be part of the nodal CTV, the interobserver topographic variability of L1 contouring was not analysed, but instead, the unintended dose was directly measured on the gold standard L1, which was considered more reliable in characterising the true volume of interest. The study did not collect the DVHs of organs at risk, therefore no correlation with L1 dose could be made. Different techniques could not be compared as all plans but one were carried out with IMRT.
The study results showed that targeting the upper axilla and the supraclavicular fossa seems not to substantially contribute to a clinically significant increase of L1 incidental dose, which relies mostly on the breast/chest wall RT fields, underlining the importance of the setup, even in case of highly conformal technique.
Footnotes
Acknowledgements
We would like to thank Simone Giovanni Gugliandolo, Johannes Lars Isaksson, and Delia Ciardo and the following collaborators of the AIRO Breast Working Group: Maria Rosa La Porta, Edoardo Petrucci, Lorenza Marino, Edy Ippolito, Alessandra Huscher, Angela Argenone, Fiorenza De Rose, Francesca Cucciarelli, Maria Carmen De Santis, Francesca Rossi, Agnese Prisco, Roberta Guarnaccia, Paola Tabarelli de Fatis, Sarah Pia Colangione, Maria Mormile, Vincenzo Ravo, Alessandra Fozza.
The Authors thank the Scientific Committee and Board of the Italian Association of Radiotherapy and Clinical Oncology (AIRO) for the critical revision and final approval of the manuscript (Nr. 3/2023)
Declaration of Conflicting Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Division of Radiotherapy IEO received research funding from AIRC (Italian Association for Cancer Research) and Fondazione IEO-CCM (Istituto Europeo di Oncologia-Centro Cardiologico Monzino) (all outside the current project). BAJF received speakers fee from Roche, Bayer, Janssen, Carl Zeiss, Ipsen, Accuray, Astellas, Elekta, IBA Astra Zeneca (all outside the current project). MCL received a speaker fee from Accuray Inc (outside the current project). SD received speakers fee from Accuray Asia (outside the current project). MGV was supported by a research fellowship from AIRC entitled “Radioablation ± hormonotherapy for prostate cancer oligorecurrences (RADIOSA trial): potential of imaging and biology”, registered at ClinicalTrials.gov NCT03940235, approved by the Ethics Committee of IRCCS Istituto Europeo di Oncologia and Centro Cardiologico Monzino (IEO-997). IEO, the European Institute of Oncology IRCCS, was partially supported by the Italian Ministry of Health with Ricerca Corrente and 5x1000 funds and by institutional grants from Accuray Inc. The sponsors did not play any role in the study design, collection, analysis and interpretation of data, nor in the writing of the manuscript, nor in the decision to submit the manuscript for publication. The remaining authors declare no conflict of interest that are relevant to the content of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
