Abstract
This study aimed to evaluate the efficacy of external fenestration using a reserved guide wire and a Viabahn-covered stent in Stanford type B aortic dissection that involves the left subclavian artery. Eight patients with Stanford type B aortic dissection involving the left subclavian artery were treated from March 2023 to May 2024 at Yinzhou People’s Hospital using a reserved guide wire external fenestration and a Viabahn-covered stent. This study compared a novel external fenestration technique (reserved guide wire + Viabahn stent) with traditional in-situ fenestration for Stanford type B aortic dissection involving the left subclavian artery. Clinical data showed that the new method achieved full technical success in vessel reconstruction, with no postoperative deaths, minimal complications, and shorter hospital stay versus the conventional approach. The fenestration complication incidence was also significantly reduced during a follow-up period of 1–3 months. Studies on larger samples can be performed in the future to verify the present results. External stent fenestration with a reserved guide wire demonstrated significant efficacy in treating large-branch artery disease, resulting in substantial improvements in prognosis in terms of complications. Therefore, this technique is worthy of further exploration.
Keywords
Introduction
Endovascular technology and instruments are constantly advancing. Common methods that are currently used for aortic dissection (AD) that involve the left subclavian artery (LSA) during thoracic endovascular aortic repair (TEVAR) surgery for the reconstruction of the aortic arch branches include chimney, in-place fenestration, in vitro fenestration, and fenestration +branch vascular stenting. 1 Reconstruction of the three aortic arch branches is often performed for obtaining a sufficient anchoring area during TEVAR.2,3 The preservation and reconstruction of the brachiocephalic trunk and the left common carotid artery (LCCA) are indisputable; however, the decision regarding closure and reconstruction of the LSA needs to be taken based on each patient’s condition. There is a risk of type I endoleak due to the difficult angle between the LSA and the aortic arch, and debris loss and embolization may occur during traditional fenestration. 4 This study addresses the critical need for safe and efficient branch artery isolation in TEVAR by introducing a reserved guide wire-assisted external fenestration technique combined with the use of Viabahn-covered stents. The reserved guide wire simplifies fenestration by aiding precise stent positioning and alignment, while Viabahn-covered stents enhance patency and reduce complications. Post-deployment balloon dilation optimizes branch vessel integration, ensuring seamless fusion with the fenestrated site. This approach prevents stent framework interference, preserves branch patency, and facilitates subsequent stent placement. By integrating the advantages of both in vitro and in-situ fenestration, this method offers a standardized solution for complex aortic pathologies, particularly Stanford type B dissections involving the LSA.
Materials and methods
Clinical symptoms
The onset of AD is often rapid; patients commonly experience tearing or knife-like chest pains and exhibit a painful face, irritability, and rapid pulse rate, often with elevated blood pressure; however, these typical symptoms are not present in all patients. Patients who do not experience the typical chest and back pain are often regarded as having acute coronary syndrome.5,6 The manifestations of AD vary according to the scope of involvement. If a Stanford type B AD (TBAD) that involves the subclavian artery is performed, it may cause disorders of the brain blood supply, coma, confusion, and other manifestations. 7
Currently, many imaging examinations are available for diagnosing Stanford TBAD that involves the LSA. Computed tomography (CT) has become the preferred screening method for suspected patients due to its near-perfect sensitivity and very high specificity. 8 Computed tomography angiography (CTA) can easily detect the Stanford TBAD rupture location and evaluate the branch contorts in the bow involved in the situation. The diagnostic accuracy of magnetic resonance imaging (MRI) for Stanford TBAD is similar to that of CT. However, a longer duration is required to acquire MRI images in patients with dissection, which makes it unsuitable for patients with acute AD.9,10 Thus, in the management of Stanford TBAD involving LSA, CTA is the optimal method for identifying the location of the dissection and detecting LSA involvement.
The novelty of this study lies in precisely locating the LSA opening and avoiding stent coverage of the branch. Combined with the flexibility and bioactive surface of the Viabahn-covered stent, the reserved guide wire using the extracorporeal windowing technique can effectively reduce the risk of endoleak and promote long-term patency of the branch vessels, especially in cases with complex anatomy in the arch of the aorta or severe stenosis in the true lumen. This technique achieves LSA revascularization through minimally invasive intervention, preserving branch function while reducing the risk of endoleak and cerebral infarction complications associated with traditional chimney techniques.
Disease progression of Stanford TBAD involving the LSA
Approximately 25%–40% of all AD patients undergo Stanford TBADs. Type B AD can be divided into complex and simple cases according to whether patients have comorbidities. Complex AD patients have higher near-term mortality rates than those with the simple AD type B. Intracavitary therapy for type B AD patients with insufficient proximal anchoring areas often needs to cover the LSA. Previous studies have shown that the aortic anchoring area can be effectively prolonged after LSA coverage. 11 The LSA generally originates directly from the aorta, and it maintains the blood supply to the left upper limb, posterior cerebral circulation, and spinal cord perfusion. Therefore, LSA without revascularization may increase postoperative complications (e.g. left upper limb ischemia, stroke, and spinal cord ischemia) and increase TEVAR mortality. 12
Stanford TBAD involves LSA treatment methods
The largest challenge in endovascular treatment is that important branches of the aortic arch that include the inconspicuous trunk artery, LSA, LCCA, right vertebral artery, and vagus right subclavian artery need to be preserved during dissection. An analysis of the reconstruction of the aortic arch branches showed that extracorporeal fenestration offers the advantages of simplicity, economy, and minimal invasion. Therefore, the reserved guide wire in vitro fenestration and Viabahn-covered stent technology was further improved on this basis.
Patient data
In total, 18 consecutive thoracic aortic disease patients who met the preoperative admission criteria were divided into the application of the new technology group, namely, in vitro fenestration with reserved guide wire and Viabahn-covered stent technology (eight cases), and the traditional in-place fenestration group (10 cases). Data on sex, age, hypertension status, and other underlying diseases were collected for all the enrolled patients. The new technology group contained four men and four women. The average age of the patients in this group was 55.8 years. There were six patients with hypertension, one with cerebral infarction sequela, and one with diabetes. All patients were diagnosed with Stanford TBAD that involved the LSA using preoperative imaging examinations that included reference of the general condition records, a specialist physical examination, and CTA (Table 1). This retrospective study used the following criteria for patient enrollment.
Preoperative baseline characteristics of the new technique and traditional in-situ fenestration groups.
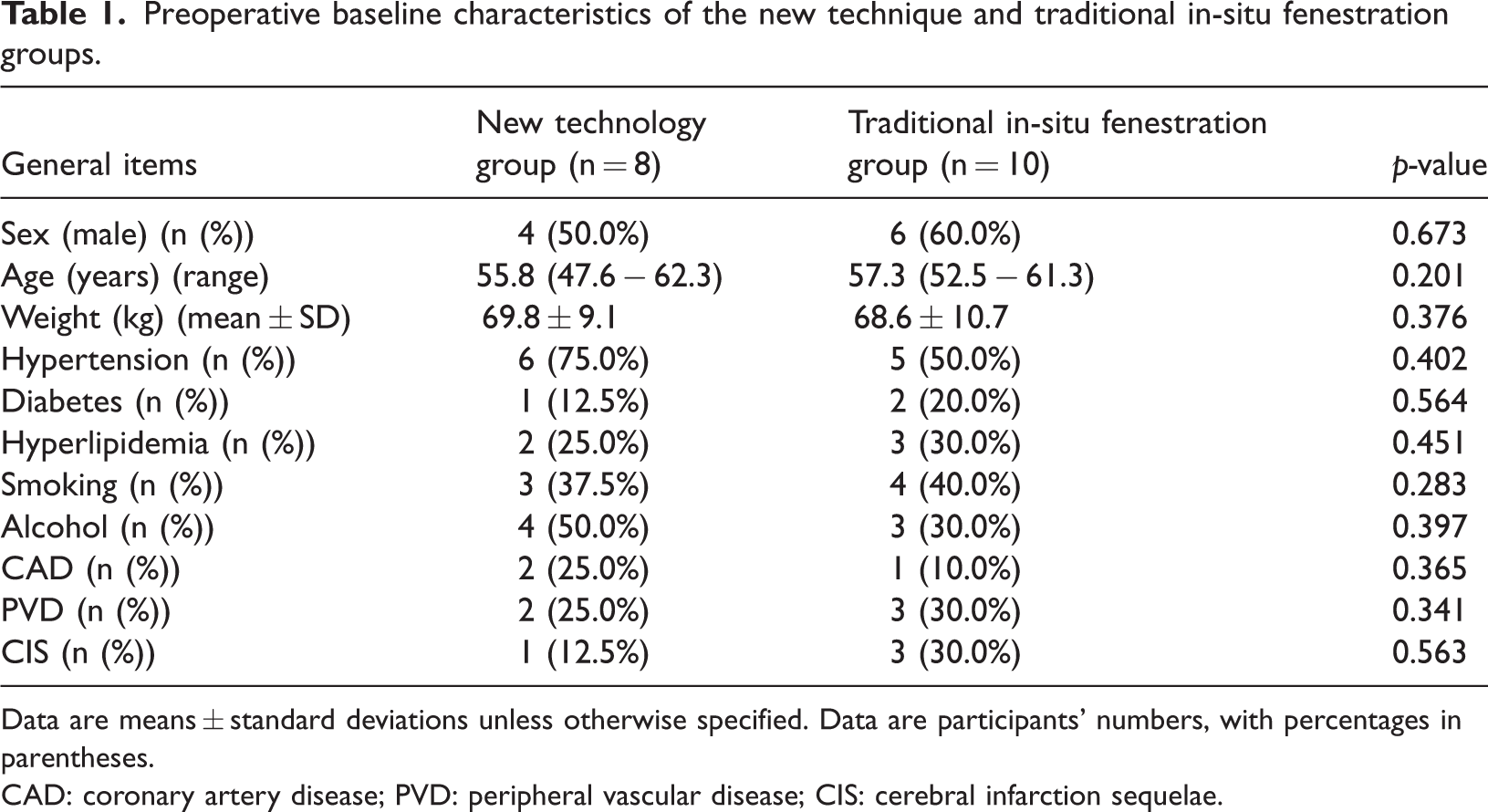
Data are means ± standard deviations unless otherwise specified. Data are participants' numbers, with percentages in parentheses.
CAD: coronary artery disease; PVD: peripheral vascular disease; CIS: cerebral infarction sequelae.
Selection criteria
Inclusion criteria
The inclusion criteria were as follows:
A patient was identified as having Stanford TBAD according to preoperative enhanced CT and CTA examinations. The preoperative CTA showed that the interlayer retrograde tear involved the LSA, and the interlayer intimal rupture distance was <15 mm. Absence of abnormal liver or kidney function. The AD scope involved at least one branch artery or the lesion scope did not involve the branch artery but still required reconstruction to ensure an adequate anchoring area.
Based on the above anatomic conditions, extracorporeal windowing techniques that preserve the LSA are preferred. In patients with persistent pain, poor organ perfusion, or rapid aortic dilatation in the acute phase, rapid, minimally invasive extracorporeal windowing is preferred for hemodynamic stabilization. The advantages of the reserved guide wire using the extracorporeal windowing technique combined with Viabahn stenting include precise localization, prevention of type I endoleak, and branch patency. Thus, the inclusion of a predominantly anatomical characterization-driven selection of indications is complemented by consideration of urgency and risk stratification as well as technical efficacy and complication trade-offs.
Exclusion criteria
The exclusion criteria consisted of the following:
The presence of a thoracic aorta-penetrating ulcer A thoracic aorta interwall hematoma or a thoracic aorta false aneurysm The presence of a severely narrowed and twisted iliac artery, abdominal aorta, or blood vessels in the diseased area that would all result in the failure of the delivery system where the reserved guide wire would be located Renal insufficiency Allergy to the contrast media Poor clotting function
Description of treatment methods
The treatment protocol for TBAD patients in this study consisted of the following four key components.
After intravenous administration of 5000 units of unfractionated heparin, fluoroscopic guidance confirmed catheter positioning within the true lumen. Imaging revealed a 1.2-cm proximal entry tear at the LSA with a stenotic true lumen. Under roadmapping assistance, the LSA and LCCA orifices were digitally marked on the fluoroscopic display. The left femoral artery sheath was exchanged for a super-stiff guidewire, which was advanced through the LSA sheath into the ascending aorta. After retrieving the exchange wire, the mean pulmonary artery pressure (MPA) catheter was removed using a snare device. With real-time recording of intravascular pressure data, the MPA catheter can be used for accurate positioning of vascular stenosis and hemodynamic evaluation in cardiovascular interventional surgery. A 34 × 26 × 200-mm stent graft was then partially deployed in vitro to create a 3-mm fenestration 1.6 cm proximal to its distal end (Figure 1(a)). The fenestrated segment was reintroduced into the delivery system, and the main stent graft was precisely aligned with the LCCA orifice (Figure 1(b)).
(a) An aperture featuring a 3-mm opening positioned 1.6 cm away from the proximal laminating stent. (b) The left subclavian artery reserved guide wire was placed 10 cm away from the opening window, and the stent was reintroduced into the sheath. (c) Left common carotid artery release and well-developed stent, with the main stent opening facing the left subclavian artery. The reserved guide wire was directed to the main descending stent, and the stent carrier was chosen. (d) A 12-Fr sheath was introduced into the aortic stent via reserved guide wire, followed by an 8-mm balloon to expand the stent; then, the sheath was advanced into the left subclavian artery for placing a 13 × 50-mm Viabahn stent. (e) DSA arteriography prior to endovascular isolation, (f) during endovascular isolation, and (g) after endovascular isolation. DSA: Digital subtraction angiography. Under controlled hypotension (systolic blood pressure (BP) <90 mmHg) conditions, the stent graft was deployed along the distal margin of the LCCA orifice, achieving 90% apposition to the aortic arch (Figure 1(c)). A 12-Fr long sheath was advanced through the fenestration into the true lumen, followed by the deployment of an 8 × 50-mm Viabahn-covered stent within the LSA. Final imaging confirmed that the Viabahn stent had a proximal 1.5-cm overlap with the main graft and distal preservation of the left vertebral artery (Figure 1(d)). Sequential balloon angioplasty (10 mm diameter) was performed at both fenestration site and Viabahn stent to optimize apposition and minimize the risk of type Ia endoleak. The key steps of the surgery are detailed in the analytical figure presented in Figure 2.
Aortic coarctation involving the left subclavian artery was treated with reserved guide wire using the extracorporeal windowing technique. The windowing window (1.6 cm from the distal end) was precisely positioned using preoperative CTA 3D reconstruction, the 34-mm main stent was partially released, extracorporeal windowing (3 mm × 1.2 cm) was accomplished with controlled hypotension (systolic blood pressure <90 mmHg), and a 12-Fr long sheath was used to implant a Viabahn 8 × 50-mm branch stent. The Viabahn 8 × 50-mm branch stent was implanted through a 12-Fr long sheath, and the final imaging shows that the main stent and branch stent overlapped by 1.5 cm, and the left vertebral artery was patent, which combined with balloon post-dilatation to reduce the risk of type Ia endoleak. CTA: computed tomography angiography; 3D: three-dimensional.


Hybrid surgery is suitable for patients with severe aortic arch distortion or other supra-aortic branch lesions (such as LCCA stenosis) and for those with rupture located <15 mm away from the opening of the LSA in whom surgical reconstruction of the LSA is required to extend the anchoring area. It is suitable for patients whose general health condition is good and who can tolerate general anesthesia.
The pre-fenestration technique is suitable for patients with natural aortic arch morphology, no severe angulation, easy and accurate identification of the fenestration location, rupture location ≥15 mm from the LSA opening, and ability to retain the LSA blood flow through in vitro fenestration. It is suitable for older patients with multiple complications (such as cardiopulmonary insufficiency) and those who cannot tolerate open surgery.
Perioperative management
Preoperative monitoring included 12-lead electrocardiogram (ECG) and arterial oxygen saturation (SpO2), with hemodynamic targets of heart rate <70 beats/min and systolic blood pressure <120 mmHg, achieved with the use of intravenous beta-blockers (e.g. metoprolol) and labetalol for hypertension control. Patients with severe chest/back pain (visual analog scale (VAS) score ≥6) received intravenous (IV) fentanyl (2–5 µg/kg) and midazolam (0.05–0.1 mg/kg) for analgesia sedation. The cumulative scope of dissection was accurately evaluated according to enhanced CT and CTA examinations of the patient. The proximal anchorage area was defined, and the diameters of the true and false lumens of the aorta as well as the distance between the AD break and left subclavicular artery were then measured. General anesthesia was administered in the operating room during the surgery. The patient’s vital signs were monitored postoperatively, and if chest pain was observed, the aortic CTA was re-examined, and warfarin anticoagulation was performed postoperatively. Regular aortic CTA was performed to evaluate the position of the stent and endoleak, and the embolism or organ ischemia was screened using ultrasound of the lower limb arteriovenous and renal function examination. The focus was on the risk of puncture point bleeding, stent displacement, and reverse tear A-type dissection.
Treatment effect evaluation and follow-up
The evaluation of treatment outcomes and patient follow-up were conducted based on the following three aspects.
Perioperative evaluation metrics
The evaluation was conducted using indices, including operation time, operation cost, fluoroscopy time, contrast agent use, and total hospital stay.
Postoperative follow-up metrics and stent assessment
Postoperative follow-up focused on stent leakage, stent morphological change, left upper limb ischemia, death, nerve injury, stent infection, and re-surgical intervention. Stent leakage was assessed using CTA or DSA to classify leakage type (type I: distal stent rupture leakage and type II: branch regurgitation) and measure leakage cavity parameters (maximum diameter ≥5 mm defined as significant leakage; volume ratio calculated as contrast leakage area relative to tumor cavity volume). Stent morphological changes were analyzed using imaging combined with three-dimensional (3D) reconstruction to identify localized depression, lateral offset (>2 mm), and overall distortion; intervention was prioritized if changes caused branch vessel compression (e.g. LSA stenosis >50%) or impaired true luminal flow (fractional flow reserve (FFR) <0.8).
Determination of remaining indicators
The remaining indicators were determined by integrating clinical symptoms and imaging findings. Within 3 months postoperatively, imaging should be rechecked every 1–2 weeks. Long-term follow-up requires maintaining blood pressure at <130/80 mmHg to reduce aortic remodeling. All patients in this study have completed follow-up.
Statistical analyses
Statistical analyses were performed using Statistical Package for Social Sciences (SPSS) software (version 24.0, SPSS Inc., Chicago, IL, USA). Different statistical methods were used for continuous variables (such as age, weight, and LSA diameter) and categorical variables (such as sex and treatment group). For continuous variables, we first evaluated their normality using the Shapiro–Wilk test. An independent sample t-test was used for continuous measurement data that conformed to a normal distribution, and data are expressed as mean ± standard deviation (SD) (x ± s) values. Non-normal data were analyzed using nonparametric tests such as the Mann–Whitney U test and expressed as median (lower and upper quartile) values. Categorical data are expressed as percentages. The discrete variables were tested using chi-square test or Fisher’s exact probability method. Statistical significance was defined as a two-sided p-value <0.05.
Results
General characteristics
A comparison of the data from the new technology and traditional in-situ fenestration groups showed no statistically significant differences in the sex, age, preoperative weight, and pre-existing diseases of patients (all p-values >0.05). Although there were proportional differences in some variables (e.g. a difference of up to 25% in the prevalence of hypertension), none of them reached the threshold of statistical significance, indicating that the baseline characteristics of the two groups were comparable, with no significant clinical imbalance. The baseline data of all enrolled patients are presented in Table 1.
Imaging features
Postoperative thoracic aortic angiography confirmed no significant leakage within the stent-covered area. The brachiocephalic trunk, LCAA, and LSA displayed continuous smooth visualization without stenosis or occlusion at anastomotic sites (Figure 1(e) to (g)). The true lumen diameter increased by approximately 30% compared with that before the surgery, while false lumen contrast retention shrank to a 5-cm segment in the lower thoracic aorta (Figure 3(a)). At the 3-month follow-up CTA, the stent graft had fully sealed the primary entry tear (1.2 cm distal to left subclavian ostium) with no residual extravasation. Quantitative analysis revealed a 60% expansion in the true lumen perfusion territory. The reserved guide wire window maintained submillimeter precision (<1 mm deviation), and the Viabahn stent showed stable positioning, with <10% lumen diameter discrepancy compared with the right subclavian artery (Figure 3(b) and (c)). Post-procedure vascular sheath removal was performed without bleeding complications, and stable hemodynamics were maintained throughout.

(a) Preoperative enhanced CT shows thoracic aortic coarctation, manifesting as a spiral tear in the intima-media sheet, with the true lumen narrowed by compression and the false lumen dilated in a cystic shape, involving the beginning of the left subclavian artery. The intima-media rupture is located on the side of the lesser curvature of the aortic arch, with peripheral appendage thrombus formation. (b) Postoperative plain CT shows that the main stent covers the diseased segment in a shuttle shape, and the stent at the opening of the left subclavian artery is in a grid-like structure, with the disappearance of the sign of contrast retention in the false lumen; the distal end of the stent is well adhered to the wall of the descending aorta with no new rupture or hematoma formation. (c) Postoperative CTA shows that the true lumen of the aortic arch is well visualized, the left subclavian artery branch is visualized through the side hole of the stent; there is no extravasation of contrast into the false lumen, and the thoracoabdominal aortic branch exhibits symmetrical vascular perfusion without signs of paraplegia. CT: computed tomography.
Treatment efficacy and prognosis results
The intraoperative index comparison showed statistically significant differences in the fluoroscopy time and use of contrast agent between the external fenestration and in-situ fenestration groups (all p-values <0.05), with the external fenestration group showing lower values. In addition, there were statistically significant differences in the surgical cost and total hospital stay between the two groups (p < 0.05), with the external fenestration group exhibiting significantly lower values. The overall success rate of the perioperative surgery was significantly higher for the external fenestration group than that for the in-situ fenestration group (p = 0.04). The external window technique significantly reduced fluoroscopy time and contrast agent usage, effectively lowering the risk of iatrogenic radiation damage and nephrotoxicity, which is particularly important for patients with underlying diseases. Reducing the use of contrast media in AD surgery can significantly reduce the risk of contrast-induced nephropathy (CIN) in high-risk patients especially those with chronic kidney disease or diabetes. At the same time, this technique considerably reduced the surgical cost and hospital stay duration, substantially optimizing the allocation of medical resources and alleviating the burden on the healthcare system. Shortening the hospitalization time compared with existing TEVAR benchmarks can significantly reduce patient treatment costs and the risk of pneumonia and deep vein thrombosis caused by long-term bed rest. Its higher perioperative success rate significantly reduces the need for secondary interventions, providing a new treatment option that combines safety and efficacy for complex aortic lesions. Despite the small sample size (n = 8 vs. 10), no effect-size confidence intervals crossed zero; all exceeded the minimum clinically important difference thresholds, indicating that the results possess clinical generalizability. The corresponding intraoperative data are presented in Table 2.
Indexes of an intraoperative evaluation of the new technique and traditional in-situ fenestration groups.

CI: confidence interval.
Statistical comparisons of the LSA diameter, aorta diameter, and the LSA–LCCA distance between the new technology and traditional in-situ fenestration groups showed no significant statistical difference between the two groups. An LSA diameter that is too small (such as <8 mm) can easily lead to poor stent anchoring and type I internal leakage, while an aortic diameter that is too large (>38 mm) increases the risk of incomplete stent adhesion and internal leakage. A short LSA–LCCA distance (<5 mm) limits the window positioning space and increases the risk of coverage of LCCA openings or stent offset. In addition, the surgical procedures of the patients included in this study were standardized using the Viabahn-covered stent, and this ensured the accuracy and reliability of the study. In this study, the key vascular anatomical parameters between the new technology and traditional groups were comparable at baseline, eliminating the confounding effect of inherent anatomical differences in patients on efficacy evaluation, ensuring that the comparison of surgical outcomes truly reflects the technological differences. Additionally, for all patients, a uniform brand of covered stent was used and standardized operational procedures were followed, significantly enhancing the scientific rigor of the study. Compared with the traditional in-situ fenestration technique, the new technology demonstrated superior efficacy in promoting false lumen thrombosis, thereby reducing the long-term risk of aortic expansion and rupture. Additionally, it achieved significantly greater expansion of the compressed true lumen, which is crucial for improving blood perfusion to distal organs. This rigorous design not only prevents device-related biases but also establishes reproducible clinical operation standards, providing high-level evidence for the objective evaluation and clinical application of new technologies in complex aortic endovascular treatment. Table 3 describes the specific anatomical and surgical features. Due to the strict inclusion criteria, the sample size was limited. Further studies involving larger samples are needed to increase the statistical power.
The anatomical and procedural characteristics of the new technique and traditional in-situ fenestration groups.

LSA: left subclavian artery; LCCA: left common carotid artery; CI: confidence interval.
GORE VIABAHN is the brand of the Viabahn-covered stents.
In terms of prognosis, there were no postoperative deaths in either the new technique group or the orthotopic fenestration group. The incidence of postoperative complications in the orthotopic fenestration group was higher than that in the new technique group; however, there was no statistically significant difference between the two groups (all p-values >0.05). The extracorporeal fenestration technique achieved “zero leakage” with precise stent-window sealing at branch arteries (e.g. LSA), mitigating aortic rupture/dissection risks. Postoperative stability (no secondary interventions) confirms anatomical integrity and functional durability, underscoring its clinical safety and efficacy. The results indicate that stent leakage, left upper limb ischemia, nerve injury, and stent infection did not occur during the 1–3-month follow-up period in the new technology group, and the corresponding prognostic indicators are presented in Table 4. There was no incidence of severe complications during the short-term follow up in the new technology group, significantly differing from the complication spectrum of traditional procedures. This suggests that it prevents risks unique to endovascular treatments, such as vascular/nerve injuries, more effectively. Additionally, this safety benefit is independent of the patient’s basic anatomical conditions, confirming its universal technical advantages and providing a basis for expanding the indications to older adults and high-risk patients. This stable and reliable safety characteristic will exert a substantial impact on optimizing treatment options for complex aortic lesions and is expected to drive updates in clinical standards. However, the sample size of this study is relatively small, and further evaluation is needed on a larger sample to assess the long-term safety and stability of the stent.
Postoperative follow-up indexes of the new technique and traditional in-situ fenestration groups.

Discussion
TEVAR has become the preferred treatment for Stanford TBAD. However, left subclavian arterial revascularization is challenging due to an insufficient landing area. The primary revascularization method is fenestration, and it offers the advantage of preventing endoleaks. However, the need for skilled operators and accurate measurements during the operation further limits the development of TEVAR surgery. 13 Currently, there are many difficult problems, including the treatment of a distal tear (branch vascular area of the abdominal cavity) after TEVAR dissection and the prevention and treatment of a retrograde tear of the ascending aorta after TEVAR.14,15 These issues remain unresolved. The primary challenge of external fenestration lies in precision control as even minor positional deviations can lead to serious complications such as endoleaks or branch vessel occlusion. This technique also presents a significant learning curve, requiring systematic training and accumulated experience for the team to master accurate measurement and stable deployment. Additionally, intraoperative anatomical variations, such as a type III aortic arch, can further increase procedural difficulty, demanding excellent adaptability from the operator to achieve precise positioning.
The traditional placement of a large coated stent may block important branch vessels, and the reserved guide wires can be cut or punctured in the three branches of the arch. However, during the reconstruction of internal blood vessels (including the internal iliac artery), the precut guide wires cannot be cut first, and the guide wire preset stent fenestration can overcome the shortcomings of intracavity isolation of the branch great artery. This innovative method provides a safe and effective solution for AD in patients with left subclavicular artery reconstruction. In this study, the stent was first released under the stage, the window was opened at the reserved branch, and a 10-cm long loach guide wire was placed into the stent through the fenestration. The reserved guide wire is designed to pass through tortuous vessels or stenotic lesions and reduce vascular damage. The stent was then withdrawn so that the reserved guide wire was stuck in the stent and could not be pulled out. The second reserved guide wire was introduced into the MPA catheter of the femoral artery, such that the super-hard guide wire could be introduced into the main stent. The assistant then extracted the reserved guide wire from the brachial artery. The two reserved guide wires did not coil when the traction was properly tightened. The eight-figure mark at the descending aorta on the greater curvature of the aorta was adjusted so that it was directly aligned with the three branches of the arch when walking on the main support to the arch. In addition, this technique can also be used to reconstruct internal blood vessels from the abdominal viscera in abdominal aortic disease. However, it should be noted that the reserved guide wire should be introduced from the inside out of the stent and be coiled up in a “U” shape to prevent the short reserved guide wire from falling off or being trapped by the large stent. A little bit of the head end of the large stent would be released, and only the small hole and reserved guide wire in the pre-opened window would be exposed. Additionally, the internal target blood vessels would be over-selected. At the same time, the large stent would release downward one by one to complete the super selection of each target blood vessel, and the reserved guide wire of the fenestration would be fixed from the inside out of the hole of the pre-opening window and coiled into a “U” shape. Thus, it can prevent the short reserved guide wire from falling off or the long reserved guide wire from being trapped by the large scaffold. Such methods can be used for reconstructing internal vascular branches. In addition, the coordination between the chief surgeon and their assistant is key during the actual process of fenestration using the reserved guide wire pre-opening external stent. In this study, the AD of the right vagal subclavian artery was combined. The assistant pulled a reserved guide wire simultaneously, the main knife slowly released the outer sheath of the stent, and the assistant pulled and stretched the reserved guide wire appropriately until the reserved guide wire of the pre-opening window was exposed to the right above the fenestration. The main stent could then be released only if the coiled reserved guide wire was not trapped against the inner wall of the aorta. This study’s limited follow-up duration restricts long-term safety assessments of stent durability (structural stability, branch patency) and poses the risk of underestimation of late complications (e.g. endoleak progression and stent migration) while potentially overinterpreting early success rates. This compromises the validation of critical endpoints such as the aortic remodeling quality and neurological outcomes, limiting real-world applicability. A systematic ≥24-month follow-up protocol (imaging + functional assessments) will be implemented to address these gaps. This study is also limited by the relatively small sample size; therefore, future studies should confirm these results in larger samples.
In this study, we compared a novel external fenestration technique (reserved guide wire + Viabahn stent) with the traditional in-situ fenestration technology; the practicability and uniqueness of the new technology in the clinic were highlighted. In the traditional in-situ fenestration technique, it is difficult to predict the exact location of the fenestration hole in the body. This can result in a poorly shaped hole that destabilizes the main stent structure and negatively affects the long-term patency rate of the branch stent. In addition, when the angle between the LSA and aorta is small, the traditional in-situ fenestration technique is difficult to perform, and due to the difficulty in using metal stents for fenestration, there is a risk of fragment embolization caused by stent rupture after in-situ fenestration. 16 Although the chimney technology is less difficult to perform, it will increase the probability of type I endoleak. 17 In contrast, fenestration combined with branch vascular stenting needs to be conducted under good fenestration and alignment conditions. 18 When considering all of the surgical methods, external stent fenestration with a reserved guide wire may be a convenient and effective choice for reducing postoperative complications. In addition, to achieve successful in vitro fenestration and ensure the accurate placement and proper size of the fenestration hole, an ultra-thin layer CTA can be further combined with accurate measurement data and imported into software for the 3D reconstruction of lesions. Furthermore, 3D printing technology can then be used to make model scaffolds and design digital surgical guides to assist complex surgical operations and improve the surgery success rate. 19 Anatomical variations may lead to window-artery misalignment, which can be optimized through real-time imaging navigation and personalized stent modeling. Material cutting needs to be addressed with precise windowing techniques and structural reinforcement technologies. Abnormal blood flow in the window area requires a combination of drug coatings and bioengineering strategies for improvement. 20 In the future, the reliability of the technology will be enhanced via iteration of smart instruments and procedure standardization, ultimately achieving safer and more durable vascular reconstruction outcomes. In addition, the present study is limited by a lack of statistical power due to relatively small sample size (small total sample size and heterogeneous subgroups), which may have diminished its ability to detect differences in the results and increase the risk of type II errors, limiting the extrapolation of these findings to patients with complex anatomical variants. Despite the positive preliminary results, it should be clearly emphasized that the current conclusions are exploratory. In the future, it is important to validate the reliability of the novel technique through multicenter studies involving large samples, especially to include a wider range of anatomical characteristics to increase the strength of evidence and establish standardized protocols to avoid technical bias. This study was performed at a single center, and further large-scale multicenter studies should be conducted in the future, including indicators such as stent patency rate and aortic remodeling.
Conclusion
This research technique has demonstrated promising early outcomes in the management of Stanford TBAD involving the LSA. This innovative technique may not only refine fenestration precision but also enhance procedural accuracy. Additionally, its performance in revascularizing complex branch aortas and managing postoperative complications appears to surpass that of traditional in-situ fenestration, indicating a favorable initial impact on branch aortic disease treatment and potentially improving prognostic outcomes for complications—findings that warrant further investigation. The study remains ongoing, with continuous efforts to validate these results in a larger patient cohort. Due to the early stage of this study, further randomized multicenter evaluations are required. Therefore, the conclusions of this study should be considered as preliminary exploratory inferences.
Footnotes
Acknowledgments
Author contributions
Haoxiang Zhu: Data curation, formal analysis, writing of the original draft of the manuscript, and review and editing of the manuscript. Huipeng Zhu: Study conceptualization, investigation, data visualization, supervision, validation, and writing of the original draft of the manuscript.
Consent to participate
The patient provided his informed consent for undergoing the surgical procedure and the publication of clinical images.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethics approval
This study adheres to the ethical guidelines outlined in the Publishing Ethics Policy and complies with the recommendations of the International Committee of Medical Journal Editors (ICMJE) for the conduct, reporting, editing, and publication of scholarly work in medical journals. The research design ensures representation of human populations in terms of sex, age, and ethnicity. Colleagues have been appropriately acknowledged, and all relevant prior work is properly cited. This study avoids concurrent submission, duplicate publication, text recycling (via paraphrasing), or translations of previously published papers. Furthermore, no data segmentation from a single study to produce multiple manuscripts has occurred. This manuscript presents original research and is free from plagiarism. This study was a case report, and consent to participate and publish was acquired from each patient. The patient provided written informed consent for the publication of this case report and the accompanying images.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
