Abstract
Background
To explore the clinical value of preoperative transrectal three-dimensional ultrasound in diagnosing the internal opening and classifying complex anal fistulas, and to evaluate its impact on surgical efficacy.
Methods
A total of 42 patients with complex anal fistulas were divided into an observation group and a control group. The control group underwent conventional transrectal ultrasound examination, while the observation group underwent three-dimensional transrectal ultrasound examination. Perioperative data were recorded and compared between the two groups.
Results
The observation group showed significantly higher diagnostic accuracy (p = 0.014) and a higher cure rate (66.67% vs. 42.86%) than the control group. Anal sphincter function improved postoperatively in both groups, with the observation group achieving superior outcomes at 6 months. The observation group also demonstrated lower Wexner incontinence scores (p = 0.015), shorter hospitalization and wound healing times, and higher SF-36 scores in bodily pain, general health, and health transition domains. However, postoperative complication rates and 6-month recurrence rates were similar between the two groups.
Conclusion
Preoperative transrectal three-dimensional ultrasound can accurately identify the internal opening and fistula tract of complex anal fistulas, which is beneficial for improving treatment efficacy.
Introduction
Anal fistula is a common chronic infectious disease in anorectal surgery. It is essentially an abnormal tract connecting the anal canal or rectum to the perianal skin, with an incidence of approximately 20/100,000 population. 1 Although most anal fistulas can be cured by surgery, complex anal fistulas, due to tortuous tracts, multiple branches, concealed internal openings, or association with deep-seated abscess cavities, often result in diagnostic difficulties, a high rate of surgical failure, and postoperative recurrence, thereby severely affecting patients’ quality-of-life. 2 The contemporary paradigm in the management of anal fistulas centers on complete excision of the fistulous tract and associated infectious foci and meticulous identification and closure of the internal opening while simultaneously preserving optimal sphincter function. 3 The cornerstone of successful treatment lies in precise preoperative anatomical delineation of the fistulous structure, with particular emphasis on accurate localization of the primary internal opening and definitive classification of the fistula according to the Parks classification system. 4
If complex anal fistulas are not diagnosed and treated promptly and accurately, serious complications such as perianal skin infection and anal incontinence may occur, causing significant negative effects on patients’ physical and mental health. 5 Traditional diagnostic methods, such as digital rectal examination and probe examination, although capable of detecting anal fistulas to some extent, have considerable limitations in accurately locating the internal opening and determining the classification of complex fistula tracts. 6 Transrectal three-dimensional ultrasound, as an emerging imaging modality, offers advantages such as high resolution, multiangle imaging, and real-time dynamic observation, providing new approaches for the diagnosis of complex anal fistulas.7,8 It enables precise delineation of the fistulous tract, accurate identification of the internal opening, and comprehensive visualization of its spatial relationship with adjacent anatomical structures.
Precise identification of the internal opening and definitive classification of the fistulous anatomy are essential prerequisites for formulating an optimal treatment strategy and selecting the most appropriate surgical approach. 9 Therefore, conducting this clinical study has important practical significance. Our study aims to comprehensively evaluate the diagnostic value of preoperative transrectal three-dimensional ultrasonography in the assessment of complex anal fistulas and to quantify its impact on surgical efficacy.
Materials and methods
Study design
This was a single-institution prospective study. All patients voluntarily selected the surgical procedure and signed written informed consent after receiving detailed information. The data were fully anonymized, complied with the Declaration of Helsinki, and were approved by the Ethics Committee of Changhai Hospital (registration number: ChiCTR2000029478; Ethics approval number: CHEC2022-092).
General data
A total of 84 patients diagnosed with complex anal fistula who were admitted to our hospital between January 2023 and June 2024 and scheduled for surgical treatment were prospectively enrolled in this study (Figure 1). The observation group (n = 42) underwent three-dimensional transrectal ultrasonography before surgery, whereas the control group (n = 42) received conventional transrectal ultrasonography. The observation group comprised 25 males and 17 females, with an age range of 21–46 years. The control group consisted of 26 males and 16 females, with an age range of 22–48 years. The baseline demographic revealed no significant differences between the two groups as presented in Table 1.
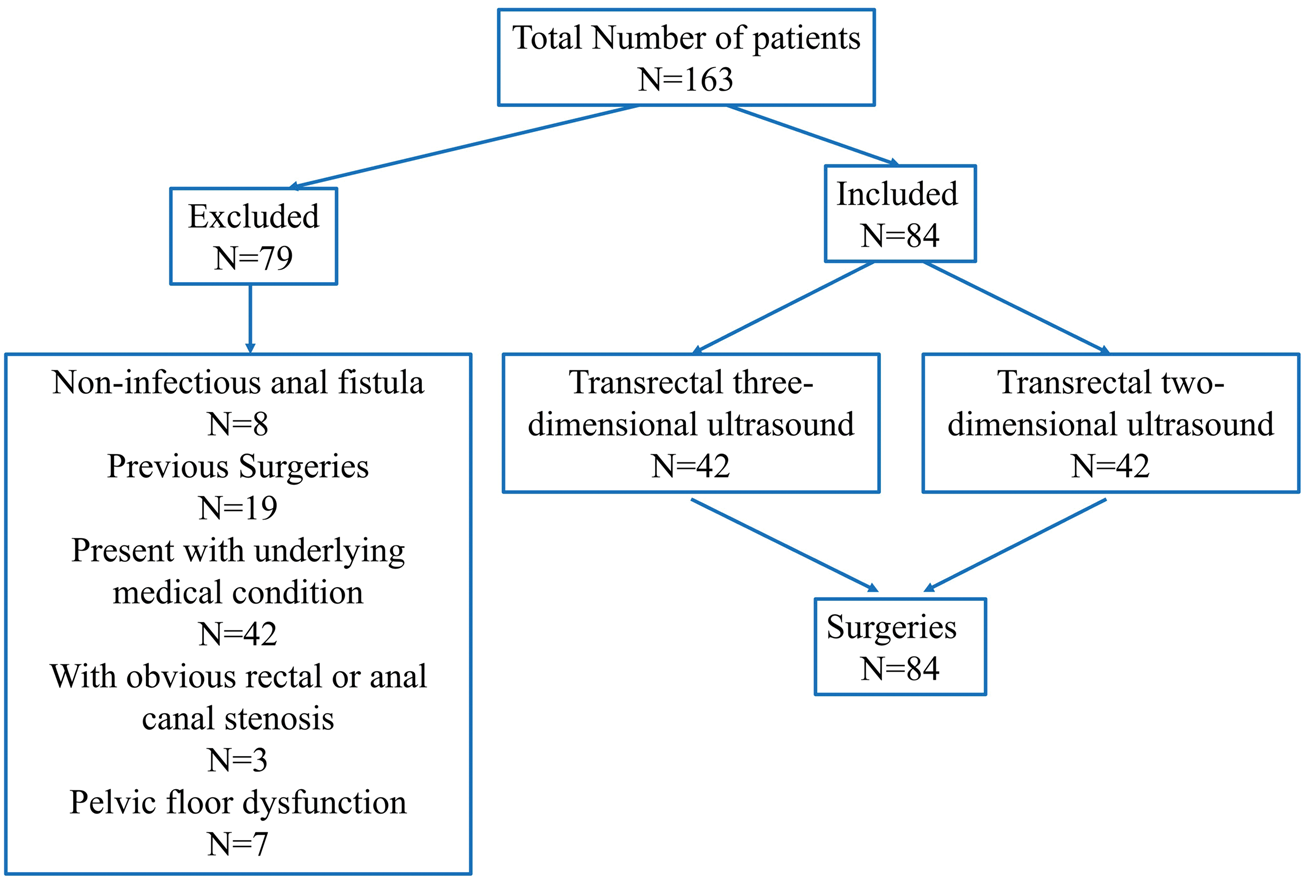
Flow chart of the study patients.
Baseline characteristics.

Age, BMI, and duration of disease were expressed as
BMI: body mass index.
Inclusion and exclusion criteria
Inclusion criteria
All study participants had a confirmed diagnosis of anal fistula and met the established diagnostic criteria for complex anal fistula. 1. Through visual inspection, digital rectal examination, anoscopy, and staining examination, two or more internal or external openings of the anal fistula, or two or more fistula tracts, branches, or blind tracts were identified. 2. Patients were scheduled to undergo surgical treatment. 3. Written informed consent was obtained from the patients or their families.
Exclusion criteria
(a) Anal fistulas caused by nonanal gland infections, such as trauma. (b) Recurrence after previous anal fistula surgery. (c) Severe cardiac, hepatic, or renal dysfunction; malignant tumors; diabetes; or mental illness. (d) Obvious rectal or anal canal stenosis. (e) Abscesses at different locations or pelvic floor dysfunction. (f) Hormone therapy within the past 3 months.
Operational methods
Preoperative anal canal examination
All patients underwent digital rectal examination and a methylene blue test before surgery. Under anesthesia, a probe was used to identify the location of the internal opening of the anal fistula and to assess the presence of branch fistula tracts.
Ultrasound examination
Control group: Routine transrectal ultrasound examination was performed before surgery. After cleansing with enema, the patient was placed in the left lateral decubitus position with both lower limbs flexed. A model 1202 color Doppler ultrasound diagnostic instrument (BK Medical, Denmark), equipped with an L8811 two-dimensional probe with a frequency of 6–16 MHz, was used. The probe was covered with a condom, and an appropriate amount of ultrasound coupling gel was applied outside the condom. The probe was then slowly inserted into the rectum and stabilized. Anal fistulas appeared as strip-shaped or irregular hypoechoic or anechoic discontinuities with fluid flow within them. Color Doppler flow imaging showed no obvious blood flow signal within the fistulas, while dotted or rod-shaped blood flow signals were observed around the fistulas. The internal orifice was characterized by interruption of mucosal continuity or partial mucosal elevation or depression with low to no echogenicity. The scanning range of the probe was adjusted according to the lesion location, and the images and data were recorded.
In the observation group, transrectal three-dimensional ultrasound examination was performed before surgery. A BK 1202 color Doppler ultrasound instrument (BK Medical, Denmark), equipped with an 8811 linear-array probe and an L8838 intracavitary three-dimensional probe with a frequency of 6–16 MHz, was used. Pre-examination preparation was the same as that for the control group. After insertion of the probe into the rectum and anal canal, routine scanning was performed. The fistulas were displayed stereoscopically, with flake-like or linear enhancement. We placed a plastic venous cannula, which was inserted into the fistulous opening. Subsequently, an appropriate volume of contrast agent (SonoVue, Bracco, Italy), dissolved in 5 mL of 0.9% NaCl, was injected to identify the internal orifice. After image acquisition, three-dimensional stereoscopic image reconstruction was performed to obtain three-dimensional stereoscopic imaging information of the lesion.
According to the Parks classification, anal fistulas were categorized as intersphincteric, trans-sphincteric, suprasphincteric, or extrasphincteric. Intersphincteric fistulas are mostly low anal fistulas, in which the tract extends within the intersphincteric space; the internal orifice is located at the anal sinus near the dentate line, and the external orifice is usually near the anal verge. Trans-sphincteric anal fistulas pass through the internal and external sphincter muscles and open onto the perianal skin. Suprasphincteric anal fistulas are mostly high anal fistulas; the tract extends upward in the intersphincteric space, passes through the levator ani muscle, and then descends through the ischiorectal fossa to reach the skin. Extrasphincteric anal fistulas extend from the perianal skin upward through the ischiorectal space and levator ani muscle, penetrate the pelvirectal space, and directly communicate with the rectum.
All ultrasound images were independently interpreted by two board-certified radiologists with 8 and 10 years of specialized experience in anorectal ultrasound. In cases of disagreement, the images were further evaluated by a third senior radiologist.
Surgical method
All patients underwent surgical treatment. For patients with intersphincteric and trans-sphincteric anal fistulas, partial fistulotomy combined with seton placement and mucosal flap advancement was performed. For patients with suprasphincteric and extrasphincteric anal fistulas, mucosal flap advancement combined with seton placement was performed. Antibiotics were routinely administered for 3 days after surgery to prevent infection. Routine dressing changes were performed 1–2 times per day. Bowel control was maintained for 3–5 days after surgery, and sitz bathing was prohibited for 7–10 days postoperatively. During surgery, exploration was conducted to record the location and number of internal openings, and the Parks classification was determined.
Observation indicators
Preoperative diagnostic accuracy of the internal opening of anal fistula and detection accuracy of Parks classification
In both groups, the preoperative ultrasound diagnosis and the Parks classification were compared with the intraoperative exploration findings to calculate and compare the preoperative diagnostic accuracy and the detection accuracy of the Parks classification between the two groups.
Efficacy judgment criteria
Cure: After treatment, the symptoms and signs of anal fistula disappeared. 10 Physical examination showed that the wound surface and the internal and external openings of the anus were completely closed, with no abnormalities in anal function. Effective: After treatment, the symptoms of anal fistula were improved. Physical examination showed that the wound surface and the internal and external openings were significantly improved but not completely closed. Ineffective: After treatment, the symptoms of anal fistula showed no improvement or worsened. Physical examination showed that the wound surface and the internal and external openings of the anal fistula remained unclosed. The cure rate = (number of cured cases/total number of cases) × 100%.
Evaluation of anal function
The Wexner incontinence scoring system 11 was used to evaluate anal function before surgery and 6 months after surgery. The score includes items assessing incontinence to gas, liquid stool, and solid stool as well as the use of sanitary pads and lifestyle alterations.
Quality-of-life questionnaire
The Short Form-36 Health Survey (SF-36) was used to assess postoperative quality-of-life in both groups at 6 months after surgery. 12 This scale includes eight dimensions: physical functioning (PF), role limitations due to physical health (RP), bodily pain (BP), general health (GH), vitality (VT), social functioning (SF), role limitations due to emotional problems (RE), and mental health (MH). Health transition (HT) was also evaluated.
Postoperative follow-up
The duration of hospitalization and the time to complete wound epithelialization were systematically documented for all subjects. Longitudinal surveillance was conducted for 6-month postoperatively in the entire cohort. Functional assessment using the validated Wexner incontinence scoring system was performed at the 6-month follow-up visit. Concurrently, the incidence of procedure-related morbidity and fistula recurrence during the surveillance period was meticulously recorded.
Statistical analysis
Data analysis was performed using GraphPad Prism version 8.0.1. Measurement data were expressed in
Results
Diagnostic results of the internal opening of anal fistula and Parks classification in the two groups
The diagnosis of complex anal fistula was surgically confirmed in 84 patients. In the observation group, intraoperative examination revealed 56 internal apertures. According to the Parks classification, the distribution comprised 22 cases of intersphincteric fistulae, 11 cases of trans-sphincteric fistulae, 7 cases of suprasphincteric fistulae, and 2 cases of extrasphincteric fistulae. In the control group intraoperative examination identified 53 internal apertures. The distribution based on the Parks classification consisted of 21 intersphincteric fistulae, 11 trans-sphincteric fistulae, 8 suprasphincteric fistulae, and 2 extrasphincteric fistulae (Table 2).
Comparison of detection accuracy rates of Parks classification between the two groups.

Data were expressed as the number of cases and percentages. The accuracy rate was calculated as (surgically confirmed cases/ultrasound-confirmed cases) × 100%. Statistical analysis was performed using the chi-square test.
Comparison of detection accuracy of anal fistula between the two groups
The preoperative diagnostic accuracy for identifying the internal opening of anal fistula was 92.86% (39/42) in the observation group and 76.19% (32/42) in the control group (χ2 = 6.012, p = 0.014).
The preoperative diagnostic accuracy for intersphincteric and extrasphincteric fistulas was significantly higher in the observation group than in the control group (p = 0.032 and p = 0.028, respectively). However, no statistically significant difference was observed between the two groups in the diagnostic accuracy for trans-sphincteric or suprasphincteric fistulas (Table 2).
Comparison of surgical efficacy and postoperative results between the two groups
The curative efficacy in the observation group was 66.67%, demonstrating a statistically significant improvement compared with 42.86% in the control group (p = 0.028) (Table 3).
Comparison of surgical efficacy and postoperative outcomes between the two groups.
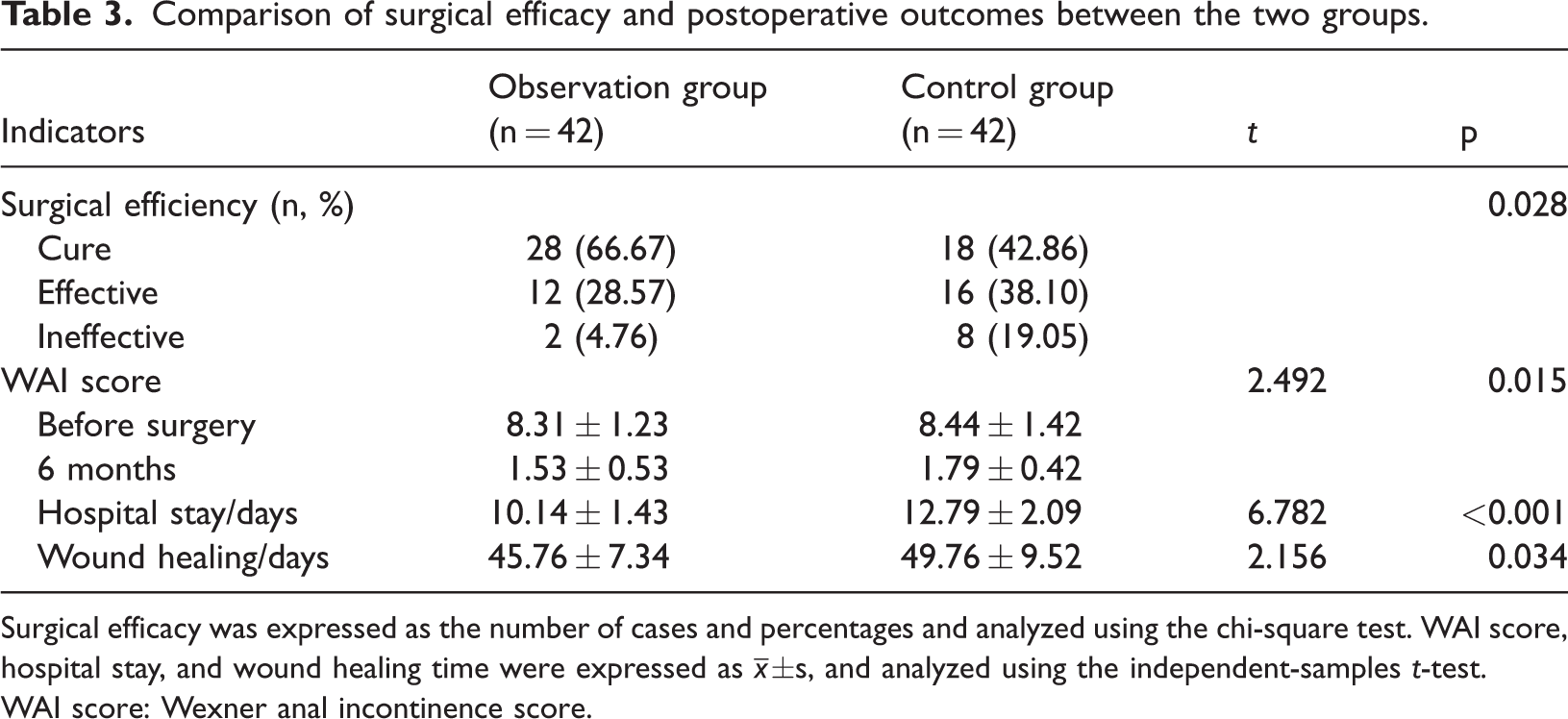
Surgical efficacy was expressed as the number of cases and percentages and analyzed using the chi-square test. WAI score, hospital stay, and wound healing time were expressed as
WAI score: Wexner anal incontinence score.
There was no significant difference in the preoperative Wexner anal incontinence score between the two groups. At the 6-month postoperative follow-up, the Wexner anal incontinence scores were significantly lower in the observation group, and the intergroup difference was statistically significant (p = 0.015). Furthermore, patients in the observation group had shorter hospitalization durations and accelerated wound healing trajectories compared with those in the control group, with both parameters demonstrating statistically significant differences between groups (p < 0.001; p = 0.034) (Table 3).
Comparison of quality-of-life surveys and complications
Upon comparative analysis of the SF-36 quality-of-life assessment, it was revealed that the observation group had significantly higher scores in the domains of BP, GH, and HT compared with the control group, and these differences were statistically significant (p = 0.001; p = 0.001; p = 0.004). Conversely, no statistically significant intergroup disparities were observed in the PF, RP, VT, SF, RE, and MH domains (Table 4).
Comparison of quality-of-life scores and complications between the two groups.

Quality-of-life scores were expressed as
There was no significant difference in the incidence of postoperative complications between the two groups (Table 4).
Comparison of postoperative anal fistula recurrence between the two groups
Within 6 months, one patient in the observation group experienced recurrence, with a recurrence rate of 3.57% (1/28), and one patient in the control group experienced recurrence, with a recurrence rate of 5.56% (1/18). There was no statistically significant difference in the 6-month recurrence rate between the two groups (χ2 = 0.104, p = 0.747).
Typical cases
A 43-years-old male patient presented with pain and discomfort in the anal region for 4 days. Ultrasound examination indicated a suprasphincteric horseshoe-shaped abscess/anal fistula located in the ischiorectal fossa. The following are two-dimensional sonographic images obtained from longitudinal section scanning along the long axis of the linear-array probe through the anorectal canal (Figure 2).

Typical cases. (a) Intersphincteric and suprasphincteric fistula (red arrow). (b) Cross-sectional view of the fistula between the puborectalis muscle, the deep part of the external sphincter muscle, and the origin of the levator ani muscle (red circle). (c) The fistula goes around the posterior edge of the puborectalis muscle and enters the ischiorectal fossa (red arrow). (d) Abscess/anal fistula in the ischiorectal fossa, running obliquely upward and outward on the cephalic side to reach the levator ani muscle (red arrow). (e) The abscess invades the levator ani muscle (red circle). (f) Reconstructed cross-sectional view at the level of the puborectalis muscle, showing the abscess/anal fistula (red circle). (g) Reconstructed oblique sagittal section view of the right ischiorectal fossa (red arrow), simultaneously showing the main trunk of the fistula and a blind-ending branch (yellow arrow). (h) Reconstructed oblique coronal section view of the posterior part of the anorectal canal, showing the abscess/fistula at two levels, deep and shallow (red arrow). (i) MRI results showed perianal abscess (white arrow) penetrating the external anal sphincter and involving the ischiorectal fossa and ischioanal fossa. Subcutaneous cellulitis is observed on the right side of the anus (white arrow). The abscess wall was slightly thickened and blurred, which might tend to be chronic. (j) Intraoperative endosonography showed that after the abscess cavity was fully drained, the pus disappeared. MRI: magnetic resonance imaging.
Discussion
Complex anal fistula holds a special position in anorectal surgery. Its definition is mainly based on the complexity of the fistula tract. 13 Compared with ordinary anal fistulas, complex anal fistulas exhibit more diverse and intricate pathological features. From a pathological perspective, the formation of complex anal fistulas is often closely related to anal gland infection. 14 The anal glands are located beneath the mucosa at the junction of the anal canal and rectum. When the anal glands are invaded by bacteria and become infected, inflammation can spread to the surrounding tissues along the gland ducts. 13 If the infection is not promptly and effectively controlled, an abscess will gradually form. 15 Persistent infection may induce fibrotic transformation of the abscess cavity wall, culminating in the formation of an abnormal epithelialized tract that establishes communication between the anorectal canal and the perianal skin, thereby constituting a fistula-in-ano. In complex anal fistulas, this infectious and fistulizing process is more extensive, often involving multiple tissue layers and anatomical spaces, which makes the condition more refractory to treatment. Traditional diagnostic methods have many limitations when dealing with complex anal fistulas. 16 Digital rectal examination, a simple and commonly used assessment method, mainly relies on the physician’s experience and tactile perception. For anal fistulas that are relatively superficial and have a simple tract, digital rectal examination may detect certain findings, such as a cord-like induration or localized tenderness.15,17 However, when the internal orifice of the fistula-in-ano is located in a deeper anatomical plane and the fistulous tract demonstrates a complex configuration, the diagnostic accuracy of digital rectal examination is substantially reduced. Probing techniques similarly present significant limitations.15,17 The tortuous and branching morphology characteristic of complex fistulous tracts often hinders advancement of the probe, preventing accurate identification of the internal opening. 18 Of greater clinical concern is the potential for iatrogenic perforation of the fistulous wall during manipulation, which may create false tracts and significantly confound accurate diagnostic assessment. 19
Transrectal three-dimensional ultrasound is an advanced ultrasonographic technique developed on the basis of conventional two-dimensional imaging and further expanded through technological innovation. It can acquire a large volume of two-dimensional ultrasound image data within a short period through a specialized probe and imaging algorithms. 20 These data encompass information from different layers and angles of the anal canal, rectum, and surrounding tissues. The collected two-dimensional images are subsequently processed and analyzed using computer software. Through complex reconstruction algorithms, the two-dimensional images are reconstructed into a three-dimensional stereoscopic image of the anal fistula (Figure 2). 21 Clinicians can then perform comprehensive multiplanar visualization of the fistula-in-ano morphology and structural configuration in the coronal, sagittal, and transverse planes as well as from various oblique perspectives. This multidimensional evaluation facilitates the identification of subtle pathological features that may not be detected with conventional two-dimensional ultrasonography.
With its high resolution and multiangle imaging capabilities, three-dimensional ultrasound can clearly display the location, shape, and size of the internal opening.22,23 In addition, the real-time dynamic observation function of three-dimensional ultrasound facilitates identification of the internal opening, thereby providing a reliable basis for Parks classification. During the examination, physicians can observe the direction of fluid flow within the fistula tract, which helps trace the location of the internal opening. 24 Conventional ultrasound provides only two-dimensional planar images and may miss or misdiagnose fistula tracts and internal openings that are deep-seated and complex in course.24,25 In this study, the preoperative localization accuracy of the internal orifice in fistula-in-ano reached 92.86% (39/42) in the observation cohort, representing a statistically significant improvement compared with the control cohort (p = 0.014).
With continued in-depth research on the treatment of complex anal fistulas and increasing sample sizes, the advantages of three-dimensional ultrasound in improving patients’ quality-of-life may become more evident. Brillantino et al. 26 reported a multicenter retrospective case-control study including 253 adult patients and suggested that preoperative evaluation with three-dimensional endoanal ultrasound could facilitate intraoperative detection of the internal orifice and improve the success rate. In our study, comparative analysis of health-related quality-of-life outcomes using the SF-36 instrument demonstrated that the observation cohort had significantly higher scores in the domains of BP, GH, and HT than the control cohort, and these intergroup differences were statistically significant. Within 6 months, one case in the observation group recurred, with a recurrence rate of 3.57% (1/28), and one case in the control group recurred, with a recurrence rate of 5.56% (1/18). There was no statistically significant difference in procedural sequelae or recurrence rate within 6 months between the two groups. Similarly, no significant difference was observed in the incidence of postoperative complications or recurrence rate between the two groups in this study.
It is important to explicitly acknowledge the key limitations of the present study, which constrain the generalizability and robustness of our findings. First, the absence of randomization between the observation and control groups represents a critical methodological limitation. The nonrandom allocation raises the potential for selection bias, as unmeasured confounding factors may have influenced both group assignment and outcomes. Second, the relatively small sample size limits the statistical power of the study, particularly for detecting small but clinically meaningful differences in outcomes such as recurrence rates or specific complication subtypes. Additionally, the single-institution design may further limit external validity and introduce institutional bias.
Conclusion
Transrectal three-dimensional ultrasound has important value in the diagnosis of complex anal fistulas. It can accurately identify the location and number of internal openings, clearly delineate the course and branches of the fistula tract, and define its relationship with surrounding tissues, thereby providing a reliable basis for Parks classification.
Footnotes
Acknowledgments
Not applicable.
Author contributions
S.X., Y.S., and E.G. wrote the initial draft of the manuscript, developed the research project, and collected the data. Z.L. performed all analyses. Q.J. and N.Z. conceptualized and designed the study. All authors revised the manuscript for final submission.
Consent for publication
Not applicable.
Date availability
All data are contained within this manuscript.
Declaration of conflicting interest
The authors declare no conflicts of interest.
Funding
This work was supported by the New Technology Project of the Naval Military Medical University (ZXJS2024C04). The funder played no role in the study design, data collection, analysis and interpretation of data, or the writing of this manuscript.
Ethics approval and consent to participate
All patients voluntarily chose and signed the surgical informed consent form after being informed of the relevant information. The data were completely anonymized, complied with the Declaration of Helsinki, and were approved by the Ethics Committee of Changhai Hospital (registration number: ChiCTR2200067105; Ethics approval number: CHEC2022–092).
