Abstract
Objective
To describe adherence to early discharge (<24 h) among patients with low-risk pulmonary embolism and to characterize barriers to its implementation.
Methods
This was a descriptive, retrospective cohort study conducted within an Anticoagulation Stewardship Program at a Colombian tertiary hospital. Low-risk pulmonary embolism was defined as simplified Pulmonary Embolism Severity Index of 0, absence of right ventricular dysfunction, and negative cardiac biomarkers. Outcomes at 30 days included mortality, major bleeding, and rehospitalization.
Results
Among 765 patients with pulmonary embolism, 62 were classified as low risk. The median age was 45.5 years (interquartile range: 32–62) and 36/62 (58.1%) were women. Early discharge occurred in 13/62 patients (20.9%), whereas 49/62 (79.1%) experienced longer hospital stay (median, 3 days (interquartile range: 2–6)). Early discharge rates increased from 9.5% (2019–2022) to 45% (2022–2024). Thirty-day outcomes were favorable, with no deaths, major bleeding events, or rehospitalizations. Five patients revisited the emergency department for pain but did not require admission. Delayed discharge was more frequent in patients with anemia, thrombocytopenia, those receiving medications associated with increased bleeding risk, and in those managed outside the internal medicine service. Documented reasons for delayed discharge included awaiting echocardiography (12/49), international normalized ratio monitoring (11/49), other comorbidities (11/49), delayed direct oral anticoagulants dispensing or authorization (9/49), and uncontrolled pain (6/49).
Conclusions
Adherence to early discharge for low-risk pulmonary embolism was limited despite excellent short-term safety outcomes. System- and process-level barriers, particularly echocardiography utilization, warfarin/international normalized ratio requirements, direct oral anticoagulants access, and pain management, may be addressed through standardized discharge pathways and diagnostic stewardship.
Introduction
Venous thromboembolic disease (VTE), including pulmonary embolism (PE) and deep vein thrombosis (DVT), remains a major global health problem. In the United States, it affects more than 300,000 people annually.1,2 European modeling studies estimated more than 500,000 VTE-related deaths in 2004, many occurring during or shortly after hospitalization. Additionally, a substantial proportion of cases are not recognized ante mortem. 1 Robust national estimates for PE in Colombia and much of Latin America are limited. One small multicenter study in Colombia reported an in-hospital mortality rate of 14.8%. 2 Despite improvements in diagnosis and management that have reduced PE mortality over the past 15 years, incidence rates have increased. The annual incidence ranges from 39 to 115 per 100,000 in the general population and increases up to eightfold in individuals older than 80 years. In the United States, hospitalizations and associated healthcare costs exceed US$2 billion annually. 3
Several risk stratification tools are available for PE, including the Pulmonary Embolism Severity Index (PESI) and the simplified Pulmonary Embolism Severity Index (sPESI). Together with imaging findings and biomarkers of right ventricular (RV) dysfunction (RVD), these parameters allow classification into high-, intermediate-high–, intermediate-low–, or low-risk categories for 30-day mortality.3,4 In derivation and validation studies, low-risk patients, typically defined as sPESI of 0 without RVD, demonstrate very low 30-day mortality (approximately 0.0%–1.6%).5,6 For these low-risk cases, international guidelines recommend early discharge, given the low rates of complications and hospital readmissions. 7 Additional benefits of early outpatient management include improved quality of life, faster return to activities of daily living, reduced length of hospital stay, lower healthcare costs, and decreased risk of nosocomial complications.3,7,8
Guidelines estimate that approximately 30%–55% of patients with low-risk PE may be candidates for early discharge; however, in clinical practice, 80%–90% remain hospitalized beyond the first 24 h. Rates of early outpatient management vary widely between centers. Reported rates are as low as 2% in Italy, 4% in France, and 14% in the United States, whereas rates of up to 50% have been reported in Canada.9,10 There is limited knowledge regarding adherence in Latin America and the factors associated with nonadherence. The reasons for noncompliance may be related to the patient, considering comorbidities and bleeding risk;11,12 to the physician, possibly due to lack of knowledge or poor application of recommended prognostic tools; 13 or to socioeconomic factors. 14
This study aimed to describe adherence to early discharge among patients with low-risk PE enrolled in an Anticoagulation Stewardship Program at a Latin American tertiary center and to characterize barriers to its implementation.
Materials and methods
We performed a descriptive observational study based on a retrospective cohort of adults with acute low-risk PE not related to coronavirus disease 2019 (COVID-19), managed within an Anticoagulation Stewardship Program at a university tertiary care hospital and national oncology referral center in Colombia (September 2019 to November 2024). Eligible cases were adults who presented to the emergency department (ED) with symptoms compatible with acute PE and in whom PE was the primary working or admitting diagnosis. PE was confirmed via computed tomographic pulmonary angiography (CTPA) or ventilation–perfusion scintigraphy when CTPA was contraindicated. We excluded patients with incidental PE, PE diagnosed more than 24 h after admission, pregnancy, and COVID-19–associated PE, as previous studies have shown that the PESI score is not adequately calibrated in patients with COVID-19 and may underestimate mortality. 15
We defined low-risk PE as follows: (a) hemodynamically stable PE; (b) sPESI score of 0; (c) absence of RVD on CTPA and/or transthoracic echocardiography (TTE); and (d) negative cardiac biomarkers, when available, in accordance with European Society of Cardiology (ESC) guidance. The sPESI includes six binary variables: age >80 years, active cancer, chronic cardiopulmonary disease, heart rate ≥110 beats/min, systolic blood pressure <100 mmHg, and arterial oxygen saturation <90%. A score of 0 denotes low risk. Patients with active cancer or age >80 years automatically receive sPESI point ≥1 and therefore were not eligible for inclusion in the low-risk cohort. Given the retrospective design and inconsistent documentation of several Hestia criteria (e.g. social support and nonmedical reasons for admission), these criteria were not considered in the definition of low risk. 16
The prespecified outcomes were 30-day mortality, major bleeding, and rehospitalization. The institutional anticoagulation registry is designed to collect 30-day outcomes, including death, major bleeding, rehospitalization, and ED revisits, through telephone follow-up and review of electronic medical records. Longer-term outcomes were not systematically available in this retrospective analysis. We also captured ED revisits. The study was approved by the institutional ethics committee (Approval Number: FM-CIE-0354-23). The ethics committee granted a waiver of written informed consent for the retrospective analysis of deidentified data; verbal consent was obtained exclusively for 30-day telephone follow-up. The study was conducted in accordance with the Declaration of Helsinki (1975), as revised in 2024. All patient data were deidentified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 17
Data were obtained from the institutional anticoagulation registry (RACHUSI) and electronic medical records using standardized data collection instruments with periodic quality audits. The anticoagulation registry includes all patients who initiate anticoagulant therapy for any indication at the hospital. Verbal consent was obtained for 30-day telephone follow-up after discharge by trained personnel using a standardized script. Sociodemographic characteristics, comorbidities, diagnostic test results, anticoagulation details, and sPESI scores were extracted from the registry, where data are systematically recorded at the point of care. Data collection and management were supported by Research Electronic Data Capture (REDCap) tools hosted at Hospital Universitario San Ignacio.18,19 Regular audits were conducted to ensure data accuracy and to identify opportunities for improvement. Missing data were supplemented through retrospective review of institutional medical records. The six sPESI variables were abstracted from the index ED assessment. When a specific variable was undocumented, it was coded as absent solely for the purpose of sPESI calculation.
For each patient, risk assessment was performed using the sPESI score and evaluation of RVD, in accordance with the 2019 ESC guidelines for the diagnosis and management of acute PE. 4 RVD was defined according to routine radiology/cardiology reports (e.g. RV/LV ratio >1, septal bowing, or an explicit statement of RVD). Continuous values for systolic blood pressure, heart rate, oxygen saturation, and cardiac biomarkers (troponin and brain natriuretic peptide) were not extracted beyond the thresholds incorporated into the sPESI score, given the retrospective design of the study.
Once low-risk patients were identified, additional information was collected on patient characteristics, including comorbidities (uncontrolled arterial hypertension, type 2 diabetes mellitus, liver disease, and chronic kidney disease), history of VTE documented in the medical history, presence of anemia (hemoglobin levels <13 g/dL in men and <12 g/dL in women), and thrombocytopenia (total platelet count <150,000/μL). Anticoagulant therapy at diagnosis and at discharge (unfractionated heparin, low-molecular-weight heparin (LMWH), warfarin, or direct anticoagulants) was recorded as well as concomitant use of medications associated with increased bleeding risk (aspirin, P2Y12 receptor inhibitors, and nonsteroidal anti-inflammatory drugs (NSAID)). Uncontrolled pain was recorded if documented in the clinical history. Bleeding risk was assessed retrospectively based on chart documentation of risk factors, including concomitant antiplatelet/NSAID use, comorbidities, and hemoglobin/platelet values; no formal bleeding risk score was applied. As this was a retrospective study, anticoagulant selection (direct oral anticoagulants (DOACs), LMWH, or warfarin) was at the discretion of the treating physician and was not guided by a standardized protocol. Socioeconomic variables included education level (categorized as high (technical education through university degree) or low (preschool through secondary/high school)), socioeconomic class (categorized as high or low according to the socioeconomic stratum defined in the public services bill), and presence of a support network, if documented in the clinical history. Physician-related variables included the treating specialty (internal medicine or other specialties) and the documented reason for delayed discharge, as recorded in the medical record.
Early discharge was defined a priori as discharge within 24 h of the initial evaluation; otherwise, discharge was classified as delayed. Because patients are administratively admitted once PE is suspected or confirmed, often remaining physically in the ED due to bed constraints, we defined early discharge strictly as discharge within 24 h from the index evaluation, regardless of physical location. Major bleeding was defined according to the International Society on Thrombosis and Hemostasis criteria as bleeding associated with hospitalization, a hemoglobin decrease >2 g/dL, transfusion of ≥2 units of red blood cells, bleeding at a critical site (intracranial, intraspinal, intraocular, retroperitoneal, intra-articular, pericardial, or intramuscular with compartment syndrome), or fatal bleeding. 20 Anemia severity was classified according to the National Cancer Institute criteria as follows: mild (hemoglobin between 10.0 g/dL and the lower limit of normal), moderate (hemoglobin 8.0–10.0 g/dL), and severe (hemoglobin <7.9 g/dL). Thirty-day mortality was ascertained through the platform of the Administration of Resources of the General Social Security Health System (ADRES), which systematically records the date of death for patients affiliated with the Colombian social security system. 21
We summarized categorical variables with counts (n) and percentages (%) and continuous variables as medians with interquartile ranges (IQRs) or means with SDs, as appropriate. The Shapiro–Wilk test was used to assess normality due to its good performance in small samples and in the presence of skewed distributions (as observed for age and length of stay). Given the moderate sample size and the descriptive objective of the study, we did not conduct formal hypothesis testing or multivariable modeling. Early discharge rates across time periods are presented descriptively. Statistical analyses were conducted using Stata version 16 (StataCorp, College Station, TX).
Results
Of the 765 patients with acute PE included in the registry, 703 were excluded, and 62 met criteria for low risk and were analyzed (Figure 1). The clinical and sociodemographic characteristics of the analyzed patients, according to early or delayed discharge, are presented in Table 1. The median age was 45.5 (IQR: 32–62) years, and 58% (32/62) were women. Chronic kidney disease was present in 4.8% (3/62), all of whom were women with lupus nephropathy. A prior history of venous thromboembolism was documented in 8.0% (5/62). No patient had a history of major bleeding. The absence of patients with active cancer or age >80 years was expected, given the requirement of sPESI of 0 for inclusion in the low-risk cohort.

Flowchart of patient selection for low-risk pulmonary embolism cohort.
Baseline characteristics of patients with low-risk acute pulmonary embolism according to early or delayed discharge.

Data are presented as n (%). No hypothesis testing was performed, and no causal inference is intended.
IQR: interquartile range; LMWH: low-molecular-weight heparin; NSAIDs: nonsteroidal anti-inflammatory drugs; VTE: venous thromboembolic disease; UFH: unfractionated heparin.
Early discharge occurred in 13/62 patients (20.9%), whereas 49/62 (79.1%) had delayed discharge, with a median length of stay of 3 days (IQR: 2–6). Early discharge rates increased over time, from 9.5% (4/22) during the September 2019 to July 2022 period to 45% (9/20) during the August 2022 to November 2024 period (p < 0.01).
All patients with chronic kidney disease were in the delayed discharge group. Among patients with comorbidities, discharge timing was influenced primarily by treatment logistics. Three individuals with systemic lupus erythematosus (SLE) and suspected antiphospholipid syndrome (APS) remained hospitalized to initiate warfarin and achieve therapeutic international normalized ratio (INR) levels; no lupus flares were documented. The three patients with chronic kidney disease overlapped with the SLE group and did not experience contrast-induced or acute renal deterioration. Of the three patients with diabetes mellitus, one required inpatient insulin adjustment. Overall, comorbidities themselves were not the principal drivers of delayed discharge; rather, delays were mainly related to anticoagulation strategy and, less frequently, metabolic control.
Among seven patients with uncontrolled chest pain, five had pulmonary infarction identified on index imaging, suggesting a plausible source of pain; one had musculoskeletal pain related to cervical disc herniation.
Initial anticoagulation was with LMWH in more than 90% of cases; one patient in the delayed discharge group received unfractionated heparin due to extensive DVT. Warfarin bridging was required in 9/62 patients (14.5%). Five patients were taking 100 mg/day of aspirin and two were taking NSAIDs prior to admission; these medications were discontinued at admission. Baseline anemia was present in seven patients (five mild, one moderate, and one severe due to abnormal uterine bleeding requiring transfusion). Except for the severe case, delayed discharge was primarily related to warfarin initiation and INR titration rather than to anemia itself.
Table 2 summarizes clinical outcomes according to early or delayed discharge. There were no in-hospital or 30-day deaths, no major bleeding events, and no rehospitalizations within 30 days. Five patients returned to the ED within 30 days, all due to chest pain: two with persistent pleuritic pain in the setting of pulmonary infarction, one with symptoms consistent with a viral syndrome, one with post-thrombotic–type limb pain, and one with nonspecific pain. Diagnostic evaluation excluded recurrent PE and major bleeding in all cases, and all patients were discharged within 24 h.
Clinical outcomes in low-risk cohort by discharge timing.

*All ED revisits were related solely to pain and did not result in admission or hospitalization.
Data are presented as n (%). No hypothesis testing was performed, and no causal inference is intended.
ED: emergency department; IQR: interquartile range.
Table 3 presents physician-related and socioeconomic characteristics potentially associated with delayed discharge. Overall, compared with the early discharge group, patients with delayed discharge had numerically higher proportions of anemia (14.2% vs. 0%), thrombocytopenia (6.1% vs. 0%), use of medications associated with increased bleeding risk (14.2% vs. 0%), management outside the internal medicine service, and lower education/socioeconomic levels. DOACs were prescribed in 84.5% of patients in the early discharge group compared with 57.0% in the delayed discharge group. Warfarin was used exclusively in the delayed discharge group (18.3%). Among patients discharged on non-DOAC therapy (n = 23), documented reasons included suspected APS (warfarin or LMWH), bleeding risk (intramuscular hematoma, n = 2), and access/logistical barriers (bridging with LMWH while awaiting DOAC authorization, n = 1). Reasons were not systematically documented for all cases.
System- and process-level characteristics by discharge timing.
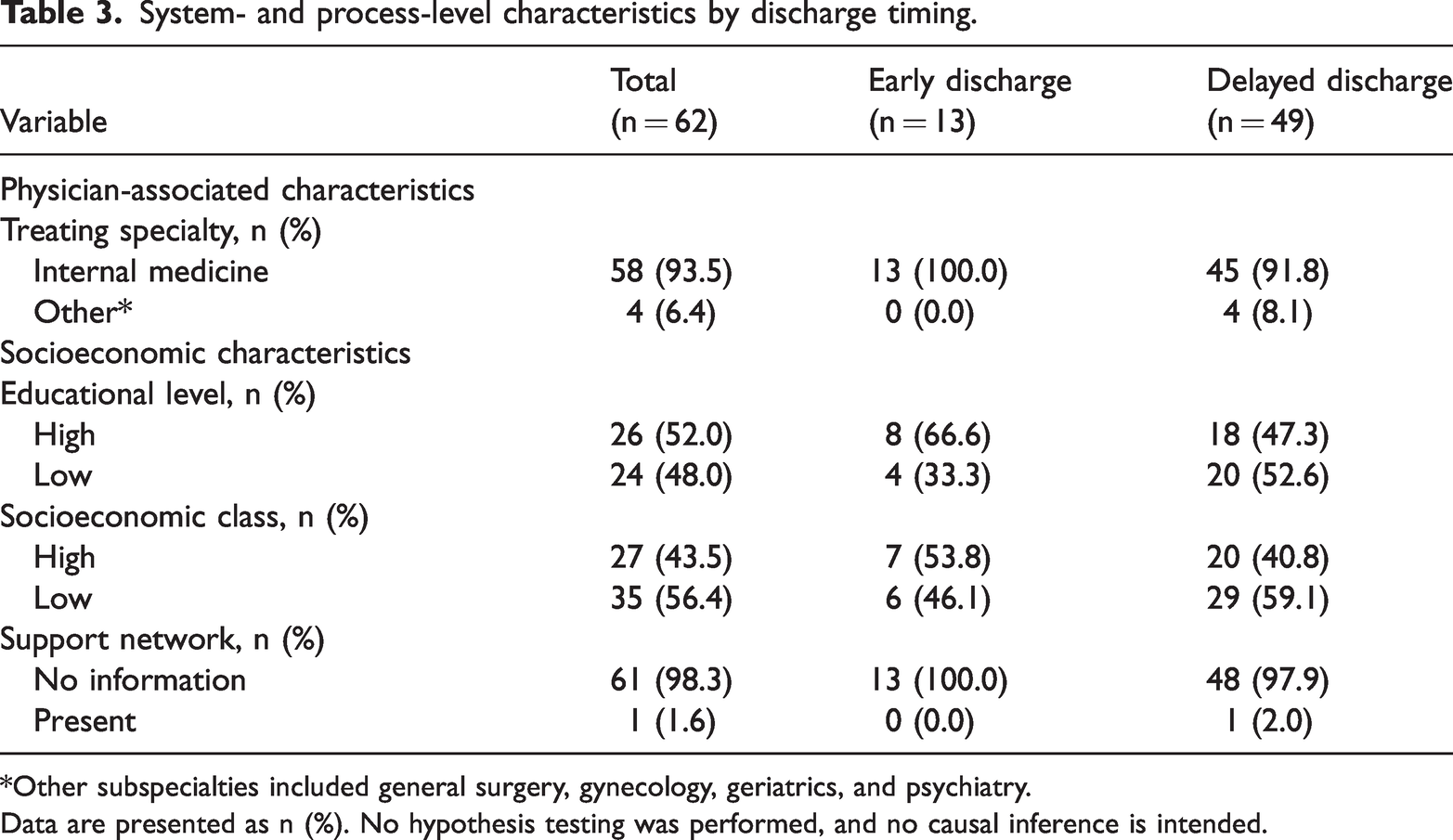
*Other subspecialties included general surgery, gynecology, geriatrics, and psychiatry.
Data are presented as n (%). No hypothesis testing was performed, and no causal inference is intended.
The most frequently recorded reasons for delayed discharge in the medical records were pending TTE (12/49; 24.5%), INR monitoring (11/49; 22.4%), management of other active comorbidities (11/49; 22.4%), delays in DOAC dispensing/authorization (9/49; 18.3%), and uncontrolled pain (6/49; 12.2%).
Discussion
This study provides insight into adherence to international guideline recommendations for early discharge in patients with low-risk PE at a Latin American tertiary care and oncology referral center. It also examines clinical, socioeconomic, and physician-related factors associated with nonadherence to these recommendations. Only 20.9% (13/62) of eligible patients were discharged within 24 h. Factors potentially contributing to delayed discharge included comorbidities such as chronic kidney disease, anemia, and thrombocytopenia; the attending physician’s specialty; lower educational attainment and socioeconomic status; pending TTE; INR monitoring; and delayed provision of DOACs.
There is robust evidence supporting the safety of early discharge in carefully selected low-risk PE patients. The HOME-PE trial randomized 26 European centers to determine eligibility for early discharge using either the sPESI or Hestia criteria and found that adverse events and 30-day mortality were close to 1% with both tools. 22 In addition, the MERCURY-PE and HoT-PE studies demonstrated similar results, with mortality, recurrent VTE, and major bleeding rates close to 1% among patients discharged early on rivaroxaban.23,24 An outpatient cohort of 109 low-risk patients managed without a formal follow-up pathway reported very low 1-year mortality, recurrence, and bleeding rates, reinforcing the longer-term safety of ambulatory care. 25
Beyond randomized and single-arm trials, recent population-based analyses are consistent with these findings. Using two large US databases (the Nationwide Emergency Department Sample and the Nationwide Readmission Database), Farmakis et al. 26 reported that among low-risk PE cases, home treatment directly from the ED occurred in approximately 31.5% of encounters and early discharge after hospital admission in 45.9%, with low 90-day rates of VTE recurrence (1.3%) and major bleeding (1.5%). In a complementary national registry analysis from Japan, Nishikawa et al. 27 found that patients with sPESI of 0 had 0% 30-day mortality; however, only 17% were managed at home. Taken together, these contemporary real-world data reinforce the safety of early discharge in appropriately selected low-risk patients. Consistent with this evidence, we observed no deaths, major bleeding events, or rehospitalizations at 30 days.
The low proportion of patients classified as low risk is consistent with the complexity of our center, where oncologic patients represent a substantial proportion of admissions, contributing to lower rates of sPESI of 0 compared with other cohorts. The rate of early discharge in our study (20.9%) was higher than that reported in Italy (2%) or France (4%), comparable to that in the United States (14%), but lower than rates reported in Canada (>50%).9,10 These findings are consistent with literature demonstrating wide variation according to local infrastructure, insurance coverage, outpatient follow-up capacity, and the presence of oncology referral centers. Despite the overall low adherence, we observed a progressive increase in early discharge over the study period, rising from 9.5% in 2019–2022 to 45% in 2022–2024, for an overall rate of 20.9%. A similar temporal trend was described by Farmakis et al., 26 who reported increasing use of home treatment for low-risk PE beginning in 2016. A Pulmonary Embolism Response Team (PERT) is available locally for high/intermediate-risk PE. Since 2018, low-risk PE has been managed through an Anticoagulation Stewardship Program that provides standardized discharge education and follow-up, which has likely contributed to the observed increase in early discharge rates. A comparable trend has been reported in Austria, where a 2.4-fold increase in early discharge was observed, from 11% in 2016 to 26% in 2019. 28 These findings underscore the importance of structured outpatient follow-up programs and continuous medical education to improve adherence to early discharge recommendations in this population.
Among factors potentially associated with delayed discharge, patients with anemia, thrombocytopenia, and chronic kidney disease were more frequently discharged late, many of whom also had a baseline diagnosis of SLE. Only two patients with anemia had moderate to severe anemia. In such cases, delayed discharge may be justified to optimize management of underlying comorbidities and address the potentially increased bleeding risk associated with anticoagulation therapy. Similar observations have been reported in other populations. For example, a RIETE registry study in Italy (2006–2013) found that 78% of patients with PE were hospitalized despite 56% being classified as low risk by PESI; anemia and a glomerular filtration rate <60 mL/min were identified as key factors influencing inpatient management decisions.11,12
Another factor associated with discharge decisions was the treating specialty: all patients discharged early were managed by internal medicine. This may reflect that certain specialties, such as internal medicine, are more familiar with and confident in the safety and benefits of early discharge. A 2014–2015 survey of European centers reported that the most frequently used prognostic approach was clinical judgment (36%), followed by validated tools (29%), such as PESI and sPESI. 13 These findings highlight the potential need for additional education and training across specialties to increase awareness and adoption of early discharge practices.
Approximately one-fourth of patients experienced delayed discharge due to pending TTE, which may not have been necessary in all cases. Prior studies have shown that RVD identified on CTPA is generally concordant with echocardiographic findings and has similar prognostic value, suggesting that routine echocardiography may not be required in all low-risk patients.29–32 A 2021 meta-analysis reported RVD in 38% of patients assessed by CTPA and in 25% assessed by echocardiography, with comparable 30-day mortality rates (1.4% vs. 2.8%).29,31 Similarly, a retrospective study found nearly identical proportions of RVD (37.8% vs. 33.8%, p = 0.61), with a strong correlation (r = 0.832, p < 0.001) and high predictive accuracy for 30-day adverse events. 32 These findings support the use of CTPA as a standalone imaging modality for risk stratification in selected low-risk PE patients. 30
Another barrier to early discharge, identified in approximately 15% of cases, was delayed authorization and provision of DOACs by patients’ health insurance, which postponed timely access to medication. These delays were primarily administrative, related to insurer authorization and pharmacy dispensing. Establishing structured outpatient PE follow-up programs with guaranteed anticoagulant access may help address this barrier. Similar challenges were described in a 2023 qualitative study of 11 US hospitals, where clinicians cited administrative delays and insurance coverage concerns for high-cost medications such as DOACs as contributors to prolonged hospital stays. 33 At our institution, partnerships with multiple insurance providers have been implemented to facilitate DOAC delivery prior to discharge, which has likely contributed to improved early discharge rates.
It is important to recognize that some factors associated with delayed discharge were clinically appropriate, such as uncontrolled pain. Pain is a common indication for inpatient management in patients with PE, particularly when intravenous analgesia is required, and it is included in the Hestia criteria for assessing eligibility for outpatient treatment. 34 In our study, six of seven patients with uncontrolled pain remained hospitalized for this reason. Persistent pain is also a frequent cause of ED reconsultation in PE populations, with reported readmission rates of approximately 3% at 30 and 90 days after discharge.35,36 Consistently, we observed five ED visits within 30 days, all due to pain. None of these patients required hospitalization beyond 24 h; after exclusion of complications and adequate pain control, all were safely discharged.
Conversely, a higher proportion of patients in the delayed discharge group had lower education level and lower socioeconomic status, suggesting that these factors may appropriately influence discharge planning. No referrals from rural areas were identified. Although lower educational or socioeconomic status may complicate logistics, our data do not allow confirmation or exclusion of an association with limited healthcare access. These comparisons are descriptive and hypothesis-generating and should be evaluated in future studies.
A 2023 qualitative study similarly identified concerns regarding outpatient follow-up and health system coverage as barriers to safe discharge. 33 The 2019 ESC guidelines also emphasize the importance of ensuring adequate outpatient care and reliable access to anticoagulation therapy, taking into account patient adherence and healthcare accessibility. 4
As noted earlier, the HOME-PE trial demonstrated that the Hestia criteria are a validated tool with safety and effectiveness comparable to sPESI. A recent meta-analysis further confirmed that both strategies carry a low risk of misclassifying low-risk PE when used to guide early discharge.22,37 Although the Hestia criteria perform similarly to sPESI and align well with practical discharge barriers (e.g. uncontrolled pain and social factors), several Hestia items, particularly “medical or social reasons for admission >24 h” and explicit documentation of social support, are inconsistently recorded in routine clinical notes at our institution. Because our study relied on a retrospective registry, we selected sPESI to ensure reproducible case ascertainment. Prospective work within our program will incorporate the Hestia criteria to better capture real-world eligibility, including patients with cancer.
The primary limitation of this study is the small sample size. Of all patients with acute PE included in the registry, only 62/765 (8.1%) met criteria for low risk and were included in the analysis, a proportion lower than that reported in previous studies.9,10,28 This likely reflects the characteristics of our patient population, as our institution is a high-complexity referral hospital and regional oncology center. Because the presence of active cancer automatically adds one point to the sPESI score, oncology patients are excluded from the low-risk category. A recent analysis of this same institutional cohort (2019–2022) showed that 8.3% of PE cases were low risk, 77.6% intermediate-low risk, 11.2% intermediate-high risk, and 2.7% high risk. 38 The low prevalence of low-risk PE is consistent with the high cancer burden in our population, in which 33.4% of patients had cancer (85.2% with active disease at the time of PE diagnosis), a rate considerably higher than the approximately 20% reported in other cohorts. 38
Our sample of low-risk patients is underpowered for inferential analyses; therefore, observed differences according to treating specialty, education level, or socioeconomic class may reflect chance, unmeasured confounding, or institutional workflow. We report these patterns descriptively to inform future, adequately powered, multicenter studies.
An additional limitation is that, in a small proportion of cases (<5% of PE cases), the six sPESI variables were not consistently documented during the initial ED evaluation. However, complete information was available for all 62 included patients, allowing accurate classification.
Despite these limitations, to the best of our knowledge, this study is the first to report adherence to early discharge recommendations and to identify implementation barriers in a Latin American population. It provides relevant insights and generates hypotheses to guide future strategies aimed at optimizing PE management.
Conclusions
In this single-center Latin American cohort, adherence to early discharge for low-risk PE was limited, despite there being no deaths, major bleeding events, or rehospitalizations at 30 days and only five ED revisits for pain. These findings highlight system- and process-level barriers—particularly the use of echocardiography in patients with sPESI of 0, warfarin/INR monitoring, and delays in DOAC access, which may be addressed through standardized discharge pathways and structured outpatient follow-up. Future multicenter studies including larger and more diverse populations across both low- and high-complexity settings are warranted to validate these findings.
Footnotes
Author contributions
All authors of the present manuscript contributed to the development of the idea and study design, data collection, data analysis, and final writing of the article. All authors read and approved the final manuscript.
Ethical considerations
The study was approved by the institutional ethics committee (approval number FM-CIE-0354-23).
Consent to participate
The ethics committee granted a waiver of informed consent for retrospective analysis of deidentified data; verbal consent was obtained exclusively for 30-day telephone follow-up and when obtaining this consent, it was established that the results of the investigation would be published in an academic journal.
Consent for publication
Not applicable.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
