Abstract
Objective
Dermatofibrosarcoma protuberans is a low-to-intermediate–grade malignancy with high postoperative recurrence risk. It is often misdiagnosed as superficial venous malformation due to nonspecific manifestations and clinical features similar to those of other soft tissue tumors. Ultrasound examination is crucial for dermatofibrosarcoma protuberans evaluation; however, its value in differentiating dermatofibrosarcoma protuberans from superficial venous malformation remains underinvestigated.
Methods
This retrospective study analyzed and compared the clinical and ultrasonographic features between 41 histopathologically confirmed dermatofibrosarcoma protuberans cases and 41 superficial venous malformation cases. Clinical data included sex, age, tumor size, and location. Ultrasound findings were categorized into five types, and the following features were evaluated: shape, boundary, pattern, invasion depth, and vascularity.
Results
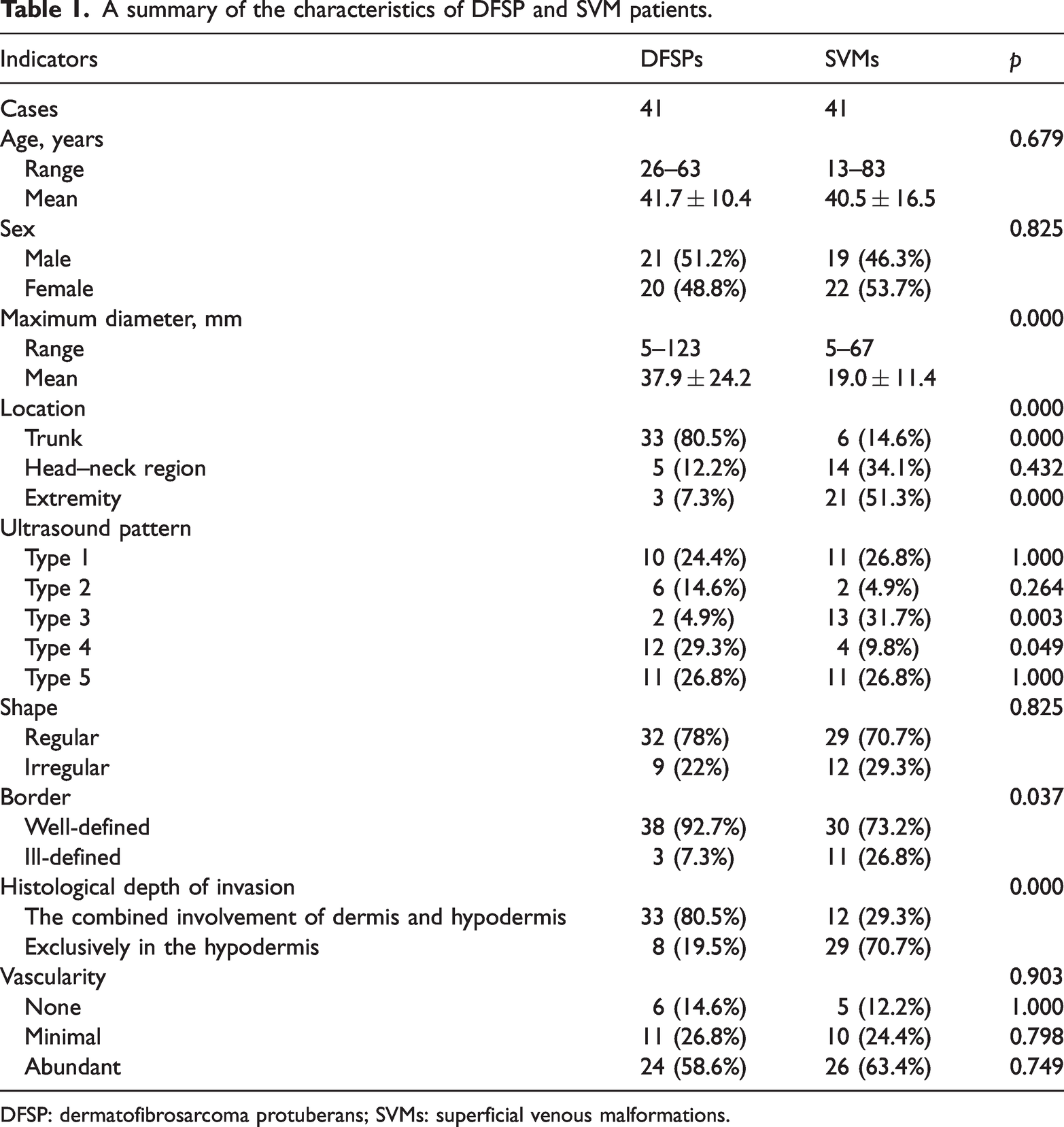
The dermatofibrosarcoma protuberans tissues had significantly larger maximum diameters than superficial venous malformations (p < 0.05). Anatomically, dermatofibrosarcoma protuberans predominantly occurred on the trunk, while superficial venous malformations were more common on the extremities (p < 0.05). Dermatofibrosarcoma protuberans tissues mostly invaded both dermis and hypodermis, whereas superficial venous malformations primarily involved the hypodermis (p < 0.05). Dermatofibrosarcoma protuberans tissues had clearer boundaries (p < 0.05) and were more likely to show hyperechoic cord-like structures (type 4), while superficial venous malformations frequently presented network structures (type 3) (p < 0.05).
Conclusions
Ultrasound can provide valuable morphological features to assist in the differential diagnosis of dermatofibrosarcoma protuberans and superficial venous malformation, improving preoperative assessment accuracy and guiding treatment planning.
Keywords
Introduction
Dermatofibrosarcoma protuberans (DFSP) is a relatively rare, low-to-intermediate–grade malignant tumor originating from the dermis, with a tendency to invade deep subcutaneous tissues, including muscle and bone. 1 Surgery with histologic margin control is necessary due to the high recurrence rate of this condition. Thus, correct preoperative assessment is important. However, the nonspecific characteristic of DFSP often leads to misdiagnosis. A retrospective study showed that >50% of DFSP patients have been misdiagnosed by physicians. 2
DFSP often presents as a cutaneous purplish-red lesion due to dermal infiltration, which can be clinically mistaken for superficial venous malformation (SVM), a more common benign subcutaneous vascular malformation characterized by normal to blue–purple surface coloration. 3 SVM was previously known as “cavernous hemangioma.” According to the International Society for the Study of Vascular Anomalies (ISSVA) classification, SVMs are congenital vascular malformations, not true neoplasms, characterized by morphological abnormalities of venous channels. 3 SVMs arise from dilated small veins and adipose tissue, forming thin-walled cystic structures that grow invasively. SVMs can be managed conservatively with venous embolization or simple excision. The standard treatment for DFSP involves meticulous surgical resection with histologic margin control (e.g. Mohs micrographic surgery); this method is superior to wide local excision in its ability to minimize recurrence risk. 4 The similar clinical manifestations lead to diagnostic confusion in primary care settings. Misdiagnosis can result in inappropriate management, highlighting the need for accurate preoperative differential diagnosis.
Ultrasound (US) is a noninvasive, rapid, and widely available modality that is increasingly being used to evaluate soft tissue masses. 5 Several studies have documented the US findings of DFSPs.6,7 However, the differential diagnosis between DFSPs and SVMs remains underexplored.
This study aimed to retrospectively analyze the US characteristics of DFSP and compare them with those of SVMs to identify distinguishing features that can improve preoperative assessment accuracy.
Material and methods
Patients
Patients with pathologically confirmed DFSPs treated at Peking University Shenzhen Hospital between November 2014 and October 2024 were retrospectively enrolled. For comparison, several consecutive SVM patients were randomly selected from 2023 to 2024. SVMs located in the skin and subcutaneous soft tissue layers were included, while intramuscular venous malformations involving only the muscle layer were excluded due to their distinct characteristics. All patients were diagnosed postoperatively. The study protocol was approved by the institutional ethics committee; written consent forms were obtained from all patients. All patients underwent the preoperative US examination, and clinical data on age, sex, and location were recorded. We have deidentified all patient details. This study was conducted in accordance with the ethical principles of the Declaration of Helsinki (1975, as revised in 2024) and followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 8
US examination
US images were captured using advanced US scanners (TOSHIBA 500, Philips IU 22 and the LOGIQ E9) equipped with 5–15 MHz linear array transducer. Two US physicians with >10 years of experience conducted the imaging assessment. Two sonographers with >10 years of experience, blinded to the histopathological diagnoses, independently assessed the images. Discrepancies were resolved by consensus. For gray-scale US images, the depth for visualizing the mass were set at 2–10 cm, and the maximum gain was set at 85%–90%. Several characteristics, including the main echotexture, echogenicity, shape, margin, border, and histological level of invasion, were evaluated. The size and the maximum diameter of the lesion was measured.
According to previous studies and routine clinical experience, the US manifestation pattern can be categorized into five types as follows:
Type 1. Homogeneous hypoechoic mass; Type 2. Solid mass with hypoechoic finger-like projection extending to the hyperechoic area; Type 3. Lesion with a hypoechoic or hyperechoic main body, containing anechoic/duct-like structures and hyperechoic walls forming a reticular network; Type 4. Predominantly hypoechoic lesion with hyperechoic cord-like structures; Type 5. Heterogeneous mixed pattern with hypoechoic and hyperechoic area (without any specific characteristics mentioned above) (Figure 1).
9
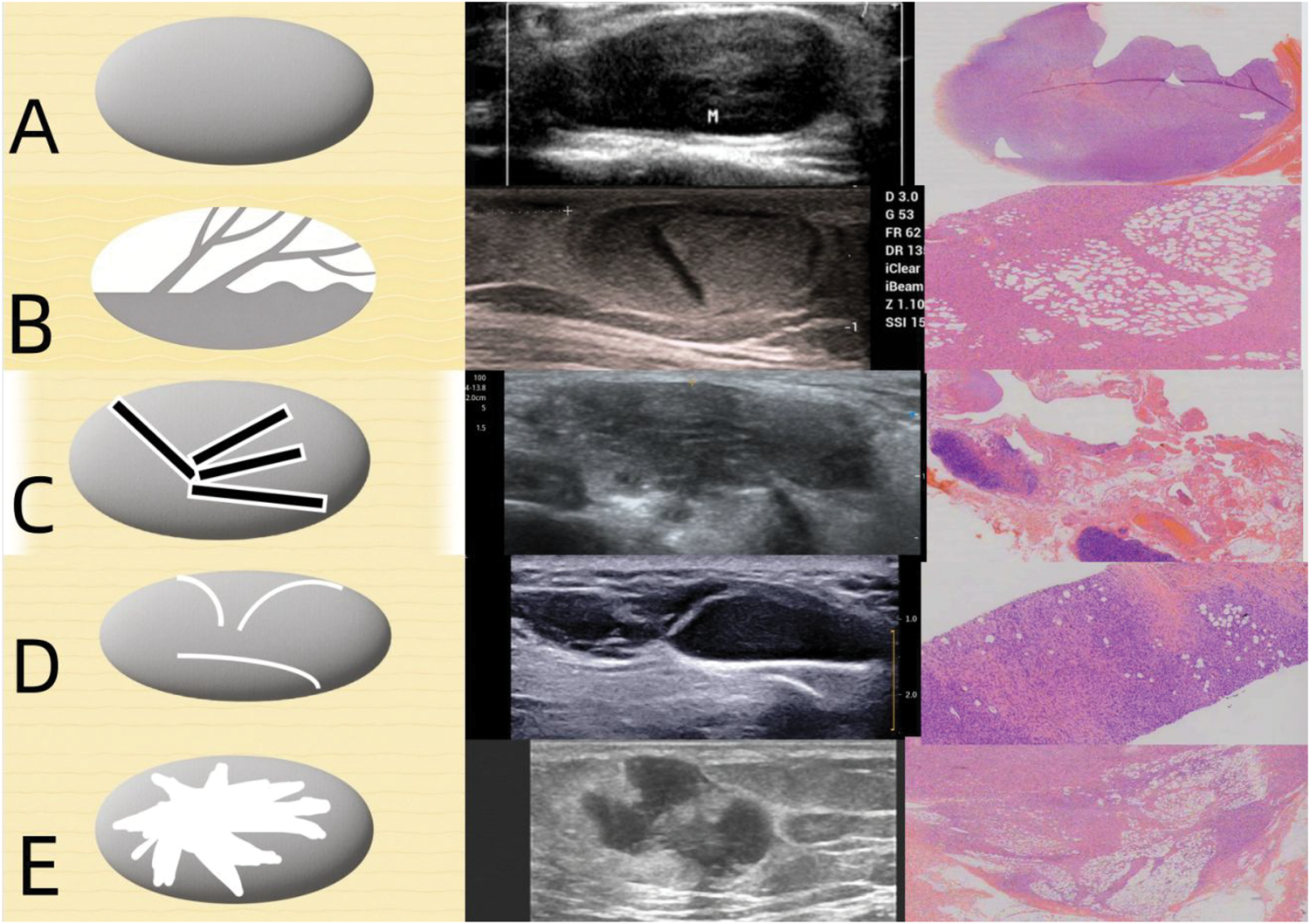
Five types of ultrasound manifestations of DFSP and SVM with corresponding pathology and schematic diagrams. (a) Type 1: Homogeneous hypoechoic mass. Pathology showed that tumor cells had clear boundaries and were evenly distributed. (b) Type 2: Solid mass with hypoechoic finger-like projection extending to the hyperechoic area. Pathology showed the proliferation of tumor cells along the septa and dispersed among fat cells (c) Type 3: Lesion with a hypoechoic or hyperechoic main body containing anechoic/duct-like structures and hyperechoic walls forming a reticular network. Pathology showed that the small veins expanded outward and formed a thin-walled cystic structure. (d) Type 4: Predominantly hypoechoic lesion with hyperechoic cord-like structures. Pathology showed that the hyperechoic cord-like structure correlates with peritumoral adipose tissue infiltration and (e) Type 5: Heterogeneous mixed pattern with hypoechoic and hyperechoic area (without any specific characteristics mentioned above). Pathology showed that the tumor cells and adipose cells were distributed in a mixed pattern. DFSP: dermatofibrosarcoma protuberans; SVM: superficial venous malformation.
For color Doppler US, the pulse repetition frequency (PRF) was set at 3–8 cm/s. The color Doppler gain was set just below the color noise threshold to visualize the low-velocity flow. The Adler blood flow classification divides blood flow into grades 0–4. 10 We considered that grade 1 indicated no blood flow; grades 2, minimal blood flow; and grades 3–4, abundant blood flow (Figure 2).

The three levels of color Doppler ultrasound: (a) no blood signal, (b) minimal blood signal, and (c) abundant blood signal.
Data analyses
Statistical Package for Social Sciences (SPSS) software (version 25.0, IBM corporation, New York) was used for statistical analyses. Qualitative variables were expressed as frequencies (%). Quantitative variables were presented as mean ± SD with 95% confidence intervals (CIs). The chi-square test or Fisher exact test was used to compare potential differences in the US findings between DFSPs and SVMs. All analyses were two-tailed, and p <0.05 was considered statistically significant.
Results
Patient characteristics
In total, 41 patients with DFSP lesions were recruited for further studies, including 21 men and 20 women with a mean age of 41.7 years. Lesions were most commonly located in the trunk (80.5%), followed by the head–neck region (12.2%) and extremities (7.3%). The average maximum diameter of DFSP lesions was 37.9 mm (range: 5–123 mm, 95% CI: 30.8–45.0 mm).
The 41 SVMs patients included 19 men and 22 women, with a mean age of 40.5 years. Lesions were most commonly located in the extremities (51.3%), followed by the head–neck region (34.1%) and trunk (14.6%). The mean maximum diameter was 19.0 mm (range: 5–67 mm, 95% CI: 15.4–22.6 mm), which was significantly smaller than that of DFSPs.
US findings
Finger-like projections were observed in six patients (14.6%). Most DFSP lesions exhibited a regular shape (78.0%) and well-defined borders (92.7%). Over half (58.6%) of the DFSP lesions showed abundant vascularity. On US, the lesions had well-defined borders in 30 (73.2%) patients with SVMs, and the lesions exhibited regular shape in 29 (70.7%) patients. Furthermore, 29 (70.7%) SVM lesions were confined to the hypodermis. Vascularity assessment showed abundant blood flow in 26 (63.4%) SVM lesions.
Comparison between DFSP lesions and SVM lesions
In total, 41 SVM patients were recruited for comparison; a summary of the characteristics of DFSPs and SVMs is shown in Table 1. No statistically significant difference in age or sex were observed between the two groups. The maximum diameter of DFSP lesions was larger than that of SVM lesions (p < 0.05). The DFSP lesions were more frequently located in the trunk, whereas the SVM lesions were more common in the extremities (p < 0.05). Regarding US pattern, the hyperechoic cord-like structures (type 4) were significantly more likely to be observed in DFSP lesions than in SVM lesions (p < 0.05). The network structure (type 3) was more likely to be observed in SVM lesions than in DFSP lesions (p < 0.05).The DFSP lesions also exhibited more well-defined border than SVM lesions (p < 0.05) (Figure 3).
A summary of the characteristics of DFSP and SVM patients.
DFSP: dermatofibrosarcoma protuberans; SVMs: superficial venous malformations.

Two typical cases of DFSP and SVMs. (a) Case 1 (DFSP): A 34-year-old male with a painless mass on the chest. Ultrasound showed a 24 × 18-mm hypoechoic mass with well-defined borders, regular shape, and internal hyperechoic cord-like structures (type 4 pattern). The CDFI showed abundant blood signal and (b) Case 2 (SVM): A 32-year-old female with a soft mass on the left hand. Ultrasound revealed an 11 × 5-mm hypoechoic mass with a reticular network structure (type 3 pattern) and abundant blood flow. DFSP: dermatofibrosarcoma protuberans; SVM: superficial venous malformation; CDFI: color Doppler flow imaging.
Discussion
US is increasingly being utilized in the diagnosis of dermatology diseases and is the first-line modality to evaluate DFSPs. Surgical excision with clear margins is the preferred treatment, utilizing techniques such as Mohs micrographic surgery. 11 SVM lesions are similar to DFSP lesions, accounting for 7%–10% of all benign soft tissue tumors; 12 however, the treatment of these two lesions is completely different. Thus, accurate preoperative assessment of DFSPs, including the extent of invasion, is important for surgical planning. In our study, we evaluated the clinical and ultrasonic feature of DFSP lesions; these lesions were typically located in the trunk, closely associated with the dermis, and often involved the hypodermis. This finding was consistent with those of previous studies. 13 In contrast, SVM lesions predominantly occur in the extremity and frequently invade the hypodermis. DFSP lesions originate from the dermis and tend to invade the deeper adjacent structures, such as the subcutaneous fat tissue, even the muscles. Serra- Guillén et al. 1 reported that in 62.6% cases, the invasion did not extend beyond the subcutaneous tissue, and in 0%–24.6%, there was infiltration into the fascia or muscle. The cellular pleomorphism of DFSP lesions was significantly associated with deep invasion, representing an important prognostic factor in metastasis and recurrence. 14 In our study, all DFSP lesions were confined to dermis and hypodermis levels; none of the lesions invaded the muscle or bone, in contrast to previous reports. As a single-center, retrospective study, our results are limited by sample size and generalizability. Crucially, the DFSP cases in this study were primarily identified from the dermatology department. The classic initial presentation of DFSP is a slow-growing, often asymptomatic cutaneous plaque or nodule that exhibits color change, which is most frequently first detected and diagnosed by dermatologists. Consequently, when these patients were referred to our center for preoperative US, the tumors were typically at a relatively early clinical stage. This observation may also be influenced by improved public health awareness and the widespread availability of diagnostic imaging. Increased patient vigilance regarding skin lesions and easier access to medical consultation can aid earlier detection before deep tissue invasion occurs.
On US, the size of SVM lesions was much smaller than that of DFSP lesions. DFSP lesions manifested as regular, well-defined hypoechoic masses. Round or ovoid tumors, on occasions with lobulated margins, seemed to be a consistent ultrasonic finding. 15 Compared with SVM lesions, DSP lesions more commonly exhibited irregular shapes and ill-defined borders, which is correlated with their aggressive pathological growth pattern.
US revealed the specific feature of DFSPs, which can help distinguish them from other soft tissue masses. In our study, the hyperechoic cord-like structure (type 4) was significantly more common in DFSP lesions than in SVM lesions. This ultrasonic feature has not been previously reported in the literature. We hypothesize that the hyperechoic cord-like structure is correlated with peritumoral adipose tissue infiltration. Finger-like projections have been documented as a typical feature of DFSPs. 16 According to the literature, 9 the distinct US characteristics were closely related to the pathological findings, representing the proliferation of spindle cells along the septa and their dispersion among fat cells. However, our findings showed no statistical significance for this characteristic, possibly due to the limited sample size and interobserver variability in identification. The network structure (type 3) was the specific sign of SVMs which represented the tortuous and dilated intertumoral vessels. 17
Both DFSP and SVM lesions exhibited abundant blood flow on color Doppler US. The SVM lesions were highly vascularized. The abundant blood flow observed in DFSP lesions is consistent with hyperplasia of small blood vessels. 18 Owing to this overlap, we could not distinguish between the lesions using color Doppler US. In further studies, we may use contrast-enhanced US to explore the differences in microvascular circulation between the two diseases.
This study had certain limitations. First, multimodal US techniques, such as contrast-enhanced US and elastography, were not used to evaluate the DFSP cases. Second, the sample size of DFSP cases was relatively small. Third, the retrospective, single-center study design may limit the generalizability of our results.
Conclusion
US is not a standalone diagnostic tool for DFSP; however, it serves as a key preoperative imaging modality for assessing lesion size, location, invasion depth, and echo patterns. These features help distinguish DFSP from SVMs, complementing clinical and pathological evaluations.
Footnotes
Acknowledgments
Not applicable.
Author contributions
Nan Zhuang: Conceptualization, Methodology, Writing–Original Draft
Zhaoling Yi: Formal Analysis, Investigation, Data Curation
Ennuo Ma: Validation, Resources, Software
Ruixiang Yin: Investigation, Visualization, Writing–Review & Editing
Yusen Zhang: Resources, Data Curation
Chenyang Zhao: Formal Analysis, Validation
Heng LV: Methodology, Software
Lu Xie: Resources, Supervision
Yun Tian: Project Administration, Funding Acquisition
Mengying Liao: Supervision, Resources
Huanyu Liu: Supervision, Resources
Desheng Sun: Supervision, Project Administration
Tan Li: Formal Analysis, Validation
Feng Wang: Supervision, Writing–Review & Editing, Funding Acquisition
Haiqin Xie: Conceptualization, Supervision, Writing–Review & Editing, Project Administration
Availability of data and material
The data underlying this article will be shared on reasonable request to the corresponding author.
Declaration of conflicting interests
The authors declare that they have no competing interests
Funding
The authors declare that financial support was received for the research, authorship, and/or publication of this article. This work was supported by the General Program for Clinical Research at Peking University Shenzhen Hospital (NO. LCYJ2021003), Sanming Project of Medicine in Shenzhen (NO. SZSM202111011), National Key Research and Development Program of China (NO. 2023YFC3402605), Guangdong Basic and Applied Basic Research Foundation (NO. 2024A1515010486), Shenzhen High-level Hospital Construction Fund, and Peking University Shenzhen Hospital Scientific Research Fund (NO. KYQD2023302).
