Abstract
Background
Coronavirus disease 2019 (COVID-19) is a serious global health threat, and some patients with COVID-19 develop post-COVID-19 symptoms, which are characterized by symptoms following acute infection. In December 2022, China relaxed its strict COVID-19 policies, which was followed by outbreaks. Therefore, it is important to investigate post-COVID-19 symptoms in China to understand their prevalence and risk factors. This multicenter study examined post-COVID-19 symptoms among Chinese healthcare workers to better understand their epidemiology.
Methods
Healthcare workers from seven hospitals who met specific COVID-19 and work criteria participated in an anonymous online survey conducted from 17 July to 19 August 2023. The collected data were analyzed using various statistical methods to identify factors associated with self-reported post-COVID-19 symptoms.
Results
Of the 5213 healthcare workers who were included in the study, 17.0% reported post-COVID-19 symptoms. Binary logistic regression confirmed male sex (odds ratio = 1.418, 95% confidence interval: 1.151–1.747), doctor/nurse position (doctor: odds ratio = 1.406, 95% confidence interval: 1.074–1.840; nurse: odds ratio = 1.516, 95% confidence interval: 1.176–1.954), chronic respiratory disease (odds ratio = 1.576, 95% confidence interval: 1.035–2.399), moderate/severe COVID-19 (odds ratio = 1.583, 95% confidence interval: 1.323–1.894), severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) reinfection (odds ratio = 1.420, 95% confidence interval: 1.219–1.655), and high self-reported COVID-19 severity (odds ratio = 1.707, 95% confidence interval: 1.456–2.001) as post-COVID-19–associated factors.
Conclusion
Our real-world multicenter study showed that 17% of healthcare workers developed post-COVID-19 symptoms following SARS-CoV-2 infection. Therefore, targeted interventions are required for healthcare workers. This study provides important insights into the prevention and management of COVID-19 in healthcare workers and the general population.
Keywords
Background
Coronavirus disease 2019 (COVID-19) poses a severe threat to human health. On 31 October 2023, the global number of individuals infected with the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) exceeded 771 million, with over 69.7 million deaths. 1 An increasing number of patients with COVID-19 have reported long-term physical and psychological after-effects, drawing significant attention to post-COVID-19 symptoms.2,3 In October 2021, the World Health Organization (WHO) defined post-COVID-19 symptoms as “long COVID,” which refers to persistent symptoms experienced by some individuals after contracting COVID-19. These symptoms typically manifest within 3 months of infection, last at least 2 months, and cannot be explained by alternative diagnoses. 4 Research on post-COVID-19 symptoms is currently widespread, with numerous studies reporting their prevalence ranging from 2.3% to 87%.2,5–12
Compared with other countries, data on long COVID in China remain relatively limited. A cross-sectional study conducted in the United Kingdom (UK) reported that the prevalence of long COVID was 83% among individuals infected with the wild-type virus and 76% among those infected with the alpha variant. 13 Furthermore, a multicenter study in Switzerland has shown a declining trend in reported long COVID cases from the wild-type to alpha/delta and then the Omicron variants. 14 Similarly, a cross-sectional study in the United States of America (US) indicated lower rates of reported long COVID during periods when the Epsilon and Omicron variants were dominant. 15
Beginning in December 2019, China implemented a zero-COVID policy. However, since December 2022, the Chinese government relaxed its strict COVID-19 containment measures, 16 signifying China’s transition from a zero-COVID policy toward “coexisting with COVID.”17,18 Given the lack of studies on post-COVID-19 symptoms in China, research on this subject is critical to better understand the prevalence of and risk factors for post-COVID-19 symptoms in the Chinese population, including healthcare workers (HCWs).
Assessment of long COVID in the general population is challenging because it primarily relies on self-reporting. In contrast, HCWs can describe related symptoms more accurately due to their medical expertise. Moreover, they face a higher risk of contracting COVID-19 and experienced greater work pressure during the pandemic, which increased their risk of developing post-COVID-19 symptoms. Therefore, post-COVID-19 studies on HCWs can provide a more precise and objective understanding of its prevalence as well as serve as an important reference for developing protective strategies and health management measures.9,19–23
Critically, despite China being one of the first countries affected by COVID-19, there remains a substantial lack of research on the prevalence and risk factors of long COVID within its population. This gap limits our understanding of the pandemic’s long-term impact in this specific context. To address this knowledge gap and generate robust, generalizable findings, we conducted a multicenter study. This design enabled the recruitment of a geographically representative sample of HCWs and application of unified standards, thereby enhancing the reliability and comparability of data on this understudied topic in China. The advantages of this design are as follows: 1. Multiple centers can collect geographically representative samples to obtain more reliable data. 2. Unified standards can reduce information bias and improve the comparability of the results.24–26
Based on this rationale, we performed a multicenter study involving HCWs infected with SARS-CoV-2 from December 2022 to February 2023 in China, using a standardized epidemiological questionnaire to ensure data quality.
Methods
Study design and data collection
We recruited HCWs from seven tertiary hospitals in Zhejiang province, China, based on the following inclusion criteria: (a) age ≥18 years; (b) confirmation of COVID-19 based on nucleic acid/antigen testing; and (c) confirmed COVID-19 occurring >5 months prior to the survey (i.e. from December 2022 to February 2023). These criteria were implemented to ensure that all participants had sufficient time (well beyond the WHO’s minimum requirement of 2 months) to develop and report persistent symptoms consistent with the definition of long COVID, thereby increasing the specificity of case identification (Figure 1). An anonymous cross-sectional population-based survey was conducted online using the WeChat-incorporated Wen-Juan-Xing platform (Changsha Ranxing Information Technology Co., Ltd., Hunan, China). The interviewees voluntarily answered a self-administered questionnaire by scanning the Quick Response code on their mobile phones from 17 July to 19 August 2023. This retrospective, multicenter cross-sectional study was performed in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Taizhou Hospital of Zhejiang Province, China (Approval Number: K20230716). Respondents’ information was kept anonymous, and the Ethics Committee of Taizhou Hospital, Zhejiang Province waived the requirement for written informed consent from participants. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 27

Flow diagram depicting participant selection.
Literature search strategy
The Cochrane Library, PubMed, and EMBASE databases were searched for relevant studies from inception to 31 October 2023. Searches included a mix of Medical Subject Headings (MeSH) and free-text terms related to the key concepts of post-COVID-19 symptoms, with no language restrictions. After screening titles and abstracts and removing irrelevant and duplicate literature, 418 related articles were identified. After reviewing the original articles, reviews, and meta-analyses, certain studies were excluded. Specifically, studies focusing on a particular symptom, acute symptoms, and those that did not use the definition of COVID-19 provided by WHO were excluded. Ultimately, 48 articles were included. We extracted the following data from the included studies using a data-extraction form: first author, study design, study period, sample size, country, and prevalence of post-COVID-19 symptoms. These data were used to make comparisons between countries/locations.
Questionnaires
The questionnaire was developed de novo for this study. The initial item pool was generated based on a comprehensive review of the scientific literature on long COVID to ensure content validity and cover all relevant domains, including demographics, medical history, vaccination status, characteristics of the acute COVID-19 episode, and a comprehensive list of potential long-term symptoms. The critical question for identifying long COVID was explicitly framed around the WHO clinical case definition.
The questionnaire underwent a pilot test involving 50 volunteer HCWs to assess its clarity, comprehensibility, completion time, and face validity. Internal consistency and reliability for the self-reported COVID-19 severity scoring scale were excellent, with a Cronbach’s alpha (α) of 0.95.
We used the questionnaire to collect the following data. First, we collected information on basic patient characteristics, including sex, age, education level, position, seniority, underlying chronic diseases, allergy history, long-term alcohol consumption history, long-term smoking history, and COVID-19 vaccination history. COVID-19 vaccination history included the number of doses, dates of administration, and vaccine type. Booster vaccination status was defined as having received three or more doses. For risk factor analysis, a participant was considered “boosted” only if the booster dose (or the complete vaccination series) was administered prior to their first confirmed SARS-CoV-2 infection. In this study, “full vaccination” was defined as the completion of a two-dose primary series, consistent with the national policy at the time. A “booster dose” was defined as a third or subsequent dose. The vaccines administered to the study population were predominantly inactivated virus platforms (e.g. Sinovac and Sinopharm), which constituted the majority of vaccines available under China’s national vaccination program. Second, we investigated clinical manifestations following COVID-19, including fever (body temperature ≥38°C), fever lasting ≥3 days, clinical diagnostic type of COVID-19, and SARS-CoV-2 reinfection. Third, we collected data regarding self-reported COVID-19 using severity rating scale, with Cronbach’s α of 0.95. We selected the point with the largest Jordon’s index as the best cutoff value point for self-reported COVID-19 severity scoring classification through receiver operating characteristic (ROC) analysis. Finally, we used the survey to ask respondents whether they met the WHO definition of “long COVID” as “new or persistent symptoms that commenced within 3 months of the initial SARS-CoV-2 infection (confirmed by a positive nucleic acid or antigen test), persisted for at least 2 months, and could not be explained by an alternative diagnosis.” The onset and duration of symptoms were calculated from the date of the initial positive test for COVID-19. Almost all questions were closed, with checkboxes provided for responses. Details of the questionnaire are provided in Additional file 1.
Statistical analyses
The primary outcome of the survey was self-reported post-COVID-19 symptoms among HCWs. Counts and frequency distributions were used to summarize classified data, and chi-square (
Binary logistic regression was performed to identify factors associated with self-reported post-COVID-19 symptoms, with results expressed as odds ratios (ORs) and 95% confidence intervals (CIs). All risk factor analyses, including vaccination status, were based on the participant’s status prior to the COVID-19 episode under investigation. Variables that were significant (
The primary outcome of this study was the prevalence of self-reported long COVID, defined strictly according to the WHO clinical case definition. A case was operationally defined as a participant who fulfilled the following criteria: (a) had confirmed COVID-19; (b) self-reported the presence of new or persistent symptoms that commenced within 3 months of the initial infection; (c) reported that these symptoms persisted for at least 2 months at the time of the survey; and (d) attributed these symptoms to COVID-19.
Patient and public involvement
This study incorporated the perspectives of HCWs, specifically focusing on the experiences and insights of medical practitioners involved in the management and treatment of post-COVID-19 symptoms. Although the study primarily focused on medical personnel, their valuable input provided crucial insights into the clinical aspects and challenges associated with prolonged recovery from COVID-19.
Results
A total of 5847 questionnaires were submitted by HCWs across the seven tertiary hospitals. Based on the inclusion criteria (age ≥18 years, confirmed COVID-19, and infection for >5 months), 5843 HCWs were deemed eligible for participation. After applying the exclusion criteria (incomplete key data), 5213 HCWs were included in the final analysis, yielding a response rate of 89.2% (5213/5843). (Figure 1). The demographic and clinical characteristics of the patients are presented in Table 1. The mean age of survey respondents was 35.0 (±8.5) years, ranging from 19 to 67 years, and 75.7% were female. Among the respondents, 50.7% were nurses and 26.1% were physicians. A total of 886 respondents self-reported experiencing long COVID, corresponding to a prevalence of 17.0% (95% CI: 16.0%–18.0%).
Sociodemographic characteristics of healthcare workers and univariate analysis of factors associated with self-reported post-COVID-19 symptoms (n

Data were expressed as numbers and frequencies (in parentheses) with HCWs’ self-reported post-COVID-19 symptoms or without self-reported post-COVID-19 symptoms.
Data on age were continuous and were expressed as mean ± SD values. The differences in the age between the willingness and unwillingness groups were evaluated using t-test.
COVID-19: coronavirus disease 2019; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; HCW: healthcare worker.
Table 1 also shows that the following characteristics were significantly associated with self-reported post-COVID-19 symptoms: male sex (
The effects of independent factors on self-reported long COVID of HCWs were examined using a multivariable binary logistic regression model. Variables that were significant
Multiple logistic regression of factors associated with healthcare workers’ self-reported post-COVID-19 symptoms (n = 5213).
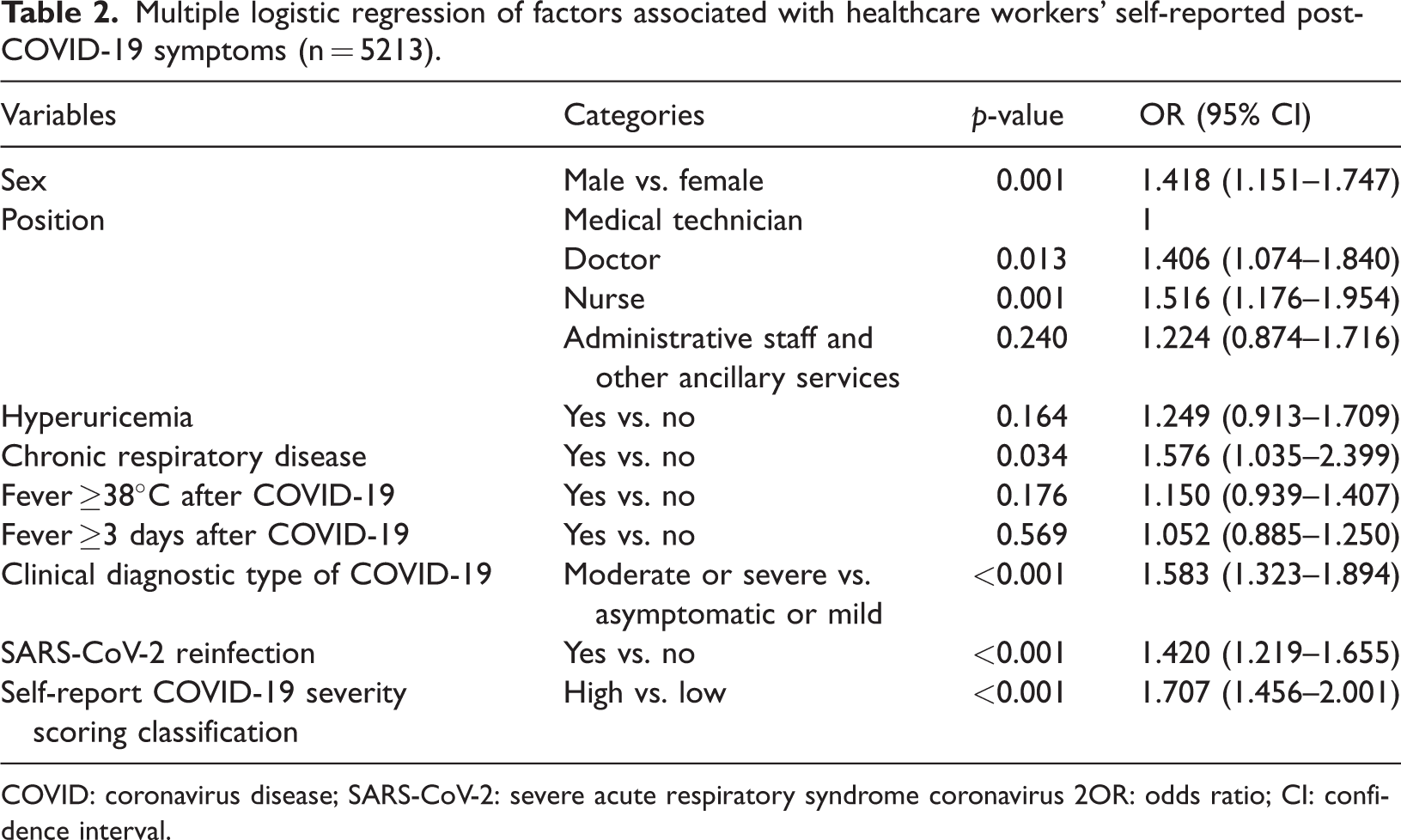
COVID: coronavirus disease; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2OR: odds ratio; CI: confidence interval.
As depicted in Table 3, the top five clinical symptoms reported by HCWs with self-reported post-COVID-19 symptoms were fatigue (68.5%), weakness (60.5%), memory loss (53.5%), discomfort after exercise (52.5%), and sleep disturbances (49.7%). The prevalence of individual symptoms is detailed in Table 3. For clearer visualization, the top 10 most frequently reported symptoms are presented in Figure 2.
Incidence of various clinical symptoms of post-COVID-19 condition in healthcare workers (n

COVID: coronavirus disease.
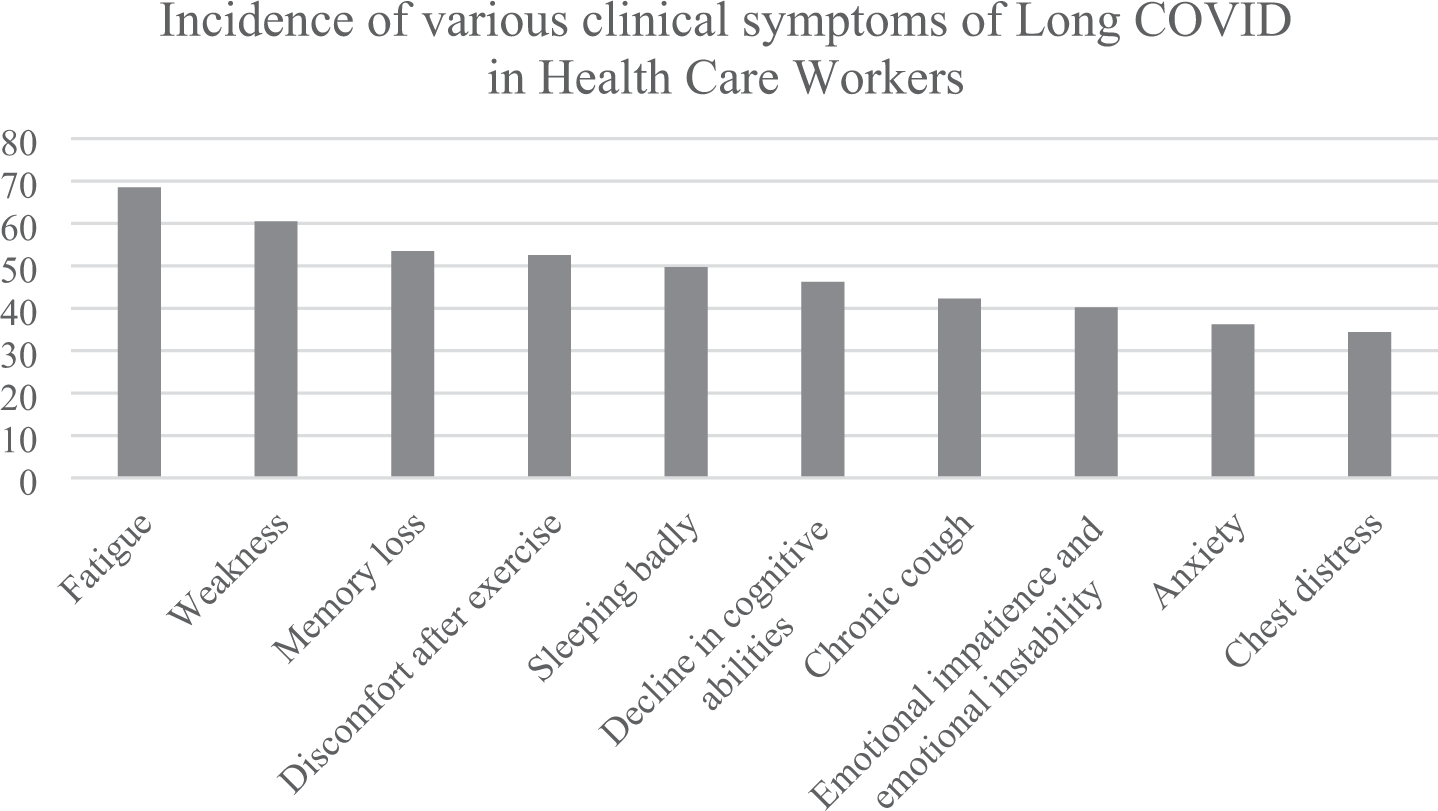
Prevalence of the top 10 most frequent symptoms among healthcare workers with self-reported long COVID (n = 886). COVID: coronavirus disease.
Discussion
In our study, 17.0% of HCWs self-reported long COVID; these findings are in contrast to the notably higher prevalence of 41% reported in a recent study conducted in France by Saade et al. 28 This discrepancy may be attributed to several factors, including differences in the study period and the dominant SARS-CoV-2 variants (Omicron era in our study vs. earlier, potentially more pathogenic variants in their study) as well as differences in the specific measurement instruments and definitions of long COVID employed.
The pathogenesis of post-COVID-19 symptoms is closely associated with the severity of acute SARS-CoV-2 infection. Established risk factors include infection with earlier viral variants, hospitalization, lower pre-infection vaccination status, and prolonged viral shedding, all indicative of a higher initial viral load and more severe inflammatory response. Our study confirms the critical role of acute disease severity, as evidenced by the strong associations with moderate/severe clinical type and high self-reported severity. Within this established framework, we aimed to investigate whether additional, context-specific factors, particularly relevant to HCWs, can further refine our understanding of post-COVID-19 symptom risk. The main risk factors included male sex, nursing/physician position, chronic respiratory disease, moderate/severe COVID-19, SARS-CoV-2 reinfection, and high self-reported COVID-19 severity. When interpreting this finding, it is most appropriate to compare the present results with those from other studies conducted on HCW populations, given the shared occupational context. For instance, studies among HCWs have reported prevalences ranging from 24.2% in South Africa to 73% in Switzerland. 29 This substantial variation underscores the influence of factors such as the dominant SARS-CoV-2 variant, study timing, and local definitions. Although the broader literature reports a highly variable prevalence (2.3%–87%) across general population studies, this wide range is cited primarily to highlight the methodological challenges in defining and measuring post-COVID-19 symptoms consistently, rather than for direct numerical comparison with our HCW-specific cohort. In contrast, larger and more recent multinational cohorts provide a critical point of comparison. For example, a study by Shukla et al., 30 which included 679 participants, reported a prevalence of 30.34%. Furthermore, a large-scale analysis that involved 5432 HCWs reported a prevalence of 16.3%. 29 The variation in prevalence across these studies as well as the present study can be attributed to differences in the dominant SARS-CoV-2 variant at the time of the study, geographical and healthcare system contexts, and the specific post-COVID-19 case definitions employed. The consistency of our estimate with the range reported in these larger, rigorous studies strengthens the validity of our findings.
At the beginning of the COVID-19 pandemic, China implemented strict control measures. By November 2022, more than 90% of China’s population had been fully vaccinated, providing protection against severe disease and deaths. After 3 years of implementing pandemic control measures, China’s capabilities in monitoring, testing, and providing clinical treatment significantly improved, laying the foundation for relaxing the COVID-19 control measures. Moreover, Chinese researchers reported that the Omicron variant was much less pathogenic and that the immunity level of residents of China was adequate against this variant. 16 Accordingly, in December 2022, the Chinese government relaxed strict COVID-19 containment measures, 16 including cancelling the centralized isolation of SARS-CoV-2–infected individuals, close contacts, and inbound travelers and eliminating regular COVID-19 nucleic acid amplification testing, lockdowns, and border control. Furthermore, residents were no longer required to present valid negative COVID-19 test results before entering public places or using public transportation. These massive changes reflected China’s transition from a zero-COVID policy toward “coexisting with COVID.”17,18 This underscores the need for better understanding regarding the prevalence of and risk factors for post-COVID-19 symptoms in China, thereby emphasizing the importance of the present study.
However, owing to the lack of consistent diagnostic criteria and clinical manifestations for post-COVID-19 symptoms, current research relies primarily on self-reported experiences and subjective factors, resulting in significant variance in reported prevalence and unclear risk factors in the literature.5,31–33 To overcome this limitation, we surveyed HCWs who not only possessed greater knowledge of post-COVID-19 symptoms but were also at higher risk of developing them.9,19–23
Research on post-COVID-19 symptoms is common. During our search for published papers on post-COVID-19 symptoms, we found different definitions of post-COVID-19 symptoms, primarily set by four organizations, including the WHO, Centers for Disease Control in the US, National Institute for Health and Care Excellence in the UK, and Association of the Scientific Medical Societies in Germany.4,34–37 We organized the literature according to the WHO’s definition of post-COVID-19 symptoms, as presented in Additional file 2. The prevalence of post-COVID-19 symptoms ranged from 2.3% to 82.9%. Six studies focused on post-COVID-19 symptoms in adolescents and children, with prevalence rates ranging from 15.2% to 79.5%.38–43 Among these, one study specifically examined children with multisystem inflammatory syndrome, whereas the other five involved previously healthy children or adolescents. One of these studies conducted an age-stratified analysis revealing that in children and adolescents aged <18 years, the prevalence of post-COVID-19 symptoms increased with age. Moreover, in studies on post-COVID-19 symptoms in adults, a higher proportion of hospitalized patients reported post-COVID-19 symptoms compared with outpatients.44–48
Studies have also evaluated post-COVID-19 symptoms in HCWs. In four such studies, the prevalence of post-COVID-19 symptoms ranged from 24.2% to 73%.49–52 A South African study involving 62 frontline HCWs reported a post-COVID-19 symptom prevalence rate of 24.2%. The present study explored the relationship between post-COVID-19 symptoms and various factors including demographics, general health status (smoking, alcohol consumption, and recreational drug use), and physical activity. In a study from Morocco that included a cohort of 118 HCWs, 47.2% reported post-COVID-19 symptoms. Chest computed tomography (CT) revealed that lung involvement was a risk factor for its occurrence, whereas no impact of SARS-CoV-2 vaccination was observed. Furthermore, a prospective cohort study from Brazil used the EuroQol-5D and EuroQol visual analog scales to investigate 289 HCWs. This study reported that 36.2% of the participants had experienced post-COVID-19 symptoms. A multicenter study in Switzerland involving 3346 HCWs who had contracted COVID-19 showed that 73% developed post-COVID-19 symptoms. Quantitative anti-S antibody (anti-S) titers were associated with high symptom scores, with acute viral symptoms serving as the best predictor of long-term COVID-19 symptoms. In addition, baseline physical activity was negatively associated with cognitive impairment and fatigue. However, research findings concerning post-COVID-19 symptoms in China are relatively limited. One previous cross-sectional study indicated that >50% of patients with COVID-19 have reported experiencing post-COVID-19 symptoms. 53
The current study found that male respondents and healthcare professionals in direct contact with patients (physicians and nurses) were more likely to develop post-COVID-19 symptoms. Contrary to a substantial body of literature from the general population, which identifies female sex as a risk factor for post-COVID-19 symptoms, our study found a significantly higher risk among male HCWs. This discrepancy may be attributable to the unique occupational context of our cohort. HCWs experience high-risk and potentially repeated viral exposures, which may result in a higher initial viral load, a proposed driver of long-term sequelae. In this professional setting, differential exposure risk by sex, possible linked to clinical role allocation, may outweigh the underlying biological sex differences. Furthermore, as medical professionals, male and female participants in our study are likely to report symptoms with high clinical accuracy, potentially mitigating the sex-based reporting bias observed in community-based studies. The specific context of the first Omicron wave in China, which placed unprecedented strain on the healthcare system, may also have contributed to this distinct risk profile. Some studies have reported that women exhibit robust T-cell responses to SARS-CoV-2 infection, which may help virus clearance and reduce the risk of COVID-19. In contrast, the immune responses in men may be slow or delayed, resulting in worse disease outcomes. 54 Moreover, estrogen may have a protective effect and reduce the damage caused to the body by SARS-CoV-2 infection, whereas testosterone may suppress immune responses. These hormonal differences may be one of the reasons for the higher susceptibility in men for post-COVID-19 symptoms. 55 Professionally, physicians and nurses who are in direct contact with patients and have initial exposure to SARS-CoV-2 virus tend to have a higher risk of developing post-COVID-19 symptoms. This may be explained by findings showing that a high initial viral load during the first infection is an independent risk factor for post-COVID-19 symptoms. 56 HCWs, due to the nature of their work, experience repeated exposure to SARS-CoV-2, which can exacerbate the course and symptoms of the disease. 57 These findings must be interpreted in the context of the evolving immune landscape. It is well-established that repeated antigen exposure builds immune memory and protects against severe acute disease, a key reason for improved outcomes during the Omicron wave. However, our data suggest that this enhanced protection against severe acute illness does not fully extend to the prevention of post-COVID-19 symptoms. Each reinfection may present a renewed risk of triggering pathological processes (e.g. viral persistence and immune dysregulation) underlying post-COVID-19 symptoms, indicating that protection from severe acute disease and susceptibility to long-term sequelae may involve distinct pathways. Additionally, frontline HCWs experience significant psychological stress, which may be related to the later development of psychological issues such as anxiety and depression, thereby affecting post-COVID-19 symptoms. 58 Our study identified that nurses, followed by physicians, had a significantly higher risk of developing post-COVID-19 symptoms compared with medical technicians. This finding is explained by the substantially higher frequency and intensity of direct patient contact among these frontline roles. This occupational exposure likely results in a higher viral inoculum during the initial infection, which is a proposed risk factor for severe acute disease and post-acute sequelae. Furthermore, nurses are predominantly responsible for performing aerosol-generating procedures and providing prolonged, close-proximity patient care, further amplifying their biological risk. Beyond the immediate viral exposure, the immense and sustained occupational stress and burnout prevalent among nursing staff during the pandemic may have contributed to this elevated risk. Chronic stress can modulate immune function and potentially hinder recovery, exacerbating the persistence of symptoms that characterize post-COVID-19 condition.
Furthermore, we found that respondents with a history of respiratory diseases, moderate/severe COVID-19, SARS-CoV-2 reinfection, and high self-reported COVID-19 severity were more likely to develop post-COVID-19 symptoms. Patients with a history of respiratory disease often have reduced lung reserve and compliance. When such patients contract COVID-19, lung damage is typically more severe, and the repair process is more challenging. This can easily lead to long-term respiratory difficulties. 59 Underlying lung diseases can increase the risk of COVID-19, leading to pulmonary interstitial fibrosis and respiratory failure. The more irreversible the lung function damage that occurs during the disease, the greater the likelihood of long-term respiratory difficulties.60,61 In addition, individuals with severe acute and repeated infections often exhibit higher viral loads, leading to severe damage to the lungs and other organs. They frequently experience a severe “cytokine storm,” in which the overly activated immune system makes it difficult for the inflammatory response to resolve naturally, thereby prolonging the disease course. Moreover, individuals with severe illness experience extended periods of hypoxia, resulting in hypoxic damage to organs such as the brain and heart, which, once established, can be difficult to reverse and can serve as a foundation for post-COVID-19 symptoms.62,63
To address these risk factors, the following measures should be implemented: (a) proper arrangement of work schedules to prevent overburdening of frontline healthcare professionals, thereby reducing the risk of repeated exposure and alleviating psychological stress; (b) strengthening health monitoring, ensuring timely isolation and treatment upon detection of infections to reduce viral transmission, and identifying post-infection complications early; (c) providing psychological interventions, such as counseling, to alleviate stress among HCWs and decreases the occurrence of depression, anxiety, and other psychological issues, thereby reducing the prevalence of post-COVID-19 symptoms and their complications; and (d) actively implementing rehabilitation therapy for healthcare professionals who have already experienced post-COVID-19 symptoms to maximize their recovery and well-being.
However, there are certain limitations in this study. First, this was a cross-sectional study; therefore, the results only represent the occurrence of post-COVID-19 symptoms during the study period. Second, the study used a retrospective design and relied on self-reported information, which may have resulted in incomplete or omitted data that can affect the assessment of post-COVID-19 symptoms. Finally, the results may have been influenced by selection bias, as individuals without post-infection symptoms may have been less interested in this study, potentially leading to less comprehensive results.
Nonetheless, this study has several strengths. First, it adopted the standard definition of post-COVID-19 symptoms as defined by WHO. Second, this was a multicenter large-scale study conducted in mainland China. Third, the use of symptom-based criteria carries an inherent risk of misclassification, as high-frequency symptoms such as fatigue are nonspecific and may overlap with other conditions, including burnout and mental health disorders.29,64,65 This could lead to an overestimation of post-COVID-19 symptom prevalence. Fourth, although we applied the WHO clinical case definition for post-COVID-19 symptoms, our study, similar to many others using this definition, did not distinguish between symptoms that persisted uninterrupted from the acute phase of the infection and those that newly developed after a negative swab test (indicating viral clearance). This distinction is methodologically challenging in large retrospective surveys; however, it is critically important as it may reflect different underlying biological mechanisms. Future prospective studies with serial testing and detailed symptom timelines are needed to elucidate the nuances of symptom onset and persistence. Fifth, as rightly noted, pre-existing anxiety and depression represent potential confounding factors, particularly for nonspecific symptoms such as fatigue, cognitive deficit, and insomnia, which are frequently reported in post-COVID-19 symptoms. Although the WHO definition used in our study requires that symptoms cannot be explained by another condition, the potential for misattribution remains a limitation of studies based on self-report, including ours. Moreover, it is also plausible that the trauma of the pandemic and the stress of COVID-19 can exacerbate or trigger new mental health symptoms, creating a complex, bidirectional relationship that is difficult to disentangle in a cross-sectional design. Future prospective studies with detailed assessments of mental health status before and after infection are needed to better delineate this relationship. Sixth, although the demographic profile of our sample was consistent with that of the broader HCW population in China and our multicenter design and high response rate strengthen generalizability, we cannot definitively confirm full representativeness for all HCWs in the participating institutions due to the lack of comprehensive institutional-level demographic data for direct comparison. This may limit the generalizability of our findings to HCWs in other regions or with different demographic structures. Finally, as this study relied on self-reporting by medical professionals who have a relatively objective assessment of post-COVID-19 symptoms, it provides a more objective evaluation of the prevalence of post-COVID-19 symptoms in HCWs.
Conclusion
In this multicenter real-world study, we investigated the prevalence of and risk factors for post-COVID-19 symptoms among HCWs with a history of SARS-CoV-2 infection. The prevalence of post-COVID-19 symptoms was 17.0%. Male sex, nursing/doctor position, chronic respiratory disease, moderate/severe COVID-19, SARS-CoV-2 reinfection, and high self-reported COVID-19 severity were significantly associated with self-reported post-COVID-19 symptoms.
For this specific group of HCWs, we recommend implementing targeted interventions, including enhancing protective measures to prevent infection, monitoring post-COVID-19 symptoms in HCWs after infection, providing psychological counseling, and offering rehabilitation therapy for health management. This study provides valuable insights into the prevention and control of post-COVID-19 symptoms, not only for HCWs but also for the general population.
Footnotes
Acknowledgments
We would like to thank the participants for their cooperation and support.
Authors’ contributions
X.D.W. and L.L. were involved in the conception and design. J.B.S., J.T.X, H.L., X.N.Y, J.H.C, and L.C.S. collected the data. X.D.W was involved in the analysis and interpretation of the data. L.L searched, sorted, and interpreted the relevant literature. J.B.S. and X.D.W. were involved in the drafting of the paper and revising it critically for intellectual content. All authors edited and approved the final manuscript and agree to be accountable for all aspects of the work.
Availability of data and materials
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare no conflict of interest. The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Ethics approval and consent to participate
This multicenter cross-sectional study was performed in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of Taizhou Hospital of Zhejiang Province, China (approval number: K20230716). Respondents’ information was kept anonymous and Ethics Committee of Taizhou Hospital, Zhejiang Province waived the need for informed consent from participants.
Funding
This study was supported in part by the Science and Technology Administration Public Interest Technology Research Project of Zhejiang Province (LGF19H030013).
Supplemental material
Supplemental material for this article is available online.
