Abstract
Objective
Although spinal anesthesia can improve the success of external cephalic version, the optimal analgesic dose of ropivacaine remains undefined, and higher doses may increase potential risks. This study aimed to determine the 90% effective dose of intrathecal ropivacaine for external cephalic version.
Methods
In this double-blind, prospective, sequential dose-finding study, 40 parturients scheduled for external cephalic version received spinal anesthesia with ropivacaine (initial dose, 2.5 mg) combined with 10 µg fentanyl. Subsequent doses were determined using a biased coin up-and-down design based on the analgesic efficacy outcome of the preceding participant. Efficacy was defined as a procedural visual analog scale pain score ≤30 mm. The 90% effective dose was estimated using isotonic regression and bootstrapping.
Results
The estimated 90% effective dose of intrathecal ropivacaine co-administered with 10 µg fentanyl was 3.22 mg (95% confidence interval: 2.80–3.36). The median (interquartile range) visual analog scale scores were 30.0 (20.0, 42.5), 30.0 (20.0, 30.0), and 10.0 (10.0, 25.0) mm in the 2.5, 3.0, and 3.5 mg dose groups, respectively (p < 0.05). Maternal and fetal hemodynamics remained stable.
Conclusions
A dose of 3.0 mg ropivacaine combined with 10 µg fentanyl provided a favorable balance between analgesic efficacy and safety for spinal anesthesia during external cephalic version and is recommended for clinical use.
Trial Registration: Chinese Clinical Trial Registration Centre (http://www.chictr.org.cn); Registration Number: ChiCTR2400084126; Registration Date: 10 May 2024
Keywords
Introduction
Breech presentation is a significant obstetric challenge, contributing substantially to the global cesarean section rate.1,2 For eligible parturients, external cephalic version (ECV) is an effective approach to achieve cephalic presentation and facilitate vaginal delivery, thereby mitigating the maternal and neonatal risks associated with surgical birth. However, the efficacy of ECV is often compromised by significant maternal pain, which reduces patient acceptance and compliance and affects procedural success rate by increasing abdominal muscle tension. This discomfort may reduce acceptance of the technique and decrease the likelihood of recommending it to other pregnant individuals, thereby limiting its widespread adoption and development.3,4 To address these limitations, the use of anesthesia has garnered increasing clinical interest as a strategy to optimize patient experience and improve the overall success of ECV.
Currently, neuraxial anesthesia is more widely used for ECV5–7 because of its significant advantages over intravenous anesthesia, which may cause maternal complications and increase the risk of fetal heart rate (FHR) decrease. Previous studies have found that although higher doses of bupivacaine provide better analgesic effects, they may also result in more severe complications, including maternal hypotension, fetal bradycardia, and emergency cesarean delivery, without improving the success rate of ECV. 8 Importantly, no consensus has been reached regarding the optimal spinal anesthetic dose for ECV. Therefore, the aim of this study was to determine the 90% effective (ED90) spinal analgesic dose of ropivacaine, co-administered with a fixed dose of fentanyl (10 µg), for ECV using a biased coin up-and-down (BCUD) sequential dose-finding method.
Methods
Ethics approval
This study was approved by the Ethics Committee of the International Peace Maternity and Child Health Hospital, Shanghai, China (Approval Number: GKLW-A-2023-038-01) on 2 November 2023. The trial was subsequently submitted for registration with the Chinese Clinical Trial Registration Centre website (http://www.chictr.org.cn; ChiCTR2400084126) in December 2023. Due to an administrative delay in the registry review process, formal trial registration was not completed until 10 May 2024. We clarify that the protocol submitted for registration in May 2024 was identical to the version approved by the ethics committee in November 2023. Patient enrollment commenced on 2 January 2024, following ethics approval but before completion of registration. The entire study protocol, including all inclusion and exclusion criteria, interventions, and outcome measures, was finalized before enrollment of the first participant and was submitted to the registry without any modifications.
This study was conducted in accordance with the principles of the Declaration of Helsinki (as revised in 2024) and was additionally designed in accordance with the Consolidated Standards of Reporting Trials (CONSORT) 2025 statement. 9 Each parturient provided written informed consent before participating in the study. No patient or members of the public were involved in the design, conduct, or reporting of this trial.
Patient recruitment
Forty-nine parturients who required ECV for breech presentation at the International Peace Maternity and Child Health Hospital between 2 January 2024 and 31 August 2024 were recruited for this study. The inclusion criteria were as follows: (a) singleton pregnancy; (b) age ≥18 years; (c) gestational age ≥37 weeks; (d) height 150–180 cm; (e) American Society of Anesthesiologists (ASA) physical status class II; and (f) body mass index ≤40 kg/m2. The exclusion criteria comprised contraindications to neuraxial anesthesia, ruptured membranes, fetal distress, allergy to local anesthetics or opioids, and parturient refusal. In addition, the following exclusion criteria were applied after recruitment: incomplete data, failed spinal puncture, and undetectable upper sensory level.
Analgesia procedure
The procedure was performed in the operating room and an 18G intravenous access was established in the left forearm of each parturient. All parturients received a β-receptor agonist (ritodrine hydrochloride) 20 min preoperatively to relax the uterus and facilitate fetal movement. Fetal presentation, placental location, spinal position, and FHR were confirmed preoperatively using ultrasound.
Parturients were placed in a supine position and connected to a monitor for continuous monitoring. Baseline vital signs, including systolic blood pressure (SBP), mean arterial pressure (MAP), heart rate (HR), and oxygen saturation, were recorded. Subsequently, the parturients were placed in the right lateral decubitus position, with the hands holding the knees and the head lowered, and an epidural puncture was performed at the L3–4 intervertebral space. A designated research assistant prepared the medication for each parturient. Specifically, 10 µg fentanyl was drawn into a 1 mL syringe, and the corresponding experimental dose of 0.75% ropivacaine (AstraZeneca AB, Södertälje, Sweden) was drawn up and diluted with normal saline to a fixed total volume of 2 mL. The prepared syringe was then provided to the anesthesiologist, who performed a spinal puncture at the L3–4 interspace and injected the medication steadily over 10 s. Following intrathecal injection, an epidural catheter was inserted 4 cm into the epidural space for supplementary analgesia if required. Cases of failed spinal puncture, defined as inability to identify the intrathecal space after three consecutive attempts by senior anesthesiologists, were excluded from the final analysis. SBP, MAP, HR, and oxygen saturation were recorded at 0, 3, 6, 9, 12, 15, and 30 min after spinal anesthesia. When the parturient’s MAP dropped >20% from baseline, 6 mg ephedrine was administered. Sensory block level was assessed via temperature discrimination using an alcohol-soaked gauze (4°C), starting 10 min after spinal anesthesia. The upper sensory level was evaluated at 5, 10, and 15 min after intrathecal injection. If the upper sensory level was undetectable, the parturient was excluded from the study. Modified Bromage scores (1: complete motor blockade; 2: almost complete motor blockade, with the patient able to move only the feet; 3: partial motor blockade, with the patient able to move the knees; 4: detectable weakness of hip flexion, with the patient able to raise a leg but unable to keep it raised; 5: no detectable weakness of hip flexion, with the patient able to raise a leg for ≥10 s; and 6: no weakness) were recorded 5 min after spinal anesthesia.
After assessment of the modified Bromage score, the obstetrician initiated the ECV procedure. The decision of whether to terminate the procedure was based on parturient intolerance, severe fetal bradycardia, or failure to achieve fetal version after multiple attempts. Ultrasound was used to confirm whether ECV was successful after the procedure. Additionally, the obstetrician recorded the visual analog scale (VAS) pain score and FHR after each attempt. VAS scores were measured using a 100-mm VAS. When the VAS score was >30 mm, 5 mL epidural lidocaine was administered. Postoperative ultrasonography was performed to confirm ECV and assess fetal well-being. ECV procedure–related indicators were recorded, including procedure duration, number of attempts, abdominal relaxation assessed by the obstetrician (0, poor relaxation; 5, optimal relaxation), parturient satisfaction (0, completely dissatisfied; 5, completely satisfied), and whether the version was successful. Adverse events related to spinal anesthesia (e.g. hypotension and nausea) and to the ECV procedure (e.g. fetal bradycardia and premature rupture of membranes) were documented and managed in real time by the attending obstetrician and anesthesiologist. Finally, delivery outcomes, including mode of delivery, neonatal birth weights, and 1-min and 5-min Apgar scores, were recorded.
BCUD design
The first parturient received 2.5 mg ropivacaine. The dose for each subsequent parturient was increased or decreased by 0.5 mg based on the response of the preceding parturient. Based on previous studies recommending a safe spinal bupivacaine dose range of 1.5–3.75 mg for labor analgesia, a ceiling dose of 5.5 mg ropivacaine (equivalent to 3.7 mg bupivacaine) was set to ensure maternal and fetal safety.10,11 Accordingly, the dose ranged from 2.5 to 5.5 mg. If the VAS score remained ≤30 mm throughout ECV, the ropivacaine dose was considered successful. According to the experimental plan, the subsequent parturient had a 1/9 probability of receiving a lower dose (decrease by 0.5 mg) and an 8/9 probability of receiving the same dose as the previous parturient. If the VAS scores exceeded 30 mm, the dose was considered a failure and the subsequent parturient received an increased dose (0.5 mg). This process continued accordingly. When the ropivacaine dose was considered a failure, 5 mL of 1% lidocaine was administered epidurally every 5 min as a rescue to ensure parturient comfort and satisfaction during ECV.
The BCUD scheme was prepared using Microsoft Excel 2016 (Microsoft Inc., Redmond, WA, USA) and implemented by a research assistant, who was the only individual with access to the software and was aware of the dose administered to each parturient.
Primary and secondary outcomes
The primary outcome was the parturient’s VAS score during ECV, which was used to determine the ED90 spinal dose of ropivacaine for ECV. Secondary outcomes included baseline characteristics of the parturients before spinal anesthesia, upper sensory level and modified Bromage scores after spinal anesthesia, procedure-related indicators, hemodynamic parameters of the parturient, adverse fetal and parturient events during ECV, and parturient delivery outcomes. It should be noted that the primary outcome was refined from an initial composite measure (sensory block ≥ T6 and VAS ≤ 30 mm) to the VAS score alone. This adjustment was made because the sensory block criterion was met in all but one participant and did not differentiate analgesic efficacy, allowing the VAS to serve as a more discriminative endpoint for dose-finding.
Randomization, allocation concealment, and blinding
The BCUD dose allocation sequence was generated in real time using a dedicated Visual Basic for Applications program (a programmed algorithm) in Microsoft Excel 2016 by a research assistant independent of all clinical assessments. Upon entering the previous participant’s dose and outcome, the program automatically generated the next assigned dose. To ensure allocation concealment, this dose was determined only after the completion of the preceding case, and the study medication was prepared in an identical nontransparent syringe by a designated research assistant. The syringe, labeled only with a study ID, was then provided to the attending anesthesiologist. The anesthesiologist who performed the spinal puncture administered the drug and subsequently recorded maternal vital signs and VAS scores, remaining blinded to the specific dose throughout the procedure. The obstetricians performing ECV and the parturients were also blinded. Thus, all individuals involved in clinical care and outcome assessment were blinded to the dose assignment.
Statistical analysis
In dose-finding studies using the BCUD design and simulation studies, enrolling 20–40 patients as a stopping rule, results in stable target dose estimates. 12 We used Wald’s sequential probability ratio to dynamically evaluate the termination criteria during participant enrollment.
where p0 = 0.5, p1 = 0.8, α = 0.05, β = 0.2, and Nmax = 40. Therefore, the sample size for this study was set at 40 parturients. 13
ED90 was defined as ropivacaine dose for spinal analgesia that provided successful analgesic effect, characterized by a VAS score ≤30 mm throughout ECV, in 90% of parturients undergoing the procedure. This dose was estimated using isotonic regression.
We analyzed the data using R software version 4.2.2. The 95% confidence interval (CI) of the estimated ED90 was obtained using a bias-corrected percentile method with 2000 bootstrap replications. Furthermore, the results were obtained using isotonic regression and bootstrapping. The Kolmogorov–Smirnov test was used to assess the normality of variables. Maternal and obstetric characteristics were presented as mean ± standard deviation (SD) for normally distributed continuous variables, median (P25, P75) for skewed continuous variables, and percentages for categorical variables. Categorical data were compared between the study groups using the χ2 test or Fisher’s exact test and were presented as percentages. Intergroup comparisons were performed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test for normally distributed data or the Kruskal–Wallis test with Dunn’s multiple comparisons correction for nonparametric data. Longitudinal changes in hemodynamic parameters within each dose group were evaluated using repeated-measures ANOVA. When the sphericity assumption was not met, the Greenhouse-Geisser correction was applied. A p-value <0.05 was considered statistically significant.
Results
Among the 49 recruited parturients, 1 was excluded because of contraindications to neuraxial anesthesia, and 4 were excluded because of membrane rupture. Ultimately, 44 parturients met the inclusion criteria and agreed to participate. Two parturients experienced difficult spinal punctures, and one was excluded because of an undetectable upper sensory level. An additional parturient was excluded owing to incomplete data. Consequently, data from remaining 40 parturients were included in the final analysis (Figure 1).

Flow chart of participant selection and progression through the sequential dose-finding study.
The sequence of effective and ineffective responses to spinal ropivacaine doses in 40 consecutive parturients is shown in Figure 2(a). The ED90 value of spinal ropivacaine was calculated to be 3.22 mg (95% CI: 2.80–3.36). These parturients ultimately formed three groups receiving 2.5, 3, or 3.5 mg of intrathecal ropivacaine. When stratified by ropivacaine dose, VAS scores exhibited a statistically significant difference across the three groups (Kruskal–Wallis test, p < 0.05). Specifically, the median (interquartile range) VAS scores were 30.0 (20.0, 42.5), 30.0 (20.0, 30.0), and 10.0 (10.0, 25.0) mm for the 2.5, 3, and 3.5 mg groups, respectively. Post hoc analysis with Dunn’s test revealed significant differences between the 2.5 mg and 3.5 mg groups (p = 0.0334) and between the 3 mg and 3.5 mg groups (p = 0.0106), but not between the 2.5 mg and 3 mg groups (p > 0.9999) (Figure 2(b)).
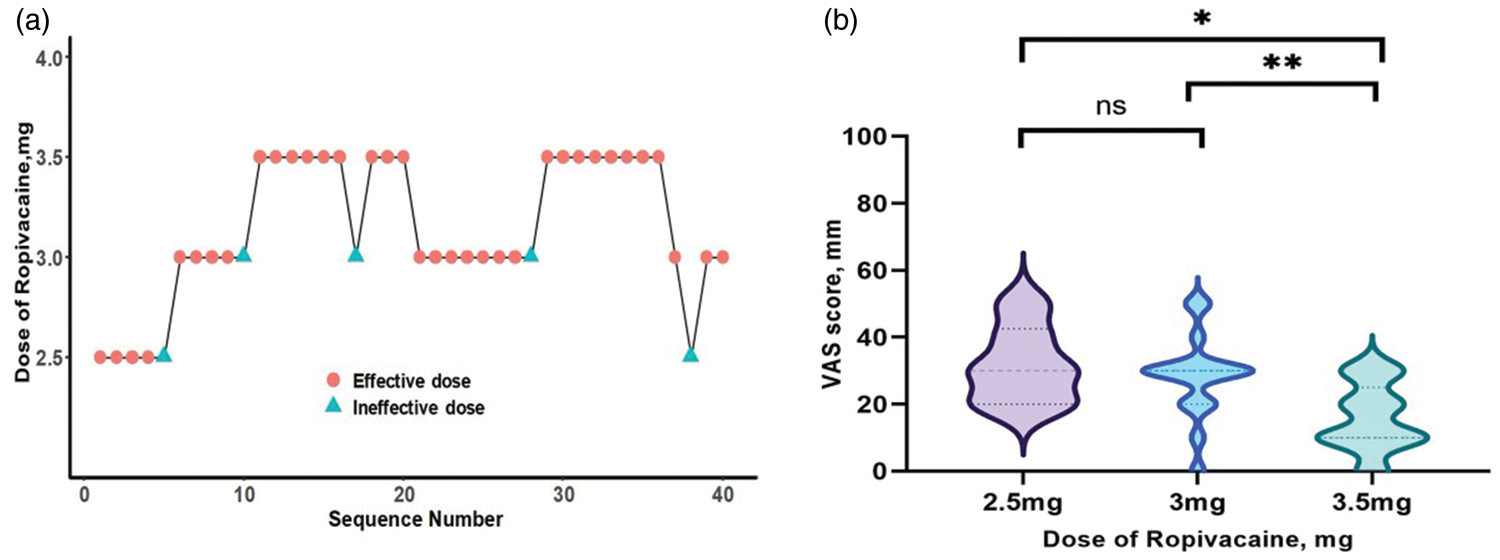
Biased coin up-and-down (BCUD) dose allocation sequence and analgesic outcomes. (a) Sequential dose allocation for each participant. Solid circles represent effective analgesia (visual analog scale (VAS) score ≤30 mm); solid triangles represent ineffective analgesia. (b) Distribution of procedural VAS pain scores across the three ropivacaine dose groups (2.5, 3.0, and 3.5 mg). Data are presented as median with interquartile range.
The demographic and obstetric general characteristics are presented by dose group in Table 1. No statistically significant differences were observed among the three groups (all p > 0.05 by one-way ANOVA or chi-square test, as appropriate).
Parturient characteristics.

Values are presented as mean (SD), number (percentage), or median (IQR).
IQR: interquartile range.
Maternal and fetal hemodynamic indicators were dynamically monitored before and up to 30 min after spinal administration. Time-course trends (mean ± SD) across four parameters were presented for the three dose groups (Figure 3). The profiles remained stable throughout the observation period. Repeated-measures ANOVA confirmed no statistically significant within-group changes over time for any parameter across all doses (all p > 0.05), demonstrating consistent hemodynamic and FHR stability during the procedure.

Hemodynamic changes during external cephalic version under spinal anesthesia. Time-course trends (mean ± SD) for the three ropivacaine dose groups (2.5, 3.0, and 3.5 mg) are shown for (a) systolic blood pressure (SBP), (b) mean arterial pressure (MAP), (c) maternal heart rate (HR), and (d) fetal heart rate (FHR).
We evaluated and recorded the outcomes of parturients and neonates after intervention. Among parturients receiving 2.5, 3, and 3.5 mg ropivacaine, the upper sensory levels were T6, T6, and T4, respectively, with a statistically significant differences (p = 0.01, p < 0.05) among the groups. The success rates were 66.7%, 64.7%, and 64.7% in the 2.5-, 3-, and 3.5-mg groups, respectively, with corresponding number of procedure attempts of 2.5, 3, and 4, and procedure durations of 8.33, 9.82, and 8.88 min. No significant differences were observed among the three groups in terms of success rates (p > 0.05), number of procedure attempts (p > 0.05), or procedure durations (p > 0.05). Abdominal relaxation scores (scale, 0–5) were 4.0, 5.0, and 5.0 in the 2.5-, 3-, and 3.5-mg groups, respectively, whereas parturient satisfaction scores were 4.5, 5.0, and 5.0, respectively. No significant intergroup differences were observed in abdominal relaxation (p > 0.05) or parturient satisfaction (p > 0.05). Hypotension occurred in three (17.6%) and four (23.5%) parturients who received 3 and 3.5 mg ropivacaine, respectively, requiring ephedrine treatment; no hypotension cases were observed in the 2.5-mg group. No statistically significant differences were observed among the three groups (p > 0.05). Fetal bradycardia was observed in four (23.5%), one (5.9%), and zero parturients who received 3, 3.5, and 2.5 mg ropivacaine, respectively, with no significant differences (p > 0.05) among the groups. No other adverse symptoms, including nausea, vomiting, or pruritus, were observed in any parturient. All parturients had a Bromage scores of 0. No emergency cesarean section, membrane rupture, or severe uterine contractions occurred during ECV (Table 2).
Parturient outcomes.

Values are presented as mean (SD), number (percentage), or median (IQR).
*p < 0.05.
NA: not available; IQR: interquartile range.
Neonatal outcomes across the three dose groups are detailed in Table 3. Birth weights were comparable among groups, with mean values of 3498 g, 3263 g, and 3418 g in the 2.5-, 3-, and 3.5-mg groups, respectively. Cephalic presentations predominated (27/40, 67.5%), with left occiput anterior being the most common orientation (17/40; distributed as 1, 8, and 8 cases across the 2.5-, 3-, and 3.5-mg groups, respectively). Breech presentations was observed for the remaining 13 cases. Neonatal Apgar scores at 1 and 5 min were 10.
Neonatal outcomes.

Values are presented as mean (SD), number (percentage), or median (IQR).
IQR: interquartile range.
Discussion
The principal finding of this prospective sequential dose-finding study is that the ED90 of spinal ropivacaine, when co-administered with 10 µg fentanyl, for providing effective analgesia during ECV is 3.22 mg, with a clinical recommendation of 3.0 mg. This dose regimen achieved effective pain control (VAS ≤ 30 mm) and maintained stable maternal and fetal hemodynamics. Our results provide a precise, evidence-based dosing reference for this specific clinical scenario.
The optimal dosage for spinal anesthesia had not been established prior to this study. Previous studies have used varying regimens, with intrathecal bupivacaine doses ranging from 2.5 to 7.5 mg, often in combination with opioids.7,14–16 Meta-analyses of randomized controlled trials on neuraxial anesthesia demonstrated that administering higher doses of local anesthetics via the subarachnoid or epidural route during breech version produces denser blocks, thereby increasing the version success rate. 14 Chalifoux et al. 2 suggested that an increased spinal bupivacaine dose does not further increase the ECV success rate; therefore, high-dose anesthesia may be unnecessary for ECV. The absence of pain signals during ECV can lead to the application of excessive force, potentially resulting in more severe complications. 17 Specifically, the possible risks associated with ECV include transient abnormal cardiotocography patterns, persistent pathological cardiotocography readings, vaginal bleeding, and placental abruption. Notably, historical data (pre-1970s) reported fewer ECV-related complications, 18 possibly attributable to the lack of neuraxial anesthesia. Therefore, minimizing the spinal anesthetic dose for ECV is clinically significant.
In the present study, we set the initial dose at 2.5 mg ropivacaine for spinal anesthesia combined with 10 µg fentanyl and used the BCUD method to identify the optimal dose for ECV. Although ropivacaine is increasingly used in spinal anesthesia, there is limited evidence supporting its application for ECV. Earlier studies recommended doses of intrathecal ropivacaine as high as 8 mg; 19 however, such doses are no longer considered appropriate in current clinical regimens. Our results showed that administering 3.22 mg ropivacaine provided satisfactory anesthesia effects, which is equivalent to 2.1 mg of bupivacaine. The ED90 identified in the present study was lower than that reported previously.14,20 Because the equivalent dose ratio of ropivacaine to bupivacaine was 3:2, our findings were consistent with those of a randomized double-blind trial conducted by Chalifoux et al. 2 The study evaluated the impact of four doses of bupivacaine (2.5, 5.0, 7.5, and 10.0 mg), combined with 15 μg fentanyl, on ECV success rate during labor and reported that bupivacaine dose >2.5 mg administered in the spinal space did not further increase the success rate or reduce the rates of cesarean section.
In the present study, the ECV success rate was approximately 65%, with a slight difference among the three dose groups. This rate was slightly higher than the previously reported average of 53%. 21 Transient FHR drops were observed in 12.5% (5/40) of parturients, all of which occurred after rather than during ECV. Unlike previous studies2,22,23 using high concentrations of local anesthetics, which reported the incidence of emergency cesarean sections of 1.1%–9.5%, no emergency cesarean sections were required in our study. Hypotension occurred in 17% of parturients, and no other adverse effects, including nausea, vomiting, or pruritus, were observed. Additionally, 62.5% of parturients had vaginal deliveries, and the Apgar scores of the neonates at 1 and 5 min were 10.
In the subgroup analysis, the VAS values and upper sensory levels showed notable differences in the 3.5 mg group (median VAS values, 10.0 mm; upper sensory level, T4). However, there was a higher hypotension rate of 23.5% and higher ephedrine usage of 23.5% in the 3.5 mg group. Therefore, we recommend 3.0 mg as an optimal spinal analgesic dose of ropivacaine combined with 10 µg fentanyl for ECV. This dose not only provided satisfactory anesthesia effects but also reduced the incidence of adverse reactions, ensuring parturient and fetal safety.
This study has some limitations. First, we did not adjust doses according to parturients heights; therefore, future studies should include a subgroup analysis based on height. Second, the intervertebral space for spinal puncture was identified by palpation. Although performed by experienced anesthesiologists, the lack of routine ultrasound confirmation may have introduced some uncontrolled variability in the exact puncture level, which is a recognized limitation of this study design. Finally, factors such as abdominal relaxation, gravidity, amniotic fluid volume, and neonatal body weight may have affected the experience and success rate of ECV. Future studies should subdivide parturients’ baseline conditions to facilitate a more accurate comparison.
Conclusions
Using the BCUD sequential method, this study determined the ED90 of intrathecal ropivacaine (combined with 10 µg fentanyl) for ECV to be 3.22 mg (95% CI: 2.80–3.36). A dose of 3.0 mg ropivacaine provided a favorable balance between analgesic efficacy and safety. These findings offer valuable guidance for clinical practice, supporting our recommendation of using 3.0 mg intrathecal ropivacaine with fentanyl for spinal anesthesia during ECV.
Footnotes
Acknowledgments
We thank all participating patients for their valuable contribution to this study.
Author contributions
Rui Ma and Jing Chai: Conceptualization. Qiao Shi, Jing Zheng, Tao Xu, and Rui Ma: Data curation. Qiao Shi, Jing Zheng, Tao Xu, and Rui Ma: Analysis and interpretation. Qiao Shi and Jing Zheng: Roles/Writing–original draft. Jing Zheng, Rui Ma, and Jing Chai: Writing–review and editing. All authors approved the final version of the manuscript.
Data availability statement
Deidentified participant data are available from the corresponding author upon reasonable request, subject to a data use agreement to protect participant privacy.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
Funding was received for this study from the International Peace Maternity and Child Health Hospital (A Three-year Action Plan for Clinical Research Program, IPMCH2024CR07).
