Abstract
Objective
Adequate anesthesia is necessary during cesarean section. We aimed to study the efficacy and safety of different doses of fentanyl combined with 10 mg bupivacaine and 0.1 mg morphine for spinal anesthesia during elective cesarean section.
Method
This double-blind randomized controlled trial enrolled pregnant women undergoing elective cesarean section between April 2019 and June 2021. Eligible women (n = 180) were randomized into six groups (C (control), F1, F2, F3, F4, or F5; n = 30 each) based on different doses of fentanyl (0, 5, 10, 15, 20, and 25 µg, respectively) for spinal anesthesia. Each patient also received 0.1 mg morphine and 10 mg bupivacaine.
Result
Sensory and motor blocks, muscle relaxation, pain intensity, intraoperative hemodynamics (every 5 min starting from the initial intrathecal injection), and adverse events were recorded. Compared with group C, groups F2–F5 had a faster onset of sensory block (p < 0.001), and groups F1–F5 had a faster onset of motor block (p = 0.015). During surgery, diastolic blood pressure levels in groups F4 and F5 were lower than those in group C between 5 and 20 min after anesthesia initiation (p < 0.05). Heart rates in group F5 were significantly higher than those in group C at 5 and 15 min of anesthesia initiation (p = 0.016 and 0.008, respectively). Groups F4 and F5 required a higher dose of phenylephrine to prevent hypotension than group C (p = 0.027). There were no significant differences in muscle relaxation, postoperative analgesia, and adverse events among the groups.
Conclusion
Intrathecal fentanyl administration at doses of 10–15 μg combined with 10 mg bupivacaine and 0.1 mg morphine provides rapid and satisfactory analgesia without additional adverse events during elective cesarean section.
This clinical trial was registered with the Chinese Clinical Registry (ChiCTR1900021721, https://www.chictr.org.cn/showproj.html?proj=35939).
Introduction
In recent decades, spinal anesthesia (SA) has become the standard procedure for elective cesarean section (CS). 1 Over the past few years, SA has become an alternative to general anesthesia during laparoscopic treatment of gynecological diseases. SA is effective in relieving postoperative pain and reducing the impact of surgical stress.2–4 Bupivacaine, with its strong sensory- and motor-blocking effects as well as good safety profile, is commonly used for SA during CS. High doses of bupivacaine were previously used to ensure adequate anesthesia;5,6 however, they carry the risk of ventricular arrhythmia, ventricular fibrillation, and even cardiac arrest. 7 Therefore, a lower dose of bupivacaine is recommended during SA. To ensure an adequate anesthetic effect, a lower dose of bupivacaine is often combined with appropriate amounts of opioids.
According to their lipid solubility, opioids can be classified as lipophilic and hydrophilic opioids. The former has a rapid onset of action; however, its duration of anesthetic effect is relatively short. The latter has a slower onset of action but can provide a prolonged anesthetic effect.8–10 Fentanyl is a lipophilic opioid, while morphine is a hydrophilic opioid. Intrathecal morphine has been shown to provide analgesia for up to 24 h postoperatively, although it has a slower onset of action. 11 Therefore, intrathecal fentanyl is recommended to improve the quality of analgesia during SA for CS.12–14 However, the optimal dose of fentanyl has not yet been determined.
In the present randomized controlled trial, we investigated the analgesic efficacy and safety of varying doses of intrathecal fentanyl when added as an adjunct to intrathecal morphine and bupivacaine in participants undergoing elective CS. Unlike previous studies,5,15,16 the target population of this study specifically consisted of pregnant Chinese women.
Methods
Study design and participants
We performed a randomized controlled trial at the Shenzhen Maternal and Child Health Care Hospital in Guangdong, China, between April 2019 and June 2021. The reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT) 2025 statement. 17 The study was conducted in compliance with the Declaration of Helsinki and relevant Chinese regulations governing clinical research. The study protocol was approved by the Ethics Committee of Shenzhen Maternity and Child Healthcare Hospital (Shenzhen, China) (Approval No. SFYLS2019082) and registered at the Chinese Clinical Trial Registry (Registration no. ChiCTR1900021721). All study participants provided written informed consent for the study. There was no patient or public involvement in the design, conduct, or reporting of this trial.
As per the inclusion criteria, pregnant women aged 18–45 years with American Society of Anesthesiology classification II, a height of 155–170 cm, weight of 55–80 kg, and full-term singleton gestation who were scheduled for elective CS were enrolled.11,18 Patients who refused SA, had contraindications to regional anesthesia or allergy to local anesthetics, or had any major comorbidity (e.g. morbid obesity, pre-eclampsia, eclampsia, coagulopathy, or serious cardiovascular diseases) were excluded. 19
Prior to the study, randomization and allocation were conducted using computer-generated random numbers, which were sealed in envelopes. Patients were assigned into different groups according to the number in the envelope. Before anesthesia induction, a researcher prepared the medication based on the random number in the envelope and stored the medication in a sterile syringe without labels. The researcher did not participate in data collection or analyses. Both patients and operators (anesthetists, intraoperative and postoperative evaluators, and recorders) were blinded to group allocation. The concentrations of bupivacaine, morphine, and fentanyl stock solutions were 0.5% isobaric, 10 mg/mL, and 0.05 mg/mL, respectively. The solutions of morphine were diluted to 1 mg/mL using saline. We selected 10 mg bupivacaine (2 mL of 0.5% solution) because intrathecal doses of bupivacaine <10 mg combined with opioids would provide less satisfactory analgesia/anesthesia. 19 The volume of all the solutions was 2.6 mL (diluted with 0.9% saline when necessary).
The following six study groups were established: control group (group C) and groups F1, F2, F3, F4, and F5. Group C received 10 mg bupivacaine and 100 µg morphine. In addition to 10 mg bupivacaine and 100 µg morphine, groups F1, F2, F3, F4, and F5 received intrathecal fentanyl at different doses (5, 10, 15, 20, or 25 µg, respectively).
Anesthesia procedure
Patients were instructed to fast overnight, and no preoperative medications were administered. Upon arrival in the operating room, oxygen saturation, electrocardiograph, and noninvasive blood pressure monitors were placed. Oxygen was delivered at a rate of 6–8 L/min through a face mask to improve the oxygen reserves in case of potential emergencies. An intravenous catheter was placed, and the patients were preloaded with lactated Ringer’s solution at 12–15 mL/kg prior to SA induction. SA was administered at the L3–L4 intervertebral space, with the patient in the lateral decubitus position. A 25-G spinal needle was inserted, and the correct position of the needle was confirmed based on the free flow of the cerebrospinal fluid after stylet withdrawal. The syringe containing the precise dose of medication was attached, and the solution was injected at 10–12 s. The needle was subsequently withdrawn, and the patient was immediately placed in the left uterine displacement position.
Data collection
The age, height, weight, and gestational week of each patient were recorded. Vital signs were monitored every 5 min throughout the surgery. The heart rate and blood pressure were recorded before anesthesia (T1) and at 5, 10, 15, and 20 min after anesthesia initiation (T2, T3, T4, and T5, respectively).
Sensory blockade was determined using the pinprick method (loss of pinprick sensation to a 23-G needle) every 2 min for the first 12 min; then at 15, 30, 45, and 60 min; and subsequently at 30-min intervals until the patients complained of pain. 20 The onset of sensory block was calculated from the time point of intrathecal injection to the time point when the sensory blockade was achieved at a T6 level (dermatomal level corresponding to the sixth thoracic spinal nerve) or higher. 21 The sensory block duration was also calculated from the time point of the maximum sensory block level to the time point of sensory block regression to the S1 level (dermatomal level corresponding to the first sacral spinal nerve). The motor block was assessed using the modified Bromage scale (Bromage score: 0, able to move the hip, knee, and ankle; 1, able to move the knee and ankle but not the hip; 2, able to move the ankle but not the hip or knee; and 3, unable to move the hip, knee, or ankle). The onset of motor block was calculated from the time point of intrathecal injection to achievement of a Bromage score of 1. 22 The motor block duration was calculated from the time point of regression to achievement of a Bromage score of 0. The number of patients with an anesthetic plane reaching the T4 level was recorded. A T4 level has been recommended by previous studies. 23 The Numeric Rating Scale (NRS; score: 0, no pain and 10, most severe pain) was employed at the time point when the fetus was removed from the uterus. 23 In case of inadequate analgesia, an additional intravenous fentanyl (0.05–0.1 mg) bolus was administered at the patient’s request.
All patients received 10 mg metoclopramide and 10 mg dexamethasone preoperatively to prevent nausea and vomiting. Hypotension (systolic blood pressure <90 mmHg or >30% decrease from the baseline value) was prevented via continuous phenylephrine infusion. The initial infusion rate was 0.5 μg/kg/min, which was adjusted to maintain normal blood pressure levels. Bradycardia (heart rate <50 beats/min) was treated with 0.3–0.5 mg atropine. The numbers of patients with intraoperative visceral traction pain and those with respiratory distress (respiratory rate <10 breaths/min) were recorded. The extent of muscle relaxation (0, perfect; 1, good; 2, fair; and 3, poor) was assessed. 24
Adverse events, including hypotension, bradycardia, nausea, vomiting, pruritus, and shivering, were recorded. The duration of surgery was documented. Postoperatively, all patients received patient-controlled intravenous analgesia (PCIA) through a pump that contained 100 mL of 10 µg/mL fentanyl, with a background flow rate of 0.5 mL/h, injection of 3 mL of bolus, and lock-out time of 20 min. During the postoperative period, sensory and motor regressions were recorded every 15 min until they regressed to the S1 level with a Bromage score of 0. The time to first PCIA demand, bolus frequency, and total volume of anesthetic solution were recorded.
Statistical analyses
The primary outcomes were the onset times of sensory and motor block as well as the incidence of intraoperative pain. The sample size calculation was based on the primary outcome of intraoperative pain incidence. According to previous studies,18,25 the incidence of intraoperative pain was 25% in the group receiving bupivacaine and morphine alone, compared with 0% in groups receiving additional fentanyl. Using PASS 2021 software (NCSS, LLC, USA), with a power (1-β) of 80% and a two-sided alpha (α) value of 0.05, a minimum sample size of 25 per group was estimated. To account for a potential 20% dropout rate during the follow-up period, we decided to enroll 30 patients in each group. As this study involved a comparison of multiple groups who were administered different doses of the same drug and a single control group, we recognized the potential for inflation of type I error. However, a gatekeeping testing strategy was applied, wherein the overall efficacy of fentanyl was first established before exploring dose-dependent effects. Bonferroni correction was considered overly conservative for this exploratory dose-finding design. Only one patient was excluded after randomization owing to a failed anesthesia procedure, and the analysis was performed on a complete-case basis. 26 We made randomized comparisons between all possible sets in the randomized controlled trials to balance known and unknown confounders, ensuring comparable groups.
SPSS, version 21.0 (SPSS, IBM, USA), was used for statistical analysis. Categorical data were described as numbers with percentages and compared using the chi-square test or Fisher’s exact probability method. Continuous data were presented as mean ± SD values and compared using analysis of variance. Post-hoc comparisons between groups were performed using unpaired Student’s t-test for continuous variables and chi-square tests for categorical variables. A p value <0.05 was considered statistically significant.
Results
Patient enrollment and characteristics
In total, 180 pregnant women were included in the study. The anesthesia procedure failed in one patient; therefore, 179 patients were analyzed (Figure 1). There were no significant differences in age, height, weight, body mass index, gestational age, and duration of surgery among the study groups (Table 1).

CONSORT flow diagram. CONSORT: Consolidated Standards of Reporting Trials.
Baseline characteristics of pregnant women receiving different doses of fentanyl.
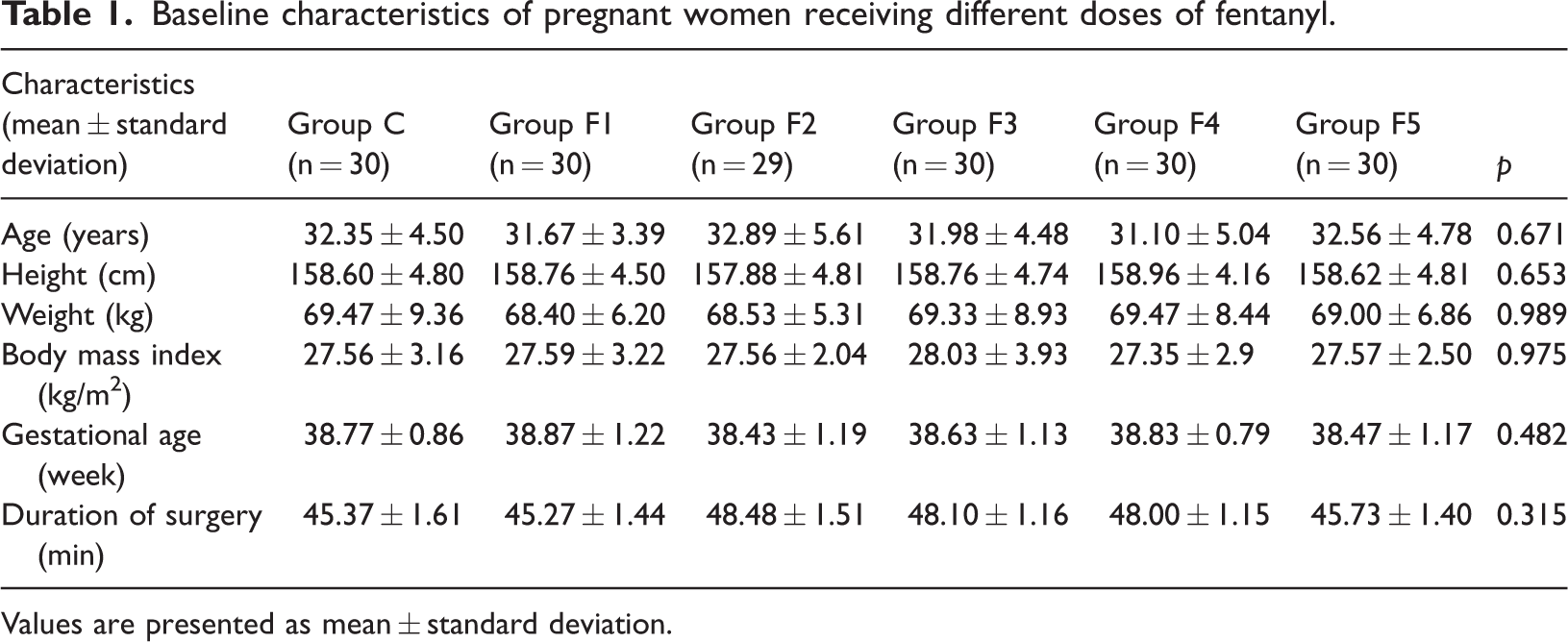
Values are presented as mean ± standard deviation.
Intraoperative characteristic comparisons
As shown in Table 2, the onset of sensory block was significantly shorter in groups F2–F5 than in group C (p < 0.001). The onset of motor block was significantly shorter in groups F1–F5 than in group C (p = 0.015). The visceral traction pain in groups F4 and F5 was lower than that in group C (p = 0.015). The doses of phenylephrine used in groups F4 and F5 were higher than those used in group C (p = 0.033 and 0.004, respectively). These results have clinical significance; however, there were no significant differences in the sensory and motor block durations, NRS score, incidence of insufficient analgesia, intraoperative use of rescue drugs, number of patients with the anesthetic plane reaching the fourth thoracic dermatome level, visceral traction pain, and respiratory distress among these groups.
Comparison of intraoperative characteristics among the study groups.
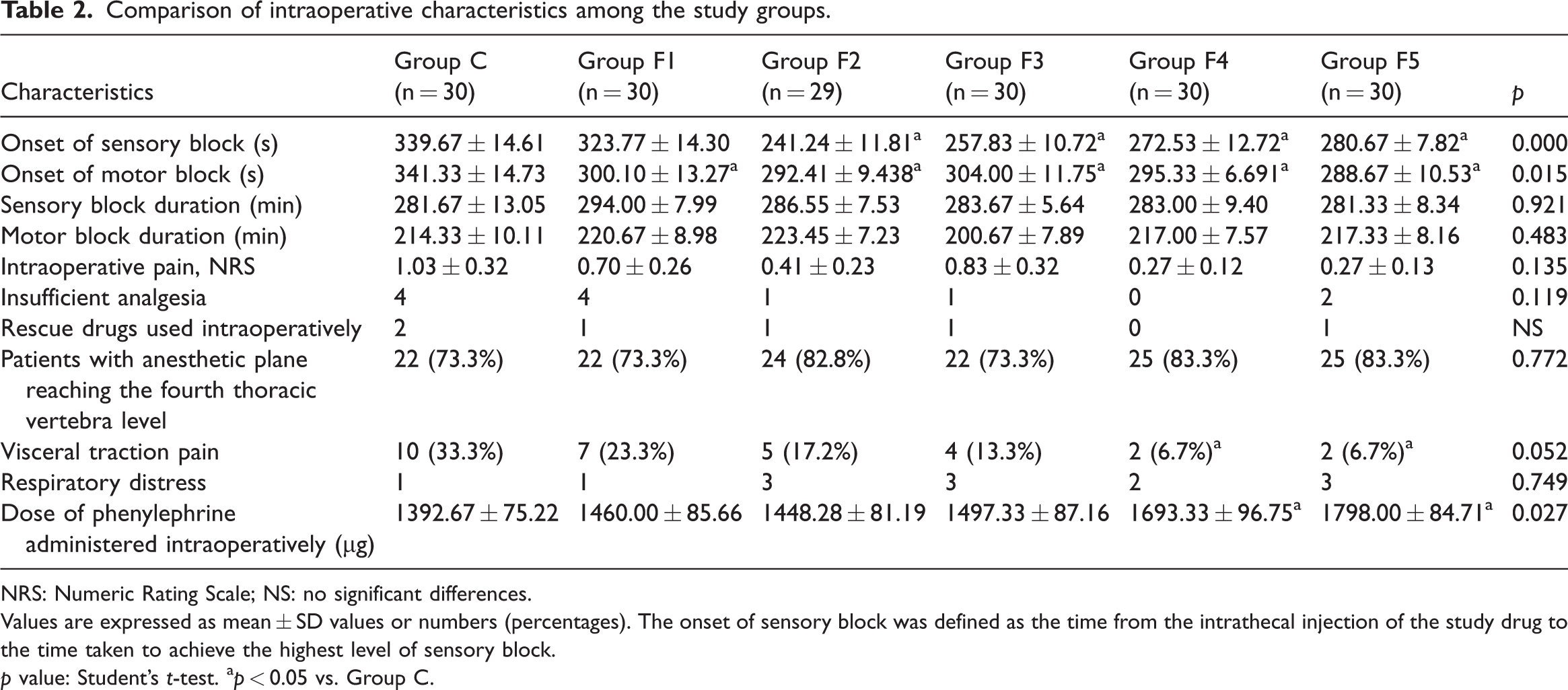
NRS: Numeric Rating Scale; NS: no significant differences.
Values are expressed as mean ± SD values or numbers (percentages). The onset of sensory block was defined as the time from the intrathecal injection of the study drug to the time taken to achieve the highest level of sensory block.
p value: Student’s t-test. ap < 0.05 vs. Group C.
During the operation, the average heart rate of patients in group F5 was significantly higher than that of those in group C at T2 (p = 0.016) and T4 (p = 0.008) time points (Figure 2(a)). The diastolic blood pressure measurements in groups F4 and F5 were lower than those in group C at T2, T3, T4, and T5 (p < 0.05) time points (Figure 2(b)). The above results have clinical significance. There was no significant difference in systolic blood pressure measurements (Figure 2(c)) or muscle relaxation among these groups (Table 3).
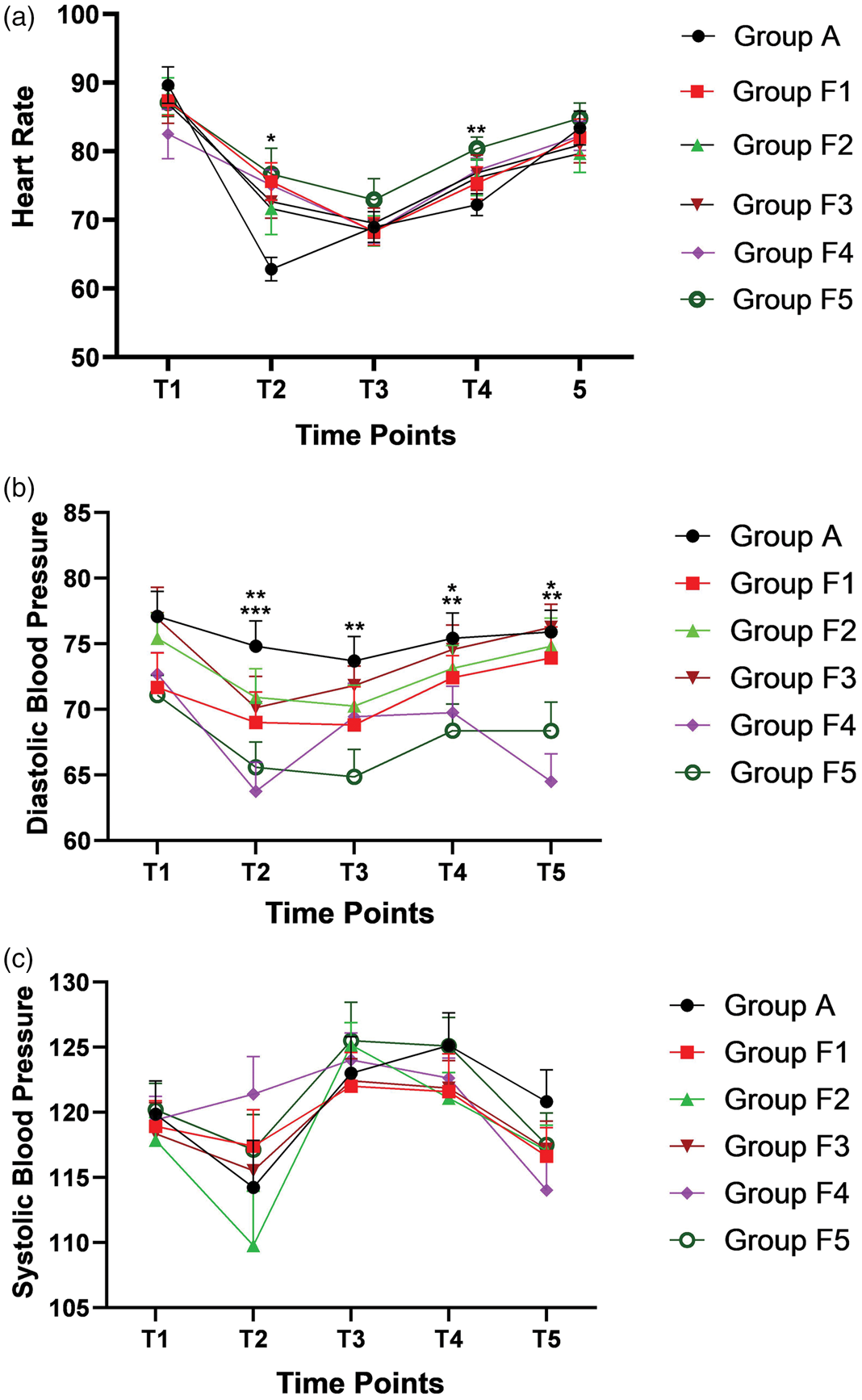
Dynamic changes in vital signs, including (a) heart rate, (b) diastolic blood pressure, and (c) systolic blood pressure. T1, before anesthesia; T2, 5 min after anesthesia; T3, 10 min after anesthesia; T4, 15 min after anesthesia; and T5, 20 min after anesthesia. *p < 0.05. **p < 0.01. ***p < 0.001.
Comparisons of muscle relaxation among the study groups.

Values are expressed as numbers (percentages). p-values were calculated using Student’s t-test.
Postoperative analgesia and adverse event comparisons
After the operation, there were no significant differences in the time to first PCIA demand, bolus frequency, or total volume of anesthetic solution among the groups (Table 4), indicating that the addition of fentanyl did not prolong the duration or improve the quality of postoperative analgesia, which was primarily sustained by intrathecal morphine administration. In addition, there were no significant differences in adverse events among the groups (Table 5).
Comparison of postoperative PCIA usage among the study groups.

PCIA: patient-controlled intravenous analgesia.
Values are expressed as numbers (mean ± SD). p-values were calculated using Student’s t-test.
Comparison of adverse events among the study groups.

Values are expressed as numbers (percentages). The p value was calculated using Student’s t-test.
Neonatal outcomes
Neonatal outcomes were evaluated using Apgar scores. The Apgar scores at 1 and 5 min were comparable across all groups, with all neonates achieving scores of ≥ 8 at 5 min. No neonates required advanced resuscitation or admission to the neonatal intensive care unit due to respiratory depression or other complications related to maternal anesthesia. The data are summarized in Table 6.
Neonatal outcomes in the study groups.

Values are expressed as mean ± SD. p-values were calculated using Student’s t-test.
Discussion
In the present study, intrathecal fentanyl (10–15 µg) combined with morphine (0.1 mg) and bupivacaine (10 mg) shortened the onset of SA without causing additional adverse events in pregnant women undergoing CS.
Our study showed that the onsets of sensory and motor blocks were faster in the fentanyl groups than in group C; however, the durations of sensory and motor blocks were similar for all study groups. Intrathecal morphine and fentanyl injections could have exerted synergistic effects, with morphine acting directly on the opioid receptors in the spinal cord and fentanyl increasing the afferent sensory block by stimulating μ1 and μ2 receptors, likely owing to its lipophilicity. 27 More recent studies have also suggested that the mechanism of fentanyl involves opening of potassium channels and inhibition of potassium release via reduced calcium channel flow. 28 Intrathecal fentanyl was recommended as a lipophilic opioid for SA to improve the quality of anesthesia. In a study of 160 patients who were administered SA for lower abdominal surgery, 20 μg fentanyl with 0.75% ropivacaine prolonged the duration of the sensation and motor blocks but did not delay postoperative recovery. 28 Cherng et al. found that epidural administration of the combination of 100 μg fentanyl and 1% ropivacaine solution accelerated the onsets of sensory and motor blocks during epidural ropivacaine anesthesia, 29 consistent with the present findings. In a prospective double-blind study, bupivacaine supplemented with 25 μg fentanyl accelerated the onsets of sensory and motor blocks in SA. However, the durations of sensory and motor blocks were longer than those in group C, 30 which is contradictory to our results. This difference might be attributed to the different combinations of drugs studied; the previous study only examined the efficacy of fentanyl used in combination with local anesthetics, while we examined the efficacy of fentanyl in combination with morphine and local anesthetics. In the present study, additional fentanyl did not delay postoperative recovery, potentially owing to the prolonged anesthesia block from morphine that had masked the effect of fentanyl. 31
By binding to the opioid receptors of the dorsal horn, fentanyl can directly inhibit the transmission of painful stimuli from the dorsal horn in the spinal cord, significantly reducing peritoneal stimulation and ameliorating the pulling response. 32 In our study, >70% of the patients had reached an anesthesia level of T4. Some patients developed visceral traction pain, although there was no statistical difference among the groups. In addition, there was no statistically significant difference in the degree of muscle relaxation among the groups. Consistent with previous studies, only seven patients in our study were dissatisfied with the degree of muscle relaxation, which was fully correlated with the degree of anesthesia block. 25 In previous studies, no serious intraoperative respiratory distress was observed with an intrathecal injection of 0.1–0.2 mg morphine alone or in combination with 25 μg fentanyl, consistent with our results. 25 Onodera et al. found that under analgesia with 0.1 mg intrathecal morphine, respiratory depression was frequently observed after CS. 25 In this study, one to three cases of respiratory depression occurred in each group, potentially associated with the administration of intrathecal morphine or a higher level of anesthesia block. To prevent traction reaction and pain, a higher anesthesia block level of T4 is necessary.33,34 When the block reaches the level of T4, thoracic spinal nerve block can occur, resulting in paralysis of the intercostal muscles and respiratory depression. 35
The potential for fetal exposure to opioids administered via SA is a valid concern. Lipophilic opioids, including fentanyl, have a rapid onset of action and are quickly absorbed by spinal tissues, resulting in low systemic plasma concentrations.36,37 In contrast, morphine is hydrophilic, has a slower onset of action, and may diffuse into the systemic circulation over time. However, the doses used in this study (fentanyl, 5–25 µg and morphine, 0.1 mg) were very low. The absence of neonatal respiratory depression or low Apgar scores in our study, consistent with extensive clinical experience using these doses, suggests minimal risk. Nonetheless, close monitoring of the neonate remains the standard of care.
In the present study, the average diastolic blood pressure levels in groups F4 and F5 were lower than that in group C, despite no significant differences in systolic blood pressure. Cohen et al. found that intrathecal sufentanil could lower blood pressure during labor analgesia due to a decrease in catecholamine levels. 38 Other studies also found that intrathecal fentanyl injection could decrease blood pressure, potentially owing to its effects on vascular tone.39–41 Spinal block–induced sympatholysis can lead to vasodilatation with subsequent hypotension in mothers. The low diastolic blood pressure in groups F4 and F5 in our study might be attributable to the peripheral vasodilatation caused by regional block after SA, decreased vascular resistance, or reduced blood return to the heart. 42 Consistent with the decreased blood pressure, the amounts of phenylephrine used in groups F4 and F5 were higher than those in group C. Phenylephrine acts on α1 receptors and has potent vasoconstrictor effects. 43 At T2 and T4 time points, the heart rates in groups F4 and F5 were higher than that in group C, which might have been caused by hyperreflexia due to low blood pressure.
We found no significant differences in the requirement for postoperative analgesia between the fentanyl groups and group C. Sibilla et al. compared the use of lipophilic and hydrophobic opioids in CS and confirmed that the quality of postoperative analgesia after the administration of 12–14 mg bupivacaine with 25 μg fentanyl was inferior to that achieved with 0.1 mg morphine. The effect of fentanyl was reported to be similar to that of placebo. 18 In addition, studies have shown no statistically significant difference in the postoperative analgesia duration between patients with and without fentanyl administration,44,45 similar to our results. This might have been related to the rapid onset of action and short duration of analgesia induced by fentanyl. The optimal approach to postoperative analgesia likely depends on the dose of long-acting morphine. 12
There were no statistically significant differences in the adverse events observed across the different groups. Nausea and vomiting associated with the intrathecal administration of opioids are believed to result from the binding of drugs to receptors in the posterior region of the brain.12,18 Several studies have shown that the combination of two opioids increases the incidence of nausea and vomiting by approximately 20%–35%.11,46–48 In previous studies, the incidences of postoperative nausea and vomiting after the administration of spinal fentanyl or morphine alone during CS were relatively low. 49 To compare the analgesic effect of spinal morphine and fentanyl in women undergoing CS, El Aish et al. performed an unmasked, parallel-group randomized controlled trial. They found that patients treated with morphine were more likely to have mild nausea and drowsiness at 6 and 12 h postoperatively, respectively. 50 A randomized controlled trial comparing the efficacy and adverse effects of bupivacaine with fentanyl or morphine for elective CS found that nausea occurred in 20.2% of the patients in the morphine group (consistent with our study) and 7.6% of those in the fentanyl group. 51 Seki et al. revealed that lipotropic opioids might reduce the occurrence of intraoperative nausea or vomiting associated with uterine surgery, while hydrophilic opioids could exacerbate these adverse effects. 52 In our study, there was no difference in morphine doses among the groups. Although we could not prove that fentanyl reduced the incidence of nausea, our findings showed that the addition of fentanyl did not increase the incidence of nausea. Moreover, we used metoclopramide intraoperatively to reduce the incidences of nausea and vomiting.
Pruritus is a common and troublesome adverse event of intrathecal opioids. 53 The addition of morphine to local anesthetics could increase the incidence of pruritus from 40% to 63%.18,54,55 Considering the differences reported in previous studies, a more recent study reported an 8.1% incidence rate for pruritus and found that most patients had mild symptoms, which required no special treatment. 51 The addition of fentanyl did not increase the incidence of pruritus. However, the overall incidence of pruritus was high, which might have been related to the use of morphine. Morphine usually causes more severe and prolonged pruritus, although most cases of pruritus were mild in this study, requiring no special treatment. 56 In this study, fentanyl did not increase the incidence of adverse events.
According to data on deliveries in the European Union in 2017, the CS rate was the highest in Cyprus, accounting for 54.8% of live births, followed by Rome (44.1%) and Bulgaria (43.1%). 51 In a recent study, Peter et al. reported a higher rate of CS in Romania, which ranged from 53.6% to 60.7%. 57 Collectively, these studies indicate a substantial requirement for adequate analgesia during CS. Better pain management can also reduce the occurrence of postpartum depression, 58 lower the hospitalization time, and reduce the resulting economic burden. Our current research findings provide an option for adequate analgesia during CS.
Limitations
This study has certain limitations. We used the same dose of postoperative analgesics. The postoperative analgesia pump was not configured as per the weight of each patient; therefore, the difference in maternal weights might have affected the analgesic effects. We performed multiple group comparisons and studied dose-dependent responses, which allowed us to identify a suitable dose range. However, this approach might have prevented the detection of subtle differences between the groups.
Conclusion
The onset of sensory and motor blocks was significantly shorter in groups F2 and F3 than in group C (p < 0.05). However, there were no significant differences in the doses of phenylephrine used or the adverse events occurring in these groups. Intrathecal administration of 10–15 μg fentanyl combined with 10 mg bupivacaine and 0.1 mg morphine could provide rapid and adequate analgesia without additional adverse events during elective CS. Future multicenter studies with a large sample size should be performed to confirm our findings. In addition, the optimal ratio for these medications based on maternal weight should be explored.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251389749 - Supplemental material for Efficacy of fentanyl combined with bupivacaine and morphine for spinal anesthesia during cesarean section: A double-blind randomized controlled trial
Supplemental material, sj-pdf-1-imr-10.1177_03000605251389749 for Efficacy of fentanyl combined with bupivacaine and morphine for spinal anesthesia during cesarean section: A double-blind randomized controlled trial by Gehui Li, Fanfang Zeng, Xiaofei Qi, Xuhong Tan, Hao Wang, Xiaolei Huang, Yuantao Li and Yun Xia in Journal of International Medical Research
Footnotes
Acknowledgements
Not applicable.
Authors’ contributions
GL, FZ, XQ, and XT collected the clinical data. HW took responsibility for the accuracy of the data analysis. GL and FZ contributed to manuscript writing and review. XH, YL, and YX contributed to the conception and design of the study. All authors contributed to data acquisition and analysis as well as reviewed and approved the final version of the manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Consent for publication
Not applicable.
Declaration of conflicting interests
We declare that we have no conflicts of interest.
Ethics approval and consent to participate
Written informed consent was obtained from all enrolled participants. The study was conducted in accordance with the Declaration of Helsinki and Chinese clinical trial research regulations. The study protocol was approved by the Shenzhen Maternity and Child Healthcare Hospital Ethics Committee (Approval No. SFYLS2019082), and the study was registered at the China Clinical Registration Center (Registration No. ChiCTR1900021721, 06/03/2019, ![]() ).
).
Funding
This work was supported by the Medical Science and Technology Research Foundation of Guangdong Province (No. A2022152) and the Shenzhen Science and Technology Program (No. JCYJ20220530155403008; No. JCYJ20240813115121029).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
