Abstract
Objective
Gastric cancer is associated with an increased risk of pulmonary thromboembolism. Although the ABO blood group system is a known thrombotic risk factor, its role in gastric cancer–associated pulmonary thromboembolism remains unclear.
Methods
This retrospective study analyzed 1488 gastric cancer patients to assess the association between ABO blood type and pulmonary thromboembolism. Cox proportional hazards models were used to identify independent predictors, and Kaplan–Meier analysis was used to compare cumulative incidence.
Results
Pulmonary thromboembolism incidence was highest in the AB group (5.49%), while that in non-AB groups was 1.81% (p < 0.01). Multivariate analysis confirmed AB blood type (hazard ratio: 2.42, 95% confidence interval: 1.09–5.39, p = 0.03), age >65 years (hazard ratio: 2.75, 95% confidence interval: 1.30–5.82, p = 0.01), and metastasis (hazard ratio: 4.03, 95% confidence interval: 1.69–9.62, p < 0.01) as significant independent predictors. Kaplan–Meier analysis confirmed a higher cumulative pulmonary thromboembolism incidence in the AB group (log-rank test, p < 0.01). However, the O versus non-O group showed no significant differences in Kaplan–Meier analysis (log-rank test, p = 0.35).
Conclusions
AB blood type is an independent risk factor for pulmonary thromboembolism in gastric cancer patients. This finding, distinct from the traditional non-O group risk model, suggests that identifying AB blood type can improve risk stratification.
Introduction
Gastric cancer is a major public health issue in East Asia, where the incidence rate is among the highest worldwide. According to recent global cancer statistics, East Asian countries such as Korea, Japan, and China account for more than 60% of the global gastric cancer burden.1–3 In South Korea, gastric cancer is the most commonly diagnosed malignancy in men and the third most common in women, with more than 27,000 new cases reported annually.4,5
Patients with gastric cancer are at increased risk of venous thromboembolism (VTE), including pulmonary thromboembolism (PTE), due to cancer-associated hypercoagulability, immobility, surgery, and chemotherapy.6–8 Identifying individual risk factors for PTE in this high-risk population is crucial for improving patient outcomes and tailoring thromboprophylaxis strategies. 9
The ABO blood group system has been increasingly recognized as a nonmodifiable risk factor for thrombotic events. Multiple studies have shown that individuals with non-O blood types (A, B, and AB) have a higher risk of VTE than those with blood type O.10–12 Factor VIII (FVIII) and von Willebrand factor (vWF) levels are higher in non-O blood types, suggesting that changes in coagulation factor levels may play a role in VTE development. 13
Although the association between ABO blood types and thrombosis has been explored in the general population and in some cancer types, limited data exist specifically for patients with gastric cancer. Given the high incidence of gastric cancer in Korea and the clinical relevance of PTE, this study aimed to investigate the relationship between ABO blood groups and the incidence of PTE in patients with gastric cancer.
Methods
Study population
This retrospective study analyzed 1488 patients aged 18 years or older diagnosed with gastric cancer, defined by International Classification of Diseases, 10th Revision code C16.x, between January 2018 and December 2022. Patients were identified from the electronic medical records (EMR) of the Gachon University Gil Medical Center in Incheon, South Korea. This study was approved by the Institutional Review Board of Gil Medical Center, affiliated with Gachon University Gil Medical Center, under approval number GDIRB2025-236. The requirement for informed consent was waived by the Institutional Review Board due to the retrospective nature of the study. This study was conducted in accordance with the Declaration of Helsinki (as revised in 2024). All patient information was anonymized and de-identified prior to analysis. The inclusion criteria were as follows: (a) a confirmed diagnosis of gastric cancer within the study period; (2) having ABO blood type recorded in the EMR; and (3) availability of at least one follow-up record after diagnosis. Patients with missing ABO blood type information or those lost to follow-up immediately after diagnosis were excluded. A total of 1488 patients met these criteria and were included in the final analysis (Figure 1). The collected data included baseline clinical characteristics and follow-up information until December 2022. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. The completed STROBE checklist is provided in Supplementary Table S1. 14

Flowchart detailing the patient selection process for the study cohort.
Outcome definition
The primary endpoint of this study was the development of PTE during the follow-up period after the diagnosis of gastric cancer, with a specific focus on differences according to ABO blood type. PTE was defined as cases confirmed via chest computed tomography (CT). All chest CT scans were interpreted by radiologists at our institution. This study aimed to identify the association between blood type and PTE incidence in patients with gastric cancer. Secondary endpoints included an exploratory analysis of clinical factors potentially associated with the occurrence of PTE.
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences software version 26 (IBM Corp., Armonk, NY, USA) and R software version 4.3.3 (R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were expressed as means ± SD for normally distributed data or as medians with interquartile ranges for skewed data. Categorical variables were presented as counts and percentages. Group comparisons were conducted using the independent samples t-test for normally distributed continuous variables and the Mann–Whitney U test for non-normally distributed variables. The chi-square test was used for categorical variables.
Time-to-event outcomes, including the primary endpoint, were analyzed using the Kaplan–Meier method and compared using the log-rank test. Follow-up time was calculated from the date of gastric cancer diagnosis to the date of PTE diagnosis, death, or last hospital visit or the study end date (31 December 2022), whichever occurred first. Patients who were lost to follow-up or were alive without PTE on the study end date were censored at the time of their last known contact or 31 December 2022, respectively. Cox proportional hazards regression analysis was performed to identify independent predictors of PTE. Variables with a p-value <0.05 in univariate analysis were included in the multivariate model. Statistical significance was set at a two-sided p-value <0.05.
Regarding missing data for baseline characteristics, we found that missing values were minimal for the variables included in the final multivariate Cox model. Therefore, analyses were conducted using available cases, excluding patients with missing data for the specific variables involved in each analysis. Given the low proportion of missing data, imputation methods were not employed.
Results
Among the 1488 patients diagnosed with gastric cancer, the distribution of ABO blood types was as follows: 588 patients (39.5%) had blood type A, 363 (24.4%) had type B, 373 (25.1%) had type O, and 164 (11.0%) had type AB (Figure 2(a)). The baseline characteristics of the patients according to blood type are summarized in Table 1. The median follow-up duration for the entire cohort was 401.5 days (interquartile range (IQR): 109.8–968.5 days). Among the four groups, patients with blood type B had the highest mean age (66.60 ± 12.45 years), followed by type O (65.83 ± 11.71 years), AB (65.84 ± 12.24 years), and A (64.51 ± 12.30 years). The proportion of male patients ranged from 61.96% for blood type AB to 72.45% for blood type B. The prevalence of comorbidities, including hypertension, diabetes mellitus, and chronic kidney disease, did not differ significantly across blood types.
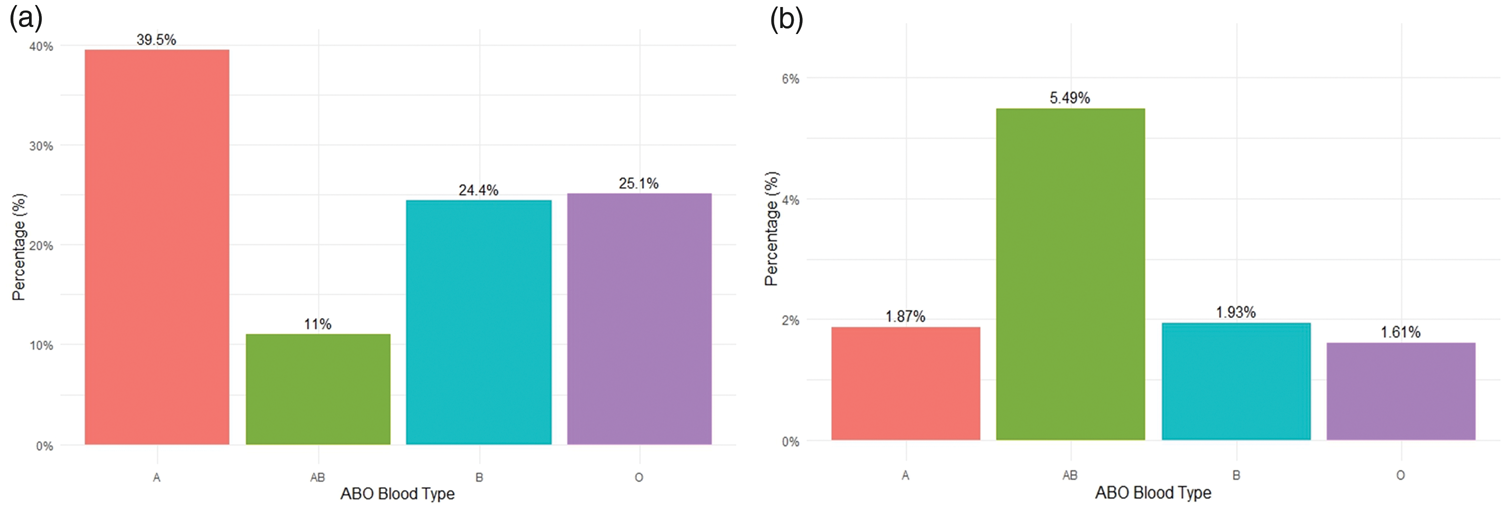
Distribution of ABO blood types and the corresponding incidence of PTE in patients with gastric cancer. (a) Distribution of ABO blood types. (b) Incidence of PTE according to ABO blood type. PTE: pulmonary thromboembolism.
Baseline characteristics of patients for each blood type.

HTN: hypertension; DM: diabetes mellitus; CKD: chronic kidney disease; AF: atrial fibrillation; PTE: pulmonary thromboembolism; BMI: body mass index.
The incidence of PTE showed notable variations among the blood groups: 5.49% for type AB, 1.93% for type B, 1.87% for type A, and 1.61% for type O (Figure 1(b)). The highest incidence rate was observed in the AB group. To explore this association in more detail, the cohort was stratified into two groups: AB (n = 163) and non-AB (n = 1325). The baseline characteristics of the two groups are shown in Table 2. No significant differences were found in age, BMI, smoking status, alcohol use, or most comorbidities. However, the prevalence of metastasis was significantly higher in the AB group (30.67% vs. 22.11%, p = 0.02). The incidence of PTE was also significantly higher in the AB group (5.49% vs. 1.81%, p < 0.01).
Baseline and clinical characteristics of patients for two groups (AB and non-AB groups).

HTN: hypertension; DM: diabetes mellitus; CKD: chronic kidney disease; AF: atrial fibrillation; PTE: pulmonary thromboembolism; CTx: chemotherapy; BMI: body mass index.
In the univariate Cox proportional hazards regression analysis (Table 3), age >65 years (hazard ratio (HR): 2.52; 95% confidence interval (CI): 1.20–5.30; p = 0.01), AB blood type (HR: 2.71; 95% CI: 1.22–6.01; p = 0.01), chemotherapy (HR: 4.16; 95% CI: 1.87–9.24; p < 0.01), and presence of metastasis (HR: 7.61; 95% CI: 3.72–15.52; p < 0.01) were significantly associated with an increased risk of PTE. In contrast, history of surgery was significantly associated with a reduced risk of PTE (HR: 0.29; 95% CI: 0.15–0.55; p < 0.01). Other variables including sex, smoking, alcohol consumption, hypertension, diabetes mellitus, and atrial fibrillation were not significantly associated with PTE development.
Univariate and multivariate analyses of variables associated with development of PTE.

PTE: pulmonary thromboembolism; HTN: hypertension; DM: diabetes mellitus; AF: atrial fibrillation; CTx: chemotherapy; HR: hazard ratio; CI: confidence interval.
In the multivariate Cox regression analysis, age >65 years (HR: 2.75; 95% CI: 1.30–5.82; p = 0.01), AB blood type (HR: 2.42; 95% CI: 1.09–5.39; p = 0.03), and metastasis (HR: 4.03; 95% CI: 1.69–9.62; p < 0.01) remained independent predictors of PTE. Although surgery (HR: 0.51; 95% CI: 0.25–1.03; p = 0.07) and chemotherapy (HR: 2.05; 95% CI: 0.83–5.11; p = 0.12) showed trends toward association, they did not reach statistical significance in the multivariate model.
Kaplan–Meier survival analysis was conducted to compare the cumulative incidence of PTE among patients with gastric cancer stratified according to ABO blood type (Figure 3). In the analysis of all four blood groups (Figure 3(a)), patients with blood type AB exhibited the highest cumulative incidence of PTE during the follow-up period. The difference in cumulative incidence among blood types was statistically significant (p = 0.02, log-rank test). The Kaplan–Meier curve (Figure 3(b)) demonstrated a significantly higher cumulative incidence of PTE in the AB group than in the non-AB group (p < 0.01, log-rank test). This result supports the findings of the multivariate analysis and highlights AB blood type as an independent risk factor for PTE development in patients with gastric cancer. Kaplan–Meier analysis comparing O and non-O blood types showed no significant difference in cumulative incidence of PTE between the two groups (Figure 4, p = 0.35, log-rank test). Similarly, comparisons of A versus non-A groups and B versus non-B groups revealed no statistically significant differences (Supplementary Figure S1, p = 0.44 and p = 0.68, respectively, log-rank test).

Kaplan–Meier analysis of the incidence of PTE in patients with gastric cancer. (a) PTE incidence according to each of the four blood types. (b) PTE incidence in AB blood type versus non-AB blood types. PTE: pulmonary thromboembolism.

Kaplan–Meier analysis of the incidence of PTE in patients with gastric cancer, comparing O and non-O blood types. PTE: pulmonary thromboembolism.
Discussion
The present study demonstrates that ABO blood type, particularly the AB group, is significantly associated with PTE risk in patients with gastric cancer. We found that individuals with blood type AB had a more than two-fold higher incidence of PTE than those with other blood groups, even after adjusting for confounders. In multivariate analysis, AB blood type emerged as an independent predictor of PTE (HR: 2.42; 95% CI: 1.09–5.39; p = 0.03), along with older age (HR: 2.75; 95% CI: 1.30–5.82; p = 0.01) and the presence of metastasis (HR: 4.03; 95% CI: 1.69–9.62; p < 0.01). Notably, no significant difference in PTE occurrence was observed between O and non-O blood types in this cohort. These findings suggest that although the overall non-O versus O distinction may not be critical in gastric cancer, the AB phenotype confers a unique thrombotic risk. Given the paucity of prior data on ABO grouping in patients with gastric cancer, our study provides novel evidence that can help contextualize PTE risk in this high-incidence cancer population.
In the general population, ABO blood type is a well-established risk factor for PTE, with individuals with non-O blood types being at higher risk than those with type O.15–17 The principal biological mechanism linking ABO blood type to VTE risk is the regulation of plasma concentrations of vWF and FVIII.18,19 Individuals with non-O blood groups have approximately 25% higher plasma vWF concentrations than those with blood group O, and approximately 30% of the variability in plasma vWF levels is determined by the ABO locus. This difference can be explained at the molecular level. After synthesis in vascular endothelial cells, vWF undergoes glycosylation, during which the ABO blood group antigens (A, B, or H) attach in the form of N-linked oligosaccharides. Blood group O expresses only the H antigen, whereas non-O groups contain glycosyltransferases that add either the A or B sugar. These additional glycan structures protect vWF from proteolytic cleavage by ADAMTS13 (A disintegrin and metalloproteinase with thrombospondin type 1 motif, member 13), thereby prolonging its plasma half-life and maintaining high circulating levels. vWF also serves as a stabilizing carrier for FVIII. When bound to vWF, FVIII is protected from proteolytic degradation, resulting in a markedly extended half-life. Consequently, individuals with non-O blood groups who have higher vWF levels exhibit significantly elevated plasma FVIII concentrations compared with those with blood group O. Elevated FVIII levels are a well-established strong and independent risk factor for VTE, making this pathway a key mechanistic link between ABO blood group and thrombotic risk. 20
In our study, no significant difference in PTE incidence was observed between individuals with blood type O and those with non-O blood types (or in the comparisons of A vs. non-A or B vs. non-B). One possible explanation is a ceiling effect attributable to underlying gastric cancer, which is itself a strong risk factor for thromboembolic events. 21 In such a high-risk population, the additional prothrombotic influence of ABO blood group–related differences in vWF and FVIII levels may be attenuated to the point of being clinically negligible. Another potential factor influencing our results is the difference in gastric cancer incidence according to ABO blood group. A previous study reported that individuals with blood group A have a higher risk of developing gastric cancer than those with other blood groups. 22
The most novel finding of our study is that despite the potential ceiling effect, blood group AB remained an independent and statistically significant risk factor for PTE. This result is further underscored by the observation that neither the A versus non-A nor the B versus non-B group comparisons yielded a significant difference in PTE risk. This suggests that the influence of ABO blood group on thrombotic risk may not follow a simple dichotomous pattern but rather exhibit a dose-dependent or synergistic effect on thrombus formation. The biological basis for this hypothesis lies in plasma concentrations of vWF and FVIII, which increase in the order O < A < B < AB, with the highest levels observed in individuals possessing both A and B antigens. 19 Although the thrombotic risk associated with blood group A or B alone might be masked by the strong prothrombotic effect of gastric cancer, the maximal prothrombotic state conferred by elevated vWF and FVIII levels in blood group AB may surpass this ceiling and emerge as a statistically significant risk factor. Our findings are also consistent with observations in other malignancies. For instance, studies in pancreatic cancer have reported an increased risk of VTE associated with non-O blood types, with some studies specifically identifying AB blood group as conferring higher risk compared with O blood group. 23 Notably, the fact that our study was conducted in a Korean cohort, a subset of the Asian population, carries important implications for the interpretation of the results. Asians have a lower baseline incidence of VTE than Western populations, suggesting that in Asian patients with gastric cancer, the thrombotic effect imposed by the disease itself may not be as high as that observed in Western cohorts. Consequently, in the context of a relatively lower baseline risk, the strong genetic predisposition conferred by AB blood group may emerge more prominently as a statistically detectable risk factor.
These findings have significant clinical implications. When assessing thrombotic risk in patients with gastric cancer, simple dichotomization into O and non-O blood groups may be insufficient. Instead, our results suggest that blood group AB should be considered an independent high-risk category. In particular, patients with AB blood group and additional risk factors, such as advanced age or metastatic disease, may warrant closer monitoring for potential development of PTE.
This study has several limitations. First, its retrospective, single-center design inherently limits the generalizability of our findings to other patient populations, geographic regions, or healthcare systems. Validation in large, multicenter cohorts is necessary before these results can be broadly applied. Second, the number of PTE events was modest (n = 33), constraining the precision of effect estimates and limiting the ability to conduct meaningful stratified analyses to explore potential effect modification or residual confounding within subgroups. Such analyses were precluded by the very small number of events within specific strata, which would have resulted in unstable models and insufficient statistical power. Therefore, although our main analysis suggests that AB blood type is an independent risk factor overall, its effect modification by factors such as disease severity requires investigation in larger datasets. Third, this study focused solely on PTE as the primary outcome. Data on deep vein thrombosis (DVT) events or overall VTE incidence were not systematically collected, precluding assessment of whether ABO blood type is also associated with DVT risk or composite VTE outcomes in gastric cancer patients. Fourth, surgery and chemotherapy were treated as baseline exposures; as these therapies occur over time, time-dependent modeling would better account for immortal-time bias and time-varying confounding factors. Fifth, residual and unmeasured confounding factors—including thromboprophylaxis strategies, performance status, central venous catheter use, and detailed anticancer regimens—cannot be excluded. Sixth, ABO phenotyping was available; however, genotyping and measurement of key intermediates (plasma vWF/FVIII, Rhesus factor) were not performed, precluding direct mechanistic mediation analysis. Finally, outcome ascertainment relied on EMR and imaging reports; therefore, misclassification of incidental, subsegmental, or asymptomatic PTE events remains possible.
Conclusion
In a large single-center cohort of patients with gastric cancer, blood group AB was independently associated with a higher risk of PTE, while no significant difference was observed between O and non-O blood groups after adjustment. Age and metastasis were also independent predictors of PTE. These findings suggest that ABO phenotyping, particularly identification of the AB group, may improve thrombotic risk stratification in patients with gastric cancer. Prospective, multicenter studies using competing-risk and time-dependent models and incorporating biomarkers such as vWF and FVIII are warranted to validate these results and elucidate the underlying mechanisms.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261417838 - Supplemental material for Association between blood type and pulmonary thromboembolism in patients with gastric cancer: A retrospective cohort study
Supplemental material, sj-pdf-1-imr-10.1177_03000605261417838 for Association between blood type and pulmonary thromboembolism in patients with gastric cancer: A retrospective cohort study by Joonpyo Lee, Youngwoo Jang, Kilyoon Pack, Taeil Yang, Yoonsun Won, Kyung Eun Ha, Young Saing Kim and Jeonggeun Moon in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605261417838 - Supplemental material for Association between blood type and pulmonary thromboembolism in patients with gastric cancer: A retrospective cohort study
Supplemental material, sj-pdf-2-imr-10.1177_03000605261417838 for Association between blood type and pulmonary thromboembolism in patients with gastric cancer: A retrospective cohort study by Joonpyo Lee, Youngwoo Jang, Kilyoon Pack, Taeil Yang, Yoonsun Won, Kyung Eun Ha, Young Saing Kim and Jeonggeun Moon in Journal of International Medical Research
Footnotes
Acknowledgments
Author contributions statement
Joonpyo Lee: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Visualization, Writing—original draft; Youngwoo Jang: Conceptualization, Data curation, Funding acquisition, Formal analysis, Investigation, Validation, Methodology, Visualization, Writing—review and editing; Kilyoon Pack: Investigation, Data curation, Writing—review and editing; Taeil Yang: Investigation, Data curation, Writing—review and editing; Yoonsun Won: Investigation, Writing—review and editing; Kyung Eun Ha: Investigation, Writing—review and editing; Young Saing Kim: Resources, Writing—review and editing; Jeonggeun Moon: Conceptualization, Funding acquisition, Supervision, Project administration, Validation, Writing—review and editing.
Data availability statement
The data supporting the findings of this study are not publicly available due to patient confidentiality and institutional policy. However, the data can be obtained from the corresponding author upon reasonable request.
Disclosures
The authors have no conflicts of interest.
Funding
This work was supported by the Gachon University Research Fund of 2024 (grant number: GCU-202409940001) to Jeonggeun Moon and Gachon University Gil Medical Center (grant number: FRD2022-05-02 and FRD2023-09-02) to Youngwoo Jang.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
