Abstract
Background
Spinal anesthesia is a commonly used anesthetic technique for a variety of surgical procedures. Despite its popularity and safety, failed spinal anesthesia remains an important clinical challenge, resulting in patient discomfort, need for repeated attempts, and/or conversion to general anesthesia. This systematic review compiles contemporary evidence from 2015 to 2025 on the predictors of failed spinal anesthesia.
Methods
A systematic search was performed using PubMed, Embase, Cochrane Library, Web of Science, and Google Scholar. Studies published between January 2015 and May 2025 reporting contributors of failed spinal anesthesia were included. Methodological quality was evaluated using the Joanna Briggs Institute tool.
Results
Twenty-one studies comprising obstetric, orthopedic, urologic, and general surgical populations were included. Failure rates ranged from 0.9% to 25.3%. Contributors of failed spinal included low local anesthetic dose, use of isobaric solutions, provider inexperience, presence of bloody cerebrospinal fluid, absence of free cerebrospinal fluid flow, lumbar puncture at the L4–L5 interspace, high body mass index, prior spinal anesthesia exposure, emergency surgery, and multiple puncture attempts.
Conclusion
Failed spinal anesthesia is multifactorial and is influenced by technical, patient-related, and contextual factors. Optimization of technique, adequate dosing, enhanced provider training, and improved patient assessment may reduce failure rates.
Introduction
Since its introduction by August Bier in 1899, spinal anesthesia has become a cornerstone of modern regional anesthesia practice due to its rapid onset of action, dense sensory–motor block, and favorable safety profile. 1 It is frequently used in obstetric, orthopedic, urologic, and general surgical procedures.2–4
Spinal anesthesia has been increasingly utilized in lumbar surgery due to its various advantages over general anesthesia; however, failure of the first dose requiring intraoperative conversion to general anesthesia occurs in as many as 3.6% of spinal anesthesia patients. 5 Advantages of spinal anesthesia include reduced postoperative nausea and vomiting, improved pain control, minimal airway manipulation, and reduced need for opioids.6–9 Additionally, spinal anesthesia has been associated with a lower 30-day mortality rate than general anesthesia. 10
Despite its safety and efficacy, spinal anesthesia can fail. Block failure may result in incomplete sensory block, inadequate motor block, inability to achieve sufficient dermatomal spread, or the need for conversion to general anesthesia.11–13 Factors such as provider inexperience, dosing issues, anatomic variability, and technical challenges may contribute to spinal anesthesia failure.14–16 Furthermore, patient position, dose of local anesthetics, and number of attempts of lumbar puncture affect the success rate of spinal anesthesia.17,18
Given the importance of successful spinal anesthesia and the variability of reported failure rates worldwide, this systematic review summarizes contemporary evidence on the predictors of failed spinal anesthesia from 2015 to 2025.
Methods
Study design
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) 2020 guidelines (Figure 1). 19 The protocol was registered in International Prospective Register of Systematic Reviews (PROSPERO) (CRD420251101582).

Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020.
Search strategy
A comprehensive search of PubMed, Embase, Cochrane Library, Web of Science, and Google Scholar was performed. Search terms included the following: “spinal anesthesia,” “failed spinal anesthesia,” “block failure,” “predictors,” “associated factors,” and “neuraxial anesthesia.”
Eligibility criteria
Observational studies (prospective or retrospective) that were published in English (2015–2025), had full-text available, and reported the incidence or predictors of failed spinal anesthesia were included.
Case reports, case series, conference abstracts, reviews, and preprints were excluded.
Study selection and data extraction
Three reviewers independently screened titles, abstracts, and full texts. Extracted data included study characteristics, definitions of failure, predictors, and anesthesia techniques.
Quality assessment
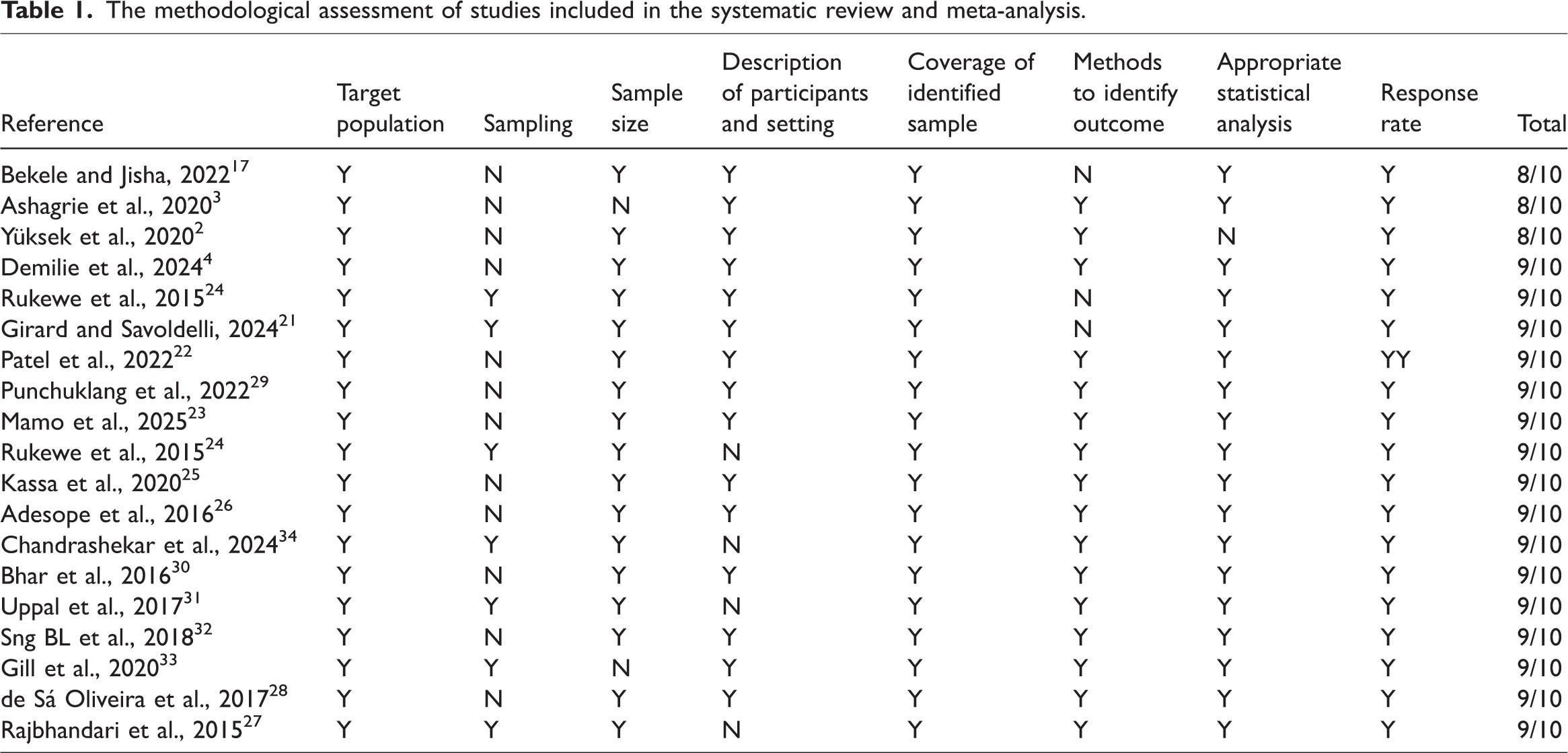
The Joanna Briggs Institute (JBI) appraisal tool was used. 20 Only studies that achieved ≥50% quality score were included (Table 1).
The methodological assessment of studies included in the systematic review and meta-analysis.
Results
Characteristics of included studies
In total, 21 studies met the inclusion criteria (Table 2); these studies included obstetric,3,9,17,24,26,27 orthopedic, 2 general surgical,4,23,35 and mixed populations.28,29 Eleven studies were prospective, and ten were retrospective. Spinal anesthesia failure rates ranged from 0.9% to 25.3% in these studies, similar to those reported previously.2,14,36 The global pooled incidence of failed spinal anesthesia is 8.36% (95% confidence interval (CI): 6.94–9.78) (Figure 2). Owing to the inclusion of a diverse array of patient populations, including those in obstetrics, orthopedics, and general surgery departments, the incidence of failed spinal anesthesia in this systematic review and meta-analysis was higher than that in a previous study. 37 A leave-one-out sensitivity analysis was conducted to evaluate the influence of individual studies on the pooled spinal anesthesia failure rate. The analysis revealed that excluding any single study did not substantially alter the overall pooled rate of 8.36% (95% CI: 6.94–9.78) (Figure 3). Furthermore, subgroup analyses by type of surgery, income of countries (Figures 4 and 5), and publication bias (Figure 6) were performed.
Characteristics of studies included in the systematic review and meta-analysis.


Pooled incidence of failed spinal anesthesia.

Sensitivity analysis.
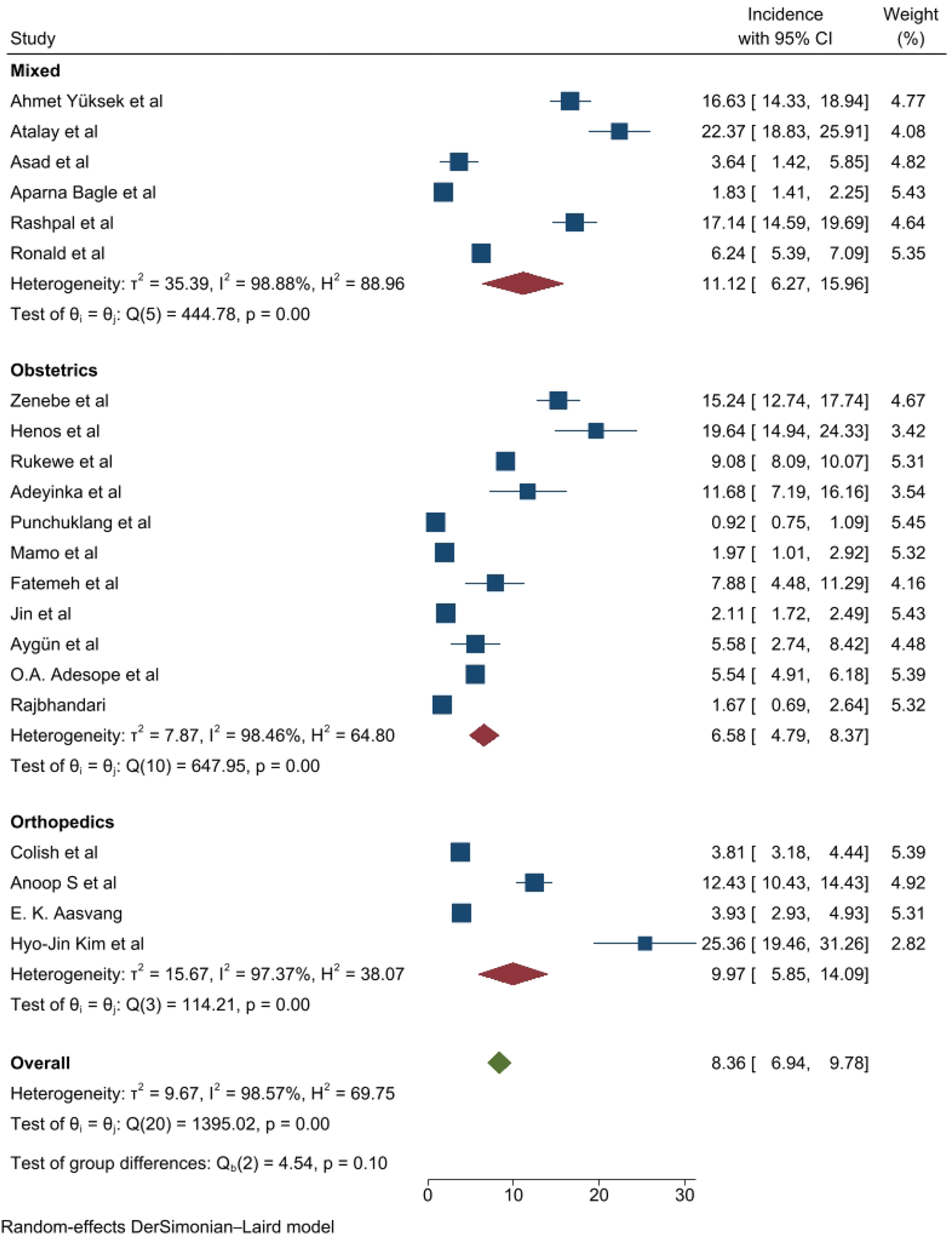
Subgroup analysis by type of surgery.
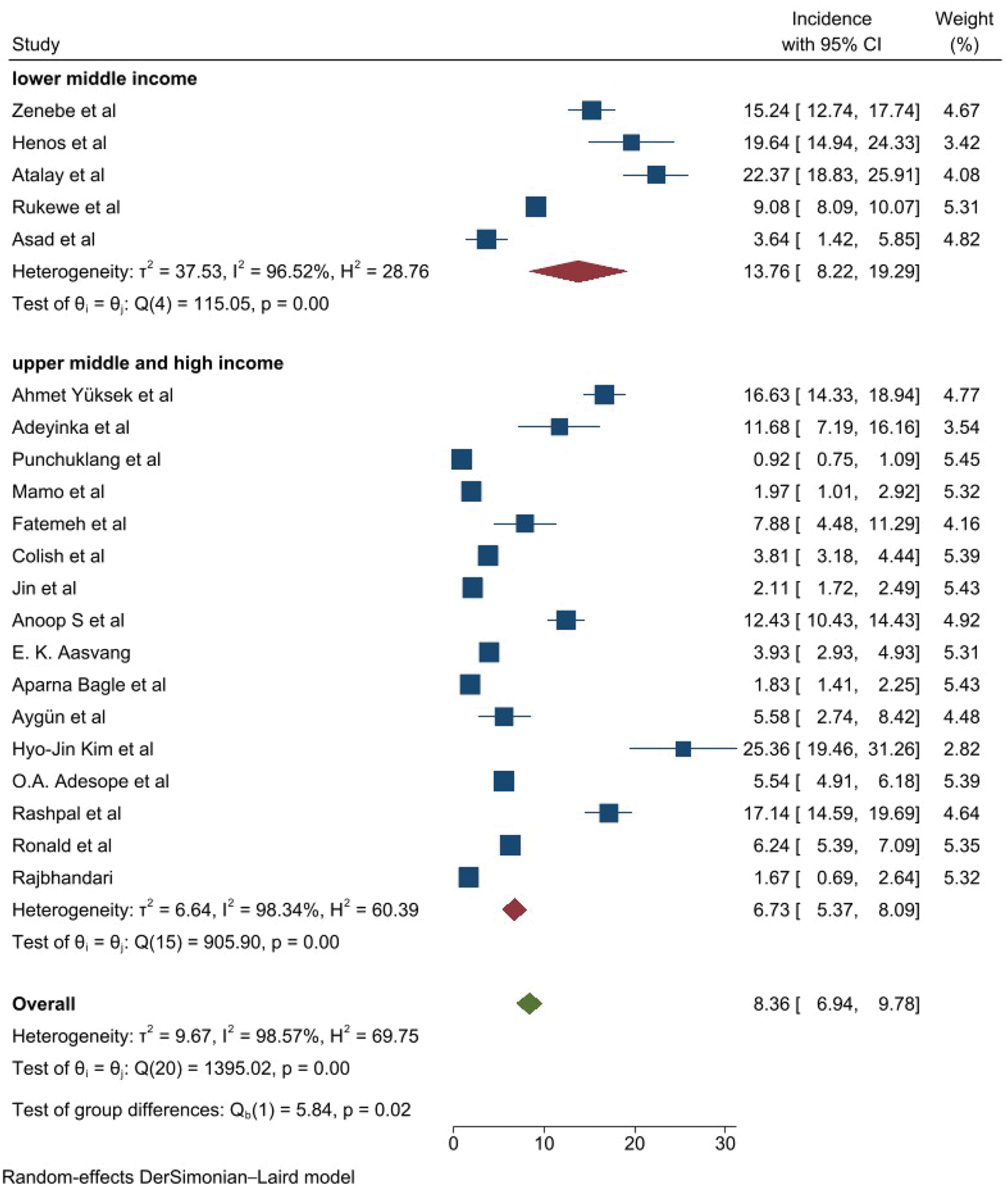
Subgroup analysis by income level of countries.

Assessment of publication bias.
Definitions of block failure included inadequate sensory level, incomplete motor block, inability to reach target dermatomal level, need for supplemental analgesia, or conversion to general anesthesia.9,14,17,24
Predictors
Key predictors of spinal anesthesia failure were the baricity of local anesthetics, provider inexperience, bloody cerebrospinal fluid (CSF), emergency surgery, patient history of anesthesia, dose of local anesthetics, body mass index (BMI), lumbar puncture performed at the L4–L5 interspace, and absence of free CSF flow (Table 3).
Predictors of spinal anesthesia failure.

BMI: body mass index; CSF: cerebrospinal fluid; LA: local anesthetics.
Local anesthetic factors
Low dose of local anesthetic
Multiple studies found that doses <10 mg were associated with increased failure, likely due to inadequate cephalad spread and shorter block duration.3,4,30 This is consistent with mechanistic descriptions showing that insufficient dosing leads to poor block quality (Figure 7).14,16
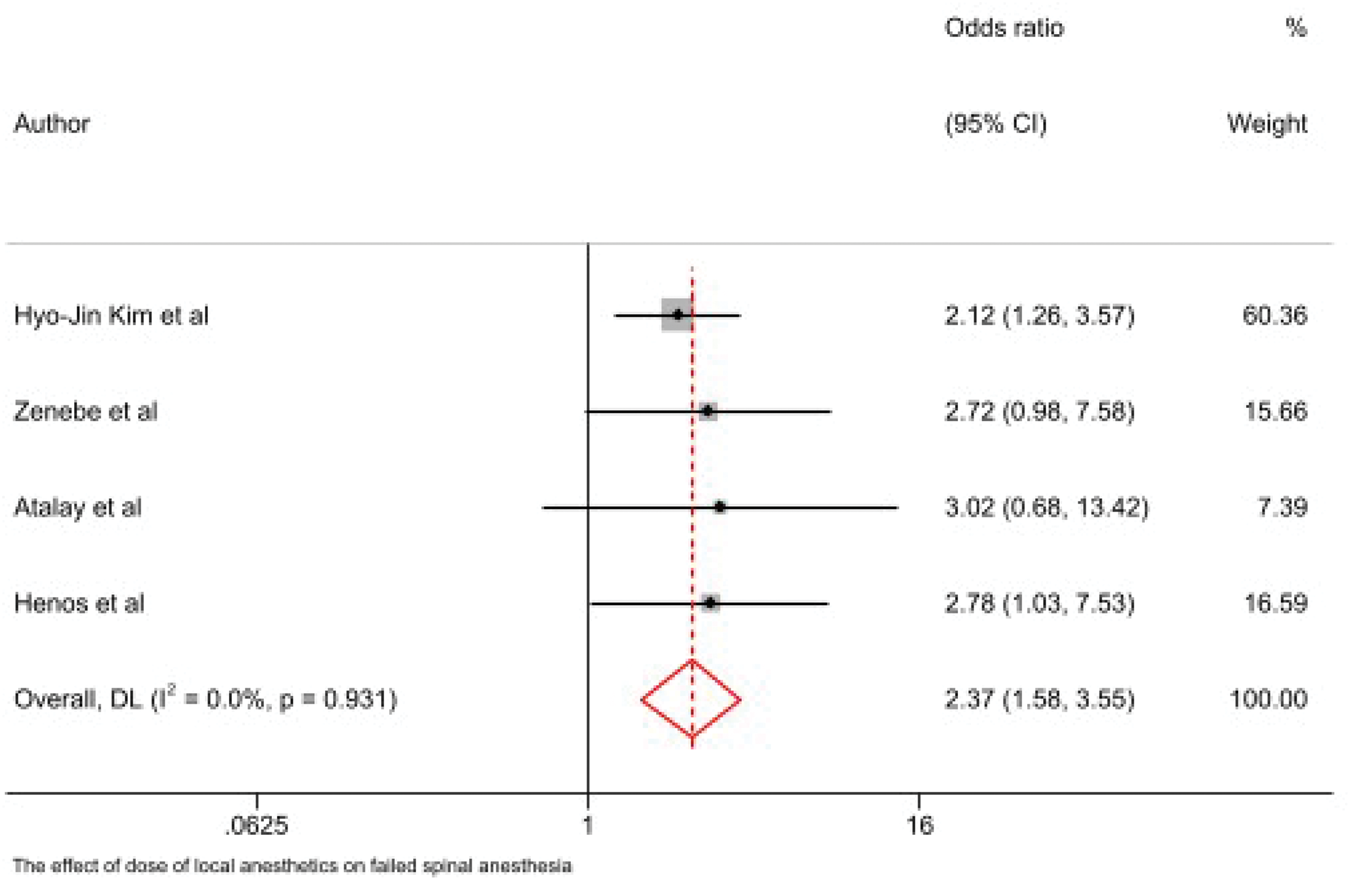
Association between dosage of local anesthetics and failed spinal anesthesia.
Baricity of solution
Isobaric bupivacaine was reported to be less reliable than hyperbaric solutions in achieving predictable block spread,31,32 supporting previous findings on the importance of baricity in CSF-dependent spread (Figure 8). 36

Baricity of local anesthetics and failed spinal anesthesia.
Provider-related factors
Provider inexperience
Novice providers (experience <1 year) had higher failure rates across several studies.3,4,17,33,35 This aligns with foundational research indicating a strong correlation between provider experience and neuraxial block success (Figure 9). 18

Impact of years of experience on failed spinal anesthesia.
Procedural/technical factors
Bloody CSF
Bloody CSF was significantly associated with spinal anesthesia failure in multiple studies,3,4,17 consistent with evidence that vascular puncture and misplacement compromise intrathecal spread (Figure 10). 36
Association between bloody CSF and failed spinal anesthesia. CSF: cerebrospinal fluid.
Absence of free CSF flow:
Poor or absent CSF flow was a strong predictor of spinal anesthesia failure across studies,3,4,17 reaffirming that free flow confirms proper intrathecal placement (Figure 11). 36
Association between the absence of free flow of CSF and failed spinal anesthesia. CSF: cerebrospinal fluid.
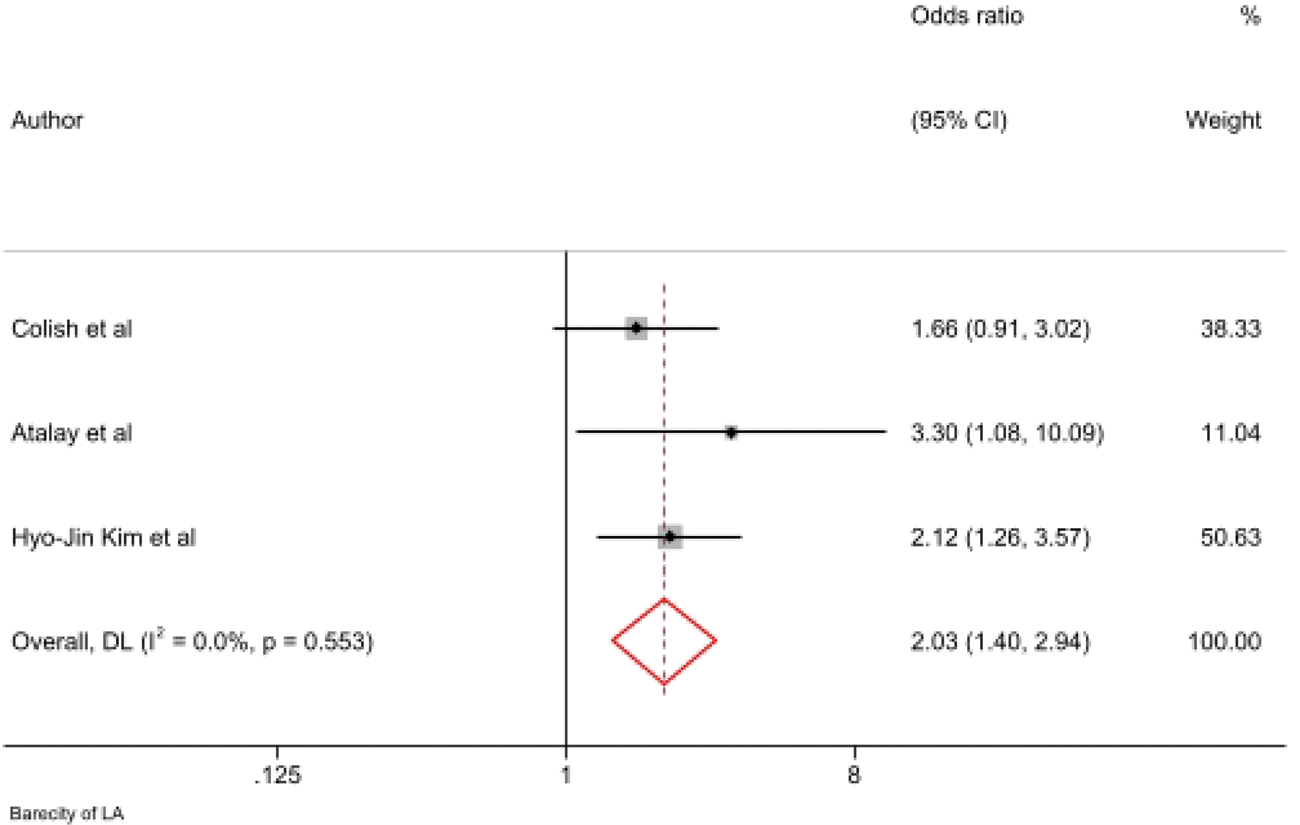
Lumbar puncture at L4–L5:
Studies demonstrated that L4–L5 puncture increased spinal anesthesia failure risk, likely due to limited cephalad spread for surgeries requiring higher block levels (Figure 12).3,4,14
Failed spinal anesthesia and site of lumbar puncture.
Multiple attempts:
Repeated attempts were associated with failure and were frequently considered a marker of technical difficulty.2,17,23,37
Patient-related factors
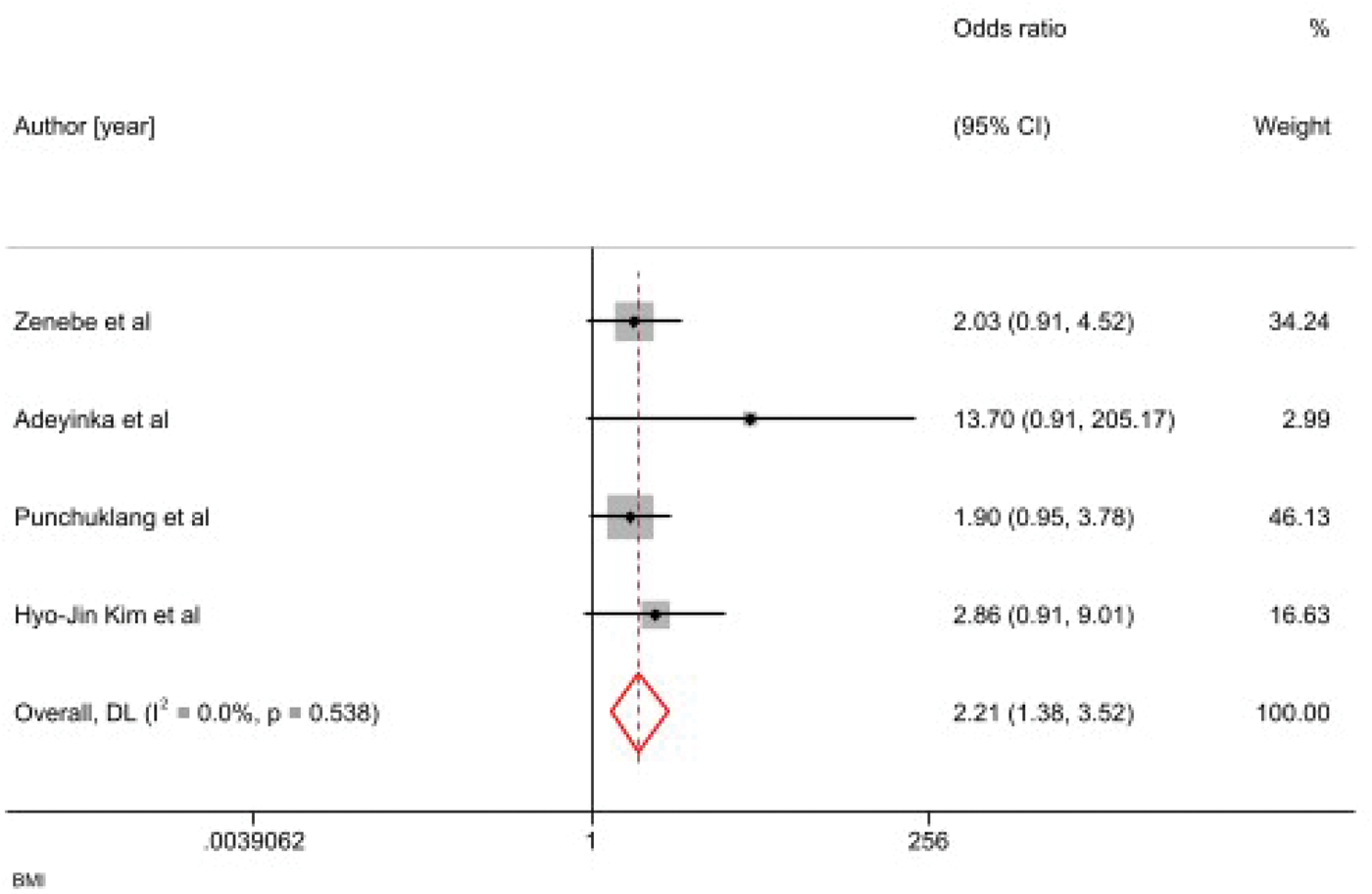
High BMI (≥30 kg/m2):
Several studies found that obesity increased failure risk due to difficult landmarks and increased needle depth.3,4,17 Related studies also support these biomechanical challenges (Figure 13). 33

The association between BMI and failed spinal anesthesia. BMI: body mass index.
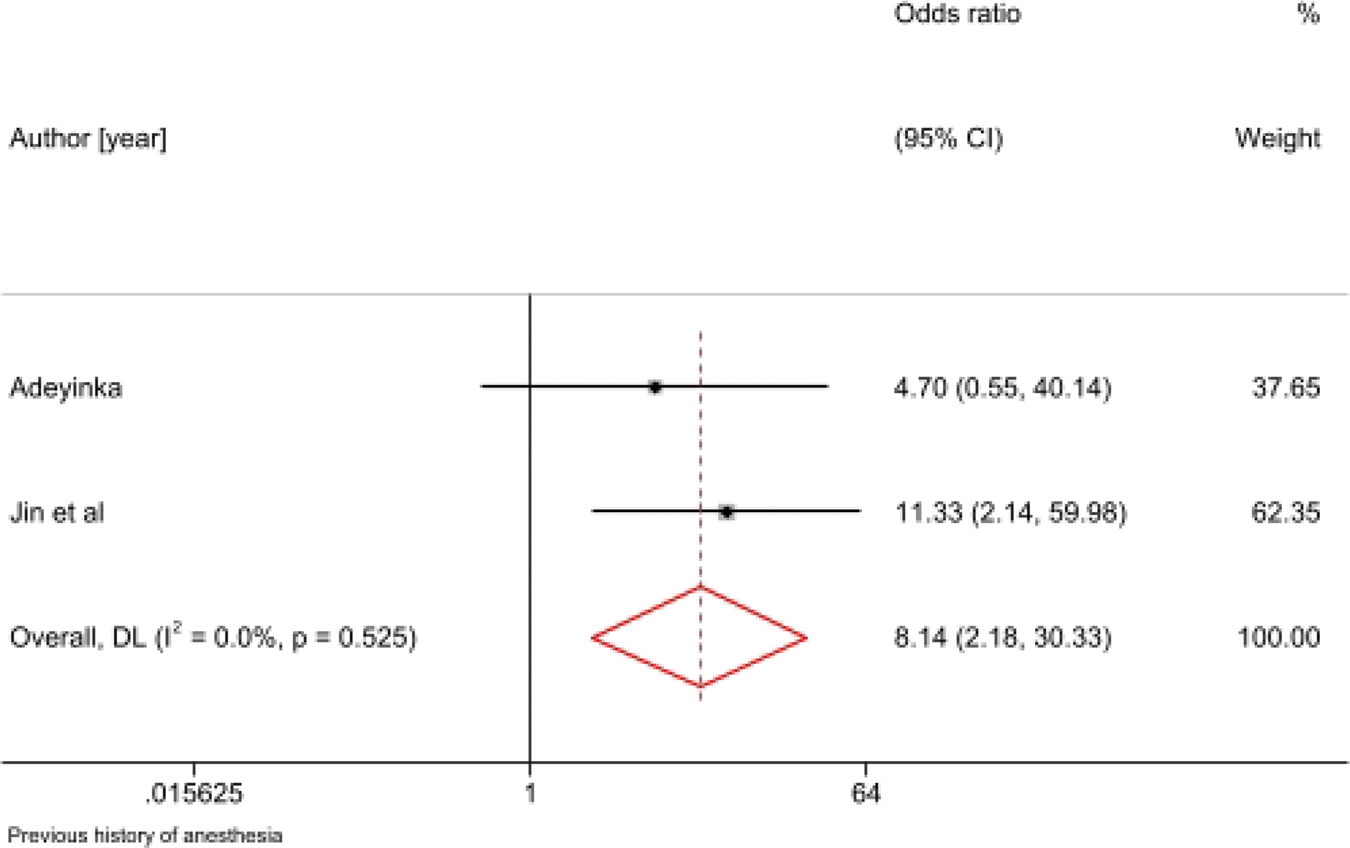
Previous neuraxial anesthesia:
Some studies reported increased failure among patients with prior spinal anesthesia, likely due to scarring, altered anatomy, or CSF flow changes (Figure 14).24,26,27
Failed spinal anesthesia and previous history.
Surgical/contextual factors
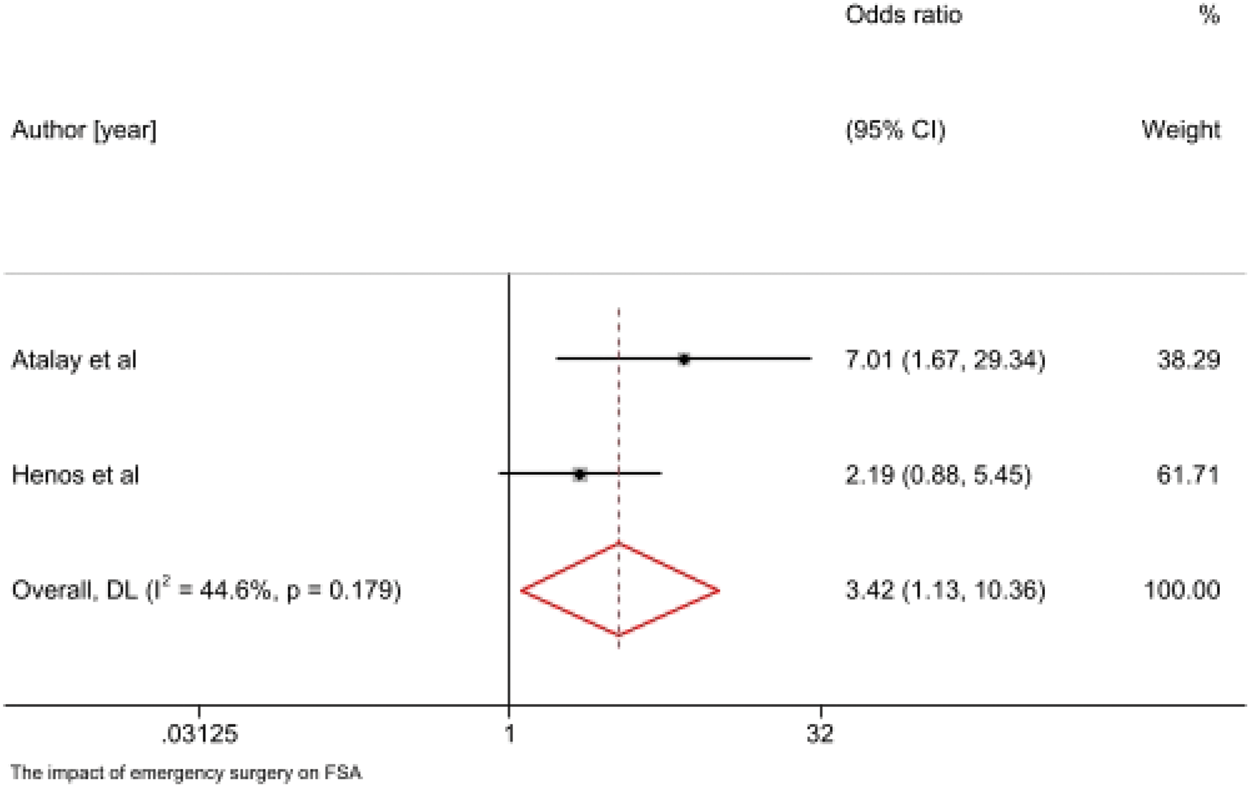
Emergency surgery:
Emergency procedures were associated with higher spinal anesthesia failure rates in several studies.3,17,24,35 Emergency settings often involve time constraints, suboptimal positioning, and less experienced providers (Figure 15). 21

Failed spinal anesthesia and emergency surgery.
Discussion
This systematic review summarizes data from 21 studies evaluating determinants of failed spinal anesthesia. Results demonstrate that spinal anesthesia failure is influenced by technical, patient-related, and provider-related factors.3,4,9,17,23–29,35
Technical factors, including inadequate CSF flow, bloody CSF, suboptimal puncture level, and multiple attempts, were among the most significant predictors. These findings support established mechanistic descriptions by Fettes et al. 36 and Parikh and Seetharamaiah 14 who emphasized the importance of proper identification of intrathecal space and adequate drug deposition.
Local anesthetic selection also played a critical role. Low-dose regimens (<10 mg) and use of isobaric solutions were repeatedly linked to block inadequacy.3,4,30–32 These observations align with pharmacological principles and previous systematic reviews highlighting differences in predictability between hyperbaric and isobaric formulations.31,32
Provider experience remains a consistent determinant, with novice practitioners demonstrating higher spinal anesthesia failure rates.3,4,17,33,35 A previous study by de Oliveira Filho et al. 18 similarly identified experience as an important predictor of neuraxial success.
Patient factors such as BMI and previous spinal anesthesia were also associated with spinal anesthesia failure, consistent with anatomical challenges documented in the literature. 33
Emergency surgery contributed to higher spinal anesthesia failure rates, likely due to time pressure, limited preparation, and the likelihood of less experienced providers performing the block. 21
Conclusion
Overall, the evidence demonstrates that failed spinal anesthesia is multifactorial. Key contributors include low anesthetic dose, isobaric drug solutions, provider inexperience, absence of free CSF flow, bloody CSF, high BMI, previous neuraxial anesthesia, emergency surgery, and suboptimal puncture levels. Improving training, standardizing the technique, and optimizing dose and baricity selection may reduce failure rates. Future studies using multivariable models are needed to elucidate independent predictors.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251403364 - Supplemental material for Contributors to failed spinal anesthesia
Supplemental material, sj-pdf-1-imr-10.1177_03000605251403364 for Contributors to failed spinal anesthesia by Sitotaw Tesfa Zegeye, Belete Muluadam Admassie, Esubalew Muluneh Aligaz, Fikadu Tadese Dires and Samuel Debas Bayable in Journal of International Medical Research
Footnotes
Acknowledgements
Artificial intelligence (AI) tools were used for writing and language improvement during the preparation of this manuscript.
Authors’ contributions
ST: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Resources, Software, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing
EM: Methodology, Resources, Software, Visualization, Writing–original draft, Writing–review & editing
SD, BM, and FT participated in Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing
Availability of data and materials
The datasets used and analyzed during the study are available from the corresponding author on reasonable request.
Consent
Not applicable.
Consent for publication
Not applicable.
Declaration of conflicting interests
There are no conflicts of interest regarding this study.
Ethical approval
Not applicable.
Source of funding
We did not receive any grant from funding agencies in the public, commercial, or not-for-profit sectors for conducting this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
