Abstract
Objective
To investigate the value of mDIXON-Quant imaging–based proton density fat fraction and diffusion-weighted imaging in detecting metabolic syndrome–related renal injury.
Methods
A total of 24 patients with metabolic syndrome and 21 age-matched healthy volunteers were prospectively enrolled in this study and underwent mDIXON-Quant imaging and diffusion-weighted imaging. The participants were divided into those with metabolic syndrome–normal estimated glomerular filtration rate (eGFR; expressed in mL/min/1.73 m2) (n = 10, eGFR ≥ 90; metabolic syndrome group 1), those with metabolic syndrome–mildly decreased eGFR (n = 14, 60 ≤ eGFR < 90; metabolic syndrome group 2), and controls (n = 21). Observer A measured radiological data, including renal fat fraction, renal apparent diffusion coefficient, liver fat fraction, and perirenal fat fraction. Statistical analyses were performed to compare the imaging parameters among groups, assess correlations with eGFR, determine the diagnostic performance of renal fat fraction using receiver operating characteristic analysis, identify eGFR predictors via multivariate analysis, and evaluate inter- and intra-observer consistency.
Results
Renal fat fraction was significantly elevated in metabolic syndrome groups compared with that in the control group (control: 3.83% ± 0.52%; metabolic syndrome group 1: 4.61% ± 0.59%; and metabolic syndrome group 2: 5.32% ± 0.47%; p < 0.001). No significant difference was observed in the apparent diffusion coefficient among the three groups (p = 0.938). Renal fat fraction exhibited an inverse correlation with eGFR (r = −0.688) across the study cohort. No significant correlation was observed between apparent diffusion coefficient values and eGFR (r = −0.104, p = 0.495). The area under the curve value was 0.94 (95% confidence interval: 0.87–1.00) for distinguishing controls from patients with metabolic syndrome. A multivariable-adjusted analysis revealed a significant negative association between the renal fat fraction and eGFR (standardized B = −12.59). The interclass correlation coefficient between observers A and B for renal fat fraction measurements was 0.86.
Conclusion
mDIXON-Quant imaging exhibits greater clinical utility than diffusion-weighted imaging in detecting metabolic syndrome–related renal injury and may serve as a promising imaging tool for monitoring renal lipid deposition and therapeutic efficacy in lipid-mediated renal injury.
Keywords
Introduction
Metabolic syndrome (MetS), a pathological state of metabolic disorders of proteins, fats, carbohydrates, and other substances in humans, is characterized by visceral obesity, hyperglycemia, dyslipidemia, and hypertension. It has been estimated that MetS affects approximately one quarter of the global population, 1 posing a significant threat to human health and public health security. Several studies have confirmed that MetS can induce changes in the renal structure and function, such as a reduction in the estimated glomerular filtration rate (eGFR) and increase in urinary microalbumin level.2,3 It is an independent risk factor for both new-onset chronic kidney disease (CKD) and the acceleration of CKD progression. 4
Among the diverse pathogenetic mechanisms underlying disease progression, lipid disorders have been well-established as one of the major risk factors for MetS and MetS-related renal injury.5,6 Compelling evidence indicates that lipid disorders are directly involved in the pathophysiology of CKD progression through lipotoxic mechanisms.7–9 Under pathological conditions, impaired lipid homeostasis leads to elevated levels of circulating free fatty acids (FFAs) and subsequent ectopic lipid deposition in nonadipose tissues such as the renal parenchyma. This process triggers lipotoxicity—a multifaceted cascade involving mitochondrial dysfunction, podocyte damage, apoptosis of renal tubular epithelial cells, and tubulointerstitial fibrosis—that collectively mediates renal injury.10,11 A lack of awareness and intervention at this stage allows renal injury to progress to advanced stages. 12 Notably, longitudinal cohort studies have demonstrated that early-phase interventions targeting lipid overload—including pharmacological modulation of adenosine monophosphate–activated protein kinase and peroxisome proliferator–activated receptor gamma—significantly attenuate histopathological damage in preclinical models.13,14 These findings underscore the clinical imperative of developing quantification methods to assess renal lipid content in patients with MetS.
Renal biopsy with histochemical staining (e.g. oil-red-O) or biochemical measurement of triglycerides remains the gold standard for evaluating renal steatosis. 15 However, its invasiveness and the associated procedural risks limit its utility for longitudinal monitoring of dynamic changes in lipid deposition or repeated assessments in clinical practice. 16 The development of functional magnetic resonance imaging (fMRI) has addressed the growing demand for noninvasive assessment of renal structure and function. Specifically, mDIXON-Quant–based proton density fat fraction (PDFF) has gained recognition for quantifying lipid content—particularly hepatic fat—in clinical practice and research. 17 It is a voxel-based quantitative technique for measuring fat percentage with good repeatability, making it suitable for small organs such as the kidney. 18 In contrast, diffusion-weighted imaging (DWI) measures the molecular Brownian motion within tissues, with quantification performed via apparent diffusion coefficient (ADC) value, 19 which is sensitive to microstructural tissue alterations—such as cellular edema, inflammatory cell infiltration, and fibrosis—that result from renal injury.20,21 As PDFF and ADC capture distinct yet consecutive pathophysiological events—with PDFF quantifying the initial metabolic insult of lipid deposition and ADC reflecting the subsequent microstructural damage—their combined application is hypothesized to provide a more integrated assessment of MetS-related renal injury. However, evidence applying this complementary approach specifically in patients with MetS remains limited.
Hence, this study aimed to quantitatively evaluate renal fat fraction (FF) and ADC values in patients with MetS using mDIXON-Quant MRI and DWI and assess their utility in detecting lipid-mediated subclinical renal impairment.
Materials and methods
Participants
In this prospective study, participants were consecutively recruited from the Health Management Center between November 2019 and July 2022. MetS was defined according to the consensus criteria jointly established by the International Diabetes Federation; American Heart Association; and National Heart, Lung, and Blood Institute. 22 A diagnosis of MetS was established when patients met at least three of the following five criteria: (a) waist circumference ≥90 cm in males or ≥80 cm in females; (b) serum triglyceride (TG) level ≥1.7 mmol/L or drug therapy for hypertriglyceridemia; (c) serum high-density lipoprotein (HDL) level <1.03 mmol/L in males and <1.29 mmol/L in females or drug therapy for low HDL cholesterol; (d) blood pressure >130/85 mmHg or drug therapy for hypertension; and (e) fasting plasma glucose (FPG) level >5.6 mmol/L or drug therapy for hyperglycemia. Considering sex-specific differences in lipid metabolism, 23 all participants in this study were male. Clinical and laboratory variables were recorded, including age, body weight, waist circumference, body mass index (BMI), TG, FPG, HDL, low-density lipoprotein (LDL), cholesterol (Cho), and serum creatinine (Scr). The eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. A total of 31 male patients were initially enrolled. Among them, seven cases were excluded for the following reasons: (a) severe respiratory motion artifacts affecting measurements due to failure to cooperate with breath-holding (n = 4) and (b) presence of simple renal cysts or other renal lesions (n = 3). Finally, 24 patients with MetS and 21 age-matched healthy volunteers were included in the study (Figure 1), all of whom were Asian. To explore the utility of fMRI in detecting early-stage renal dysfunction, patients with MetS and eGFR <60 mL/min/1.73 m2 were excluded. All participants were classified into three groups: (a) control group (n = 21), healthy volunteers; (b) MetS group 1 (MS-G1, n = 10), cases with eGFR ≥90 mL/min/1.73 m2; and (c) MetS group 2 (MS-G2, n = 14), cases with eGFR ≥60 mL/min/1.73 m2 but <90 mL/min/1.73 m2, which was considered a mild decline according to the Kidney Disease: Improving Global Outcomes (KDIGO) criteria. Healthy volunteers were excluded if they had a history of hypertension, diabetes, vascular diseases, or primary CKD. Detailed inclusion and exclusion criteria are summarized in Figure 1.

Study flowchart. MS: metabolic syndrome; eGFR: estimated glomerular filtration rate.
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of The Third Affiliated Hospital of Southern Medical University (Approval No.: 201708011, Date: 4 August 2017). Written informed consent was obtained from all participants prior to the examination. Patient identifiers were removed from all data to ensure anonymity. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 24
MRI examinations
All MRI examinations were performed using a 3T whole-body system (Ingenia, Philips Healthcare, Best, The Netherlands) with a 32-channel phased-array torso receive coil. Each examination included localizing coronal and axial T2-weighted single-shot fast spin-echo sequences as well as coronal mDIXON-Quant sequences acquired within a ≤15-s breath-hold period, with an automatic reconstruction of PDFF maps. The parameters for mDIXON-Quant imaging were as follows: repetition time (TR) = 5.6 ms; echo times (TE) = 0.95, 1.65, 2.35, 3.05, 3.75, and 4.45 ms; slice thickness = 3 mm; number of slices = 60; matrix size = 144 × 144; number of excitations (NEX) = 1; field of view (FOV) = 360 × 360 mm; bandwidth = 2345.8 Hz/pixel; acquisition time = 15 s. The acquisition volume was centered on the kidneys, with at least the lower pole of each kidney included. The parameters for DWI were as follows: TR/TE = 1529/55 ms; FOV = 300 × 360 mm2; matrix size = 128 × 128; slice thickness =5 mm; 2 b-values = 0 and 800 s/mm2.
Image analysis
Clinical data were collected by a nephrology resident with 2 years of clinical experience. Image analysis was performed by two radiology residents (with 3 and 5 years of experience, respectively) under the supervision of a senior abdominal radiologist with 10 years of professional experience. Avoiding the collecting system and perinephric or renal sinus fat and vessels, the observers selected two representative central slices on the coronal sections near the renal hilum through each kidney and manually segmented the renal parenchyma using a free-hand region-of-interest (ROI) tool. All ROIs were close to the segment center but away from the renal edges. On each parameter map, three ROIs were drawn in each kidney, two in the liver, and one in the perirenal fat on each side. Attention was paid to possible areas of artifacts such as the boundaries along the perinephric or renal sinus fat. We did not measure the FF in the renal cortex and medulla separately, as it is difficult to distinguish the cortex from the medulla in some patients with MetS and atrophic renal parenchyma. ADC maps were generated based on the mono-exponential fitting model. For the measurement of renal ADC, five hand-drawn ellipses were placed on the central two slices of diffusion-weighted images (b = 800 s/mm2), on each side of the kidney. Observer A (5 years of experience) measured these data and was blinded to all clinical information. As the biochemical parameters measured represent the overall function of the bilateral kidneys, the FF and ADC values of the two kidneys were averaged. The mean liver FF and perirenal FF were obtained by averaging the measurements obtained from all ROIs (Figure 2(a)). To assess intra- and interobserver consistency in renal FF measurement, 23 patients were randomly selected. After a 1-week washout period, the renal FF was independently evaluated by Observer A and Observer B (3 years of experience).
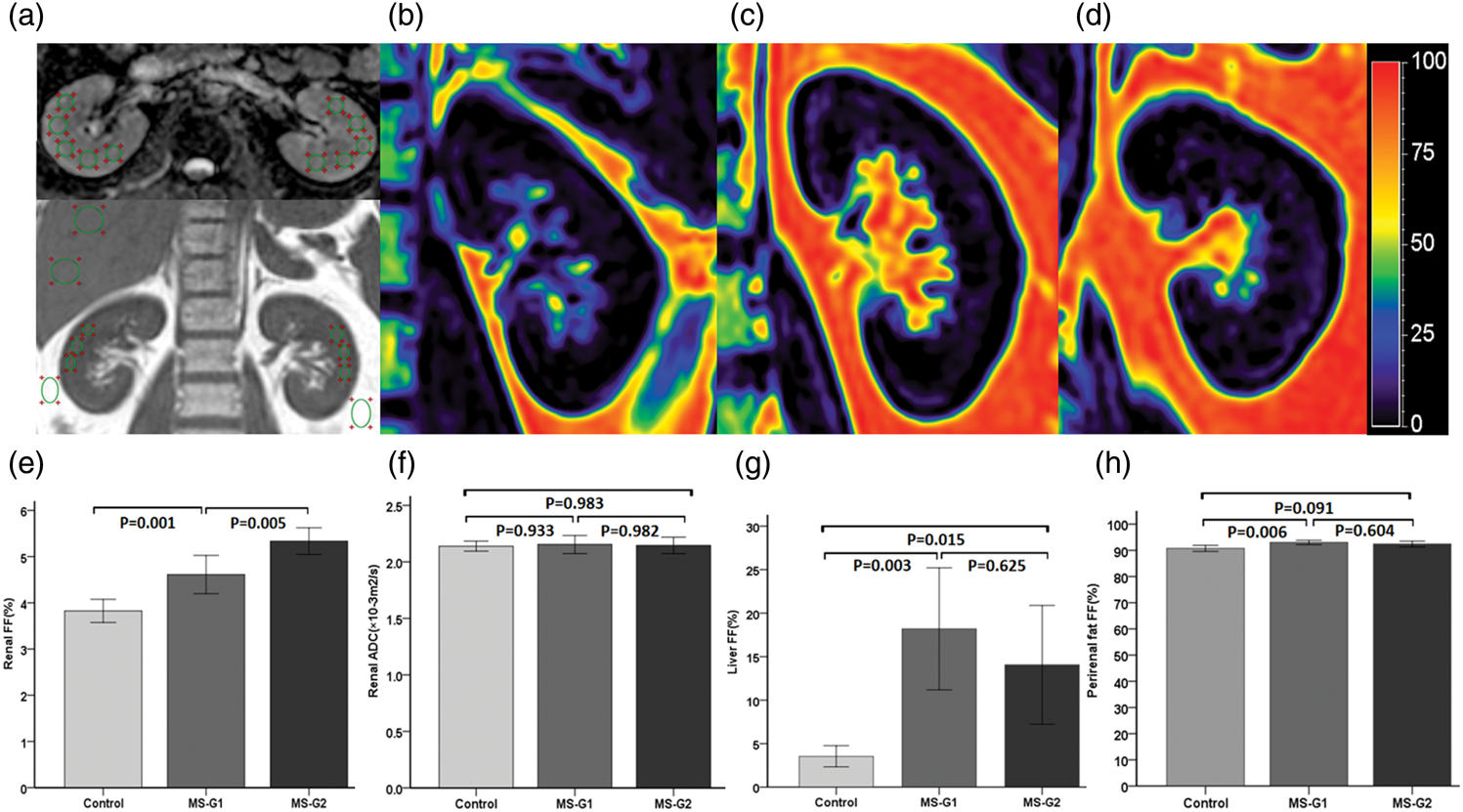
(a) Schematic illustration of ROI placement for the axial ADC map (upper) and the coronal PDFF map (lower). ROIs were manually drawn within the renal parenchyma, carefully excluding the renal sinus, collecting system, and major vessels. (b–d) Color-coded FF maps: (b) a 25-year-old male in the control group, (c) a 28-year-old male in MS-G1, and (d) a 40-year-old male in MS-G2. The average renal FF values in the three groups were 3.34%, 4.41%, and 5.71%, respectively. (e–h) Comparisons of (e) renal FF, (f) renal ADC, (g) liver FF, and (h) perirenal FF among the three cohorts. Renal FF was highest in MS-G2 and lowest in the control group, while no significant differences in ADC values were observed among the three groups.
Statistical analysis
Statistical analyses were performed using SPSS (version 24.0, IBM Corporation, Armonk, NY, USA) and GraphPad Prism (version 9.0, GraphPad Software, Inc., La Jolla, CA, USA). All data were expressed as mean ± standard deviation. Demographic, clinical, laboratory, ADC, and FF data were tabulated and summarized according to the MetS status and eGFR groupings. Data normality was assessed via the Shapiro–Wilk test, and homogeneity of variance was assessed via Levene’s test. One-way analysis of variance (ANOVA) was performed for intergroup comparison of quantified renal FF and ADC values. If the ANOVA result was significant, post-hoc analyses were conducted using Tukey’s honestly significant difference (HSD) test for data with equal variance or Games–Howell test for data with unequal variance, as determined by the Levene’s test. Spearman’s rank correlation analysis was used to evaluate correlations between MRI parameters (Dixon, DWI parameters) and eGFR. Receiver operating characteristic (ROC) curve analysis was performed to evaluate the diagnostic performance of renal FF in distinguishing between the study groups. To examine the nonconfounded effect of renal FF, eGFR (response variable) was modeled using a multivariate linear regression model, with age, body weight, waistline, BMI, TG, FPG, HDL, LDL, Cho, liver FF, perirenal FF, and renal FF as predictor variables. The stepwise method was used for screening predictor variables except for age and BMI. Repeatability of renal FF measurements was assessed via Bland–Altman analysis, and intraclass correlation coefficient (ICC) determination was performed. For all statistical analyses, p < 0.05 was considered statistically significant.
Results
Demographic and anthropometric characteristics
The study included 24 male patients with MetS divided into 2 groups: MS-G1 (n = 10, normal eGFR) and MS-G2 (n = 14, mildly decreased eGFR). Moreover, 21 healthy male volunteers were included in the control group. No significant intergroup difference in age was observed (p = 0.716). The mean BMI in MS-G2 (31.76 ± 5.07 kg/m2) was significantly higher than those in both MS-G1 (28.56 ± 2.06 kg/m2) and the control group (22.21 ± 2.45 kg/m2; p < 0.001). The FPG levels also differed significantly among the three groups (p < 0.001), with MS-G2 exhibiting the highest values (6.54 ± 1.54 mmol/L), followed by MS-G1 (5.51 ± 0.93 mmol/L) and control group (4.90 ± 0.49 mmol/L). HDL levels were significantly lower in both MetS groups than in the control group (control: 1.53 ± 0.40 mmol/L; MS-G1: 1.28 ± 0.27 mmol/L; and MS-G2: 1.20 ± 0.34 mmol/L; p = 0.027). Significant intergroup differences were also observed in body weight (p < 0.001), waist circumference (p < 0.001), TG (p < 0.001), Scr (p < 0.001), and LDL (p = 0.004). No significant difference was detected in total Cho (p = 0.596). Complete demographic and anthropometric data are presented in Table 1.
Characteristics of the study population.
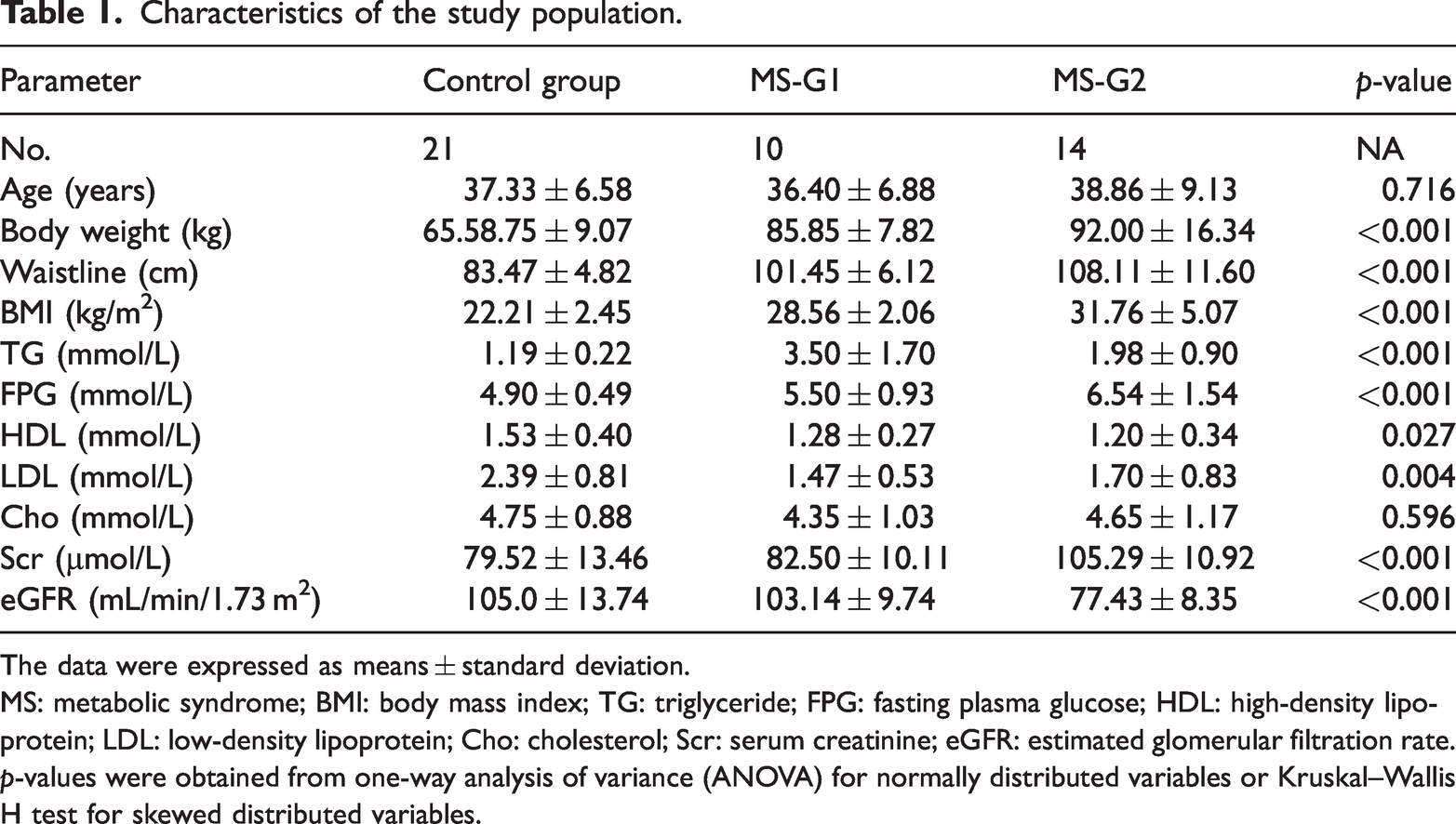
The data were expressed as means ± standard deviation.
MS: metabolic syndrome; BMI: body mass index; TG: triglyceride; FPG: fasting plasma glucose; HDL: high-density lipoprotein; LDL: low-density lipoprotein; Cho: cholesterol; Scr: serum creatinine; eGFR: estimated glomerular filtration rate.
p-values were obtained from one-way analysis of variance (ANOVA) for normally distributed variables or Kruskal–Wallis H test for skewed distributed variables.
Radiological data by group
Significant intergroup differences were observed in the renal parenchymal FF (p < 0.001), with a progressive increase from the control group (3.83% ± 0.52%) to MS-G1 (4.61% ± 0.59%) and MS-G2 (5.32% ± 0.47%; p < 0.05 for all comparisons). Standardized ROI placements in the kidneys, liver, and perirenal fat are illustrated in Figure 2(a), with representative color-coded renal FF maps shown in Figure 2(b) to 2(d). Comprehensive data for renal FF, renal ADC, hepatic FF, and perirenal FF are summarized in Table 2 and visually presented in Figure 2(e) to 2(h). No significant differences were detected in the renal ADC values among the three groups (p = 0.938), with comparable mean values in the control group (2.14 ± 0.10 × 10−³ mm2/s), MS-G1 (2.15 ± 0.11 × 10−³ mm2/s), and MS-G2 (2.15 ± 0.13 × 10−³ mm2/s).
Functional MRI measurements.

The data were expressed as means ± standard deviation.
MS: metabolic syndrome; G1: group 1; G2: group 2; FF: fat fraction; ADC: apparent diffusion coefficient; MRI: magnetic resonance imaging.
Correlation between renal FF/ADC and eGFR
Spearman correlation analysis revealed a strong inverse correlation between renal parenchymal FF and eGFR across the entire study cohort (r = −0.688, p < 0.001). In contrast, no statistically significant correlation was detected between eGFR and renal parenchymal ADC values (r = −0.104, p = 0.495). These relationships are visually depicted in the scatter plots (Figure 3).
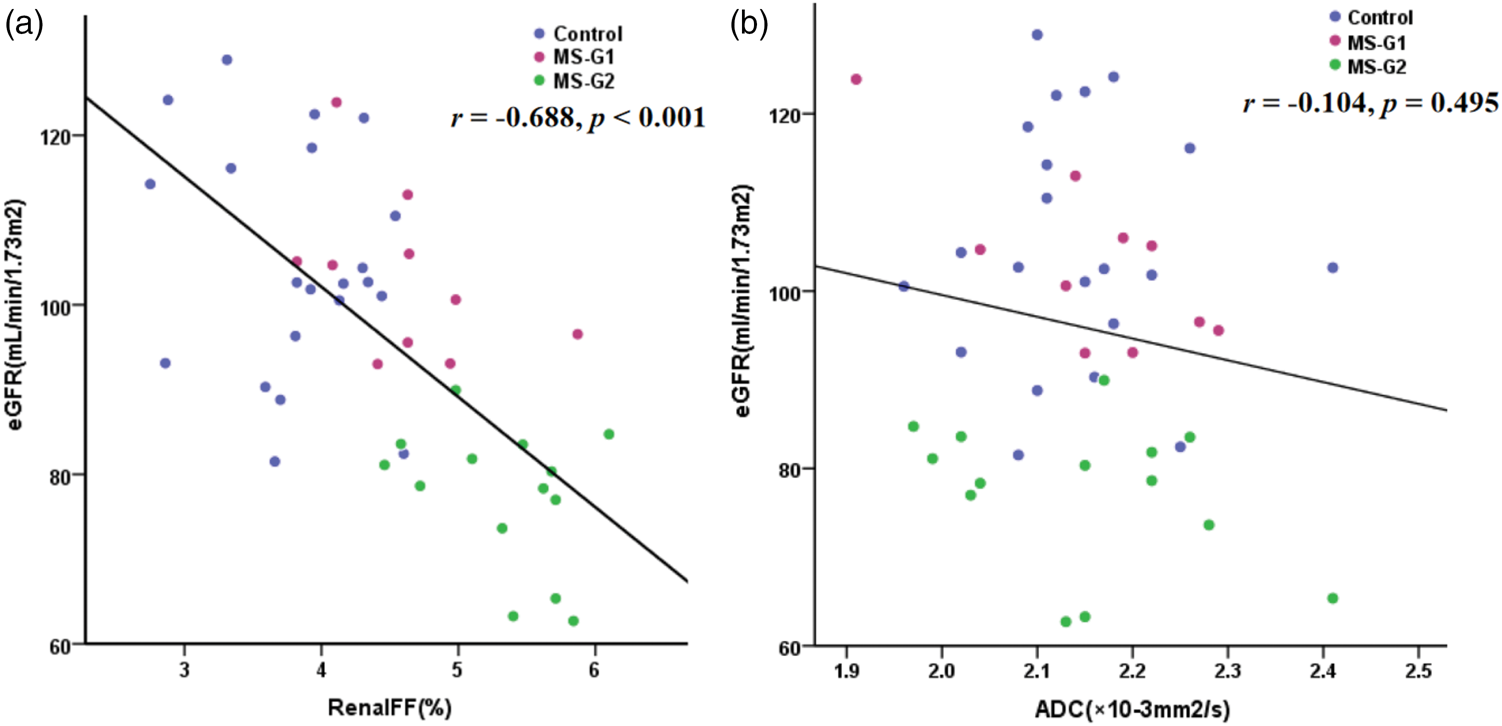
(a) Significant negative correlation between eGFR and renal FF in all participants (r = −0.688, p < 0.01) and (b) no significant correlation between eGFR and ADC values. eGFR: estimated glomerular filtration rate; FF: fat fraction; ADC: apparent diffusion coefficient.
Diagnostic performance of renal FF
The diagnostic performance of renal FF in differentiating between the study groups was evaluated using ROC curve analysis (Figure 4). The area under the curve (AUC) values, along with the optimal cutoff value, sensitivity, and specificity, are presented in Table 3.

Diagnostic performance of renal FF in differentiating study groups. The ROC curves depict the discriminatory power of renal FF for the following comparisons: red curve, healthy controls vs. the entire MS cohort; green curve, healthy controls vs. MS with normal eGFR; and blue curve, MS with normal eGFR vs. MS with mild eGFR decline. FF: fat fraction; MS: metabolic syndrome; ROC: receiver operating characteristic; eGFR: estimated glomerular filtration rate.
Diagnostic performance of renal FF.

AUC: area under the curve; CI: confidence interval; MS: metabolic syndrome; G1: group 1; G2: group 2; FF: fat fraction.
Multivariate analysis of eGFR determinants
In the multivariate linear regression model adjusted for age and BMI, renal FF demonstrated a significant inverse association with eGFR (β = −0.619, standardized B = −12.59 per 1% increase in FF; p = 0.001). No significant associations were observed between eGFR and other variables including renal ADC, hepatic FF, or metabolic parameters (TG, FPG, or HDL) in this model (Table 4).
Multivariate regression analysis for eGFR.

eGFR: estimated glomerular filtration rate; FF: fat fraction; BMI: body mass index.
Reproducibility of renal FF measurements
Reproducibility analysis revealed good intra- and interobserver ICCs for FF measurements: an interobserver ICC of 0.86 (p < 0.001) between Observer A and Observer B, and an intra-observer ICC of 0.93 (p < 0.001) for Observer A based on two repeated measurements with a 1-week washout period. Bland–Altman plots illustrating the reproducibility of renal FF values in 23 randomly selected patients are presented in Figure 5. For the two measurements by Observer A, the mean bias was 0.04% (95% confidence interval (CI): −0.59% to 0.66%). For measurements between Observer A and Observer B, the mean bias was −0.06% (95% CI: −0.88% to 0.77%).

(a) Bland–Altman plots of renal FF values from two repeated measurements by Observer A and (b) Bland–Altman plots of renal FF values between Observer A and Observer B (in 23 randomly selected patients). FF: fat fraction.
Discussion
In this study, we evaluated the clinical utility of renal FF and ADC for detecting lipotoxicity-mediated subclinical renal impairment and demonstrated the feasibility of mDIXON-based noninvasive quantification of renal lipid deposition in patients with MetS. Renal FF was significantly elevated in MetS subgroups (MS-G1 and MS-G2) compared with that in the control group, with the highest FF observed in MS-G2, which exhibited mild eGFR decline. A significant negative correlation was identified between renal FF and eGFR in all participants (r = −0.688, p < 0.001). Furthermore, regarding renal FF, the AUC value was 0.94 for distinguishing controls from patients with MetS. In contrast, no significant differences in ADC values were noted between the MetS and control groups. Furthermore, no significant correlation was found between ADC values and eGFR in the study population. These findings indicate that mDIXON-based PDFF allows quantitative assessment of renal lipid deposition in patients with MetS. Moreover, compared with DWI, the mDIXON-PDFF technique exhibits higher sensitivity in detecting lipid-mediated renal injury and enables earlier identification of structural and functional renal alterations.
Our data demonstrated that renal lipid deposition is present in patients with MetS and correlates with reduced eGFR. Renal lipotoxicity driven by elevated plasma FFAs and their impaired metabolism has been proposed to play a crucial role in the relationship between CKD and MetS. 25 Excessive accumulation of FFAs in the kidney can induce damage to podocytes, proximal tubular epithelial cells, and tubulointerstitial tissue through multiple mechanisms—particularly by enhancing the production of reactive oxygen species, promoting mitochondrial damage, and exacerbating tissue inflammation. 9 However, the preclinical pathological diagnosis of renal injury in patients with MetS is challenging, as individuals without obvious clinical symptoms are often reluctant to undergo renal biopsy—an invasive and risky procedure. In this context, our findings demonstrate that renal FF, as measured by mDIXON-Quant, is significantly elevated in patients with MetS—even in those with normal eGFR—and increases further with a mild decline in the renal function. Importantly, this study adds to the existing knowledge by moving beyond the established role of mDIXON-Quant imaging. By implementing a combined PDFF and ADC imaging protocol, we provide comparative evidence that PDFF holds superior clinical utility for detecting subclinical renal injury in MetS compared with ADC. This positions renal FF not only as a fat deposition biomarker but also as a pivotal noninvasive tool for identifying early, lipid-driven renal pathology.
The current measurements of renal fat content using the DIXON sequence exhibit substantial variability across studies.21,26,27 When positioned within the spectrum of established renal FF ranges, our findings in patients with MetS—with mean FF values of 4.61% in MS-G1 and 5.32% in MS-G2—occupy an intermediate position. These values are considerably higher than those reported for healthy volunteers (e.g. 1.11% in the cortex and 1.10% in the medulla) 21 but are generally lower than those observed in cohorts with diabetic nephropathy (DN). For instance, a study on DN reported cortical and medullary FF values of 1.99% and 2.21%, respectively, 21 whereas other studies on type 2 diabetes mellitus (T2DM) have reported median values as high as 4.89% 26 or mean values reaching 5.6% among study participants. 27 This gradient—from healthy to MetS and further to overt DN—suggests a potential continuum of renal lipid accumulation that parallels disease severity. This discrepancy between our data and the lower FF values reported in certain DN studies 21 is likely attributable to methodological differences. Specifically, in the abovementioned study, ROIs were positioned at the upper and lower poles as well as in the renal cortex adjacent to the outer border of the kidney—an area where Dixon imaging typically shows hypointense regions due to MRI-related artifacts, leading to underestimated measurements. Such artifacts are inherent to chemical shift–based MRI techniques and occur when water and fat protons coexist within the same voxels. 28 Additionally, our focus on the renal polar region helped mitigate potential underestimation of measurements caused by phase-shift artifacts. 29 Despite these methodological variations, our core conclusion remains consistent: Dixon-based MRI reliably detects renal lipid deposition, which is significantly associated with CKD progression. Future studies with expanded sample sizes and standardized methodologies are crucial to definitively establish reference ranges across the entire CKD spectrum, from health to MetS to diabetes, thereby providing a clear benchmark for evaluating lipotoxicity-related kidney injury.
The ADC is a widely accepted indicator for evaluating various renal pathological states. 30 However, our observations revealed no significant differences in the ADC values among the three groups; moreover, we did not identify a significant correlation between ADC values and eGFR. Possible explanations for these findings are as follows. In previous studies,31,32 irreversible pathological changes (e.g. glomerulosclerosis, interstitial fibrosis, and renal tubular atrophy) in the kidneys restrict the free movement of water molecules in intracellular and extracellular spaces, leading to decreased ADC values. Notably, significant differences in ADC values among subjects in these studies were primarily observed in individuals with severely impaired renal function (e.g. those with DN). In the present study, no significant difference in renal ADC values was found between patients with MetS and healthy volunteers. This suggests that although lipid deposition may induce renal function changes, its impact on water molecule diffusion and directional flow is negligible. Consequently, ADC appears insensitive to renal lipid deposition assessment prior to the onset of glomerulosclerosis and interstitial fibrosis. In contrast, mDIXON-Quant–based PDFF can detect lipid deposition before the occurrence of irreversible renal pathological changes, facilitating early clinical intervention and potentially preventing progression to irreversible kidney damage.
Renal fat infiltration (also termed fatty kidneys or renal steatosis) has been recognized since the 1900s. 33 Ectopic fat deposition in the renal parenchyma is linked to lipotoxicity, inflammation, and fibrosis, which contribute to CKD. However, the precise role of lipid deposition in renal functional decline remains incompletely elucidated. Recently, human and animal studies have provided insights into how lipid accumulation in various renal cell types may alter their structure and function, thereby contributing to the pathophysiology of lipotoxicity-related renal injury.11,12 In this study, we observed a significant negative correlation between eGFR and renal FF across all participants. Notably, renal FF values were significantly higher in patients with MetS and mild eGFR decline than in both patients with MetS and normal eGFR and the control group—a finding rarely reported in the literature. Multivariate linear regression analysis (Table 4) revealed that renal FF is a significant independent factor influencing eGFR: even after adjusting for age and BMI, increased renal FF was associated with a significant decline in eGFR. In contrast, ADC values, liver FF, perirenal adipose FF, body weight, BMI, TG, FBG, HDL, LDL, and Cho showed no significant effects on eGFR. These findings suggest that renal lipid deposition may directly contribute to reduced eGFR, and renal FF measured via the mDIXON sequence can evaluate early or subclinical renal injury as well as dynamically monitor the therapeutic responses in lipotoxicity-related renal damage.
Consistency of a technique is critical for longitudinal monitoring of changes. Previous studies have demonstrated high reproducibility of the mDIXON-Quant sequence in measuring PDFF in the liver, spleen, and pancreas, with ICC values approaching 0.9. 34 However, the utility of mDIXON-Quant imaging for assessing renal FF in patients with MetS remains unexplored. Therefore, we conducted validation and repeatability assessments of renal FF measured using this sequence. The results showed strong interobserver agreement between repeated measurements by Observer A and Observer B, with an ICC of 0.86 (p < 0.001). This finding suggests that the quantitative mDIXON sequence enables a reliable assessment of renal fat content and has high clinical utility.
This study has several limitations. First, the small sample size precluded a stratified analysis of differences in renal fat deposition based on the number of MetS components. Second, to avoid potential confounding effects of sex-related differences in lipid metabolism, only male participants were included. Notably, numerous experimental models of kidney disease have established the renoprotective effects of female sex hormones. Specifically, female mice are protected against renal lipid accumulation and exhibit significant resistance to lipotoxicity-induced renal dysfunction compared with their male counterparts.35,36 Therefore, future studies should expand the sample size to analyze the effects of individual MetS components on renal lipid deposition and include female participants to investigate sex-specific differences in renal lipid accumulation patterns. Such a study is currently underway at our institution. Third, all measurements were performed using the same scanner model from a single vendor. Although we did not directly validate this in our study, a previous multi-vendor reproducibility study on hepatic steatosis assessment suggests that scanner differences are unlikely to significantly affect PDFF quantification. 37 Fourth, the cross-sectional nature of this study indicates that the observed statistical correlations do not imply causality. Longitudinal studies are required to establish a causal relationship between renal lipid deposition and renal function. Fifth, our analysis measured global rather than compartment-specific renal FF, precluded by severe steatosis that obscured the corticomedullary junction. Future studies using advanced imaging are needed to delineate the distinct roles of cortical and medullary lipid deposition. Finally, renal FF measurements were not validated by pathological analysis, as all participants were recruited from the Health Management Center: these individuals had either normal renal function or only mild eGFR reduction, and none of them met the criteria for CKD requiring a kidney biopsy.
Conclusion
mDIXON-Quant imaging may detect renal lipid deposition, which leads to renal injury, with high interobserver consistency. This sequence exhibits greater clinical utility than DWI in detecting MetS-related subclinical renal injury and may serve as a promising imaging tool for monitoring renal lipid deposition and therapeutic efficacy in lipid-mediated renal injury.
Footnotes
Acknowledgements
We would like to thank Professor Hequn Zou from Southern Medical University for his assistance in participant recruitment and his overall support for our research institute.
Author contributions
Xiaodong Zhang: Conceptualization, Resources, Data Curation, Supervision.
Yongqiang Li: Conceptualization, Resources, Data Curation, Supervision.
Shisi Li: Formal analysis, Writing–Original Draft, Writing–Review & Editing.
Yanjun Chen: Investigation, Resources.
Yingjie Mei: Investigation, Resources.
Xianfu Mo: Investigation.
Jialing Chen: Investigation.
Chijian Li: Investigation.
Xiran Chen: Investigation.
Dong Han: Formal analysis.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
All authors declare that they have no conflicts of interest.
Funding
This study received funding from the National Natural Science Foundation of China (81801653).
