Abstract
Objective
Palmoplantar warts are among the most common types of warts and are often challenging to treat. Currently, only a few effective treatments exist for palmoplantar warts. This study aimed to compare the efficacy and safety of 0.05% topical calcipotriol ointment and liquid nitrogen spray in the treatment of palmoplantar warts.
Methods
Fifty-eight adult patients with palmoplantar warts were randomly assigned to two groups. Thirty patients in Group A were treated with 0.05% topical calcipotriol twice daily, while twenty-eight patients in Group B received liquid nitrogen spray biweekly. Both treatments were administered for 8 weeks.
Results
Statistically significant reductions in wart number and size were observed in both groups at weeks 4 and 8; however, the differences between the two groups were not statistically significant. Most adverse events in the calcipotriol group were mild, while those in the liquid nitrogen group included pain (82.14%), vesicular lesions (78.57%), erythema (42.86%), bleeding (3.57%), and edema (3.57%).
Conclusions
Both treatments demonstrated comparable efficacy; however, topical calcipotriol was safer than liquid nitrogen spray for treating palmoplantar warts. The 0.05% topical calcipotriol ointment may represent a safer alternative to liquid nitrogen spray for the treatment of palmoplantar warts.
Introduction
Warts caused by human papillomavirus (HPV) infection are highly prevalent worldwide. Approximately one-third of warts are persistent and resistant to treatment, particularly those on the palmar and plantar surfaces and in periungual areas. 1 This is attributed to their location at pressure points and deep penetration beneath thick skin—a phenomenon known as the “iceberg effect.”2,3 Palmoplantar warts can cause pain, discomfort, and functional impairment, significantly affecting patients’ quality of life. 4 Additionally, their esthetic appearance may contribute to psychological distress. Warts also pose a substantial economic burden; approximately two million people in the United Kingdom receive treatment for plantar warts annually, at a cost exceeding US$40 million. 5 Although various treatment modalities exist, none have been established as superior, and recurrence rates remain high. 6 Topical treatments are increasingly preferred owing to their noninvasive nature, cost-effectiveness, and convenience for home use. 7 Recently, vitamin D has attracted growing attention as a potential treatment option for warts. Intralesional vitamin D injections have demonstrated efficacy but require invasive procedures in medical facilities.8–11 A randomized controlled clinical trial reported that topical calcipotriol was significantly more effective than placebo in treating warts and was also considered a safe treatment method. 12 Liquid nitrogen cryotherapy remains a widely used treatment option for palmoplantar warts. However, it requires repeated clinical visits and is associated with adverse effects, including pain, erythema, blistering, infection, and scarring. 4 Because clinical studies evaluating the efficacy and safety of topical calcipotriol, a vitamin D analog, in treating palmoplantar warts are limited, identifying an optimal treatment remains challenging for physicians.
This study aimed to evaluate the efficacy and safety of topical calcipotriol versus liquid nitrogen spray in the treatment of palmoplantar warts.
Materials and methods
This prospective, randomized study was conducted at the Ho Chi Minh City Hospital of Dermato-Venereology. Ethical approval was obtained from the hospital’s medical research ethics committee (No 534/CN-BVDL, 18 March 2024), and written informed consent was obtained from all patients. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki (1975), as revised in 2024.
Patient selection
Fifty-eight adult patients (aged >18 years) from the outpatient department of our hospital participated in the study. Palmoplantar warts were diagnosed based on clinical evaluation. The recruitment period extended from March 2024 to December 2024.
The exclusion criteria were as follows: (a) the presence of >20 lesions; (b) the use of any wart treatment within the past 2 months; (c) the presence of local infection, diabetes-related foot pathology, and immunocompromised conditions; and (d) a history of hypersensitivity to vitamin D. Pregnant or lactating women were also excluded. 12
The sample size was calculated to compare two proportions using the standard formula for trials. Efficacy rates of 85.7% for calcipotriol (p1) and 44% for liquid nitrogen cryotherapy (p2) were assumed.12,13 With α = 0.05 (two-sided) and β = 0.05 (power = 95%), the calculation yielded n ≈ 28 patients per group.
During the recruitment period, patients meeting the inclusion criteria were randomized sequentially until each group included at least 28 participants.
Study design
A coin-flip method was used to randomly categorize the patients into two groups. Group A (n = 30) was asked to apply 0.05% topical calcipotriol ointment (Daivonex 30 g, LEO Laboratories Limited, Ireland) twice daily. Patients were instructed to apply a 1-mm thick layer of ointment to the lesions, with a maximum dose of 100 g/week.
Group B (n = 28) received liquid nitrogen biweekly. The implementation process followed the “Procedure for Treating Skin Diseases with Liquid Nitrogen Spray” and the “Procedure for Improving Liquid Nitrogen Spray Technique in the Examination Department” at the Ho Chi Minh City Hospital of Dermato-Venereology.
All patients were treated until complete lesion clearance or for a maximum of 8 weeks. Follow-up assessments were conducted every 4 weeks to evaluate the clinical outcomes.
Clinical evaluation
Photographs were taken at baseline and at the first (week 4) and second (week 8) follow-up visits. Global treatment response was independently evaluated by the researcher, patient, and blinded investigator using photographs taken before and after treatment. Treatment response was rated using a quartile grading scale based on the reduction in lesion area (1, mild improvement, <25%; 2, moderate improvement, 25%–49%; 3, significant improvement, 50%–74%; and 4, marked improvement, ≥75%). Treatment was considered effective in patients who exhibited significant or marked improvement (≥50% reduction in wart size). Conversely, patients demonstrating mild or moderate improvement (<50% reduction in wart size) were classified as having an ineffective response to treatment. 14 The number of lesions remaining after treatment at weeks 4 and 8 was also recorded as an evaluation parameter.
Adverse effects, including pain, erythema, swelling, blistering, bleeding, and other side effects, were documented. Patients evaluated these side effects at each session and during the first and second follow-up visits.
Patients rated their satisfaction with each type of combination therapy using a four-point Likert scale with the following categories: extremely satisfied, satisfied, partially satisfied, and dissatisfied.
Statistical analyses
Data were entered into a computer and analyzed using JASP software version 0.19.0 (supported by the University of Amsterdam). All patient details were fully deidentified to ensure confidentiality and prevent any possibility of identification. The significance of the results was assessed at a 5% level. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 15
Results
Patient demographics
This study included 58 adult patients aged 18–60 years. No significant differences were observed in the demographic or clinical characteristics between the two groups (Table 1).
Comparison of demographic and clinical data between Groups A and B.

Chi-squared test.
Mann–Whitney U test.
Fisher exact test.
Treatment effectiveness
Number and size of warts after treatment
In Group A, statistically significant reductions in the number (p = 0.045 and p < 0.001, respectively) and size (p = 0.006 and p < 0.001, respectively) of warts were observed at weeks 4 and 8 compared with baseline.
In Group B, statistically significant reductions in the number (p = 0.022 and p = 0.002, respectively) and size (p = 0.020 and p = 0.015, respectively) of warts were observed at weeks 4 and 8 compared with baseline.
However, no statistically significant difference in cure rates was observed between the two groups at weeks 4 and 8 (Table 2).
Number and size of warts after treatment.
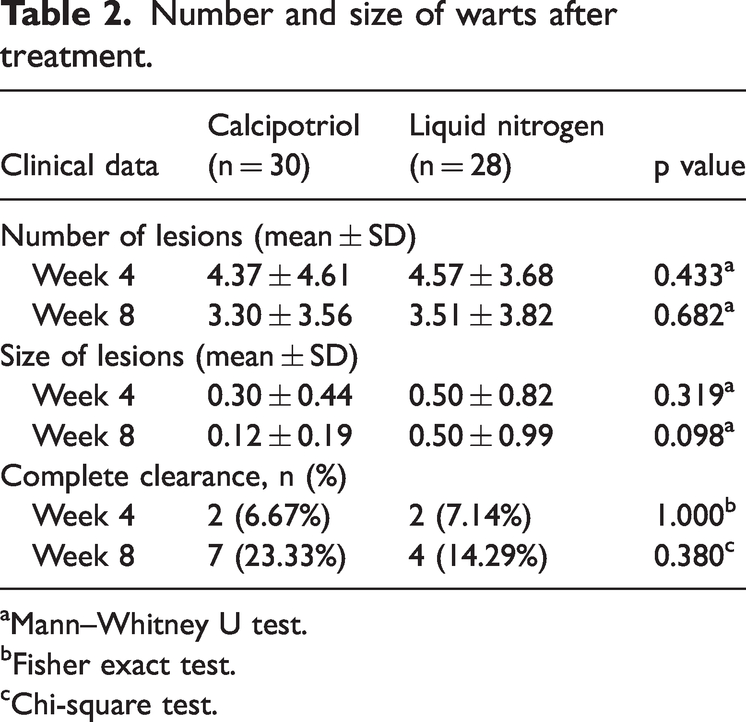
Mann–Whitney U test.
Fisher exact test.
Chi-square test.
Treatment response and efficacy at week 4
At week 4, no statistically significant differences in treatment response (Figure 1) or efficacy (Figure 2) were observed between the two methods, as assessed by the researcher, patient, and blinded investigator.

Treatment response at week 4.

Treatment efficacy at week 4.
Treatment response and efficacy at week 8
At week 8, no statistically significant differences in treatment response (Figure S1) or efficacy (Figure S2) were observed between the two methods, as assessed by the researcher, patient, and blinded investigator.
A statistically significant difference in calcipotriol treatment effectiveness was observed at week 4 (p = 0.027) based on disease duration, with none (0.00%) of the eight patients whose lesions lasted >12 months achieving a treatment response. However, by week 8, this difference was no longer significant (p = 0.207), as determined using Fisher exact test. Furthermore, no associations were observed between calcipotriol treatment efficacy and other demographic or clinical characteristics at either week 4 or week 8.
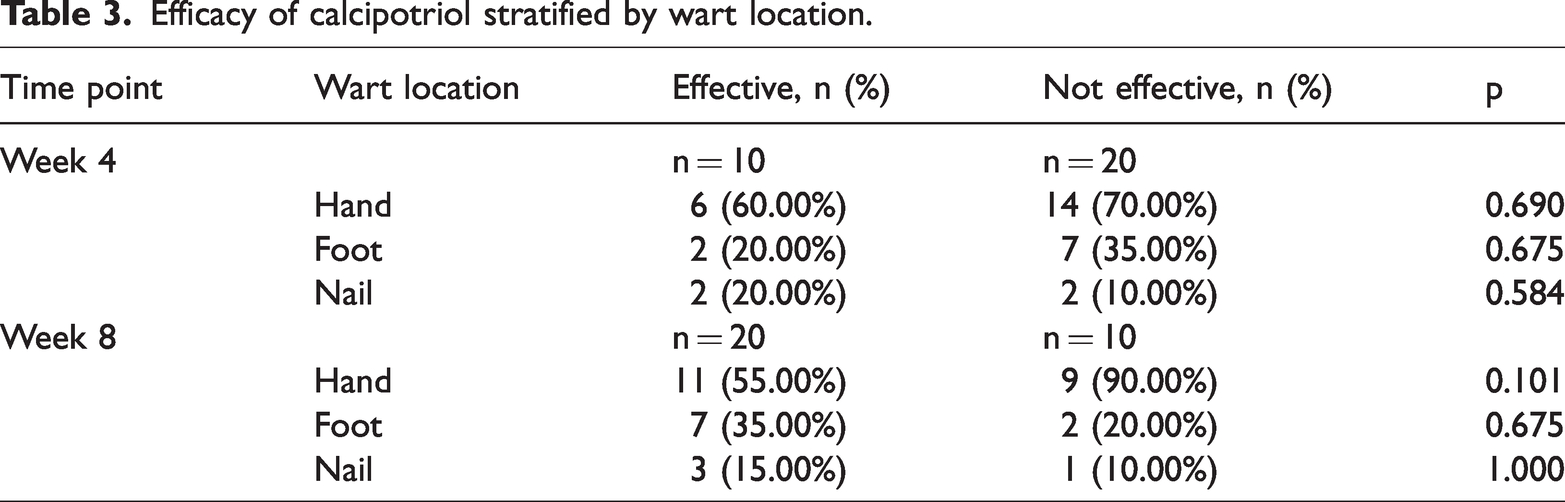
Because the wart location was recorded as a multiple-response variable, we created binary variables for each location. Associations between each location and treatment response were examined using the Fisher exact test. At both weeks 4 and 8, there was no statistically significant association between lesion site and treatment outcome (p > 0.05) (Table 3).
Efficacy of calcipotriol stratified by wart location.
Safety assessment
A statistically significant difference in treatment side effects was observed between Groups A and B (p < 0.001) (Table 4). The most common side effects in both groups were blisters/vesicles (p < 0.001), pain (p < 0.001), and erythema/irritation (p = 0.001).
Treatment side effects.

Pain was recorded in patients who experienced pain lasting at least 1 day after treatment, excluding cases of transient pain occurring during liquid nitrogen spray.
When calcipotriol safety was assessed based on lesion location, no statistically significant difference was observed for warts in the nail area (p = 0.690). However, a significant difference was observed for warts on the hand (p = 0.02); none (0.00%) of the patients with lesions on the hand experienced adverse events, whereas all (100%) of the five patients with adverse events had lesions on the foot (p < 0.001). Additionally, no associations were observed between calcipotriol safety and other demographic or clinical characteristics.
Patient satisfaction
No significant difference in patient satisfaction with treatment was observed at week 4 (p = 0.481) (Figure S3). However, a statistically significant difference was observed between the two groups at week 8 (p = 0.014), as determined using the chi-square test. Nearly all patients (96.66%) in Group A reported being “very satisfied” (53.33%) or “satisfied” (43.33%), and most patients in Group B (57.14%) reported being “satisfied.”
Discussion
The palmar, plantar, and periungual areas are challenging to treat because of their persistence and high recurrence rates of warts in these areas. This study aimed to compare the efficacy, safety, and patient satisfaction associated with topical calcipotriol and liquid nitrogen spray treatments. No significant differences were observed in the baseline data between Groups A and B, and these findings differ significantly from those reported in previous studies.12–16 Topical calcipotriol is preferred because of its noninvasive and safer profile compared with vitamin D injections.
Group A received treatment twice daily for 8 weeks, demonstrating efficacy at weeks 4 (33.33%) and 8 (66.67%). Armaghan et al. 12 reported that 0.05% calcipotriol, applied twice daily for 4 months, significantly reduced wart size and number compared with placebo, with no significant difference in adverse events. Similarly, Almuhyi et al. compared 0.05% topical calcipotriol applied twice daily with intralesional vitamin D3 for the treatment of cutaneous warts. Their results showed that topical vitamin D3 demonstrated greater efficacy, faster response, fewer side effects, and lower risk of recurrence than intralesional vitamin D3. 17 The delayed efficacy of calcipotriol in our study may be attributed to its time-dependent immunomodulatory effects, such as cytokine production and toll-like receptor activation, which enhance antiviral peptide synthesis but require time to reach peak activity. 18
In Group B, 75.00% of the patients exhibited an ineffective response at week 4, which decreased to 50.00% at week 8. Yousaf et al. reported a 43.33% success rate for cryotherapy in the treatment of plantar warts. Conversely, Amin et al. reported that 92% of patients in the liquid nitrogen group achieved treatment success when therapy was administered at 3-week intervals for up to three sessions, followed by a 6-week follow-up period. The differences between our findings and those of previous studies may be attributed to variations in sample size, treatment protocols, or follow-up duration.
Despite the observed differences in the response rate and treatment efficacy assessments among the researcher, patients, and blinded investigator at weeks 4 and 8, the results demonstrated similar trends across all groups. This variation may be attributed to subjective factors in image-based assessment and differences in professional expertise between physicians and patients.
No statistically significant difference was observed in the efficacy of topical calcipotriol when patients were stratified by smoking status at weeks 4 and 8. This finding contrasts with that of El-Taweel et al., who reported smoking as a poor prognostic factor for the response to intralesional vitamin D injections. This discrepancy may be attributed to ethnic differences, variations in smoking prevalence, or other contributing factors, warranting further investigation. 19 Additionally, we observed a statistically significant difference in treatment efficacy based on disease duration at week 4, but not at week 8. This finding aligns with that of El-Taweel et al., who reported no significant difference in vitamin D injection efficacy when patients were stratified by disease duration. These findings suggest that lesions persisting for >12 months may require an extended treatment duration to achieve an optimal therapeutic response. 19 Wart location was categorized into three groups for analysis: hands, feet, and periungual sites. Our findings are consistent with those of Amin et al., who reported no significant difference in treatment outcomes across different locations. 15 Similarly, El-Taweel et al. reported no difference in efficacy among palmar, plantar, and common warts treated with intralesional vitamin D injections. 19 These results suggest that calcipotriol may be effective across most wart sites.
The incidence of adverse events was significantly higher in Group B (85.71%) than in Group A (16.67%). This disparity may be explained by the mechanism of action of liquid nitrogen, which induces lesion destruction and activates a cell-mediated immune response via local inflammation. Although effective, this approach also increases the risk of damage to the surrounding healthy skin. 20 In contrast, Group B experienced fewer side effects, likely because of the treatment’s less invasive mechanism, which regulates epidermal proliferation and differentiation rather than directly destroying lesions. Additionally, the ability of vitamin D to induce antiviral peptides may contribute to a more targeted treatment approach, minimizing its effects on surrounding tissue. 18
A significant difference was observed in the incidence of side effects between the palmar and plantar warts. Specifically, side effects occurred less frequently in palmar warts and more frequently in plantar warts. This may result from prolonged drug contact and increased absorption in foot lesions because footwear can trap medications against the skin. Conversely, frequent hand movements may reduce drug retention and decrease the likelihood of side effects. To the best of our knowledge, no prior studies have compared side effects by wart location, highlighting the need for further research.
At week 8, most patients in Group A reported being “very satisfied” or “satisfied,” with only one patient expressing dissatisfaction. In contrast, 21.43% of patients in Group B were “dissatisfied,” and 3.57% were “very dissatisfied.” This difference may be attributable to the comparable efficacy of calcipotriol, which is associated with significantly fewer side effects than liquid nitrogen. Additionally, the need for frequent hospital visits for cryotherapy may have contributed to the lower satisfaction in Group B.
This study has certain limitations. It was conducted with limited available data; hence, the sample size was small, and the treatment duration was short. Future studies with larger sample sizes, longer treatment durations, and improved study designs are required to enhance statistical power and minimize bias. In addition, extended follow-up periods (≥3–6 months) are recommended to evaluate recurrence rates and post-treatment recurrence times.
Conclusions
Topical calcipotriol and liquid nitrogen sprays demonstrated comparable clinical efficacy in treating palmoplantar warts. However, topical calcipotriol exhibited a more favorable safety profile than liquid nitrogen spray. These findings may encourage the use of a noninvasive, cost-effective, and easy-to-use alternative to conventional methods. When selecting the most appropriate treatment, factors such as disease duration, lesion location, and personal factors (e.g. time commitment and travel costs) should be considered.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251396708 - Supplemental material for Topical calcipotriol versus liquid nitrogen spray in the treatment of palmoplantar warts: An observational study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251396708 for Topical calcipotriol versus liquid nitrogen spray in the treatment of palmoplantar warts: An observational study by Nguyen Dac Khoi Nguyen, Tu Nguyen Anh Tran and Hao Trong Nguyen in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank the staff at the Outpatient Department, Ho Chi Minh City Hospital of Dermato-Venereology, for their help with the data collection.
Author contributions
Nguyen Dac Khoi Nguyen: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Writing–original draft, Writing–review & editing.
Tu Nguyen Anh Tran: Data curation, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing.
Hao Trong Nguyen: Conceptualization, Data curation, Formal analysis, Methodology, Supervision, Validation, Visualization, Writing–original draft, Writing–review & editing.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
No funding was received for this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
