Abstract
Background
Acute type A aortic dissection is more common in acute and severe cases, and hyperglycemia on admission is associated with the risk of adverse outcomes in acute myocardial infarction. However, the impact of admission hyperglycemia on postoperative outcomes in patients who underwent acute type A aortic dissection surgery remains unclear.
Objective
To assess the prognostic value of admission hyperglycemia for postoperative outcomes, particularly the development of acute kidney injury, in nondiabetic patients undergoing acute type A aortic dissection surgery.
Methods
This retrospective study included 322 nondiabetic patients who underwent acute type A aortic dissection surgery at Wuhan Asian Heart Hospital between October 2018 and March 2021.
Results
There was no significant difference in the incidence of postoperative death between the case and control groups. Sixty-nine patients developed postoperative acute kidney injury, and the incidence of postoperative acute kidney injury was significantly higher in the case group (29.2%) than in the control group (17.2%) (p = 0.012). Univariate analysis suggested statistically significant component differences in preoperative stress-induced hyperglycemia, systolic blood pressure, blood urea nitrogen, and intraoperative aortic cross-clamp time between the two groups. Further screening performed by including the above variables in a multifactorial analysis suggested that stress-induced hyperglycemia and systolic blood pressure might be independent risk factors for postoperative acute kidney injury.
Conclusion
This study suggests a correlation between preoperative stress-induced hyperglycemia and the incidence of postoperative acute kidney injury in nondiabetic patients who underwent acute type A aortic dissection surgery.
Keywords
Introduction
Previous research has shown a correlation between preoperative stress-induced hyperglycemia (SIH) and the incidence of postoperative acute kidney injury (AKI) in nondiabetic patients undergoing acute type A aortic dissection (AAAD) surgery, 1 and the only treatment currently available is surgical repair. 2 Although surgical and anesthesia techniques have improved, the prognosis of patients with AAAD remains poor. 3 Frequent progression of accumulated aortic branch vessels causes poor perfusion of the corresponding organs, with the kidneys being one of the most frequently involved organs, leading to several serious complications. Prolonged extracorporeal diversion, intraoperative hypoperfusion, and ischemia–reperfusion injury cause further damage to the kidneys, making AKI the most common complication in the early postoperative period (within 30 days after surgery) of AAAD. 4 Therefore, early identification of risk factors associated with a poor prognosis is clinically significant.
Several studies indicate a close connection between abnormal glucose metabolism and the progression and outcome of cardiovascular diseases. Therefore, the blood glucose (BG) level on admission, often referred to as admission BG (ABG), has been used as a measure of SIH in most studies.5–8 Moriyama et al. 9 reported that high BG level on admission was an independent predictor of AKI in 664 Japanese patients with acute myocardial infarction (AMI), one-third of whom had diabetes mellitus (DM). This association can be explained by several pathophysiological mechanisms, including oxidative stress, 10 insulin resistance, and increased free fatty acid levels. 11 Excessive free fatty acid levels exert toxic effects on infarcted and ischemic myocardium. Another potential mechanism is endothelial dysfunction that occurs in hyperglycemic patients on admission. 12 As a clinical emergency, the pathophysiological changes associated with AAAD induce a strong stress response, which may destroy the stability of the body’s intracellular environment, thereby leading to glucose metabolism disorders. However, the impact of admission hyperglycemia on AAAD prognosis has not yet been reported.
The purpose of this study was to evaluate the predictive value of admission hyperglycemia for the in-hospital prognosis of patients undergoing AAAD surgery.
Methods
Population
This retrospective observational study included consecutive patients who underwent AAAD surgery at Wuhan Asia Heart Hospital between October 2018 and March 2021, all of whom were diagnosed preoperatively using computed tomography angiography. All patient data were anonymized and de-identified prior to analysis to ensure confidentiality. The following patients were excluded from the study: (a) patients with DM, autoimmune diseases, fever of unknown causes, or malignant tumors at the time of admission; (b) pregnant women; (c) patients who had not undergone a BG test within 24 h of admission or had an incomplete medical record; (d) patients who were lost to visit within 30 days of surgery; and (e) patients with a prior diagnosis of DM, those receiving antidiabetic medications, or those with glycated hemoglobin (HbA1c) level ≥6.5% 13 during hospitalization (to ensure the inclusion of only nondiabetic patients). Many study participants took beta-blockers before the surgery, which may have inhibited the occurrence of tachycardia, thereby affecting the relationship between tachycardia and stress hyperglycemia. The control of heart rate by beta-blockers during and after surgery may be one of the reasons for no detection of tachycardia in our study participants. According to the definition of hyperglycemia mentioned in the Joint Statement on Glycemic Control in Hospitalized Patients of the American Society of Clinical Endocrinology and the American Diabetes Association, 14 the eligible case group included patients with ABG >140 mg/dL (7.8 mmol/L) and the control group included those with ABG ≤140 mg/dL (7.8 mmol/L). The study was approved by the Ethics Committee of Wuhan Asian Heart Hospital (Approval No. 2023-B022). All procedures were conducted in accordance with the Declaration of Helsinki (1975) and its most recent revision in 2024.
Data collection
All data were collected by four uniformly trained investigators, including baseline and preoperative characteristics as well as intraoperative and postoperative data. In addition, details of intensive care unit (ICU) and hospital stays were recorded.
Primary endpoint
The occurrence of postoperative AKI was defined according to the Kidney Disease: Improving Global Outcomes (KDIGO) criteria.
Secondary endpoints
Secondary endpoints included in-hospital mortality and other postoperative complications.
Timing
Admission glucose was measured within 24 h of hospital admission, and postoperative renal function was monitored within 48 h and up to 30 days postoperatively.
Definition
We used the American Diabetes Association’s diagnostic criteria for DM, i.e. Hba1c level ≥ 6.5%. 13
According to the 2008 American Heart Association (AHA) Scientific Statement on Hyperglycemia and Acute Coronary Syndromes, a recognized diagnostic criterion for SIH is an ABG level >7.8 mmol/L. 14
In-hospital mortality was defined as all-cause death that occurred during hospitalization or within 30 days after discharge. 15
Stroke was defined as an acute onset of focal neurological deficit caused by cerebral blood supply disturbance and lasting ≥24 h. 16
According to the KDIGO guidelines, 17 postoperative AKI is defined as a rise in the serum creatinine (SCr) level to more than 1.5 times the baseline value, an increase from the baseline value to 26.5 μmol/L within 48 h, or urine output ≤ 0.5 mL/kg/h within 6 h. In the present study, we used the patient’s SCr level on day 1 of hospitalization as the baseline value and compared it with their SCr level during the first 48 h of the postoperative period to determine whether the patient had developed AKI after the surgery.
Statistical analyses
Based on a Gaussian distribution, continuous variables were expressed as mean ± SD or median (interquartile range) values, and categorical variables were expressed as numbers/percentages. Continuous variables were analyzed using analysis of variance (ANOVA) or Kruskal–Wallis test, and categorical variables were analyzed using chi-square test or Fisher’s exact test. Multivariate logistic regression analysis was performed to assess the relationship between ABG and postoperative adverse events of AAAD. Statistical Package for Social Sciences (SPSS, IBM-SPSS version 25.0) was used for data processing. p < 0.05 indicated that the difference was statistically significant. 18
Results
Baseline characteristics
From October 2018 to March 2021, 453 patients underwent AAAD surgery at our hospital. Of these, 131 were excluded (73 with diabetes, 8 with cardiac surgery history, 5 with autoimmune diseases, 3 with tumors, 16 with unexplained fever, 1 who was pregnant, 2 with long-term use of steroids or immunosuppressants, and 23 with incomplete clinical data); finally, 322 patients were enrolled. The flowchart of patient enrollment is shown in Figure 1.

Flowchart of the patient cohort. AAAD: acute type A aortic dissection.
Table 1 presents the baseline data of the two groups of patients; there were 209 patients (64.9%) in the control group and 113 (35.1%) in the case group. Patients in the case group, on an average, were older, had higher body mass index, and had longer ICU stay and hospitalization than those in the control group; there was no significant difference in the preoperative laboratory tests and percentage of aortic branch involvement between the two groups. The incidence of postoperative adverse events in the two groups is shown in Table 2. In total, 29 patients (in-hospital mortality rate of 9.0%) died during hospitalization; those who died after discharge or transfer were excluded from mortality analysis. There was no significant difference in the in-hospital mortality between the control and case groups (p = 0.195). AKI occurred postoperatively in 69 patients (21.4%), representing a significantly higher prevalence in the case group than in the control group (29.2% vs. 17.2%, p = 0.012). In addition, there was no statistically significant difference in the prevalence of gastrointestinal bleeding, pulmonary infection, aortic dissection, cardiac insufficiency, and neurologic complications between the two groups (p > 0.05).
Baseline data grouped according to admission blood glucose levels.

Data are expressed as number (%) for categorical variables and as mean (SD) for continuous variables in case of normal distributions and median (interquartile range (IQR)) for non-normal distributions.
Unpaired Student’s t-test (for normal distributions) and Mann–Whitney U test (for non-normal distributions) were used for quantitative variables, and chi-square test was used for qualitative variables.
BMI: body mass index; COPD: chronic obstructive pulmonary disease; EF: ejection fraction; SBP: systolic blood pressure; DBP: diastolic blood pressure; WBC: white blood cell; BUN: blood urea nitrogen; CPB: cardiopulmonary bypass; ACC: aortic cross-clamp; ICU: intensive care unit; CT: computed tomography.
Postoperative data grouped according to admission blood glucose levels.

Unpaired Student’s t-test (for normal distributions) and Mann–Whitney U test (for non-normal distributions) were used for quantitative variables, and chi-square test was used for qualitative variables.
Univariate analysis of postoperative AKI occurrence in patients who underwent AAAD surgery
According to the results in Table 2, the postoperative occurrence of AKI was used as a grouping variable, and preoperative baseline data and laboratory test data were analyzed univariately. Systolic blood pressure (SBP) (p = 0.044), blood urea nitrogen (BUN) (p = 0.010), and aortic cross-clamp (ACC) time (p < 0.001) showed statistically significant differences between the two groups.
Multifactorial logistic regression analysis of the postoperative incidence of AKI in AAAD patients
The results, as shown in Table 3, were further screened by including the above four variables in a multifactorial analysis, suggesting that SIH (odds ratio (OR) = 1.803, 95% confidence interval (CI): 1.029–3.161, p = 0.039) and SBP (OR = 0.982, 95% CI: 0.966–0.998, p = 0.026) may be independent risk factors for postoperative AKI. Subgroup analyses stratified by SBP quartiles revealed that the association between SIH and AKI remained significant in patients with SBP <120 mmHg (OR = 2.15, 95% CI: 1.22–3.81, p = 0.008), suggesting a synergistic effect of hypotension and hyperglycemia on renal injury. The corresponding forest plot is shown in Figure 2.
Analysis of crude and adjusted OR associations between SIH and AKI via logistic regression.

OR: odds ratio; CI: confidence interval; SIH: stress-induced hyperglycemia; AKI: acute kidney injury; SBP: systolic blood pressure; BUN: blood urea nitrogen; ACC: aortic cross-clamp.
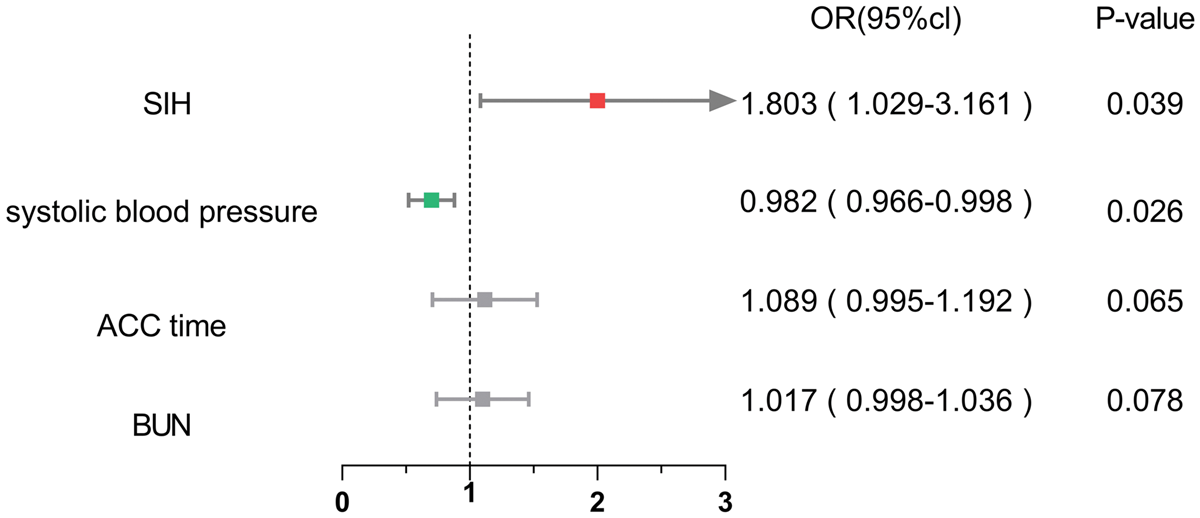
Forest plot of multivariate logistic regression for AKI risk factors (adjusted for age, BMI, and hypertension grade). AKI: acute kidney injury; SIH: stress-induced hyperglycemia; OR: odds ratio; SBP: systolic blood pressure; BUN: blood urea nitrogen; ACC: aortic cross-clamp; ICU: intensive care unit; BMI: body mass index.
Discussion
In this study, we evaluated the predictive value of SIH for the postoperative development of AKI in patients who underwent AAAD surgery. Our results showed that when SIH was present preoperatively in patients who underwent AAAD surgery, it was significantly associated with the development of postoperative AKI. In addition, we found that within a certain range, SBP was an independent risk factor for the development of postoperative AKI.
SIH typically refers to hyperglycemia that occurs for a short period during an episode of illness and is usually limited to patients with no prior evidence of diabetes. 19 Currently, there is no uniform definition of SIH. Most early studies have defined hyperglycemia using the first available BG value or ABG. The most acceptable description of ABG is the first BG level obtained within 24 h of admission. In 2008, the AHA Scientific Statement on Hyperglycemia and Acute Coronary Syndromes recommended the use of an ABG level >140 mg/dL (7.8 mmol/L) to define hyperglycemia in this setting. 14 The evidence underlying this recommendation is primarily derived from retrospective observational studies, and ABG value is an important indicator of the response to acute events in patients without DM.
Several studies have indicated that SIH exacerbates disease conditions and influences the prognosis of patients, especially those with cardiovascular disease. This condition occurs in response to an acute event that increases oxidative stress and free radical production, leading to endothelial, vascular, and immune dysfunction. 9
Previous studies have examined the relationship between newly discovered hyperglycemia and mortality in patients with AMI or acute coronary syndromes (ACSs).
In a previous meta-analysis, 5 the pooled unadjusted relative risk (RR) of in-hospital mortality after myocardial infarction in 1856 nondiabetic patients with SIH on admission was 3.9 (95% CI: 2.9–5.4) compared with that in normoglycemic nondiabetic patients. Compared with nondiabetic patients, the mortality risk in 688 diabetes patients with hyperglycemia was 1.7 (1.2–2.4) times that in diabetes patients with normoglycemia. Another meta-analysis 20 showed that in patients without diabetes, SIH was associated with a higher risk of death after stroke (pooled RR: 3.07, 95% CI: 2.50–3.79). Several possible explanations have been proposed for the association between admission hyperglycemia and increased risk of cardiovascular events. Systemic immune activation, changes in platelet function and the thrombo-fibrinolytic system, abnormal autonomic tone, increased oxidative stress, endothelial dysfunction, and impaired myocardial contractility appear to play a role in myocardial injury.21,22
After cardiac surgery, AKI is a common complication, and the kidneys are extremely sensitive to ischemia and hypoxia, especially in AAAD surgery; cardiopulmonary bypass and deep hypothermic circulatory arrest are reported to aggravate ischemia–reperfusion injury, resulting in renal dysfunction. 23 A previous study found that in diabetic patients with AMI, high ABG values increased the risk of AKI. 24 Another study investigated the effect of ABG on AKI in hospitalized patients and found a significant relationship of high ABG with AKI and mortality in nondiabetic patients. 25 Overall, these reports are consistent with our findings, suggesting that SIH at the time of visit, rather than non-SIH in diabetic patients, causes kidney damage. Parallel dichotomies regarding the effect of hyperglycemia on the outcomes between patients with SIH and diabetic patients without SIH have been reported in other clinical conditions, such as cerebrovascular events 26 and AMI, 27 underscoring the important role of stress response and damage to other organs during AKI induction. In addition, our multifactorial regression analysis showed that admission hyperglycemia was an independent risk factor for AKI development and may be an important predictor of AKI.
Another notable finding of our study is that admission SBP was a risk factor for postoperative AKI. Based on previous studies,28,29 hypotension may be associated with poor prognosis because blood pressure is considered critical for organ perfusion. 30 Toru et al. 31 showed that the pre-hospital SBP of patients can serve as a strong predictor of early AKI after trauma. When the pre-hospital SBP is <125 mmHg, the incidence of AKI shows a 7.75-fold increase. As per another study, 32 compared with AAAD patients with SBP of 81–100 mmHg and 101–150 mmHg, the incidence of acute renal failure was significantly higher in patients with SBP >150 mmHg and ≤80 mmHg (p < 0.004). However, high SBP was not independently associated with poor outcomes, possibly because hypertension is medically easier to treat, mitigating its impact on patient outcomes in AAAD. These assumptions are speculative and need to be tested in future prospective studies. This study shows that within a certain range, the higher the pre-hospital SBP, the lower the incidence of postoperative AKI.
Despite rigorous exclusion of patients with established diabetes, our study has certain limitations that require careful interpretation. First, the potential influence of undiagnosed glucose metabolism disorders (including impaired fasting glucose and prediabetes) cannot be entirely excluded because oral glucose tolerance tests were not routinely performed during preoperative screening. Second, the assessment of perioperative glycemic variability was constrained by nonuniform sampling intervals in the retrospectively collected data, potentially obscuring critical fluctuations relevant to renal outcomes. These methodological limitations inherent to single-center retrospective studies underscore the need for validation in prospective cohorts with protocolized glucose monitoring.
Another limitation of our study is that prognostic biomarkers such as D-dimer and troponin, which are strongly associated with outcomes in aortic dissection, were not consistently available in our retrospective dataset. Therefore, we could not evaluate their correlation with stress hyperglycemia. Future prospective studies with complete biomarker data are warranted to clarify these relationships.
Furthermore, although we excluded chronic corticosteroid/immunosuppressant users, ABG levels may still be confounded by unmeasured variables. Pre-admission medications affecting glucose metabolism (e.g. atypical antipsychotics), dietary patterns, and circadian variations in cortisol secretion could collectively influence acute BG measurements. Future studies should incorporate detailed pharmaco-dietary histories and standardized admission timing to determine the specific effect of surgical stress on hyperglycemia.
The dual nature of SIH, evolutionarily conserved as an acute adaptive mechanism yet detrimental in chronic exposure, may explain our paradoxical findings. Although transient hyperglycemia can provide metabolic support during surgical stress, 21 our data suggest that when it exceeds a critical threshold (as reflected by admission BG levels), it may precipitate renal injury in AAAD patients. This aligns with emerging evidence showing that the clinical impact of SIH follows a J-shaped curve, where both extremes of glucose variability portend poor outcomes. Although we observed a significant association between SIH and postoperative AKI, a causal relationship cannot be confirmed. We speculate that elevated BG is a result of the body’s stress response, which in turn activates the sympathetic nervous system, leading to renal vasoconstriction and renal hypoxia, thereby exacerbating AKI development.
Our findings establish ABG as an independent AKI predictor in patients who have undergone AAAD surgery (adjusted OR: 1.32 per 1 mmol/L increase, 95% CI: 1.15–1.52). However, the following three critical questions remain unresolved: (a) Optimal BG thresholds for perioperative intervention; (b) temporal patterns of glucose dysregulation most predictive of organ injury; and (c) the mechanistic interplay between hyperglycemia-induced oxidative stress and aortic dissection pathophysiology. Multicenter trials comparing stringent versus moderate glycemic control, coupled with biomarkers of mitochondrial dysfunction (e.g. cell-free DNA), could help bridge these knowledge gaps.
The pathophysiological link between SIH and AKI may involve endothelial dysfunction mediated by oxidative stress. Elevated levels of interleukin-6 and tumor necrosis factor-alpha in hyperglycemic states could exacerbate renal ischemia–reperfusion injury, as demonstrated in AMI populations. 33 Future studies should measure these biomarkers to clarify the mechanistic pathways.
Conclusion
Hyperglycemia at admission was significantly and independently associated with the incidence of postoperative AKI in patients who underwent AAAD surgery. The current findings provide guidance for the prevention of postoperative adverse events in patients who have undergone AAAD surgery. The latest European Society of Cardiology guidelines for the treatment of ACS and ST-elevated myocardial infarction recommend that it is reasonable to maintain a BG level <11.1 mmol/L. 34 However, there is no clear research guidance on intervention thresholds and treatment targets for SIH, especially for patients who have undergone AAAD surgery. Previous studies have suggested that in patients who underwent AAAD surgery, when BG levels exceed 11.1 mmol/L and there is no underlying DM, glycemic control should be aggressively pursued, and fluctuations should be regularly monitored. In addition, high-risk clinical factors for SIH should be carefully evaluated. In the future, multicenter large-scale randomized controlled trials are needed to clarify treatment goals and potential clinical benefits.
Footnotes
Acknowledgments
None.
Authors’ contributions
All authors contributed significantly to this manuscript and endorsed the final version.
Consent for publication
Not applicable.
Data availability
Data supporting the results of this study were available from the Wuhan Asian Heart Hospital, but the availability of these data was limited, and these data were used under license for this study and therefore not publicly available. However, data may be obtained from the corresponding author, with permission from the Wuhan Asian Heart Hospital.
Declaration of conflicting interests
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Wuhan Asian Heart Hospital (Approval No. 2023-B022). All procedures were conducted in accordance with the Declaration of Helsinki (1975) and its most recent revision in 2024, and the requirement for informed consent was waived by the Ethics Committee of Wuhan Asian Heart Hospital because the study was a retrospective analysis of previous clinical data.
Funding
None.
