Abstract
Backgrounds
The Systemic Inflammatory Index, a comprehensive marker of blood inflammation linked to multiple diseases, was examined for its association with adult asthma in this large-scale study.
Methods
We analyzed data from the National Health and Nutrition Examination Survey (2009–2020) to explore the relationship between adult asthma (defined by self-report/medication use) and the Systemic Inflammatory Index score (calculated using blood cell counts). We employed weighted logistic regression analysis and restricted cubic splines to examine associations and nonlinear relationships. Subgroup analyses were conducted to evaluate potential interactions among subgroups stratified by sex, age, race, educational level, poverty–income ratio, smoking status, and alcohol consumption.
Results
There were 23,176 participants in this study; of these, 3840 (15.02%) had asthma. Logistic regression analysis showed that high Systemic Inflammatory Index (log2-transformed) scores were associated with increased asthma prevalence. Subgroup analysis revealed that there is no significant interaction effects in the association between the two entities across subgroups (p for interaction >0.05). Restricted cubic spline plots showed a consistent upward trend in asthma prevalence with increasing Systemic Inflammatory Index scores.
Conclusions
Overall, higher Systemic Inflammatory Index scores were associated with higher prevalence of adult asthma in this study. As a cost-effective and readily accessible composite inflammatory marker, the Systemic Inflammatory Index holds potential clinical value in the diagnosis and prevention of adult asthma.
Keywords
Introduction
Asthma is a long-term inflammatory respiratory condition that causes chest tightness, wheezing, coughing, and shortness of breath. The number of asthma sufferers is increasing annually, with more than 360 million people worldwide diagnosed with asthma. 1 The global variation in prevalence is large, with the rate ranging from 0.2% to 21% in adults and 2.8% to 37.6% in children. 2 In China, asthma has become the second most common respiratory disease. 3 Asthma imposes a heavy economic burden worldwide and is a public health problem. Economic losses attributable to asthma exceed US$82 billion in the United States alone 4 and are expected to rise in the coming years. 5 Asthma diagnosis continues to be a challenge, owing to cases of underdiagnosis and overdiagnosis. In the United States, 20%–70% of asthma patients in the community remain undiagnosed and 30%–35% are overdiagnosed. 2 In Vietnam, >80% of children, especially girls, with asthma symptoms remain undiagnosed. 6 Currently, the diagnosis of asthma mainly relies on symptoms and pulmonary function tests. Symptoms are subjective, and pulmonary function tests require a high level of patient understanding and cooperation. There is an urgent need for more objective indicators to aid asthma diagnosis.
Blood cell composition is a common, objective, economical, and easy to obtain indicator; changes in the blood cell composition correlate with the inflammatory responses in the body, making the association with the occurrence and prognosis of respiratory diseases a widely researched topic.7–9 Elevated neutrophil counts and higher neutrophil-to-lymphocyte ratios (NLRs) are strongly associated with acute aggravation of chronic obstructive pulmonary disease (COPD) and may lead to a poor prognosis.10,11 The NLR is also a good predictor of pneumonia onset, intensive care unit admission, and poor discharge outcomes. 12 Elevated eosinophil counts are positively associated with asthma risk. 13
In addition to the above blood cell indices, the Systemic Inflammatory Index (SII) is being used as a novel integrative biomarker for extensive exploration of its correlation with various inflammatory diseases and has proven to be a valid prognostic factor for diseases with inflammation-related etiologies. 14 The SII score is calculated as follows: peripheral neutrophil × platelet count/lymphocyte count. 15 The body's immune system is active at high SII scores, leading to the activation of tissue inflammation; therefore, the SII can be used to monitor disease activity and efficacy.14,16 Satis 17 retrospectively analyzed the SII scores of 109 rheumatoid arthritis patients and 31 normal controls and found that the SII scores of rheumatoid arthritis patients were higher and correlated with disease activity. Kelesoglu Dincer and Sezer 16 concluded that the SII is a simple, practical, cost-effective, and readily accessible tool for monitoring disease activity and treatment efficacy in patients with psoriatic arthritis. In summary, SII may become a novel indicator to aid the diagnosis and monitoring of asthma.
No large-sample studies have examined the relationship between SII scores and asthma occurrence. The National Health and Nutrition Examination Survey (NHANES) is a program in the United States that studies the health and nutrition status of the population. A complex stratified, multi-stage sampling design was adopted to obtain a nationally representative sample. 18 We used the NHANES data to explore the association between SII scores and asthma occurrence and provide strong clinical evidence.
Methods
Study population and design
The NHANES is conducted in 2-year cycles and evaluates approximately 5000 individuals nationwide each year. The database includes questionnaire, biospecimen, and physical examinations data. We used data from six cycles from 2009 to 2020.
Study participants signed informed consent forms developed by the National Center for Health Statistics Research Ethics Review Board. This study involves secondary analysis of de-identified public data; therefore, additional institutional review board (IRB) approval was not required (https://www.cdc.gov/nchs/nhanes/about/erb.html.). The details of the NHANES-relevant content are publicly available at https://www.cdc.gov/nchs/nhanes/. Our study was performed according to the relevant Enhancing the Quality and Transparency of Health Research (EQUATOR) guidelines. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 19
Exclusion criteria
The following exclusion criteria were applied: (a) age <20 years (N = 27,186); (b) pregnant women (N = 404); (c) tumor diagnosis (N = 3792); and (d) incomplete clinical information (N = 10,695) (Figure 1). The distribution of missing values by parameters was follows: poverty–income ratio (PIR, 3951), body mass index (BMI, 2051), insurance status (61), diabetes (20), hypertension (45), educational level (52), marital status (24), smoking status (23), alcohol consumption (4646), and SII score (3088). As a single participant could have missing data for two or more variables, the total number of missing entries was 10,695 (rather than the sum of individual missing counts). In total, 23,176 participants were ultimately included in the analysis, of which 3480 had asthma. Then, according to the requirements for the use of the NHANES database, we chose the mobile examination center (MEC) weights 1/6 × wtmec2yr for weighted analysis. As a cross-sectional observational study, the sample size was calculated using the following formula:
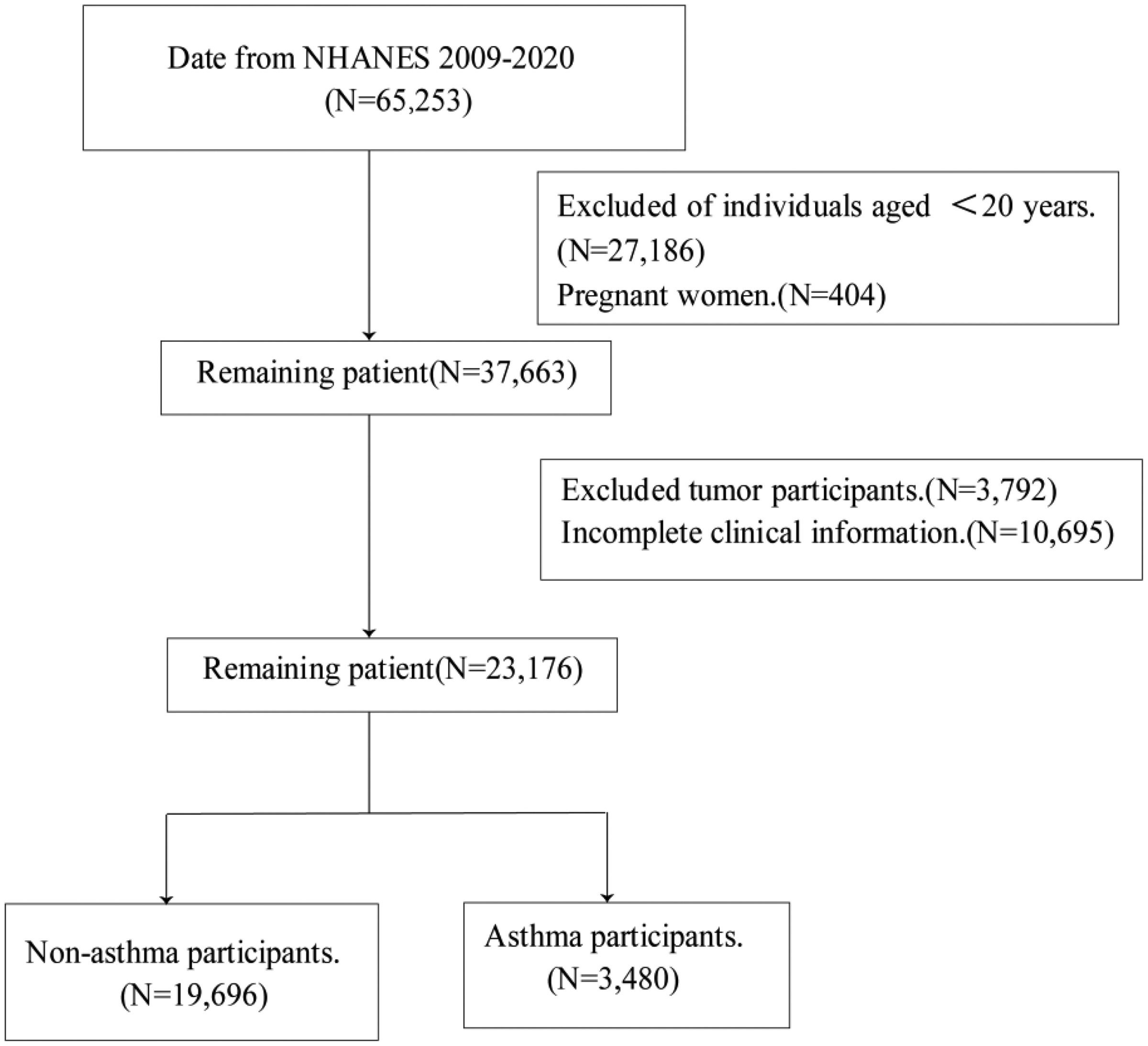
NHANES 2009–2020 participant screening flowchart. NHANES: National Health and Nutrition Examination Survey.
Definition of SII
The MEC of the NHANES uses the Beckman Coulter counting and molecular weight assays to derive parameters for whole blood counts. The formula for calculating SII scores is as follows: platelet count ×neutrophil count/lymphocyte count, as reported in previous studies. 15 In addition, histogram visualization revealed a left-skewed distribution of the SII (Figure 2(a)), likely attributable to substantial interindividual variability in the SII scores. To address this non-normality, we applied logarithmic transformation to SII, which successfully normalized the distribution for subsequent analyses (Figure 2(b)).

(a) The distribution of SII scores and (b) The distribution of log2-transformed SII scores. SII: Systemic Inflammatory Index.
Definition of asthma
Our study utilized data from the NHANES, which employs the following standardized criteria for asthma classification (participants meeting either criterion were considered as having asthma):
Affirmative response (“Yes”) to the survey question: “MCQ010: Have you ever been told by a doctor or other health professional that you have asthma?” Self-reported use of anti-asthma medications (leukotriene modifiers, inhaled corticosteroids, selective phosphodiesterase-4 inhibitors, or mast cell stabilizers) among participants aged <40 years without a smoking history, chronic bronchitis, or emphysema.
This asthma definition has been widely adopted in previous studies.22–24
Covariates
These covariates were identified through self-reported questionnaires, physical examinations, and laboratory measurements. Covariates affecting asthma were included in our study to the extent possible, including sex (Male, Female), age (<65 years, ≥65 years), race (White, Black, Mexican American, and Other), educational level (High school diploma, Lower than high school, and More than high school), PIR (<2, ≥2), smoking status (Never, Ever, Current), alcohol consumption (No, Yes), BMI (<30, ≥30), insurance status (Yes, No), hypertension (Yes, No), and diabetes (Pre-diabetes, Yes, No). The PIR was calculated by dividing the household income by the poverty guideline for a given survey year. Less than 100 cigarettes, more than 100 but not smoking currently, and more than 100 cigarettes and currently smoking were the criteria for never, former, and now smoking, respectively. 25 BMI was calculated by dividing the weight in kilograms by the square of the height in meters (kg/m2); a BMI ≥30 kg/m2 was considered to indicate obesity. Whether or not household insurance was obtained was referred to as the insurance status.
Statistical methods
All analyses were performed using R software (version 4.3.2; The R Foundation for Statistical Computing, Vienna, Austria). The NHANES database uses complex, multi-stage sampling. Appropriate weights were selected based on the study variables, and the results of our statistical analyses are based on the weighting of the data. All continuous variables were converted to categorical variables based on appropriate criteria and expressed as weighted percentages. We used the chi-square test to compare the various groups. The distribution of SII data was significantly uneven and left-skewed. Therefore, we performed a log2 transformation on the values before performing the statistical analysis. To reduce the bias caused by extreme values, we analyzed the log2-SII values after quartile division.
Then, we analyzed the relationship between log2-SII values and adult asthma using a weighted multiple logistic regression model and calculated odds ratios (ORs) and 95% confidence intervals (CIs). The crude model was not adjusted for any variables. Model 1 was adjusted for age, sex, educational level, and race. Model 2 was adjusted for PIR, smoking status, and insurance status based on model 1. Model 3 adjusted for diabetes, hypertension, and BMI based on model 2. In addition, we plotted a restricted cubic spline (RCS) to explore the relationship between log2-SII values and adult asthma.
Finally, we performed further subgroup analyses of interactions based on age, sex, race, educational level, PIR, insurance status, diabetes, hypertension, and BMI. Statistical test results were considered significant if the bilateral p value was <0.05.
Results
General characteristics of the study population
In total, 23,176 people were included in the study, of whom 3480 were diagnosed with asthma. The clinical characteristics of the log2-SII quartile participants are shown in Table 1. After weighting according to the actual population, we found that high log2-SII values were more common in women, older individuals, current smokers, those of White ethnicity, and those with PIR <2 and high school educational level. Furthermore, high log2-SII values were more likely to be associated with obesity, pre-diabetes or diabetes, and hypertension. When participants with higher log2-SII values were compared to those with lower log2-SII values, we also found statistically significant differences in age, sex, race, educational level, PIR, diabetes, hypertension, and BMI (all p < 0.05), except for alcohol consumption and insurance status (p > 0.05).
Weighted baseline characteristics of all participants in the NHANES 2010–2020.

NHANES: National Health and Nutrition Examination Survey.
The relationship between adult asthma and log2-SII values
Table 2 shows that after multifactorial adjustment, the ORs of log2-SII quartiles in the final model were 1 (reference), 1.03 (0.86, 1.23), 1.23 (1.04, 1.47), and 1.26 (1.07, 1.49). The ORs of asthma occurrence tended to increase with log2-SII quartile transformation (p for trend <0.0001). Analysis of continuous variables also demonstrated a stable positive correlation between the log2-SII values and adult asthma (OR: 1.16, 95% CI: 1.08–1.24, p < 0.0001), indicating that a 1-unit increase in the log2-SII value was associated with a 16% increase in the prevalence of adult asthma.
Log2-SII value and asthma-weighted multivariate logistic analysis.
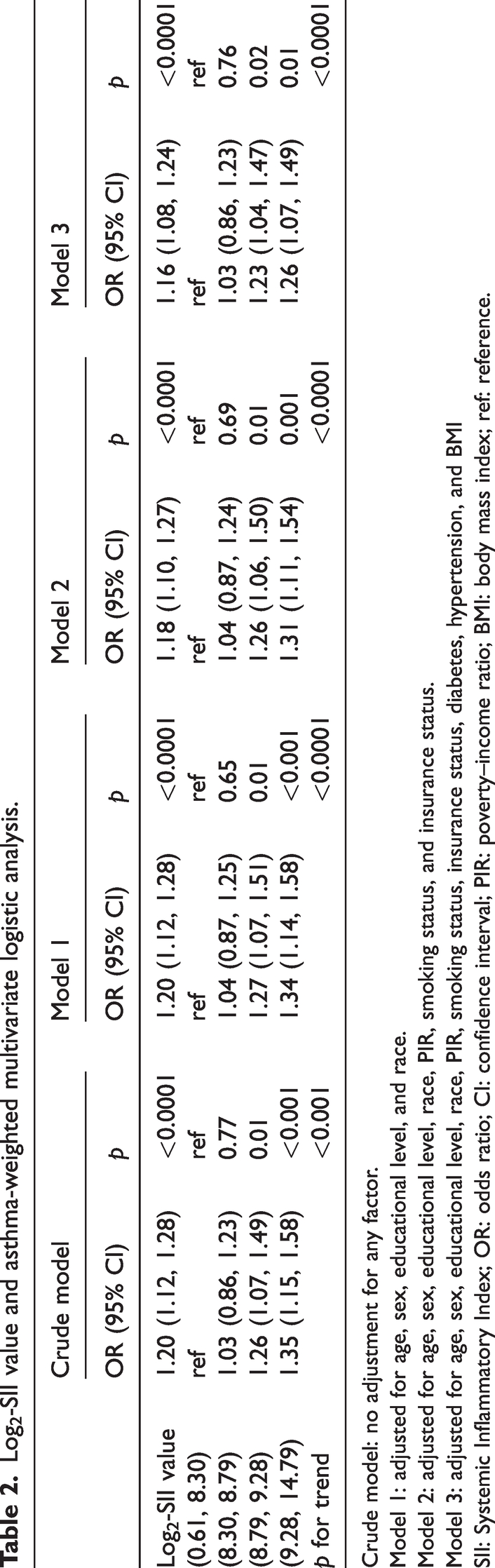
Crude model: no adjustment for any factor.
Model 1: adjusted for age, sex, educational level, and race.
Model 2: adjusted for age, sex, educational level, race, PIR, smoking status, and insurance status.
Model 3: adjusted for age, sex, educational level, race, PIR, smoking status, insurance status, diabetes, hypertension, and BMI
SII: Systemic Inflammatory Index; OR: odds ratio; CI: confidence interval; PIR: poverty–income ratio; BMI: body mass index; ref: reference.
The RCS plot showed a linear correlation between log2-SII values and asthma risk after adjusting for multiple covariates (p overall < 0.0001, p for nonlinear = 0.163, Figure 3). As the log2-SII value increased, the prevalence of asthma correspondingly rose.

The fully adjusted relationship between the log2-SII value and adult asthma in the NHANES 2009–2020 data. SII: Systemic Inflammatory Index; NHANES: National Health and Nutrition Examination Survey.
Stratification of subgroup analysis results and interaction tests
We found that asthma was associated with elevated log2-SII values in most subgroups (Table 3, p < 0.05), except among nondrinkers (Table 3, p > 0.05). In addition, interaction tests showed no significant effect of sex, age, race, educational level, BMI, and PIR on this association (Table 3, all interactions p > 0.05). Additionally, we conducted further stratified analyses to evaluate sex-specific associations between log2-SII values and adult asthma within each racial subgroup. The results demonstrated no significant interaction effects between sex and log2-SII values in association with asthma prevalence across all racial groups (p for interaction >0.05, Figure 4). These findings confirm that the association between log2-SII values and adult asthma remains robust irrespective of sex or racial background. Furthermore, the relationship between sex and this association was unaffected by race.
Subgroup analysis of the relationship between the log2-SII value and adult asthma.

Log2-SII: Q1 (0.61, 8.30), Q2 (8.30, 8.79), Q3 (8.79, 9.28), Q4 (9.28, 14.79).
SII: Systemic Inflammatory Index; Q: quartile; ref: reference.
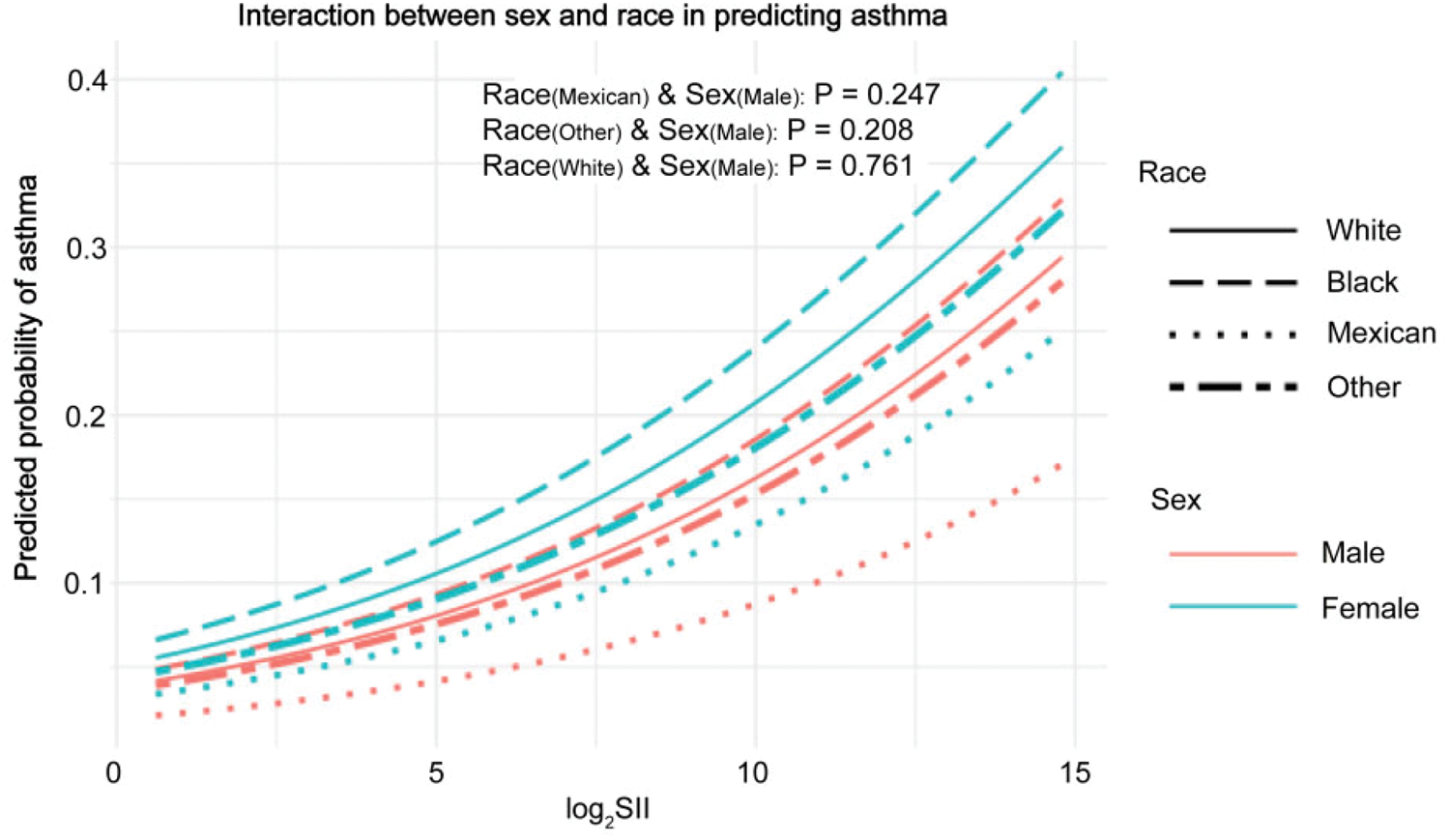
The influence of sex on the association between the log2-SII value and adult asthma across racial groups. SII: Systemic Inflammatory Index.
Discussion
This study ultimately included 23,176 participants from the NHANES database across six cycles from 2009–2020, including 11,508 men and 11,668 women. Among them, 3480 patients had asthma. The findings suggest that people with asthma have higher SII scores than those without asthma. SII scores were significantly associated with asthma. Moreover, in the final model, this relationship was linear, with asthma prevalence increasing with the SII scores.
Asthma is a common respiratory disease that affects more than 360 million people worldwide. Although most asthma deaths are preventable, nearly 400,000 people died from asthma in 2015. 26 Currently, there is no complete cure for asthma, and it can only be prevented or minimized to the greatest extent possible; however, some patients are unable to achieve adequate disease control, resulting in a very poor quality of life. Asthma is very difficult to diagnose and treat, with high instances of overdiagnosis, misdiagnosis, and undertreatment. The diagnosis of asthma is usually based on subjective symptoms. In one study, compared with non-obese patients, obese patients reported more asthma-like symptoms and may have been falsely diagnosed with asthma. 27 Asthma in older individuals is more likely to be misdiagnosed as COPD, resulting in inadequate treatment. 28 In addition, some patients suffer from inadequate asthma treatment owing to economic constraints, lack of understanding of the disease, and fear of adverse drug reactions.28,29
Overall, the diagnosis and treatment of asthma remain challenging, especially accurate diagnosis; therefore, there is an urgent need for objective indicators to aid diagnosis. Asthma is a heterogeneous disease characterized by chronic airway inflammation and hyperresponsiveness. The pathogenesis of asthma remains unclear and is affected by many factors. Blood cell components are objective and common indicators. Among them, neutrophils, eosinophils, lymphocytes, and platelets are closely related to inflammatory response. Neutrophils activate and mobilize the innate immune system and secrete elastase to mediate chronic inflammation. 30 Lymphocytes are associated with adaptive immunity. Platelets influence inflammatory response function by binding to leukocytes and endothelial cells. 31 At present, considerable attention is being paid to the relationship between blood cell composition and asthma.32–34 A study has shown that eosinophil and basophil counts are increased, monocyte counts are decreased, and the eosinophil–lymphocyte, eosinophil–neutrophil, eosinophil–monocyte, and platelet–lymphocyte ratios are increased in patients with asthma. 32
The SII is a composite inflammatory marker derived from platelet, neutrophil, and lymphocyte counts. Our study demonstrates a significant association between elevated SII scores and increased asthma prevalence. While the precise underlying pathophysiological mechanisms for this relationship remain unclear, several potential pathways may contribute to it. First, elastase secreted by neutrophils can induce airway hyperresponsiveness (AHR) and promote airway epithelial–mesenchymal transition (EMT), leading to airway remodeling.35–37 Allergen-activated neutrophils release α-defensins, which stimulate human bronchial epithelial cells to secrete interleukin (IL)-8. 38 IL-8 enhances bronchial smooth muscle cell migration and proliferation, induces AHR, and stimulates airway EMT, thereby promoting airway remodeling.39,40 Furthermore, IL-8 is closely associated with asthma severity. 41 NADPH oxidase in neutrophils generates reactive oxygen species, amplifying allergic airway inflammation.42,43 Neutrophil depletion reduces AHR and remodeling in murine asthma models. 44 Second, lymphocytes are closely associated with asthma development. T helper type 2 cells (Th2) cells secrete IL-4, IL-5, and IL-13 to activate eosinophils and induce asthma. 45 T helper type 17 (Th17) and Th2 cells may share common immune responses. Basic research indicates that IL-6 monoclonal antibodies or retinoic acid-related orphan receptor gamma t inhibitors can reduce Th17 and Th2 immune responses in mice, thereby decreasing neutrophil and eosinophil inflammation in the airways.46,47 Moreover, Th17 cells exhibit greater multifunctional plasticity and instability than Th2 cells. During the acute phase of allergic asthma, the initial polarization of Th17 cells fails to stably migrate into the lungs, subsequently transforming into more pathogenic IL-17–producing Th2 cells that sustain persistent mixed eosinophilic and neutrophilic inflammation in the chronic phase of asthma. 48 Furthermore, the molecular expression levels of distinct lymphocyte subsets correlate with asthma phenotypes/severity. 49 Finally, platelets play multifaceted roles in asthma pathophysiology. 50 Activated platelets can aggregate around thymic stromal lymphopoietin–stimulated dendritic cells (DCs) to induce DC maturation and CCL17 production, playing a critical role in allergen sensitization. 51 Furthermore, platelets directly interact with eosinophils to mediate eosinophil recruitment in the lungs of mouse models of asthma. 52 Research indicates platelet granules contain abundant mediators associated with allergic asthma, including adenosine triphosphate, histamine, serotonin, transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF), and vascular endothelial growth factor (VEGF).53–55 Additionally, factors such as TGF-β, PDGF, and VEGF in platelets can cause epithelial damage, goblet cell hyperplasia, subepithelial thickening, airway smooth muscle hyperplasia, and angiogenesis, thereby playing a significant role in airway remodeling.50,56 Thus, SII and asthma share underlying pathophysiological mechanisms.
In addition, systemic inflammation is associated with increased airway neutrophilia in patients with asthma, possibly owing to its impact on the expression of genes involved in innate immune, defense, and inflammatory responses, which leads to hyperactivation of relevant signaling pathways. 57 Moreover, the SII is a cost-effective indicator that assesses the degree of systemic inflammation. Several studies have demonstrated that SII is associated with the occurrence and progression of many diseases. One study found that SII scores can predict the risk of diabetic nephropathy in patients with type 2 diabetes, making it an affordable and simple method that may help detect diabetic nephropathy. 58 Li et al. 59 showed that high SII scores were associated with increased overall cardiovascular disease mortality in the general population. He et al. 60 further explored the relationship between SII scores and all-cause mortality in people with atherosclerotic cardiovascular disease (ASCVD) and found that for every 1 unit increase in ln-SII, there was a 24% increase in the risk of all-cause mortality in ASCVD patients. The SII also shows unique significance in women, especially postmenopausal women, in whom high SII scores suggest a higher likelihood of osteoporosis. 61 Another study 14 found that the SII can be used to determine the prognosis of patients with psoriasis and psoriatic arthritis. In addition, SII has been shown to correlate with cancer prognosis.14,62,63 In conclusion, the SII has demonstrated its utility in several aspects of a wide range of diseases and has the potential to be a predictor of asthma.
However, there are no studies on the association between the SII and asthma. We explored the relationship between the SII and adult asthma for the first time based on the NHANES database. We found that the prevalence of adult asthma increases with higher SII scores, and a linear relationship exists between SII scores and adult asthma prevalence. Similar relationships also existed in different subgroups, with no interaction effect. These results indicate that SII is linearly correlated with asthma across age, sex, race, educational levels, and lifestyle habits. This finding suggests that modulating systemic inflammation may contribute to asthma prevention and management, while offering potential diagnostic value for adult asthma.
This study offers the following advantages. First, it employs a nationally representative sample. Second, because the NHANES database consists of multi-stage complex sampling data, we analyzed it using a weighted logistic regression model and adjusted for other covariates, enhancing the accuracy and reliability of our results. Finally, we performed log2 transformation for SII before the analysis to ensure a normal distribution.
Limitations
This study has certain limitations. First, this was a cross-sectional study; therefore, we cannot establish a causal relationship. Second, owing to the small sample size, we were unable to consider all potential confounders, such as family history of asthma, which may have led to biased results. Third, the results of this study may not apply to younger patients because our study did not include participants aged <20 years. Furthermore, while we stratified our analysis using a clinically meaningful age cutoff (<65 vs. ≥65 years), an age quartile-based approach might have provided more granular details regarding distributional characteristics across the age spectrum. Finally, most of the data we used were collected through interviews or self-reported questionnaires; therefore, undiagnosed asthma cases may exist, introducing recall and selection bias. Future studies should incorporate spirometry or biomarker data (e.g. fractional exhaled nitric oxide) to mitigate selection bias.
Conclusion
To the best of our knowledge, this is the first study to explore the relationship between SII and asthma and provide novel insights into the prediction of asthma occurrence. Findings have shown that the SII score is positively correlated with the occurrence of asthma and may contribute to its clinical diagnosis to a certain extent. With the advantages of low cost as well as simplicity and ease of use, the SII is a promising and convenient inflammatory marker for predicting adult asthma. We believe that the SII can become a useful predictor for several diseases.
Footnotes
Acknowledgements
Not applicable.
Author contributions
Yuting Fan: Conceptualization, Methodology, Writing–review and editing. Youli Wen: Conceptualization, Data curation, Writing–review and editing. Ying Yang: Investigation, Writing–Original Draft Preparation, Writing–review and editing. Wenqiang Li: Formal analysis, Data curation, Writing–review and editing. Zhiping Deng: Methodology, Supervision, Writing–review and editing. Qian Huang: Methodology, Writing–Original Draft Preparation, Writing–review and editing.
Consent for publication
Not applicable.
Data availability statement
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Ethical considerations and consent to participate
The NHANES database is administered by the National Center for Health Statistics (NCHS) of the Centers for Disease Control and Prevention (CDC). All NHANES protocols were reviewed and approved by the NCHS Research Ethics Review Board (ERB), and all participants provided written informed consent prior to survey participation. Furthermore, the NHANES data used in this analysis are publicly available, de-identified secondary data, and therefore did not require additional human research ethics approval.
Funding
This study was supported by the 2022 Scientific Research Project of Zigong Academy of Medical Sciences (No. ZGYKY22KF001, No. ZGYKY22KF005, and No. ZGYKY22KF009).
