Abstract
Purpose
This review aimed to compare the efficacy of various sympathetic nerve regulation techniques in the treatment of cold sensations or abnormal sweating in hemiplegic limbs following stroke, providing guidance for managing autonomic nervous system dysfunction in post-stroke patients.
Methods
A comprehensive search of the PubMed and China National Knowledge Infrastructure databases was conducted using the keywords “cerebral infarction,” “cerebral hemorrhage,” “stroke,” “stellate ganglion block,” “sympathetic nerve block,” “sympathetic neurolysis,” and “radiofrequency sympathectomy.” Relevant studies published in English on the use of sympathetic nerve regulation to treat cold sensations or abnormal sweating in hemiplegic limbs after stroke were selected and rigorously reviewed. The collected literature was then categorized based on content and reviewed along with experimental results and clinical case evidence.
Results
Sympathetic nerve regulation significantly improves cold sensation and abnormal sweating in hemiplegic limbs by inhibiting sympathetic activity, reducing sympathetic tone, increasing collateral and peripheral circulation, and enhancing skin and muscle blood perfusion, thereby increasing limb temperature and improving patient quality of life.
Conclusion
Computed tomography–guided thoracic/lumbar sympathetic radiofrequency ablation has demonstrated significant and lasting efficacy, with minimal complications. Therefore, it is recommended for treating cold sensations or abnormal sweating in hemiplegic limbs post-stroke.
Keywords
Background
Autonomic nervous system (ANS) dysfunction is a common complication following stroke, with research indicating a bidirectional relationship between them. 1 ANS dysfunction not only impairs patients’ quality of life but also influences prognosis, potentially leading to disability in severe cases. 2 The underlying mechanisms may involve damage to the central autonomic network—including the frontal and parietal cortices and brainstem—or impairment of the autonomic pathways, extending from the hypothalamus through the midbrain, pons, and medulla to the spinal cord. 3 Clinical manifestations vary widely, which include cold sensation or abnormal sweating in the affected limbs; cardiovascular autonomic dysfunction, such as arrhythmias and impaired blood pressure regulation; and bladder, bowel, and sexual dysfunction.4,5 Cold sensation or abnormal sweating on the hemiplegic side is commonly persistent or irreversible 6 and significantly impacts limb functional recovery. Rehabilitation outcomes are generally poor owing to compromised circulation in the affected limbs, often combined with hypertonia and spastic pain; disability may occur in severe cases. Early and effective treatment of post-stroke cold sensation or abnormal sweating in the hemiplegic limb not only enhances warmth perception but can also accelerate the rehabilitation process and boost the patient’s confidence regarding further treatment, a crucial factor affecting the disability rate. 7 Furthermore, parasympathetic impairment is more prominent than sympathetic impairment in ANS dysfunction following stroke, especially ischemic stroke,8–10 and can lead to multisystem complications such as cardiovascular abnormalities, including arrhythmias, electrocardiogram changes, and blood pressure dysregulation; respiratory issues; gastrointestinal problems, including gastroparesis and stress ulcers; sudomotor dysfunction; and genitourinary disorders.11–14 Elevated circulating catecholamine levels due to sympathetic overactivation may be a key contributing factor. 11 Current pharmacological treatments and functional training have demonstrated limited treatment efficacy. However, sympathetic nerve modulation techniques have shown significant potential in the treatment of post-stroke cold sensation and sweating abnormalities in hemiplegic limbs.15–19 Vagus nerve stimulation (VNS) has also emerged as a promising treatment approach during the hyperacute phase of stroke. 1 This review summarizes the literature on sympathetic nerve modulation for treating cold sensations and abnormal sweating in hemiplegic limbs post-stroke, based on a review of articles published between 1 January 2000 and 30 June 2024, retrieved from the PubMed and China National Knowledge Infrastructure (CNKI) databases. This research was performed in line with the principles of the Declaration of Helsinki (as revised in 2024). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.20,21
Cold sensation in hemiplegic limbs
Stroke can lead to altered temperature sensitivity in patients,22,23 with significantly lower skin temperatures observed on the hemiplegic side than on the unaffected side, particularly in the feet. 24 Infrared (IR) thermography can confirm this phenomenon. 25 In addition to the limbs, asymmetric skin temperatures can occur in the chest. This asymmetry is often detected in the early stages after stroke; however, most patients do not begin to feel cold until months later. These symptoms may manifest only when the individual is exposed to cold or hot temperatures in their “normal life.” The most significant temperature differences are observed in the upper limbs, hands, calves, and feet, with an astounding difference of 6°C–10°C. This symptom not only reduces patients’ quality of life but also hinders rehabilitation owing to poor blood flow.
The mechanisms underlying cold sensations in hemiplegic limbs are complex and may be related to pyramidal tract lesions or reflex sympathetic dystrophy. In cases of medullary infarction, the coldness in hemiplegic limbs may be caused by damage to the medullary vasomotor center. Damage to higher regulatory centers weakens the inhibitory effect of the sympathetic nervous system, leading to abnormal sympathetic excitation, increased vascular tone, reduced skin blood flow, and decreased skin temperature on the hemiplegic side. 26 Poor blood flow to the affected limb directly affects the efficacy of rehabilitation therapy.
Sympathetic nerve modulation therapy
The peripheral nerve ganglia are commonly targeted in clinical treatment because of the anatomical characteristics of the sympathetic nervous system. Techniques such as stellate ganglion block (SGB), thoracic/lumbar sympathetic chemical neurolysis, and thoracic/lumbar sympathetic radiofrequency ablation have demonstrated significant clinical efficacy with minimal surgical trauma in the treatment of conditions including head and facial pain, hyperhidrosis, cold limbs, lower-limb arterial occlusion, and Raynaud syndrome. These treatments are well-tolerated by patients and have thus been widely used in clinical practice, generally in combination with traditional rehabilitation therapies.16,27–35 We have categorized the sympathetic nerve modulation techniques based on their invasive and noninvasive nature (Table 1).
Classification of sympathetic nerve modulation techniques.
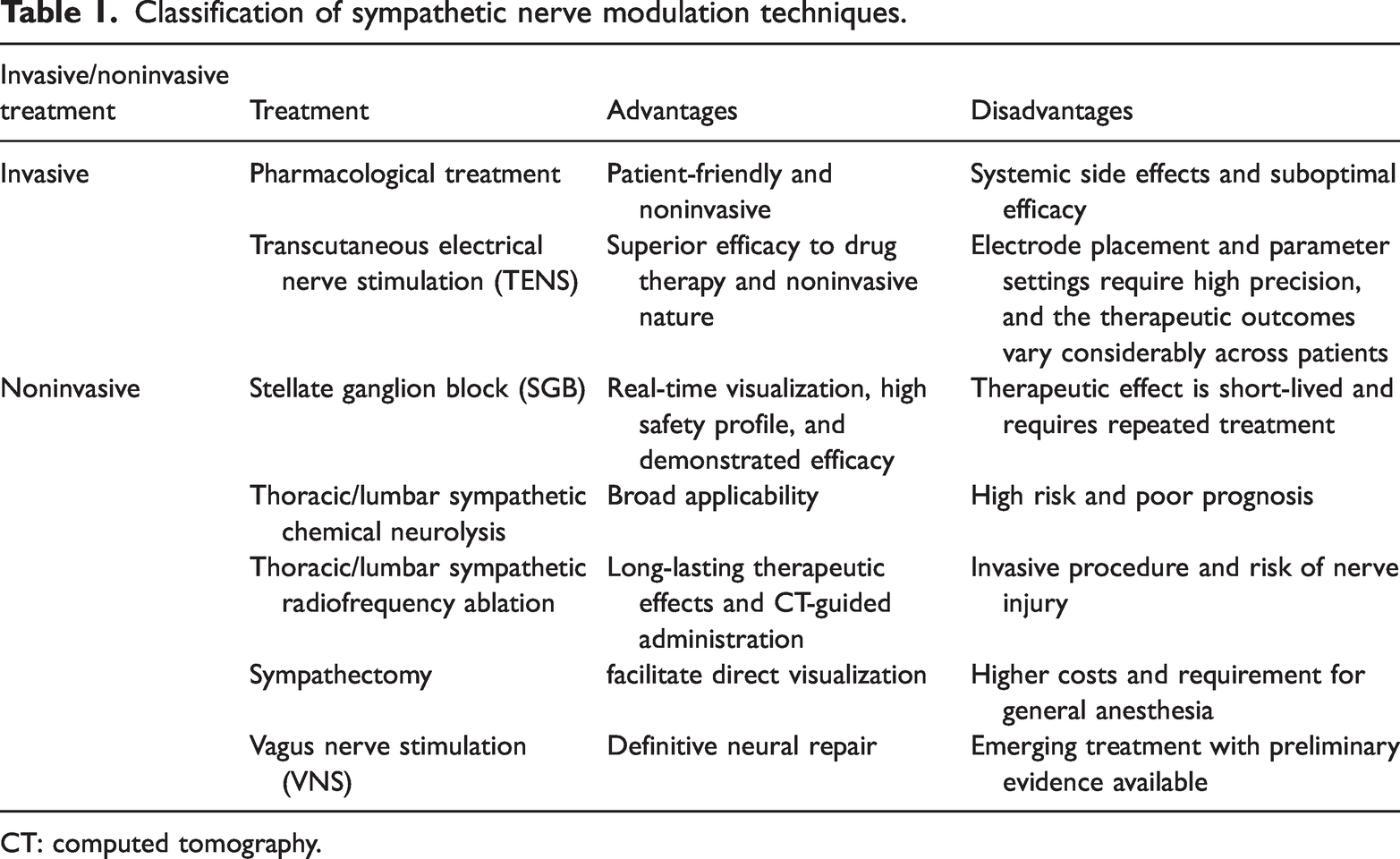
CT: computed tomography.
Pharmacological treatment
Pharmacological treatment is a widely utilized, safe, and noninvasive approach. Its fundamental pharmacological mechanism involves counteracting excessive sympathetic nerve excitation, dilating blood vessels, and enhancing blood flow, thereby alleviating symptoms associated with cold limbs. In contemporary clinical practice, α-adrenergic receptor blockers (e.g. phentolamine) and β-adrenergic receptor agonists (e.g. isoproterenol) are commonly employed. 36 Pharmacological treatment often serves as a crucial foundational and adjunctive therapeutic strategy for managing post-stroke cold limb symptoms. 37 Clinicians frequently integrate pharmacotherapy with physical rehabilitation and invasive interventions. However, it is essential to remain vigilant about the potential systemic adverse effects of pharmacological treatment. 26
Transcutaneous electrical nerve stimulation (TENS)
TENS is a noninvasive technique for sympathetic nerve modulation that minimizes the risk of infection, bleeding, and tissue damage; it is associated with minimal adverse effects and excellent patient tolerance. 38 It is markedly effective in treating or alleviating cold sensations in the limbs following a stroke, which are often accompanied with hyperalgesia and allodynia. 39 The mild-to-moderate local vasodilation induced by TENS can enhance microcirculation to a certain extent. However, it is challenging to predict the efficacy of TENS owing to factors such as electrode placement, parameter settings, and individual patient differences. Additionally, the therapeutic effects are frequently temporary; in some cases, the benefits diminish rapidly after the cessation of stimulation. In cases of severe vasospasm and coldness resulting from excessive sympathetic drive, the local vasodilatory effect of TENS may be insufficient to achieve a clinically significant increase in the temperature, reducing its therapeutic effectiveness.
SGB
The stellate ganglion is formed by the fusion of the inferior cervical ganglion and T1 ganglion and is located between the C6 and C7 vertebrae. It contains preganglionic sympathetic fibers that supply the head and neck and postganglionic sympathetic fibers, which in turn supply the upper limbs and heart. SGB is widely used in the treatment of various painful and nonpainful conditions.40–42 SGB involves the injection of a local anesthetic around the stellate ganglion and surrounding tissues, including the cervical sympathetic trunk, cervical sympathetic ganglia, and preganglionic and postganglionic nerves, to block sympathetic innervation to the head, face, neck, shoulders, upper limbs, anterior chest, and back. This helps regulate the sympathetic and autonomic nervous systems as well as the circulatory, endocrine, and immune systems while maintaining a dynamic balance in the body. The mechanism of SGB primarily involves the regulation of thalamic activity for homeostasis maintenance. Additionally, by blocking the pre- and postganglionic fibers, sympathetic functions in the distribution area are inhibited, thereby aiding the treatment of related diseases.43,44 Ultrasound-guided SGB allows real-time visualization of the procedure, enabling the operator to avoid blood vessels and nearby vital organs while monitoring the spread of the anesthetic (Figure 1). This significantly enhances the safety and efficacy of the technique. SGB has recently gained recognition from both doctors and patients, and its clinical application prospects are promising. 45 Cold sensation or abnormal sweating in the head, face, or upper limbs on the hemiplegic side can be treated with ultrasound-guided SGB. In this technique, a puncture needle is precisely inserted into the stellate ganglion, and a mixture of 1 mL of lidocaine hydrochloride, 1 mL of mecobalamin, and 5 mL of saline is repeatedly injected as needed based on the patient’s condition. The appearance of Horner syndrome indicates successful blockade. As chemical neurolysis or radiofrequency ablation of the stellate ganglion can cause long-term Horner syndrome, these treatments are contraindicated. However, pulsed radiofrequency combined with SGB yields better results. The main limitation of this technique is its short-lived effect, which necessitates repeated treatment cycles.

Real-time visualization of the procedure using ultrasound-guided SGB. SGB: Stellate ganglion block.
Thoracic/lumbar sympathetic chemical neurolysis
The lower sympathetic centers are located in the intermediate lateral nucleus of the gray matter of the spinal cord, from the T1 to L2 or L3 segments. The thoracic sympathetic chain is located in front of the rib heads, with 10–12 paravertebral ganglia on each side. The lumbar sympathetic chain is located on the anterolateral side of the lumbar vertebrae between the psoas major muscle and each side of the lumbar sympathetic ganglia. Postganglionic fibers return to the thoracolumbar spinal nerves via the gray rami and are distributed to the blood vessels, sweat glands, and pilomotor muscles in the chest wall and lower limbs. Thoracic/lumbar sympathetic chemical neurolysis and radiofrequency ablation have been widely applied in clinical practice for treating hyperhidrosis, cold limbs, Raynaud syndrome, and lower-limb arterial occlusive disease.46,47 Anhydrous ethanol is a common neurolytic agent; however, owing to its lack of control, it is associated with a risk of severe complications, such as spinal artery embolism, leading to paraplegia. During thoracic sympathetic chemical neurolysis (often at the T3 level), anhydrous ethanol may spread upward to the first rib, causing permanent Horner syndrome; saline injection into the stellate ganglion can dilute ethanol and serve as a rescue measure. During lumbar sympathetic neurolysis (usually at the L2 level), ethanol may spread upward to the L1 level and cause ejaculatory dysfunction. Moreover, ethanol diffusion can damage the sciatic and intercostal nerves. Owing to the high level of risk and poor outcomes associated with anhydrous ethanol, thoracic/lumbar sympathetic radiofrequency ablation is increasingly being used in place of chemical neurolysis in clinical practice.48–50
Thoracic/lumbar sympathetic radiofrequency ablation
Thoracoscopic sympathetic radiofrequency ablation provides a direct view of the thoracic sympathetic nerves, offering an effective treatment strategy for hyperhidrosis and other conditions. Despite being minimally invasive, thoracoscopic surgery involves endotracheal intubation and general anesthesia, and is associated with high medical costs, postoperative chest pain, and compensatory hyperhidrosis, which limit its use in patients with hyperhidrosis.51,52 Computed tomography (CT)–guided thoracoscopic sympathetic radiofrequency ablation eliminates the need for chest surgery. This technique involves the insertion of a blunt, 10-cm long 20-G needle, with a 1-cm exposed tip, in the front of the rib head under local anesthesia (Figure 2). The advantages of this technique include minimal trauma, rapid recovery, 53 and high treatment efficacy.7,30,50

CT-guided thoracoscopic sympathetic radiofrequency ablation and puncture path planning (with the needle tip positioned at the target site). CT: computed tomography.
The procedure for CT-guided thoracoscopic sympathetic radiofrequency ablation (including T4 sympathetic nerve ablation as an example) involves positioning the patient prone on the CT table, monitoring vital signs in real time, and locating the T3–T4 intervertebral space using CT. After selecting the optimal puncture plane and point, a blunt needle is advanced through the costotransverse joint to the upper edge of the T4 rib. After confirming appropriate needle positioning by testing the electrical resistance, the temperature is set to 95°C and ablation is performed for 300 s. Surgical success is indicated by a significant increase in the perfusion index and hand temperature. Furthermore, IR thermography and computed tomography angiography (CTA) performed before and 24 h following the procedure show marked differences (Figure 3).

Comparison of preoperative and postoperative infrared thermography and CTA findings. CTA: computed tomography angiography.
For lumbar sympathetic radiofrequency ablation, the patient is placed in the prone position, and the L3 vertebrae are scanned. An appropriate needle path, angle, and depth are selected to avoid nerves, blood vessels, and organs (Figure 4). Once confirmed via CT, the needle tip is advanced to the target, and ablation is initiated at 95°C for 300 s. Successful outcomes are indicated by significant improvements in postprocedural IR thermography and CTA (Figure 5). Notably, performing this technique on the right side is more challenging and is associated with a lower success rate during lumbar ablation. 54
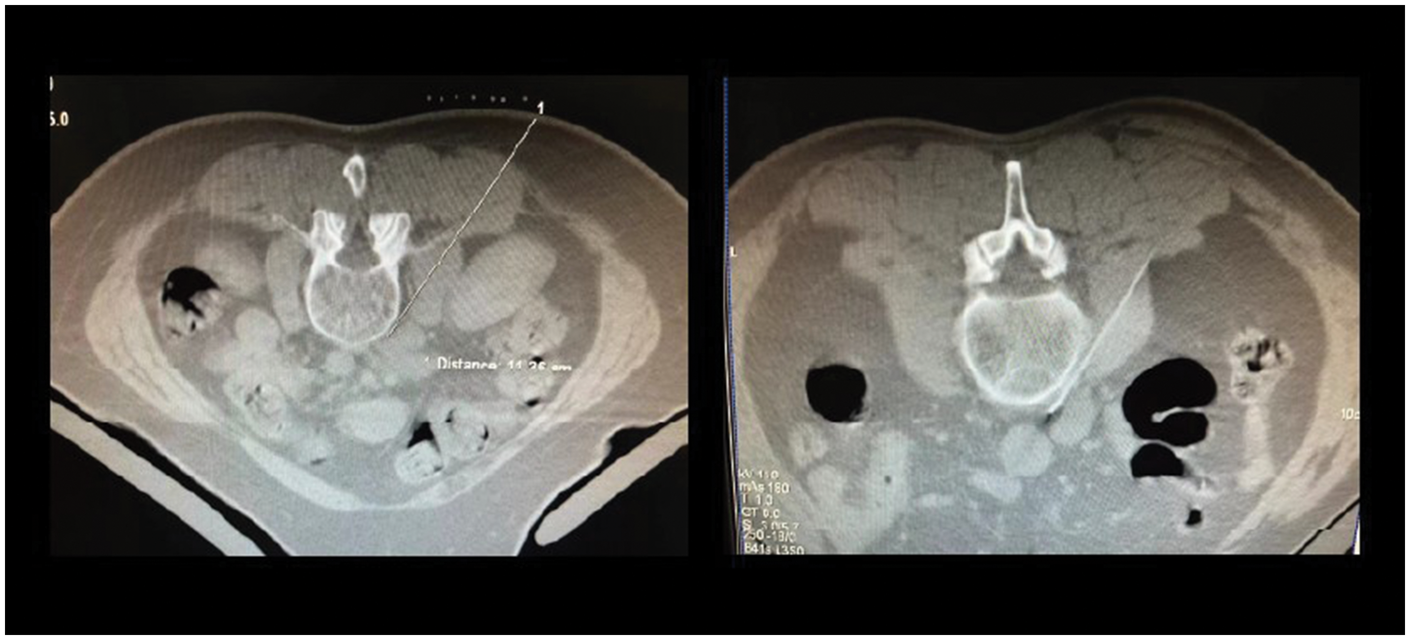
CT-guided puncture path planning for lumbar sympathetic radiofrequency ablation and lumbar sympathetic radiofrequency (with the needle tip positioned at the target site). CT: computed tomography.
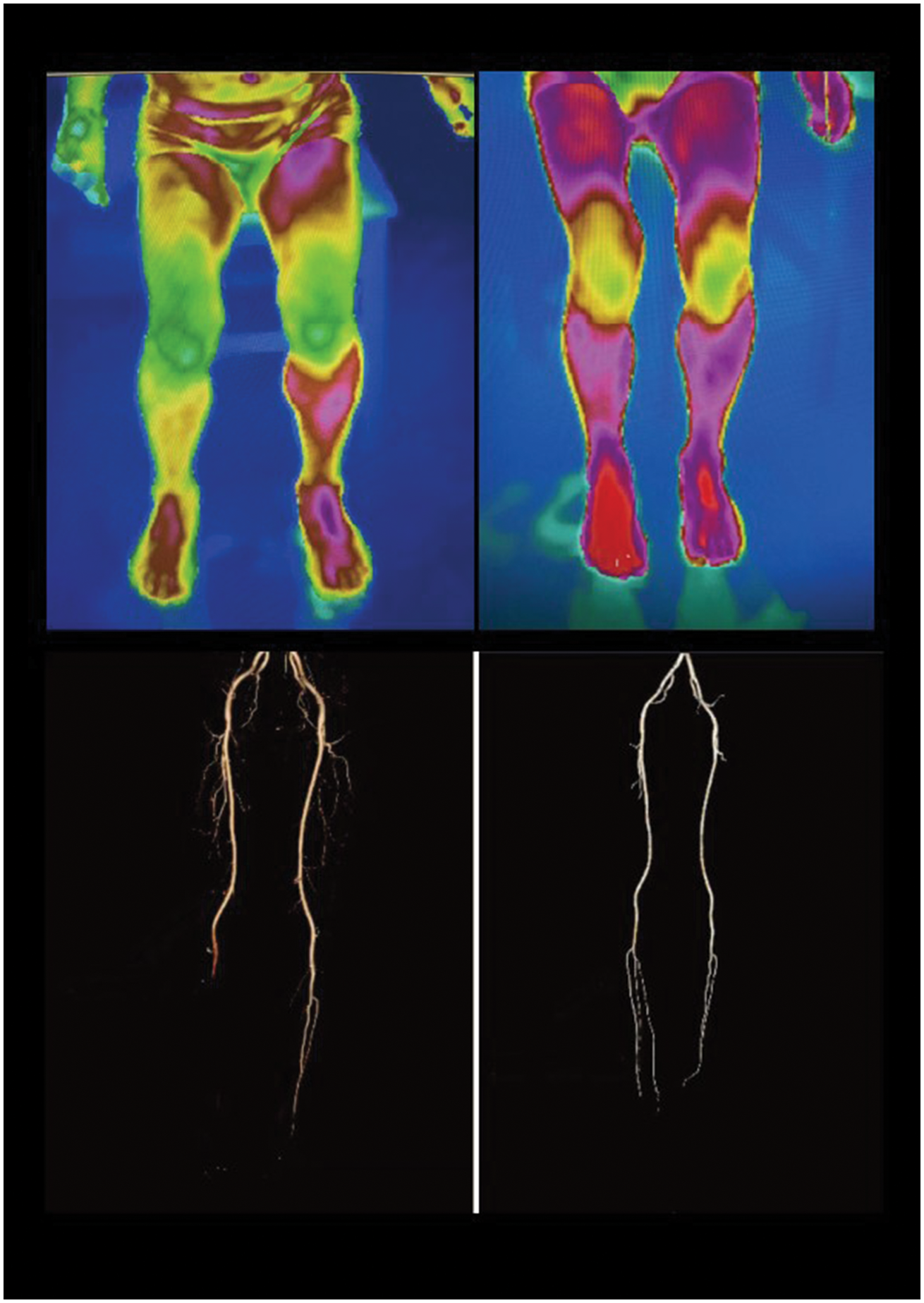
Comparison of preoperative and postoperative infrared thermography and CTA findings.
Following CT-guided thoracic/lumbar sympathetic radiofrequency ablation, stroke patients with cold limbs experience warmth in the affected limb, with significant changes observed on IR thermography and CTA. Blunt needles can be used for puncture (Figure 6), followed by a “withdrawal method” during insertion, which significantly reduces the risk of complications such as pneumothorax, hematoma, and nerve damage.

Technique demonstrating the use of a blunt needle and the “withdrawal method” during insertion.
VNS
According to several studies, VNS may be a promising approach for treating the hyperacute phase of stroke.55,56 VNS does not directly target the peripheral sympathetic nerves; instead, it stimulates the afferent fibers of the vagus nerve, activating brainstem nuclei such as those of the solitary tract and thalamocortical circuits. This activation promotes a significant release of norepinephrine, acetylcholine, and brain-derived neurotrophic factor in the brain, which modulate neural activity. When combined with rehabilitation training, these neurochemicals can markedly enhance and accelerate the remodeling of brain neural networks, a process known as neuroplasticity. This approach holds the potential to fundamentally repair the autonomic regulatory centers damaged by stroke, such as the insula and anterior cingulate cortex, thereby restoring their normal inhibitory control over sympathetic nerve output. Consequently, it may simultaneously improve vascular constriction (cold sensation) and abnormal sweat gland secretion (sweating). Additionally, VNS modulates the immune system via the cholinergic anti-inflammatory pathway, reducing the release of proinflammatory cytokines and exhibiting neuroimmunomodulatory effects. However, to date, no clinical research has directly demonstrated the exact efficacy of VNS on post-stroke autonomic symptoms, such as cold sensation and abnormal sweating. Moreover, the efficacy of VNS is highly contingent upon its combination with specific rehabilitation training, which necessitates high patient compliance and a relatively complex treatment process. Owing to variations in the location and size of stroke lesions as well as individual neural plasticity, there is considerable variability in the therapeutic response to VNS among different patients. Therefore, although VNS has demonstrated potential for modulating vagus nerve activity, it is still in an exploratory phase and warrants further research. 57
Discussion
Cold sensation and abnormal sweating in hemiplegic limbs after stroke can be effectively treated using sympathetic nerve regulation techniques. Mechanical, chemical, or thermal ablation of the sympathetic nerve decrease the sympathetic tone, leading to vasodilation, reduced peripheral vascular resistance, increased collateral and peripheral circulation, and enhanced blood perfusion to the skin and muscles. These events increase the temperature of the limbs. Techniques such as SGB, thoracic/lumbar sympathectomy, chemical neurolysis, and radiofrequency sympathectomy have demonstrated notable clinical success and are therefore widely used in the treatment of sympathetic nervous system-related disorders.
For stroke patients with cold limbs, if coldness or sweating occurs in the head and face, an ultrasound-guided SGB followed by CT-guided T3 sympathetic radiofrequency ablation is recommended. In case of coldness or sweating in the upper limbs, CT-guided T4 sympathetic radiofrequency ablation is recommended. For coldness or sweating in the lower limbs, CT-guided L3 sympathetic radiofrequency ablation is advisable.
Footnotes
Acknowledgments
I would like to express my deepest gratitude to all those who have supported me during the research and preparation of this literature review. First, I express my sincere thanks to my supervisor, Professor Yong Fei, for his invaluable guidance, constructive criticism, and continuous encouragement throughout this process. His insightful comments and suggestions were instrumental in shaping the direction and depth of this work. I am also grateful to the faculty and my fellow colleagues at the Zhejiang Chinese Medical University and Jiaxing University Affiliated Hospital for their stimulating discussions and supportive companionship. My special thanks to the authors of all the cited publications. Their pioneering research provided the essential foundation upon which this review was built.
Author contributions
Mingqing Yu contributed the most to the literature review, including reference screening, and drafted the initial manuscript. Ping Xu contributed to the literature review and was responsible for writing the Conclusion section. Hanrui Fan participated in the literature search and provided the relevant figures. As the corresponding author, Yong Fei conceived the study framework, oversaw the entire research process, and is responsible for manuscript correspondence.
Data availability statement
This manuscript is a review article and does not report original data. All data discussed in this review are sourced from the published literature cited within.
Declaration of conflicting interests
The authors declare no conflict of interest.
Ethical compliance,human and animal rights,and informed consent
All reported studies/experiments involving human or animal participants have been previously published and complied with all applicable ethical standards including the Helsinki declaration and its amendments.
Funding
No funding or sponsorship was received for this study or publication of this article. The Rapid Service Fee was funded by the authors.
