Abstract
Background
Isolated cardiac sarcoidosis is a distinct entity of sarcoidosis characterized by disease confined exclusively to the heart, without involvement of other organs. While cardiac sarcoidosis can lead to severe complications such as arrhythmias and heart failure, isolated cardiac sarcoidosis remains understudied, and no systematic review has previously addressed this condition.
Key findings: A systematic review of the literature from 1978 to 2025 identified 14 relevant studies, primarily case series. Isolated cardiac sarcoidosis affects both sexes equally, with most patients aged 55–65 years. Compared with multi-organ cardiac sarcoidosis, isolated cardiac sarcoidosis is associated with lower left ventricular ejection fraction and poorer survival. Variability in diagnostic criteria and potential selection bias were key limitations.
Conclusion
Isolated cardiac sarcoidosis is a distinct and severe clinical entity with limited available data. Its poorer prognosis underscores the need for specific diagnostic and management guidelines. Future multicenter studies are essential to improve understanding and care for patients with this condition.
Objective
Sarcoidosis is an inflammatory disease characterized by the formation of non-caseating granulomas in various organs, most commonly the lungs, heart, eyes, muscles, skin, and brain. Although pulmonary involvement is the most common, sarcoidosis can affect nearly any organ. 1 Clinically, sarcoidosis is often asymptomatic or may present with general symptoms such as fever, fatigue, and weight loss. In more severe cases, symptoms depend on the affected organ. The disease is driven by the accumulation of activated T cells and the transformation of epithelial cells into non-necrotizing granulomas, likely in response to unidentified antigens. 2
Isolated cardiac sarcoidosis (ICS) is a distinct and complex condition that has garnered increasing attention in recent years. While numerous studies have investigated various aspects of cardiac sarcoidosis (CS), important questions remain regarding the epidemiology, diagnosis, treatment, and prognosis of ICS in particular. Several studies have explored its prevalence, with differing results across populations. For instance, Tavora et al. conducted an autopsy study from 1995 to 2008 and found that although 73% of the examined CS cases had no known extracardiac involvement, only 29% were ultimately confirmed as true ICS cases based on clinical assessments. 3 Similarly, in Finland, Kandolin et al. initially identified 71 of 110 patients as having ICS based on clinical findings and X-rays; however, further evaluation using fluorodeoxyglucose positron emission tomography/computed tomography (FDG-PET/CT) imaging revealed that 30 of these patients had extracardiac involvement. 4 A study in Japan also suggested that approximately 27% of patients with CS could be classified as having ICS, underscoring the variability in diagnostic approaches and population differences. 5
Regarding prognosis and disease severity, some evidence indicates that ICS may present with more advanced cardiac dysfunction compared with other forms of CS. For example, one study reported that 69% of ICS patients showed signs of left ventricular (LV) systolic dysfunction, whereas this figure was 41% among all CS patients. Tavora et al. also observed a higher association between ICS and mortality. 3 Nonetheless, these findings are based on relatively small sample sizes, and more extensive data are needed to draw firm conclusions. Early diagnosis and treatment in non-isolated cases might also contribute to improved outcomes, further complicating the interpretation of prognosis in ICS. In terms of diagnosis, while endomyocardial biopsy (EMB) can confirm the presence of granulomas with high specificity, its sensitivity is limited. Kandolin et al. found that only 35% of ICS patients could be diagnosed through biopsy alone, although combining it with cardiac magnetic resonance imaging (CMR) significantly improved diagnostic yield. 6
Based on these findings, several key questions remain unanswered regarding the epidemiology, diagnosis, treatment, and prognosis of ICS. Addressing these gaps is essential to deepen our understanding of the disease and to develop more precise diagnostic and therapeutic strategies. Accordingly, the present study was designed to systematically review the available evidence on ICS and compare it with other forms of CS.
Methods
This systematic review was conducted in accordance with the PRISMA 2020 guidelines, with registration ID IR.IUMS.FMD.REC.1402.185, approved by the Research Ethics Committee of Iran University of Medical Sciences, Tehran, Iran. 7 The objective was to summarize evidence on the prevalence, clinical features, diagnostic methods, treatment strategies, prognosis, and risk factors in patients with ICS.
Search strategy
A comprehensive literature search was performed in PubMed, Scopus, and ISI Web of Science from 1978 to May 2025, using predefined keywords and Medical Subject Headings (MeSH) terms related to “isolated cardiac sarcoidosis,” “prevalence,” “diagnosis,” “treatment,” “clinical symptoms,” and “prognosis.” Search strategies were adapted for each database. All retrieved references were managed using EndNote for deduplication and organization.
Article screening, inclusion, and exclusion
The initial search retrieved 404 records (PubMed, 85; ISI Web of Science, 135; Scopus, 167). After removing duplicates, 215 records remained for screening. A total of 171 articles were selected for full-text review. Of these, 157 articles were excluded. Exclusion criteria included reviews, case reports, conference abstracts, and studies focusing on systemic sarcoidosis without isolated cardiac involvement.
Ultimately, 14 articles met all inclusion criteria and were included in the qualitative synthesis (Supplementary Material 1) (Table 1).5,6,8–19
Comprehensive summary of reviewed articles.
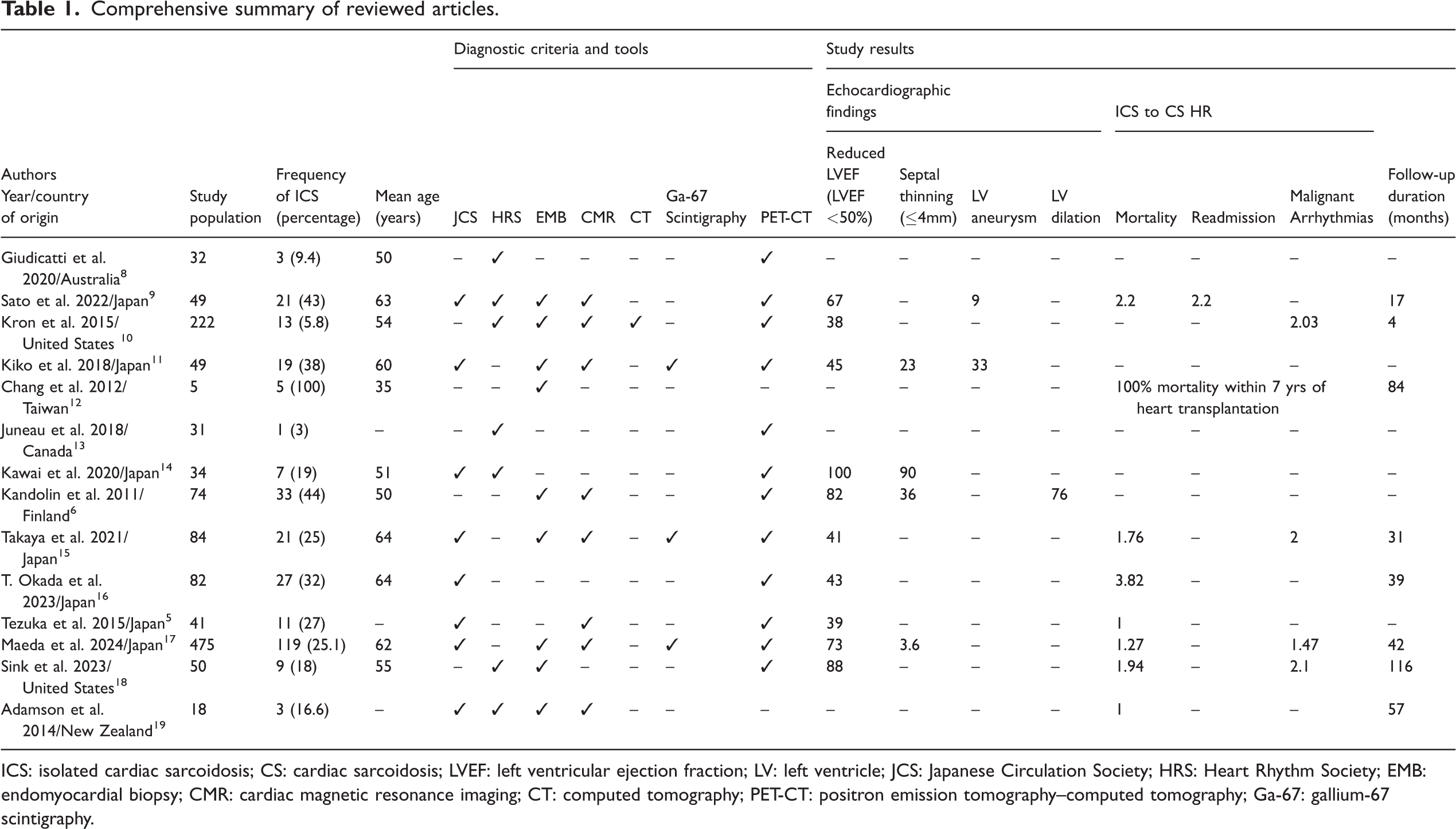
ICS: isolated cardiac sarcoidosis; CS: cardiac sarcoidosis; LVEF: left ventricular ejection fraction; LV: left ventricle; JCS: Japanese Circulation Society; HRS: Heart Rhythm Society; EMB: endomyocardial biopsy; CMR: cardiac magnetic resonance imaging; CT: computed tomography; PET-CT: positron emission tomography–computed tomography; Ga-67: gallium-67 scintigraphy.
Study selection and data extraction
Two independent reviewers (a cardiologist and an epidemiologist) screened titles, abstracts, and full texts for eligibility. Discrepancies were resolved by consensus. Data were extracted using a standardized form capturing study characteristics, diagnostic criteria, clinical findings, management approaches, and outcomes.
Risk of bias assessment
The Newcastle–Ottawa Scale was used to assess the quality of observational studies. Cross-sectional studies were evaluated using version 12.4.65 of the AXIS tool. Each study was rated as having low, moderate, or high risk of bias.
Epidemiology and demographics
Sarcoidosis demonstrates marked geographic and ethnic variation, with the highest incidence in African Americans and the lowest in Asians, typically presenting between ages 40 and 55 years of age, with earlier onset in men. Its prevalence, severity, and outcomes are influenced by racial, geographic, and socioeconomic factors, and the disease is associated with increased morbidity, including a 70% higher 5-year mortality risk and elevated rates of cardiovascular disease, autoimmune conditions, and treatment-related infections.20,21 Cardiac involvement is a major manifestation, affecting approximately 30% of patients in the United States and over 50% in Japan. A distinct subtype, ICS, is defined by granulomatous inflammation confined to the myocardium without extracardiac disease, requiring histological confirmation. The prevalence of ICS among patients with CS varied considerably across studies, ranging from 3% to 60%. When pooling data from all included cohorts, the overall non-weighted prevalence of ICS was 23%. This summary estimate highlights that while ICS constitutes a substantial minority of CS cases, its reported frequency differs markedly depending on diagnostic criteria, study design, and geographic population. These findings underscore both the importance of rigorous diagnostic evaluation and the heterogeneity of published literature (Figure 1).4,6,22

Epidemiological relationship of sarcoidosis, CS, and ICS. The outer circle represents all sarcoidosis cases (100%), the middle circle represents CS (approximately 30% in the United States and >50% in Japan), and the inner circle represents ICS. Across all included studies, the overall non-weighted pooled prevalence of ICS among CS patients was approximately 23%, although reported estimates varied widely from 3% to 60% depending on study design, diagnostic criteria, and population. CS: cardiac sarcoidosis; ICS: isolated cardiac sarcoidosis.
Across the included studies, sample sizes ranged from 5 to 475 patients, comprising both CS and ICS cases. In most reports, CS was more prevalent than ICS (approximately 60%), although a minority of studies demonstrated an ICS predominance. The proportion of ICS cases among all patients ranged from 3% to 100%; excluding two outliers, this range narrowed to 5.8%–60.8%. Analysis of sex distribution from 14 studies indicated a male predominance in 9 studies, with male proportions between 56.0% and 70.4%. Conversely, three studies reported a higher prevalence among females (57.9%–78.0%). Overall, ICS appeared slightly more common in men, though the difference was modest. Age distribution was relatively consistent across studies, with mean ages ranging from 50 to 64 years, except for one study limited to five post-heart transplant patients. 12 These findings suggest that ICS is typically diagnosed in middle-aged individuals (50–60 years).
Pathophysiology and etiology
Sarcoidosis arises from a complex interplay of genetic and environmental factors. Genetic susceptibility is strongly associated with specific human leukocyte antigen (HLA) class II alleles, particularly HLA-DR3, and epidemiological studies indicate that a family history increases the risk of disease by two- to fourfold. 23 Environmental exposures also play an important role, with increased risk observed among farmers, individuals exposed to pesticides or insecticides, those in contact with silica particles, and people exposed to smoke, chemicals, or certain bacterial infections. 24 Immunologically, sarcoidosis is considered a type IV hypersensitivity disorder, characterized by the formation of non-caseating granulomas. This process is driven by hyperactivation of Th1 and Th17 helper T cells, which secrete excessive interferon–gamma and recruit macrophages and antigen-presenting cells. Overexpression of transcription factors such as T-bet and signal transducer and activator of transcription 1 (STAT1) further amplifies T-cell activity, while dendritic cell-mediated antigen presentation through Toll-like receptors (TLRs) sustains chronic inflammation (Figure 2(a)).21,25 Within granulomas, a high frequency of TRAV2.3+TRBV22+CD4+ T cells has been identified, typically activated by HLA-DR3-restricted antigen-presenting cells. Molecular analyses suggest that these antigens may be self-derived, such as vimentin or adenosine triphosphate (ATP) synthase fragments, supporting a possible autoimmune component.21,26 Disease course varies, with many patients achieving spontaneous remission, while others progress to chronic disease. Current models propose that outcomes are influenced by the balance between immune regulatory mechanisms and persistent antigen availability (Figure 2(b)).21,27

(a) Cellular and molecular mechanisms in sarcoidosis and (b) disease progression or remission in sarcoidosis.
Clinical and paraclinical findings
A retrospective cohort study in Japan of 55 patients with CS and 27 with ICS reported a higher prevalence of left ventricular ejection fraction (LVEF) below 50% in ICS compared with CS (74.1% vs. 43.6%). 16
In a Finnish multicenter study of 71 patients with clinically diagnosed ICS, the most common initial presentation was symptomatic atrioventricular (AV) block (48%), followed by ventricular tachycardia or fibrillation (38%), with abnormal electrocardiograms (ECGs) observed in 88% of cases. In this cohort, analyzed by Kandolin et al., only 14% of all CS patients had a normal ECG, with the most frequent abnormality being second- or third-degree AV block (45%), followed by right bundle-branch block (37%) and left bundle-branch block (21%). These conduction disturbances tended to be more common in ICS than in patients with known extracardiac sarcoidosis, although the differences were not statistically significant. Echocardiography in the Finnish study revealed LV dilatation in 42% of patients and septal thickening or thinning with LV dysfunction in 70%. Among patients undergoing fluorine-18 FDG PET (18F-FDG PET), focal myocardial FDG uptake and FDG uptake in mediastinal lymph nodes were the most frequent findings (71%). Together, these data underscore the high prevalence of conduction system disease and structural myocardial involvement in CS, supporting the use of ECG, echocardiography, and PET imaging in diagnosis. 4
A recent large Japanese multicenter cohort study clearly differentiated clinical findings at diagnosis and post-treatment in 356 CS and 119 ICS patients. Abnormal ECGs were present in 27.1% of CS and 23.2% of ICS patients. Heart failure was more common in ICS (31.5% vs. 19.1%). Serum angiotensin-converting enzyme (ACE) and soluble interleukin-2 receptor (sIL-2R) levels were significantly higher in CS, whereas ICS patients had a greater prevalence of atrial fibrillation, implantable defibrillator use, and lower LVEF. 17
In another Japanese study involving 21 ICS patients, compared with CS and other cardiac conditions, ICS patients exhibited higher rates of arrhythmia, particularly sustained ventricular tachycardia. Both ICS and CS groups had more frequent LV geometric abnormalities and late gadolinium enhancement (LGE) on CMR than other cardiac patients. Regional wall motion abnormalities (RWMA) were more common in ICS, while LVEF was higher in CS. FDG-PET abnormalities combined with LGE were detected in all ICS and 95% of CS patients. Echocardiography showed greater LV end-diastolic and end-systolic dimensions and volumes, but lower LVEF, in ICS compared with CS. 9
A Finnish series reported that 91% of ICS patients were hospitalized due to high-grade AV block, heart failure, or ventricular tachyarrhythmia. All but two patients had echocardiographic abnormalities, and nine had extracardiac sarcoidosis identified on physical examination or chest imaging. 6
Data from the Taiwanese heart transplant registry (1987–2011) identified 5 ICS cases among 411 transplants. All patients had dilated cardiomyopathy with patent coronary arteries. The most common clinical features were AV block, ventricular arrhythmias, and ST-segment elevation with severely reduced LVEF (17%–23%). Heart transplantation was effective in end-stage disease, and post-transplant survival was slightly, but not significantly, better than in other transplant recipients. 12
In a Japanese cohort study of 55 CS and 27 ICS patients, ACE, lysozyme, and sIL-2R levels were significantly higher in CS than in ICS. 16 Another study found elevated ACE, serum lysozyme, and calcium in at least 30% more CS patients. 4 One small study of 19 ICS cases showed that ACE, sIL-2R, B-type natriuretic peptide (BNP), and troponin were higher in CS, with B-type natriuretic peptide (cTnI) predicting fatal arrhythmia in sarcoidosis but not in ICS. 11
Echocardiographic findings in ICS generally suggest more severe disease outcomes, with most differences being statistically significant (P < 0.05). The most frequently reported features include basal interventricular septal thinning and septal thinning. Across multiple studies, LV systolic dysfunction (EF <50%), RWMA, ventricular aneurysm, and LV dilation have also been observed, although the frequency varies between cohorts. Some studies reported additional findings, such as LV wall thickening, but these were less consistently reproduced in larger ICS populations. 28
Reported prevalences vary: basal interventricular septal thinning occurred in 38%–95% of patients, LV dysfunction (LVEF <50%) in 43%–82%, and ventricular aneurysm in up to 47%. RWMAs were common, reaching 90%–95% in some series. Mean LVEF values across different studies ranged from approximately 38% to 52%, indicating a substantial burden of systolic dysfunction. LV dilation was found in up to 76% of cases, and interventricular septal abnormalities (thinning or thickening) were observed in about two-thirds of patients in some cohorts. Overall, echocardiography consistently highlights structural and functional LV involvement in ICS, with septal abnormalities being a hallmark finding. Despite low sensitivity, echocardiography remains useful for screening and follow-up due to its accessibility and low cost.4–6,9–11,14–16,29
Histopathological studies have suggested a characteristic pattern of myocardial involvement in CS, which may also apply to ICS. A systematic review of 33 autopsy and cardiac transplant studies in patients with CS-related sudden cardiac death found that LV involvement was most often subepicardial (98%) and midmyocardial (65.3%), with subendocardial and transmural involvement present in 53.1% and 63.3% of cases, respectively. Multifocal lesions were significantly more frequent than unifocal ones, and notable distribution patterns were observed across LV segments and levels. 30
No single definitive diagnostic test exists for ICS; however, updated criteria from the Japanese Circulation Society (JCS) now provide specific guidelines for both CS and ICS. Between 2011 and 2025, studies have increasingly relied on advanced imaging—particularly 18F-FDG PET-CT and CMR—for diagnosis. 18F-FDG PET-CT is often considered the gold standard in recent studies, as echocardiography and ECG can yield false-negative results in patients with true disease.31,32 CMR offers a high negative predictive value, making it valuable for excluding CS when results are negative. 33 FDG-PET can also identify metabolically active inflammatory lesions. The most effective diagnostic approach combines advanced imaging with complementary tests to rule out other cardiac conditions, 34 ideally conducted by a multidisciplinary team with expertise in systemic sarcoidosis, heart failure, electrophysiology, advanced cardiac imaging, cardiovascular genetics, and cardiac pathology. 31
Diagnostic criteria and challenges
ICS is a diagnostically challenging condition, characterized by granulomatous inflammation confined to the myocardium without systemic involvement. Patients often present with unexplained ventricular arrhythmias, conduction abnormalities, or LV aneurysm in the absence of coronary artery disease or myocarditis. 35
Definitive diagnosis of CS requires histological confirmation of myocardial granulomas and exclusion of systemic involvement. However, EMB, while highly specific, has limited sensitivity due to the patchy distribution of lesions and sampling error, driving ongoing efforts to refine diagnostic approaches. 36 The concept of ICS remains controversial, as studies indicate that many patients with imaging-defined ICS demonstrate subclinical extracardiac disease on further evaluation. Overdiagnosis without biopsy confirmation risks unnecessary treatment or missed alternative diagnoses. 37 Moreover, growing evidence suggests that a significant proportion of patients harbor underlying cardiomyopathy-associated genetic variants, even when an inflammatory trigger is evident. In this context, genetic testing has led to the reclassification of some cases previously labeled as ICS into genetic cardiomyopathies, with important implications for both diagnosis and management. 38
Serum biomarkers such as troponin T, lysozyme, and ACE have been investigated, but none demonstrate sufficient sensitivity or specificity for routine clinical use. 39
Noninvasive imaging is central to evaluation. Echocardiography can reveal segmental wall thinning with high specificity but low sensitivity. CMR and FDG-PET/CT improve detection of structural changes and active inflammation. Despite these advances, careful clinical assessment and exclusion of alternative diagnoses remain essential, as findings suggest that FDG-PET and CMR are useful tools for the noninvasive evaluation of CS, providing high sensitivity but limited specificity. 40
In a cohort, the majority of patients referred for imaging did not ultimately have CS, underscoring the low diagnostic yield and the nonspecific nature of PET/CT findings in this setting. Advanced imaging is further limited by its inability to reliably distinguish CS from other forms of inflammatory or genetic cardiomyopathy, many of which can present with overlapping clinical and imaging features. Finally, the diagnosis of CS remains dependent on integration with clinical criteria, emphasizing that PET/CT cannot serve as a standalone tool for confirming or excluding ICS. These limitations highlight the importance of comprehensive, multidisciplinary evaluation and the incorporation of genetic testing when assessing patients with suspected CS. 41
Diagnostic frameworks differ between regions. The Heart Rhythm Society (HRS) 2014 consensus primarily addresses patients with histologically proven extracardiac sarcoidosis, requiring either myocardial biopsy or extracardiac biopsy combined with specific cardiac manifestations. HRS does not provide a pathway for diagnosing ICS without extracardiac involvement, and imaging findings such as FDG-PET or LGE are supportive but not definitive. In contrast, the JCS 2016 guideline explicitly includes ICS, allowing a clinical diagnosis when cardiac FDG-PET uptake is positive alongside at least three other major cardiac criteria—such as conduction abnormalities, structural changes, LV dysfunction, or LGE—after exclusion of extracardiac sarcoidosis through clinical and imaging evaluation. This reflects the more permissive, imaging-integrated approach of JCS 2016 compared with the histology-dependent HRS 2014 framework (Figure 3).42–46

(a–b) Two-dimensional and M-mode echocardiography demonstrating basal septal thinning, which subsequently progresses to LV dilation and dysfunction (c–d), accompanied by secondary mitral Continued.regurgitation (not shown). 18F-FDG PET-CT MIP and axial fused PET-CT images showing (e) focal-on-diffuse hypermetabolic activity within the LV myocardium, consistent with CS, and (f) complete resolution of myocardial metabolic activity following immunomodulatory therapy. (g–i) CMR demonstrating subendocardial LGE in the hypertrophied septum and lateral LV wall, consistent with active myocardial inflammation, edema, and focal fibrosis (four-chamber view, red arrows; three-chamber view, green arrows). (h–j) Additional foci of increased signal in the basal septum, basal mid-anterior, basal inferior, inferolateral, and subendocardial septal walls indicate sarcoid-related inflammation (two-chamber view, yellow arrows; short-axis view, blue arrows). LV: left ventricle; 18F-FDG PET-CT: fluorine-18 fluorodeoxyglucose positron emission tomography–computed tomography; CS: cardiac sarcoidosis; MIP: maximum intensity projection; CMR: cardiac magnetic resonance imaging; LGE: late gadolinium enhancement. Graphical abstract. Graphical abstract summarizing epidemiology, clinical features, diagnosis, treatment, prognosis, and research needs in isolated cardiac sarcoidosis.
An important historical development in the diagnosis of ICS has been the shift from biopsy-dependent frameworks to imaging-integrated criteria. While the HRS 2014 consensus relied on histologic confirmation, the diagnostic yield of EMB in real-world practice has been low, making definitive diagnosis extremely difficult. To address this limitation, the JCS published updated guidelines in 2019 (formulated in 2016), which for the first time permitted a clinical diagnosis of ICS without histologic proof, provided that FDG-PET was positive and at least three major cardiac criteria were fulfilled. Even prior to this, Sohn et al. (2018) had proposed a scoring system that integrated PET findings (1–2 points depending on extent) with other major features, requiring ≥4 points for diagnosis. While both approaches share similarities, the JCS criteria are stricter in mandating PET positivity and allow LV systolic dysfunction as a major criterion, a point that remains debated due to its nonspecific nature. These developments underscore the evolving nature of diagnostic strategies for ICS and the ongoing need for consensus to balance sensitivity and specificity.42,47
According to the American Heart Association (AHA) scientific statement on 2023, the diagnosis of ICS is based primarily on the JCS 2016 criteria, as other international guidelines, such as HRS, require extracardiac involvement and therefore do not apply to ICS. Histological confirmation can be made when EMB or surgical myocardial tissue demonstrates non-caseating granulomas. When biopsy is negative or not performed, a clinical diagnosis of ICS can be established if there is abnormal tracer uptake on gallium-67 (67Ga) scintigraphy or FDG-PET in the heart, together with at least three major criteria: high-degree AV block or fatal ventricular arrhythmia, basal septal thinning or abnormal ventricular wall anatomy, LV systolic dysfunction, and LGE on CMR. In addition, several prerequisites must be fulfilled: there should be no clinical evidence of sarcoidosis in extracardiac organs, no abnormal tracer uptake outside the heart on PET or gallium imaging, and no chest CT findings consistent with pulmonary sarcoidosis, such as lymphatic tract shadows or hilar/mediastinal lymphadenopathy greater than 10 mm. 48
Table 2 summarizes the major societal guidelines (HRS 2014, JCS 2016, AHA 2024) focusing on the diagnostic approach to CS in the absence of extracardiac sarcoidosis. Importantly, while HRS 2014 requires myocardial biopsy demonstrating granulomas for diagnosis, JCS 2016 provides an imaging-integrated pathway (PET positivity plus ≥3 major criteria), and the AHA 2024 Scientific Statement recommends a multimodality approach combining advanced imaging, cardiac manifestations, and exclusion of mimics. The AHA also emphasizes biopsy whenever possible, along with three-generation pedigree analysis and genetic testing. If biopsy is negative, application of the JCS 2016 criteria is advised (Table 2).
Diagnostic pathways for CS in the absence of extracardiac sarcoidosis.

CS: cardiac sarcoidosis; ICS: isolated cardiac sarcoidosis; EMB: endomyocardial biopsy; PET: positron emission tomography; CMR: cardiac magnetic resonance; LV: left ventricle; RV: right ventricle; VT: ventricular tachycardia; AV: atrioventricular; LGE: late gadolinium enhancement; JCS: Japanese Circulation Society; HRS: Heart Rhythm Society; AHA: American Heart Association.
Treatment and management
Management of ICS aims to suppress granulomatous inflammation, preserve cardiac function, and prevent adverse outcomes. Systemic corticosteroids are the first-line therapy, typically initiated with oral prednisolone 0.5–0.75 mg/kg/day for 4 weeks, followed by gradual tapering according to clinical response and disease severity. 49 In prolonged treatment, steroid-sparing immunosuppressants such as methotrexate, azathioprine, mycophenolate mofetil, or cyclosporine may be used to minimize corticosteroid-related toxicity. 50 Large cohort studies have reported azathioprine as the most frequently prescribed adjunct, with other agents used in selected patients. 4
Cardiac complications require targeted management. Device-based therapies, including implantable cardioverter-defibrillators (ICDs), pacemakers, or cardiac resynchronization therapy with defibrillator (CRTD), are indicated for high-risk patients with sustained ventricular tachycardia, advanced conduction disease, or reduced LVEF. 35 Observational data suggest CRTD use is more common in ICS than in systemic CS. 18 However, the indication for ICD implantation in ICS remains controversial. Recent reviews demonstrate that while patients meeting class I guideline recommendations (such as those with reduced LVEF or sustained ventricular arrhythmias) have a higher incidence of appropriate ICD therapy, even patients with preserved LVEF and clinically apparent CS remain at significant risk of ventricular arrhythmia and sudden cardiac death. This raises concern that current recommendations may underestimate risk in certain subgroups, suggesting a potential need for revision of guideline thresholds. Moreover, predictors of ICD benefit extend beyond LVEF alone, with cardiac imaging findings, ventricular characteristics, and demographic factors also influencing outcomes. At the same time, adverse events such as inappropriate shocks, device complications, and the relatively younger age of CS patients compared with other ICD cohorts highlight the risks of device therapy. Taken together, these findings underscore the importance of individualized decision-making, integrating arrhythmic risk, imaging markers, comorbidities, and patient preferences when considering ICD implantation in ICS. In refractory or advanced cases, ventricular tachycardia ablation, orthotopic heart transplantation, or durable LV assist devices may be necessary. Due to the rarity of ICS and the absence of large randomized trials, treatment strategies remain largely guided by expert consensus and observational data. Optimal care requires a multidisciplinary approach involving cardiology, electrophysiology, and rheumatology specialists, with therapy tailored to disease activity, cardiac function, and patient tolerance.49–52
Prognosis and outcomes
Evidence on the prognosis of ICS comes mainly from retrospective cohorts, with follow-up ranging from 3 to 116 months, limiting direct comparisons. A large multicenter national study from Finland, using multivariable Cox regression to adjust for confounders, found worse outcomes in ICS compared with CS. 4
A prospective study of 59 untreated CS patients (approximately 40% ICS) showed that ventricular arrhythmia increased the risk of major adverse cardiac events (MACE) 4.75-fold, and right bundle-branch block (RBBB) was another adverse prognostic factor. Due to small numbers, ICS-specific analysis was not feasible. 53
In a Japanese cohort (median follow-up 31 months), 76% of ICS patients versus 38% of CS patients experienced malignant ventricular arrhythmia. ICS, older age, and New York Heart Association (NYHA) class III–IV independently predicted adverse events, with ICS patients having a 2.83-fold higher risk of death or hospitalization. Over 46 months, approximately 50% of ICS patients versus 22% of CS patients experienced death, hospitalization, or heart failure. 15
Heart transplantation data from Taiwan (1987–2011) identified 5 ICS cases among 411 recipients. All had dilated cardiomyopathy, were approximately 10 years younger than other recipients, and survived up to 8 years post-transplant without recurrence or graft failure, suggesting orthotopic heart transplant is effective in end-stage ICS. Inflammatory markers, including ACE and sIL-2R, were higher in CS than ICS, but their prognostic role in ICS remains unclear.11,12
Overall, ICS consistently demonstrates a poorer prognosis than CS across studies, with higher rates of mortality, hospitalization, and ventricular arrhythmia. While statistical significance was sometimes lacking due to small sample sizes, the direction of effect was consistent across countries. Progression from ICS to CS is rare, with only isolated cases reported.
Future directions and research gaps
Current evidence suggests that ICS typically presents at 55–60 years of age, shows no sex predilection, and is associated with a worse prognosis compared with systemic CS. Management should prioritize early detection and treatment of ventricular arrhythmias and tachycardia, with corticosteroids such as prednisolone and immunosuppressants like azathioprine as the main therapies, alongside beta-blockers for symptomatic control. Diagnosis relies on clinical assessment, exclusion of alternative cardiac diseases via tests such as ECG, and confirmation with advanced imaging—particularly 18F-FDG PET-CT.
Despite these insights, significant research gaps remain. Future studies should leverage ongoing national and international cohort databases and conduct nested case–control analyses to better define disease patterns. Rigorous evaluation of current treatments is needed to establish their efficacy in ICS. The diagnostic performance of commonly used tests, including FDG-PET/CT, CMR, and biopsy, should be systematically validated in this population. Additionally, prospective studies should aim to identify prognostic factors that can guide risk stratification and optimize long-term management strategies.
Finally, it should be noted that the HRS is currently revising its 2014 consensus statement, with a new version expected in 2026 that is anticipated to include specific guidance on ICS. While our systematic review is based on the presently available guidelines (HRS 2014, JCS 2016, AHA 2024), future updates will need to incorporate the forthcoming HRS recommendations once published.
Conclusion
ICS is a distinct but high-risk entity, presenting predominantly in middle age and often leading to severe arrhythmias, conduction disease, and LV dysfunction. Compared with systemic CS, ICS is consistently associated with poorer outcomes, including higher mortality and hospitalization rates. Diagnosis is hindered by nonspecific findings and the limited sensitivity of endomyocardial biopsy, making advanced imaging—particularly CMR and 18F-FDG PET—central to detection. Current treatments rely on corticosteroids, adjunctive immunosuppressants, and device therapy yet are supported only by observational evidence. Robust, multicenter prospective studies are urgently needed to refine diagnostic criteria, identify prognostic markers, and establish evidence-based management strategies.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251392454 - Supplemental material for Isolated cardiac sarcoidosis: A systematic review
Supplemental material, sj-pdf-1-imr-10.1177_03000605251392454 for Isolated cardiac sarcoidosis: A systematic review by Shokoufeh Hajsadeghi, Elham Piryaie and Shayan Mirshafiee in Journal of International Medical Research
Footnotes
Acknowledgments
None.
Author contributions
S.H. conceptualized the study, designed the research approach, and provided overall supervision throughout the project. E.P. conducted data curation, extraction, and analysis and contributed to the initial drafting of the manuscript. S.M. contributed to writing the manuscript and performed the final editing and critical revisions for intellectual content.
Data availability statement
The data supporting this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
None.
Funding
None.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
