Abstract
A 64-year-old female complaining of unrelieved chest pain for 2 days was admitted to the Emergency Room of the Beijing Anzhen Hospital, Beijing, China. After definitive diagnosis, a percutaneous coronary intervention was implemented, but immediately after embedding the stent in the distal area of the right coronary artery, an acute coronary and aortic dissection was found. Cardiologists immediately gave the patient conservative management. At the same time, another smaller stent was immediately embedded in the proximal area of the right coronary artery and plunged into the ascending aorta by 2 mm, with the intention of covering the tear of the dissection. Repeated coronary angiography showed that a 40% stricture of the distal right coronary artery remained and less contrast agent had been extravasated. The patient was then transferred to the Department of Cardiac Surgery and received emergency surgery consisting of right coronary artery bypass grafting and ascending aorta replacement. The patient remained in the intensive care unit for 18 days after the surgery. The patient recovery was acceptable and she was discharged with a small amount of bilateral hydrothorax, moderate malnutrition oedema and iron deficiency anaemia.
Introduction
Iatrogenic acute aortic dissection (IAAD) is a fatal complication that can occur during open cardiac surgery and coronary interventions, 1 such as off-pump coronary artery bypass grafting, 2 thoracic endovascular aortic aneurysm repair, 3 trans-aortic valve replacement 4 and complex percutaneous coronary intervention (PCI). 5 Among these procedures, the cumulative incidence of catheter-induced aortic dissection has been estimated at approximately 0.062% during both diagnostic and therapeutic procedures. 1 However, the follow-up treatment strategies (conservative or surgical treatment) and relevant standards remain controversial.
Case report
A 64-year-old female complaining of unrelieved chest pain for 2 days was admitted to the Emergency Room of the Beijing Anzhen Hospital, Beijing, China on 31 October 2016. Evaluation of an electrocardiogram with her laboratory examinations and symptoms clearly indicated the diagnosis of acute non-ST segment elevation myocardial infarction. She was then admitted into one of our cardiology general wards.
Ten years previously, the patient underwent PCI (one coronary stent in the left anterior descending branch, and another in right coronary artery), and about 2 years ago, the patient underwent PCI again (one stent in the posterior branch of the left ventricle) because of similar symptoms. Throughout the last 10 years after the first surgery, she suffered from intermittent chest pain, which had become more intense in the last 3 months. The patient also had a history of high blood pressure for 30 years and had been receiving regular drug therapy during the last 10 years. Her blood pressure was usually controlled at approximately 140/80 mmHg during a resting state. She was never definitely diagnosed with type 2 diabetes mellitus or other diseases.
After admission onto the ward, bedside echocardiography was performed and showed acceptable cardiac function with mild mitral regurgitation. Routine laboratory investigations also eliminated liver and kidney dysfunction. With complete preoperative preparation, the patient underwent coronary angiography (CA) and PCI that night. The CA was performed smoothly and showed a 95% in-stent stricture in the remote area of the right coronary artery (RCA) (Figure 1a). During PCI, right after embedding the stent in the distal area of the RCA, we observed by CA that contrast agent was stranded in the proximal area of the RCA and spread outside of the aortic wall. At this time, the patient immediately felt a foreign body sensation in the throat with mild chest tightness, and thus, a coronary and aortic dissection was suspected (Figure 1b). Cardiologists immediately controlled her blood pressure with an intravenous method and gave her analgesic treatment and rehydration therapy. At the same time, another smaller stent was immediately embedded in the proximal area and plunged into the ascending aorta by 2 mm, with the intention of covering the tear of the dissection. Repeat CA showed that a 40% stricture of the distal RCA remained (the real degree of stricture may be higher than this because of coronary aortic dissection) and that there was less contrast agent extravasation (Figure 1c), demonstrating that there was no expansion of the dissection. The symptoms of the patient were also markedly relieved. During the entire process of the operation, the patient’s blood pressure fluctuated from 140/70 mmHg to 150/90 mmHg, the heart rate was approximately 70 beats/minute, and oxygen saturation was approximately 100% throughout the procedure.
Coronary investigations of a 64-year-old female who had unrelieved chest pain for 2 days and a history of hypertension and previous symptoms requiring percutaneous coronary intervention (PCI). (a) A coronary angiography was performed before the PCI and showed a 95% in-stent stricture in the remote area of the right coronary artery (RCA). (b) A coronary and aortic dissection was suspected. The left arrow in the figure shows the contrast agent outside of the aortic wall. The right arrow in the figure shows the stranded contrast agent in the right coronary sinus. (c) A 40% stricture of the distal RCA remained (the real degree of stricture may have been higher than this because of coronary aortic dissection) and that there was less contrast agent extravasation.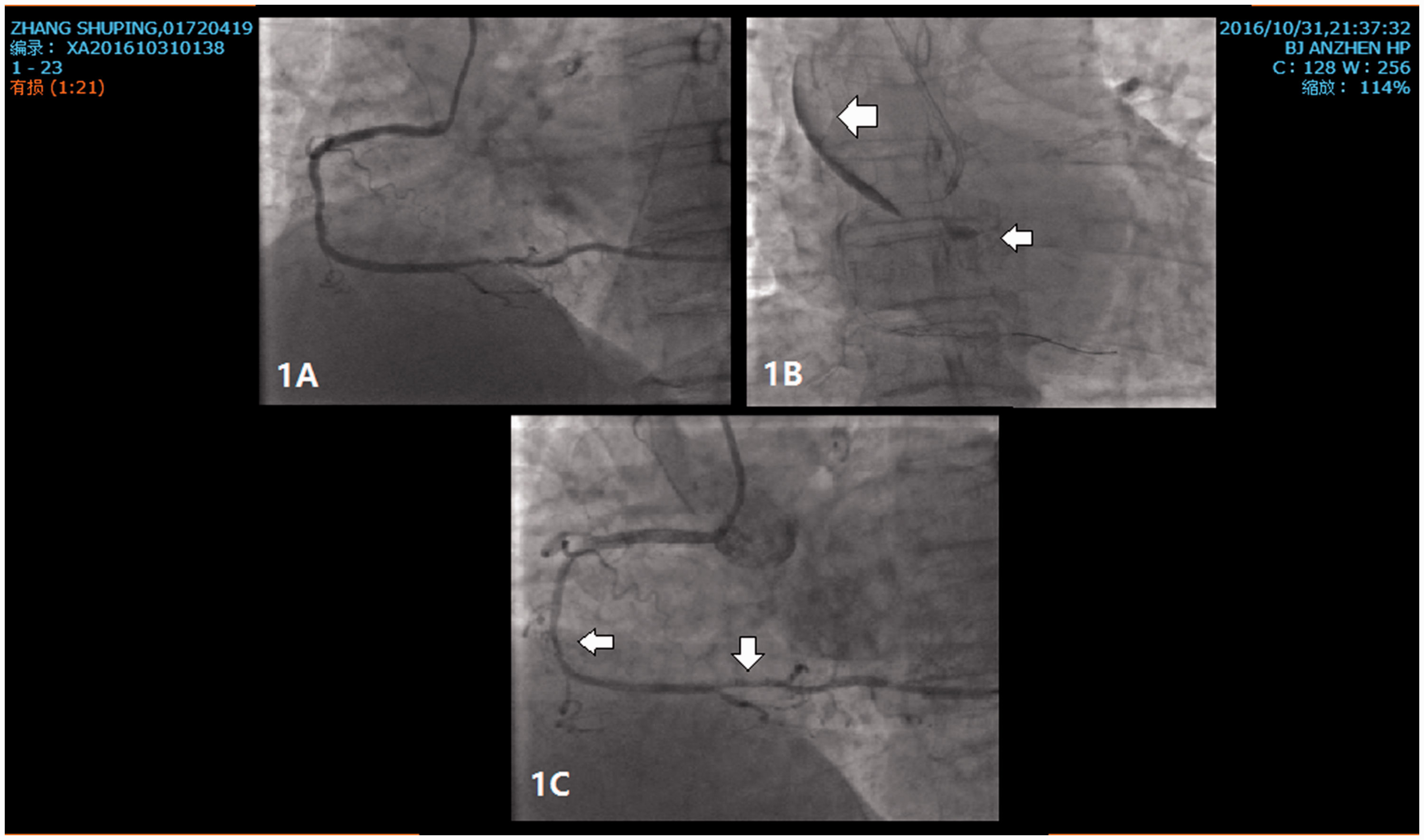
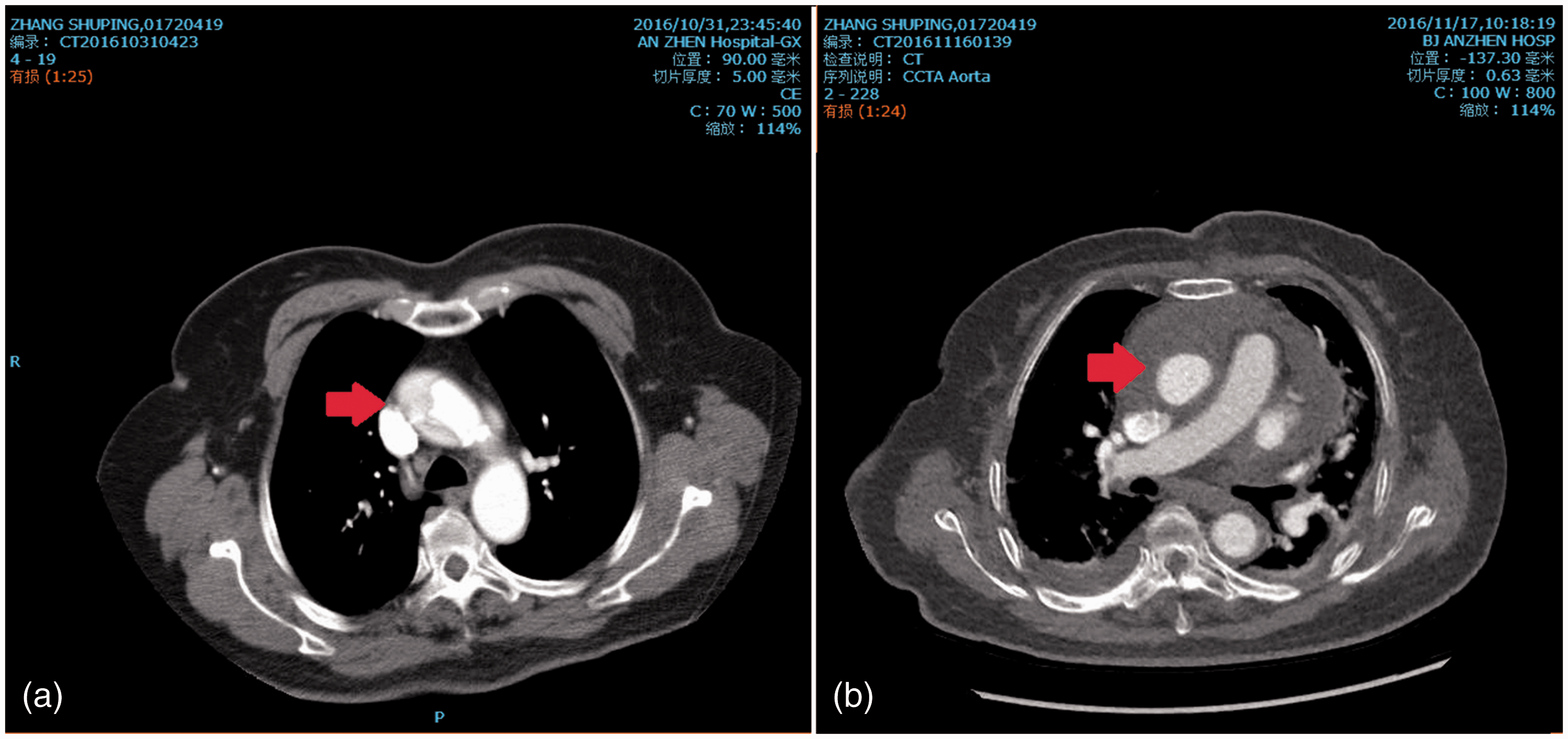
In case of worsening of the dissection, the interventional physicians asked for immediate consultation for cardiac surgery. An urgent bedside transthoracic echocardiography was also performed, and the aortic valve showed a mild regurgitation without pericardial infusion. Urgent aortic computed tomography angiography (CTA) showed that the dissection expanded to the ascending aortic artery and innominate artery, but no other part of the aortic arch was involved (Figure 2 and 3a). The above-mentioned results led to a diagnosis of ‘acute aortic dissection (Stanford A)’. When the patient was taken back to the ward and the clinical consultation proceeded, the patient started to feel an unrelieved thoracic backache again, followed by a discontinuous fall of blood pressure, and the effects of rehydration and cardiac therapy gradually became worse. The patient was then transferred to the Department of Cardiac Surgery, Beijing Anzhen Hospital, Beijing, China during that night, then received an emergency operation. Transoesophageal cardiography was performed during surgery, and the result was consistent with the preoperative echocardiogram. We established a cardiopulmonary bypass, took out the right coronary stent and performed right coronary artery bypass grafting and ascending aorta replacement surgery. The surgery was rather difficult because the haemostatic process was complicated by the excessive weight of the patient and preoperative dual anti-platelet therapy. However, cardiac function during the operation was acceptable, which may have been due to the immediate stent implantation. The patient woke up 7 days after surgery with stable blood circulation and without definite neurological lesions in a head computed tomography scan. We speculated that her delayed awakening might have been related to the untreated innominate artery. After staying in the intensive care unit for 14 days, she was returned to the general ward. Subsequent echocardiography and aortic CTA did not show any abnormalities (Figure 3b). Finally, the patient was discharged with a small amount of bilateral hydrothorax, moderate malnutrition oedema and iron deficiency anaemia 18 days after the surgery.
Urgent aortic computed tomography angiography before surgery showed that the dissection expanded to the ascending aortic artery and innominate artery. The red arrow shows the involved innominate artery. The colour version of this figure is available at: http://imr.sagepub.com. (a) Urgent aortic computed tomography angiography (CTA) before surgery showed that the dissection expanded to the ascending aortic artery. The red arrow shows the involved ascending artery. (b) Subsequent aortic CTA after surgery showed that the false lumen had been eliminated and there was no residual leak. The colour version of this figure is available at: http://imr.sagepub.com.
Discussion
Most IAADs are caused by complicated lesions of the cardiovascular system, such as chronic total occlusion, 6 acute myocardial infarction, 7 and chronic heart failure caused by valvular heart disease. 4 The vast majority of cases are urgent and lack systematic surgical preparation and management, such as the continued use of combined anticoagulant drugs, uncontrolled hyperglycaemia and chronic obstructive pulmonary dysfunction. These factors explain why operations related to IAAD are high risk and are associated with high mortality rates. 1 Thus, experimental treatment and effective cooperation between departments are essential in improving the survival rate of this kind of disease.
The diagnosis of IAAD during PCI
Chest pain is the most frequent symptom of IAAD. 8 It may manifest in a sharp, ripping, tearing, knife-like pain, which is typically different from chest pain of other causes, and the abruptness of its onset is the most specific characteristic. 9 However, in this present case, the foreign body sensation in the throat with mild chest tightness was the main symptom during the onset of dissection, as demonstrated by CA, which has also been mentioned to be the only symptom in other cases.10,11 It suggests that a sore throat during CA or PCI may be a significant symptom of IAAD.
Imaging examinations are most important in the diagnosis of IAAD. The National Heart, Lung, and Blood Institute classifies coronary-induced dissections into six types (A–F) based upon their CA appearance: 12 (A) Minor radiolucent areas with little or no persistence of contrast; (B) Parallel tracts or a double lumen with little or no persistence of contrast; (C) Contrast outside of the coronary lumen with persistence of contrast; (D) Spiral dissection with filling defects; (E) Persistent filling defects in the lumen; and (F) Total occlusion without distal flow. In this present case, we were able to diagnose the patient with a typical type C coronary dissection. Except for the CA appearance during interventional operation, aortic CTA could be the gold standard in the diagnosis of coronary and aortic dissection.
Laboratory tests can also provide strong evidence for the diagnosis of IAAD, especially coagulation-related indicators, such as platelet counts, D-dimer and fibrinogen/fibrin degradation products (FDP). Authorities have proven that a dramatic elevation of D-dimer and FDP are indicative of AAD. 13
How should cardiac interventional doctors treat IAAD?
First, when the diagnosis of aortic dissection is suspected, the operator should minimize the contrast media injections since this may promote further expansion of the dissection since the contrast media will preferentially circulate in the false lumen. Secondly, following basic principles, the injury in the coronary artery should be sealed after a definitive diagnosis has been made. That is why we embedded a smaller stent in the proximal area during PCI and repaired the right coronary ostium during the surgery.
We believe that those are essential procedures for both the short-term and long-term prognosis of patients. In this present case, the successful implantation of another coronary stent after dissection could be beneficial in several aspects. First, the coronary stent could restore coronary blood flow as much as possible, which was significant for surgical safely. Secondly, covering the tear of the dissection with a stent in a timely matter could effectively slow down the progression of the dissection (we could determine that the dissection was confined in the ascending aorta during the operation). Thirdly, it could effectively relieve the pain caused by the dissection.
What should cardiac surgeons do when dealing with IAAD?
First, it is important to quickly determine the surgical indication. Medical and conservative management is definitely a better choice for this type of patient, but for this particular case, there were several reasons why we chose a risky surgery. On the one hand, the in-stent stricture in the remote area of the RCA was not solved; although her right heart function might be sustained for some time, the intermittent chest pain could not be resolved, which was rather difficult to distinguish from the symptoms of aortic dissection development. We believed that was a very risky hidden danger for her long-term recovery. On the other hand, although bedside transthoracic echocardiography showed a mild regurgitation of the aortic valve without pericardial infusion, this patient began to feel unrelieved thoracic backache after the analgesic treatment was stopped, the patient’s blood pressure began to fall discontinuously after getting off the operating table, and the effects of rehydration and cardiac therapy gradually became worse when the patient returned to the general ward. After clinical consultation and after talking to the patient’s family members, we decided to perform the surgery.
Secondly, the operative strategy should be selected properly. Different treatment strategies should be performed based on the region of the different lesions and the patients’ systemic condition. In this present case, we dealt only with the ascending aorta, which not only eliminated the risk of aortic rupture, but also avoided the risk of moderate hypothermic circulatory arrest as well, 14 which proved to be an advisable choice. It is worth emphasizing that we removed the sealed stent during surgery, considering two relative problems. The first one being that we needed to remove the stent and ensure cardioplegic solution perfusion through the coronary ostial during surgery. The second problem concerned the potential to develop a coronary dissecting aneurysm during the long-term follow-up because the sealed stent was removed during surgery.
Conclusion
Iatrogenic acute aortic dissection caused by PCI occurs in approximately 0.062% of diagnostic CA and aortic CTA procedures, and often causes serious complications. Therefore, more experience in dealing with IAAD caused by PCI would be beneficial. First, it is important to make a quick and definite judgement based on symptoms, laboratory tests and imaging examinations, as some of them are distinct and might be used for the differential diagnosis. Secondly, it might be a feasible treatment to embed a stent to cover the tear of the dissection immediately when IAAD is first diagnosed, as it could restore coronary blood flow as soon as possible and cover the tear of the dissection in a timely manner to slow down the progression of the dissection.
Footnotes
Acknowledgements
We acknowledge Hui Ai and Mei Wang (Department of Cardiology, Beijing Anzhen Hospital, Capital Medical University, Beijing, China) for their assistance in reviewing the manuscript during its development.
Declaration of conflicting interests
The authors declare that there are no conflicts of interests.
Funding
This study was financially supported by grants from the National Science Foundation of China (no. 81470580 and no. 81600362), the General Programme of Science and Technology Development Project of Beijing Municipal Education Commission of China (no. KM201610025019), and the Clinical and Basic Science Cooperation Projects of Capital of Medical University (no. 17JL71).
