Abstract
Objective
We aimed to compare Demodex positivity before and during the coronavirus disease 2019 pandemic.
Methods
Patients who underwent a standardized skin surface biopsy and direct microscopic examination for Demodex species in the prepandemic or pandemic period at a tertiary care center were included. The positivity rates in the two periods were compared across age, sex, and months using a proportional Z-test. A p value <0.05 was considered statistically significant.
Results
In total, 573 and 523 patients were included in the pandemic and prepandemic periods, respectively. Demodex positivity was significantly higher in the pandemic period (68.9%) than in the prepandemic period (50.5%) (p < 0.00001). Demodex positivity increased significantly among male (p = 0.00386) and female groups (p < 0.00001) in the pandemic period compared with that in the prepandemic period. Positivity rates increased in all age groups, with significant increases observed in those aged 10–25 (p = 0.00006), 36–45 (p = 0.0012), and 46–55 (p = 0.01242) years.
Conclusion
Demodex positivity was higher in the pandemic period than in the prepandemic period, with significant increases observed during colder months. Furthermore, the increase in positivity was most prominent in patients aged 10–25 years, who spend more time in schools or crowded work places. Therefore, a possible association of face mask use and higher Demodex positivity must be explored through future controlled studies.
Introduction
Demodicosis is a parasitic infestation caused by Demodex brevis and Demodex folliculorum; it may cause blepharitis, dermatitis, and otitis, especially in immunosuppressed patients. 1 These ectoparasites can affect individuals of all age groups; however, the incidence tends to be higher in older individuals. 2
As parasitic diseases are closely associated with the immune status of patients, it is interesting to study the frequency and severity of these infestations during the coronavirus disease 2019 (COVID-19) outbreak. 3 Contradictory results have been reported in the literature about the effect of parasitic co-infections with COVID-19; some studies have reported that parasites disrupt the immune system and increase the morbidity and mortality associated with COVID-19, while others have suggested that they exert protective effects against COVID-19.4–7
When evaluating the association of parasitic infestations with COVID-19, various entities including the immune status, drug regimens, isolation conditions, mode of infestation transmission, site of parasitic infestation, and pathogenicity of parasite must be considered. 3 Therefore, reports on this topic in the literature are limited and contradictory. Moreover, most studies have analyzed the effect of parasites on COVID-19 outcomes; only a few have examined the status of parasitic infestations during the outbreak. Limited data are available on cutaneous infestations such as demodicosis during the COVID-19 outbreak. 8 In this study, we aimed to compare the demographic features and seasonal changes associated with Demodex infestation between the prepandemic and pandemic periods.
Material and methods
Patients
In this retrospective study, we reviewed the microbiological test results of patients consecutively admitted to a tertiary university hospital who underwent a standardized skin surface biopsy followed by a direct microscopic examination for the diagnosis of demodicosis between January 2018 and March 2020 (27 months) (in the prepandemic period) and between March 2021 and December 2022 (22 months) (during the COVID-19 pandemic). We identified these periods because the COVID-19 pandemic started in December 2019 and according to the Health Ministry of Turkey, the first case in Turkey was detected on 11 March 2020. After a few weeks, the infection was detected in eastern Anatolia. The test was not available between April 2020 and February 2021 because only patients with emergent or severe chronic conditions were referred during this period.
This cross-sectional retrospective study was approved by the Yüzüncü Yıl University Ethics Committee on 14 April 2023 (under 2023/04-12) and was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. All patient details have been de-identified in the manuscript. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 9
Diagnosis of Demodex infestation
We used a standardized skin surface biopsy for the detection of Demodex spp. in our laboratory. In this method, after a drop of cyanoacrylate adhesive is applied to a cellophane tape, the band is stuck over a possible infected region in the face. After allowing the cyanoacrylate adhesive to dry for approximately 1 min, the band is removed and fixed onto a slide. After the application of xylene, the slide is examined for the presence of Demodex mites and their eggs under a microscope with objective lens (10× magnification) (Figure 1).

Demodex mites visible under a light microscope at a magnification of 40×.
Statistical analyses
Minitab Statistical Software, version 14, was used for statistical analysis. Continuous variables were presented as mean ± SD, minimum, and maximum values. Categorical variables were expressed as numbers and percentages. A Z test was used for comparison of proportions. p > 0.05 was considered to indicate statistical significance.
Results
In total, 523 consecutive patients (aged 45.7 ± 14.6 (12–90) years) were examined for Demodex spp. in the prepandemic period (27 months) and 573 (aged 42.9 ± 14.6 (11–89) years) in the pandemic period (22 months). Demodex was detected in 264 (50.5%) patients in the prepandemic period and 395 (68.9%) in the pandemic period. Demodex positivity was significantly higher in the pandemic period than in the prepandemic period among all patients (p < 0.00001). Furthermore, females exhibited a significantly higher positivity rate in the pandemic period than in the prepandemic period (69.2% (308/445) vs. 50.4% (191/379)). A similar trend was observed for male participants (68% (87/128) vs. 50.7% (73/144)) (p < 0.00001 and p = 0.00386, respectively) (Table 1).
Distribution of the study patients according to sex, age groups, and months in the prepandemic and pandemic periods.

The Demodex positivity rate in females (60.6% (499/824)) was slightly higher than that in males (58.8% (160/272)) for the entire duration; however, the difference was nonsignificant (p = 0.61006). In addition, the difference in Demodex positivity rates between females and males was nonsignificant in both prepandemic (50.4% (191/379) vs. 50.7% (73/144)) and pandemic (69.2% (308/445) vs. 68.0% (87/128)) periods (p = 0.95216 and p = 0.78716, respectively).
According to the months, during the prepandemic period, Demodex positivity was highest in September (58.6%), March (57.1%), and May (56.0%), while it was lowest in November (37.8%). For the pandemic period, this rate was highest in May (80.9%), February (78.9%), and January (76.3%), while it was lowest in October (56.4%). During all months, there was an increase in the positivity rate; however, this increase was statistically significant only in January (49.0%–76.3%), April (53.2%–74.5%), May (56.0%–80.9%), June (39.3%–74.3%), and November (37.8%–63.8%) (p < 0.05) (Table 1, Figure 2).

The distribution of Demodex positivity according to months in the prepandemic and pandemic periods (%).
Demodex positivity in both prepandemic and pandemic periods generally increased with patient age. The highest positivity rates were observed in patients aged >56 years (66.7% in the prepandemic and 80.5% in the pandemic period) and those aged 36–45 years (57.9% in the prepandemic and 76.4% in the pandemic period). Patients from all age groups exhibited an increase in the positivity rate in the pandemic period; this increase was statistically significant in those aged 10–25 (p = 0.00006), 36–45 (p = 0.0012), and 46–55 (p = 0.01242) years (Figure 3).

The distribution of the Demodex positivity across age groups in the prepandemic and pandemic periods.
The Demodex positivity rate among patients with seborrheic dermatitis, allergic dermatitis, irritant contact dermatitis, and rosacea decreased in the pandemic period, while that in patients with contact dermatitis increased; the differences in positivity rates between the prepandemic and pandemic periods were −0.15%, −0.22%, −0.37%, −9,33%, and +0.25%, respectively. A comparison of Demodex positivity among patients with dermatological diseases in the prepandemic and pandemic periods is detailed in Figure 4.
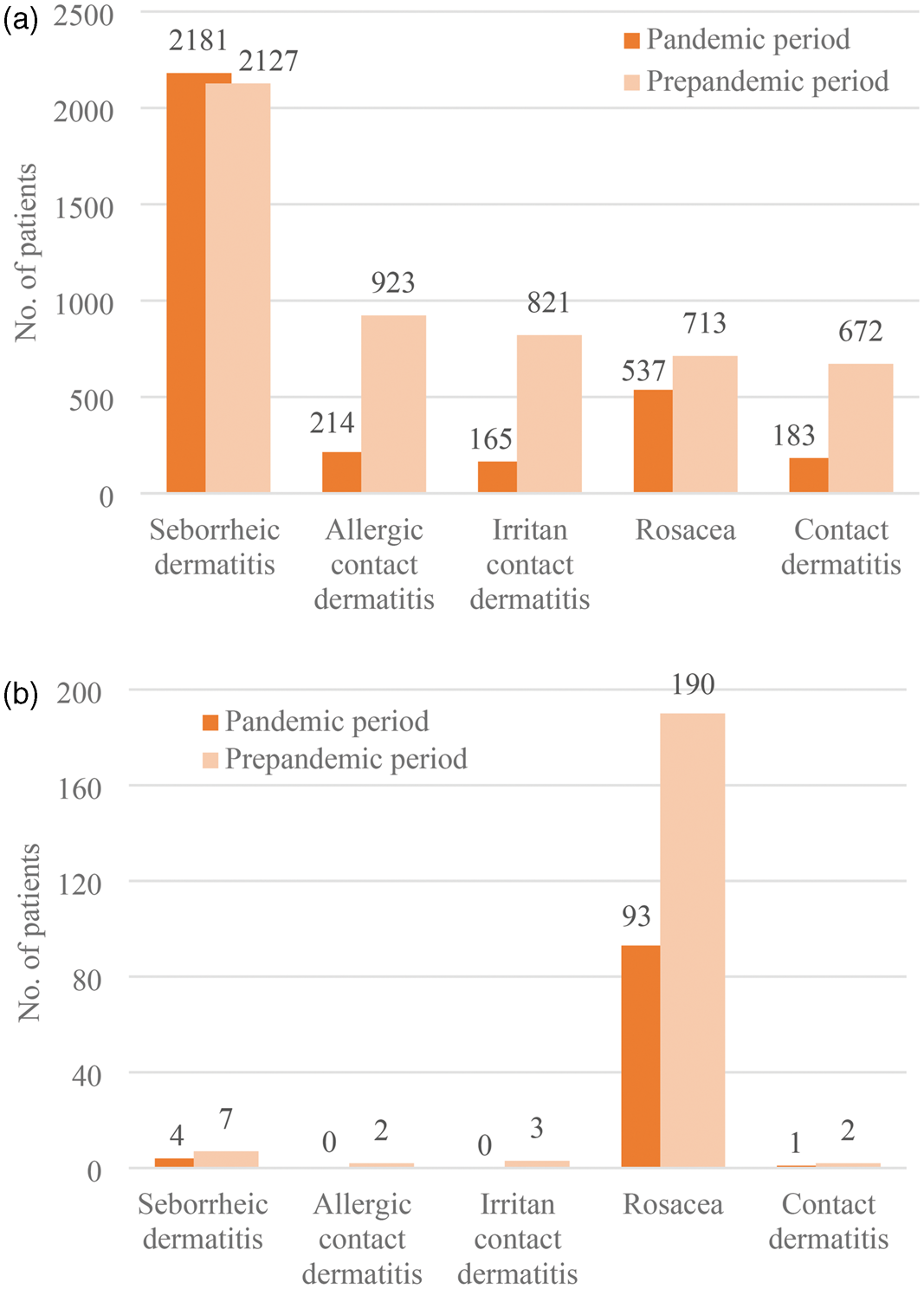
(a) Distribution of all patients who were diagnosed during the prepandemic and pandemic period and (b) distribution of Demodex-positive patients across other disease groups.
Discussion
Demodex infestation is usually observed when the immune system is disrupted by a comorbidity or immunosuppressant drugs. 10 Parasitological diseases may exert a protective effect against COVID-19; this assumption is based on the fact that fewer COVID-19 cases and COVID-19–related deaths were reported in Africa, where infestations are very common. This suggests that immunomodulation mechanisms associated with parasitic infestations may lead to milder COVID-19. 4 The effect of COVID-19 on parasitic infections is an interesting topic. Demodex infestation is closely associated with the immune status of patients; therefore, we compared the demographic features of demodicosis patients between the prepandemic and pandemic periods.
There are contradictory results about the difference in the rates of cutaneous parasitic infestations between the prepandemic and pandemic periods. Several reports have been published on scabies incidences during the pandemic and nonpandemic periods in different locations. In some countries, including Italy 11 and the Netherlands, 12 there was an increase in this incidence, while in other countries such as USA 13 and France, 14 a decrease was noted. Nonsignificant differences in the incidence of scabies and tinea capitis/barbae were also reported in Sweden. 15 Roongpisuthipong et al. studied 20,915 patients with skin lesions from 2019 to 2021 and concluded that the rates of viral and fungal skin infections, urticaria, psoriasis, acne, and autoimmune diseases significantly increased during the COVID-19 pandemic, while the rates of pigmentary disorders, xerosis cutis, other skin infections (syphilis and parasitic infections), and nail and hair disorders decreased. They stated that the rates of other skin infections were 1% in the prepandemic period and 0.6% in the pandemic period (p < 0.001). 16 However, in our study, we found a statistically significant increase in the average monthly number and rate of Demodex-positive patients.
A higher proportion (5.7 times) of Demodex mites are reportedly found in the skin of patients with rosacea than in the skin of controls. This association is attributed to the fact that the food source of mites is sebum or protein. It was also suggested that Demodex mites induce and aggravate the immune response in patients, contributing to inflammation due to rosacea.17,18 A possible association of daily mask use with rosacea incidence has been examined previously. Trave et al. compared the rate of papulopustular rosacea relapse between the pandemic and postpandemic periods among 30 patients who reportedly used a face mask daily during the pandemic period. They found no statistically significant difference and concluded that daily mask use was not associated with an increased risk of papulopustular rosacea relapse. 19 Consistent with this study, rosacea was identified in an average of 26.4 patients per month during the prepandemic period and 24.4 patients per month in the pandemic period in our study. There was a slight decrease in the monthly average number of rosacea patients in the pandemic period compared with that in the prepandemic period. Paichitrojjana reported the case of a 46-year-old woman who was diagnosed with demodicosis after wearing a face mask for >8 h daily for 3 weeks; the patient had no history of this condition or recent use of topical drugs or cosmetics. 20 This potential association may be due to decreased skin hydration, increased sebum secretion, and higher skin temperature owing to daily use of face mask. 21 Consistent with these data, the average monthly number of Demodex-positive patients in the prepandemic period was 9.8 (264 patients over 27 months) and that during the pandemic period was 17.9 (395 patients over 22 months) in our study. Approximately two-fold increase was detected in the average monthly number of Demodex patients in the pandemic period compared with that in the prepandemic period; this was not correlated with the nonsignificant difference in the number of rosacea patients. Thus, we believe that the increase in the number of Demodex-positive patients must be attributed to another condition.
Various studies have shown that Demodex infestation in more common during autumn and winter seasons.8,22–24 In our study, the highest rate was detected in September, March, and May during the prepandemic period and May, February, and January during the pandemic period. The lowest rate was detected in November during the prepandemic period and October during the pandemic period. Consistent with the findings of the abovementioned studies, the lowest rate was detected in summer (45.3%) followed by winter (51.4%), autumn (53.4%), and spring (55.4%) during the prepandemic period. In contrast, during the pandemic period, the highest rate was detected in summer (68.0%) followed by autumn (71.9%), winter (72.3%), and spring (76.4). However, the highest rates in both prepandemic and pandemic periods were observed in spring (55.4% and 76.4%, respectively). We believe that this inconsistency in the results is attributable to the colder climate and higher altitude of our region because low air temperatures are usually also observed in the spring season. The lowest difference in the positivity rate between the prepandemic and pandemic periods was detected in September, October, and August while the highest was found in June, January, and November. In particular, the increases in the positivity rate during November, December, January, and February (24.1%) were more prominent. We believe that during cold months, people tend to stay at home or spend more time at their workplace where they are required to use face masks more commonly. However, during hot months, people go outside or visit rural areas more often, where there is minimal mask use. Furthermore, long school holidays during hot months could reduce the rate of daily mask use, supporting our finding. However, we cannot provide a rationale for the highest increase in the positivity rate in June.
The sex-based differences in the positivity rate found in our study is similar to that reported previously, with no significant difference.8,23,25,26 Moreover, the increase in the positivity rate among men and women (+17.3% vs. +18.8%) between the prepandemic and pandemic periods was similar.
The highest rate of infestation was detected among patients aged >65 years in both prepandemic and pandemic periods, consistent with a previous study. 23 However, the highest difference in the positivity rate between the prepandemic and pandemic periods was noted in patients aged 10–25 years and lowest difference was noted among those aged 26–35 years. This finding may support the theory of face mask use as a risk factor for Demodex infestation. This is because younger patients, who tend to spend more time in schools or crowded work places, use face masks daily and more commonly than older people, who are usually retired and spend more time at home. However, no objective results were obtained from our comparison of patients from the same age group who did and did not use face masks on a daily basis.
In a literature review in Iran, the researchers assessed patients with scabies and Demodex infestation and reported a significant decrease in the positivity rates of both ectoparasites during the COVID-19 pandemic. They also compared the decline in the scabies incidence in İran and European countries with the outbreak of scabies in Turkey during the COVID-19 pandemic. They attributed this to the high number of individuals per household and the movement of people to rural areas during the pandemic in Türkiye. 8 Another study from Turkey reported an average increase of 1038% in scabies incidence between 2017 and 2021. 27 This may also be due to the high number of individuals per household, which leads to more interactions among people, and the aforementioned movement of people to rural areas, where poor socioeconomic conditions lead to poor hygiene. However, we could not suggest such an association for Demodex infestation.
One of the major limitations of our study is the unequal observation durations for the prepandemic and pandemic period (27 vs. 22 months), which may have affected our results. In addition, confounding factors, such as skin hygiene, underlying diseases, and changes in healthcare-seeking behavior, which are closely associated with the infestation status were not studied. Although we found some results that may support an association of Demodex infestation with daily face mask use, this hypothesis must be further investigated in more detailed, prospective studies, which should also examine confounding factors using more robust statistical analyses such as logistic regression in controlled groups for similar observation periods.
Conclusion
Data on daily mask use and cutaneous parasitic infestations remain contradictory and insufficient. We found a significant increase in Demodex positivity rates, with the increase being highest in the cold months and lowest in the hot months. Furthermore, the increase was most prominent in patients aged 10–25 years. These findings may support the theory that daily use of face mask use is associated with an increased risk of Demodex infestations. However, larger, multicenter studies must be conducted to confirm this association.
Footnotes
Acknowledgements
None.
Author contributions
Data availability statement
The dataset of this study is available on request from the corresponding author.
The authors declare that this paper is original and not under consideration in elsewhere.
Declaration of conflicting interests
There is no conflict of interests.
Funding
No funding was received for this study.
Informed consent statement
The study employed a retrospective design; therefore, informed consent was not required. All patient details have been de-identified in the manuscript.
